Enhanced Uptake of Fe3O4 Nanoparticles by Intestinal Epithelial Cells in a State of Inflammation
Abstract
:1. Introduction
2. Results and Discussion
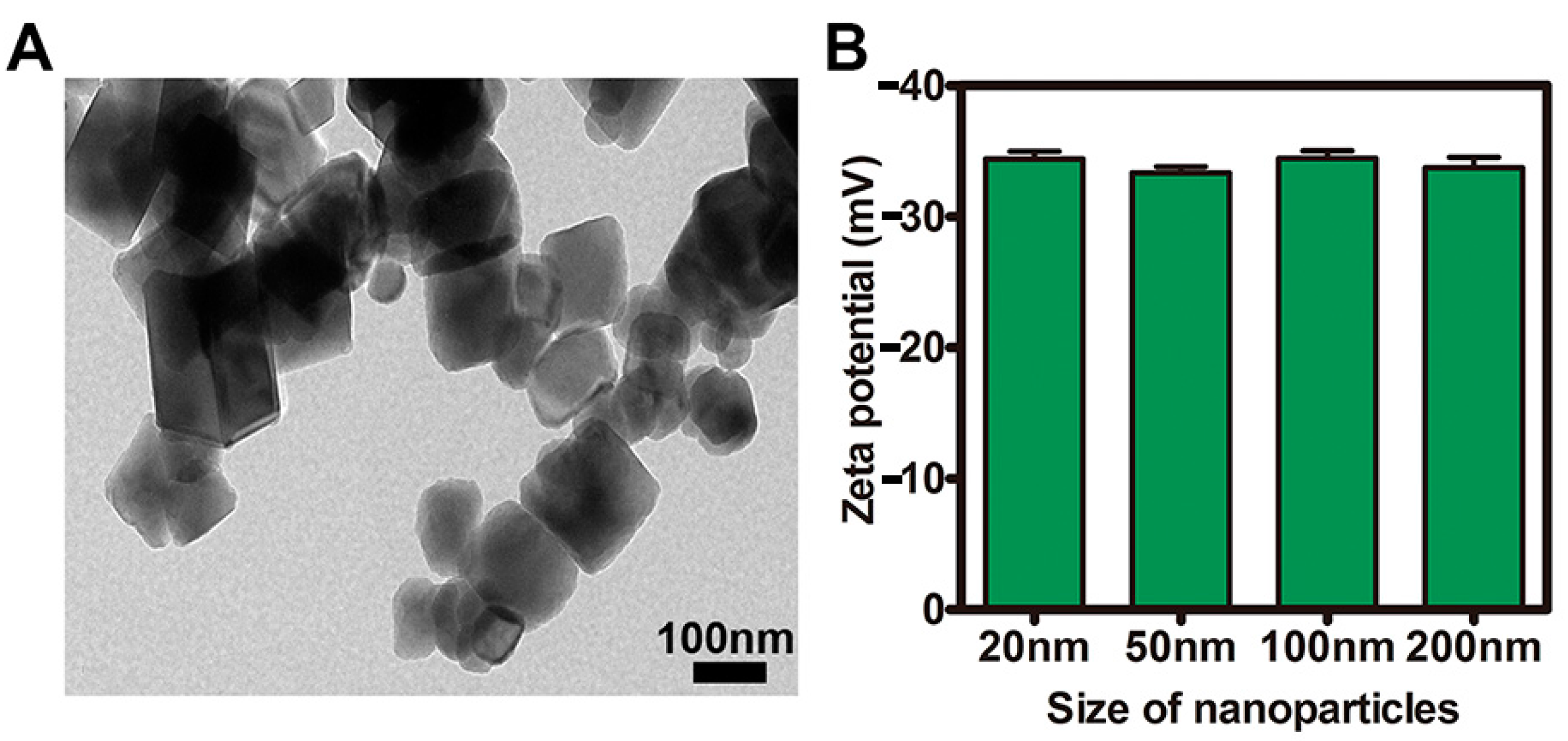
2.1. Characterization of NPs
2.2. CCK-8 Assays and Measurement of Transepithelial electrical resistance (TEER) and Inflammation
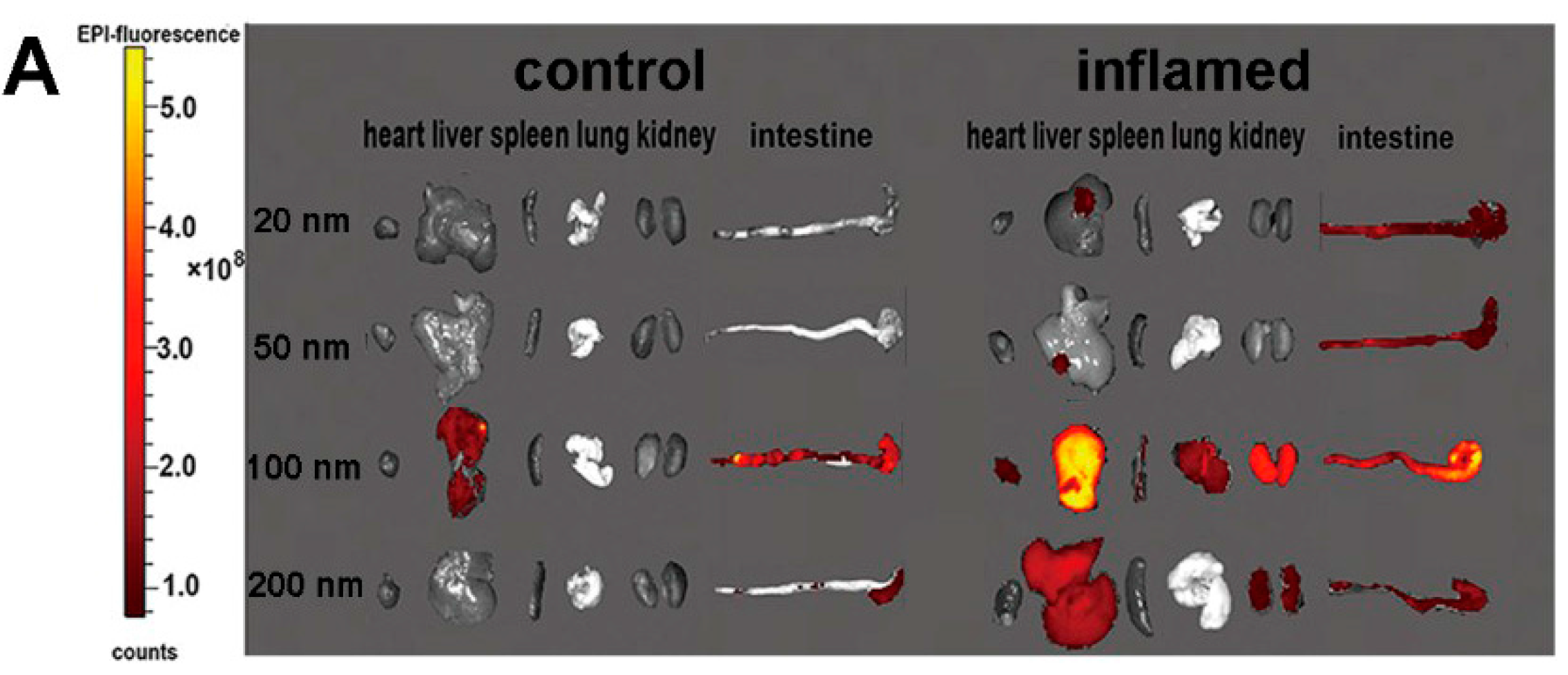
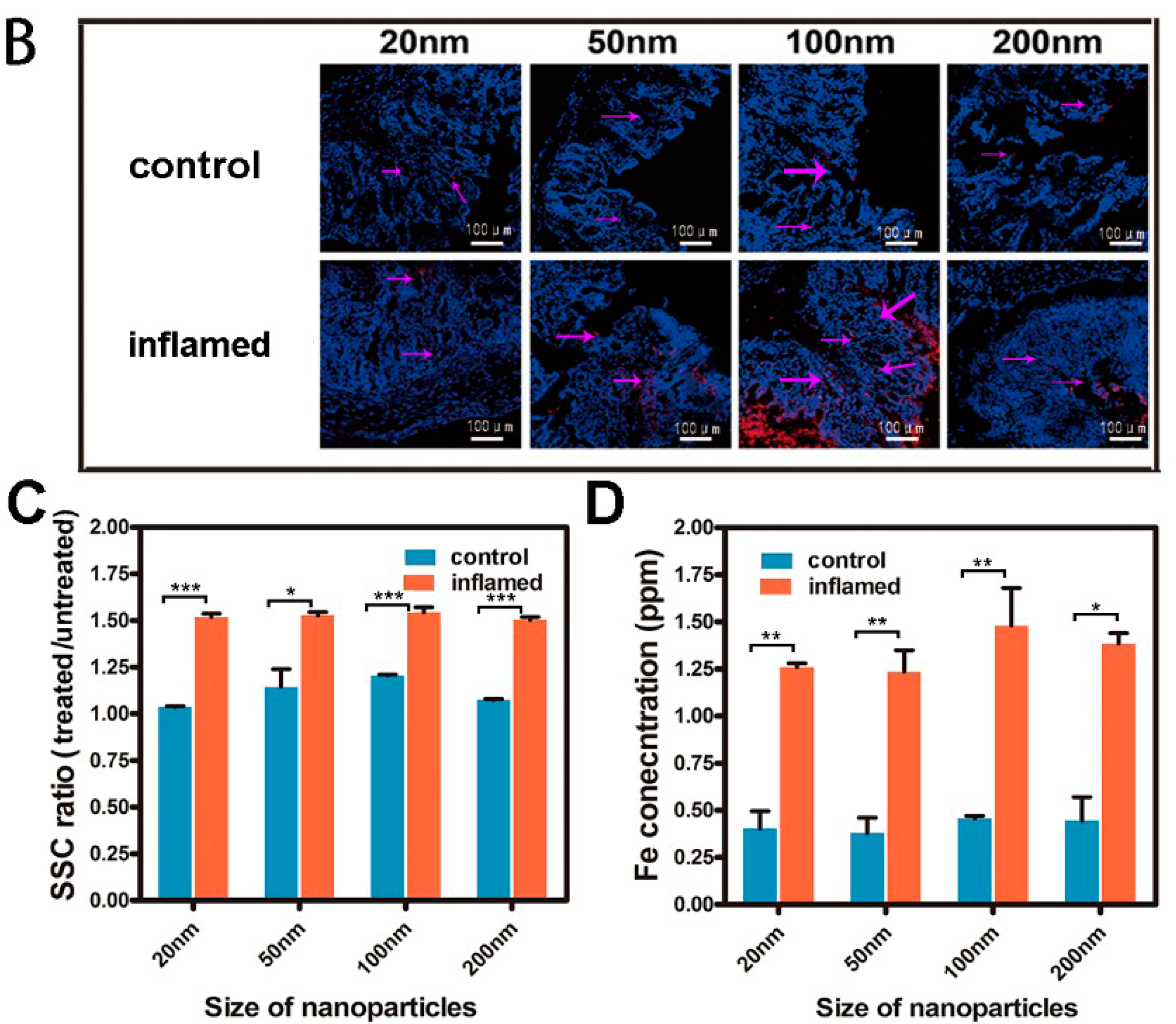
2.3. Tissue Distribution and Cellular Localization of Fe3O4 NPs
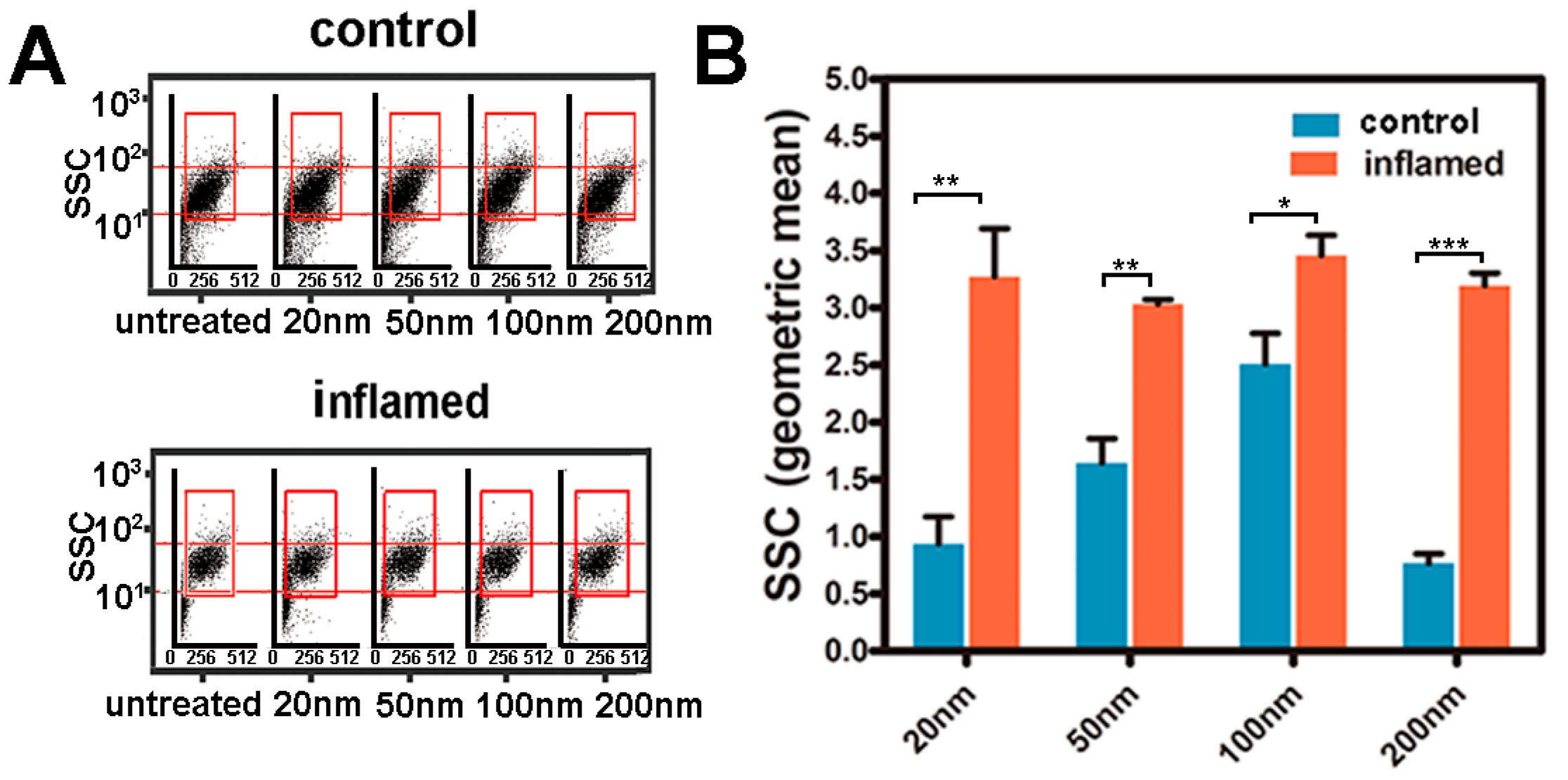
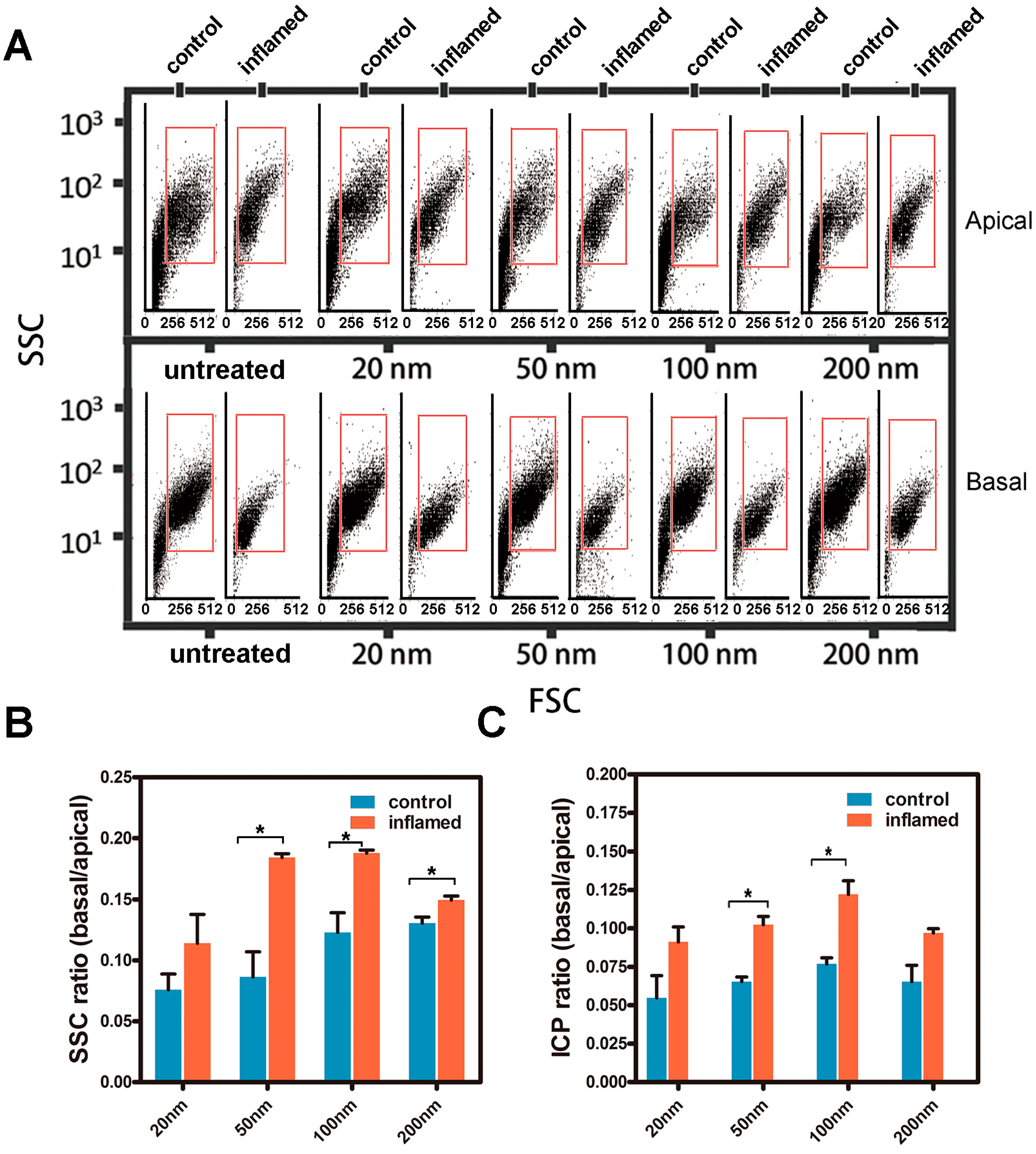
2.4. Investigation of the Uptake of Fe3O4 NPs in Caco-2 Cells
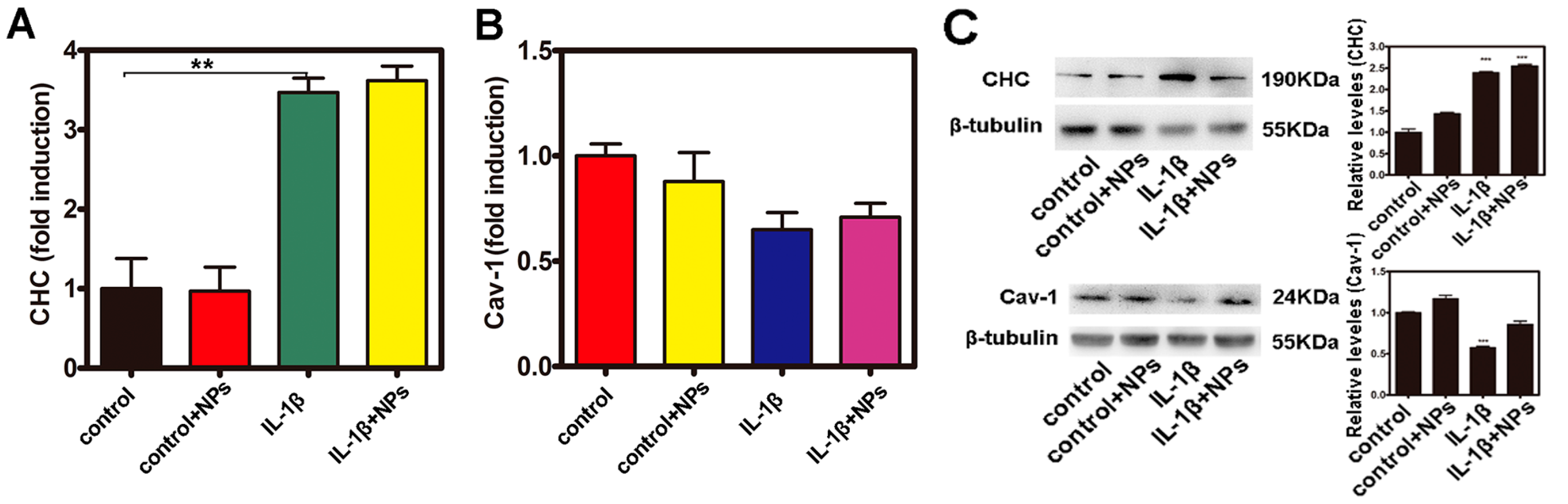
2.5. Investigation of the Uptake Features of Fe3O4 NPs in Caco-2 Cells
2.6. Transport of the Fe3O4 NPs in Caco-2 Cell Monolayers
3. Materials and Methods
3.1. Materials
3.2. Characterization of NPs
3.3. Cell Culture
3.4. Cell Viability Assay
3.5. Bio-Distribution and Cellular Location of NPs
3.6. Quantitative Analysis of the Internalized NPs in Intestinal Epithelial Cells
3.7. Quantitative Analysis of the Fe3O4 NPs Transport across Caco-2 Cell Monolayers
3.8. Western Blotting
3.9. Quantitative Real-Time PCR
- β-actin sense: 5′-GGTGTGATGGTGGGAATGGG-3′;
- β-actin antisense: 5′-ACGGTTGGCCTTAGGGTTCAG-3′.
- TNF-α sense: 5′-CCCAGGGACCTCTCTCTAATCA-3′;
- TNF-α antisense: 5′-AGCTGCCCCTCAGCTTGAG-3′.
- CHC sense: 5′-TGAGGCGACTGGGCGGAGTT-3′;
- CHC antisense: 5′-CCGGGGACGCAGGAAACTGG-3′.
- CAV-1 sense: 5′-GCCAACTACCAGCGTGAC-3′
- CAV-1 antisense: 5′-ATGCCCGCACCTGAGTAA-3′
3.10. Transmission Electron Microscopy
3.11. Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saraiva, C.; Praca, C.; Ferreira, R.; Santos, T.; Ferreira, L.; Bernardino, L. Nanoparticle-mediated brain drug delivery: Overcoming blood-brain barrier to treat neurodegenerative diseases. J. Control. Release: Off. J. Control. Release Soc. 2016, 235, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Elzoghby, A.O.; Hemasa, A.L.; Freag, M.S. Hybrid protein-inorganic nanoparticles: From tumor-targeted drug delivery to cancer imaging. J. Control. Release: Off. J. Control. Release Soc. 2016, 243, 303–322. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wu, M.; Pan, L.; Shi, J. Tumor vascular-targeted co-delivery of anti-angiogenesis and chemotherapeutic agents by mesoporous silica nanoparticle-based drug delivery system for synergetic therapy of tumor. Int. J. Nanomed. 2016, 11, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Al Faraj, A.; Shaik, A.P.; Shaik, A.S. Magnetic single-walled carbon nanotubes as efficient drug delivery nanocarriers in breast cancer murine model: Noninvasive monitoring using diffusion-weighted magnetic resonance imaging as sensitive imaging biomarker. Int. J. Nanomed. 2015, 10, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Win, K.Y.; Feng, S.-S. Effects of particle size and surface coating on cellular uptake of polymeric nanoparticles for oral delivery of anticancer drugs. Biomaterials 2005, 26, 2713–2722. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar] [CrossRef]
- Neves, A.R.; Queiroz, J.F.; Costa Lima, S.A.; Figueiredo, F.; Fernandes, R.; Reis, S. Cellular uptake and transcytosis of lipid-based nanoparticles across the intestinal barrier: Relevance for oral drug delivery. J. Colloid Interface Sci. 2016, 463, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Beloqui, A.; des Rieux, A.; Preat, V. Mechanisms of transport of polymeric and lipidic nanoparticles across the intestinal barrier. Adv. Drug Deliv. Rev. 2016, 106, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Gao, H.; Bao, G. Physical principles of nanoparticle cellular endocytosis. ACS Nano 2015, 9, 8655–8671. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Qi, J.; Gogoi, R.; Wong, J.; Mitragotri, S. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. J. Control. Release Off. J. Control. Release Soc. 2016, 238, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Bhamidipati, M.; Fabris, L. Multiparametric assessment of gold nanoparticle cytotoxicity in cancerous and healthy cells: The role of size, shape, and surface chemistry. Bioconjug. Chem. 2017, 28, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Win, K.Y.; Teng, C.P.; Ye, E.; Low, M.; Han, M.Y. Evaluation of polymeric nanoparticle formulations by effective imaging and quantitation of cellular uptake for controlled delivery of doxorubicin. Small 2015, 11, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Lin, P.; Jia, Z.; Du, W.; Qu, W.; Yuan, L.; Dai, W.; Zhang, H.; Wang, X.; Wang, J.; et al. The transport mechanisms of polymer nanoparticles in caco-2 epithelial cells. Biomaterials 2013, 34, 6082–6098. [Google Scholar] [CrossRef] [PubMed]
- Chai, G.-H.; Xu, Y.; Chen, S.-Q.; Cheng, B.; Hu, F.-Q.; You, J.; Du, Y.-Z.; Yuan, H. Transport mechanisms of solid lipid nanoparticles across caco-2 cell monolayers and their related cytotoxicology. ACS Appl. Mater. Interfaces 2016, 8, 5929–5940. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Morishita, Y.; Hata, T.; Kondoh, M.; Yagi, K.; Gao, J.Q.; Nagano, K.; Higashisaka, K.; Yoshioka, Y.; Tsutsumi, Y. Cellular internalization, transcellular transport, and cellular effects of silver nanoparticles in polarized caco-2 cells following apical or basolateral exposure. Biochem. Biophys. Res. Commun. 2017, 484, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Peterson, L.W.; Artis, D. Intestinal epithelial cells: Regulators of barrier function and immune homeostasis. Nat. Rev. Immunol. 2014, 14, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Luissint, A.C.; Parkos, C.A.; Nusrat, A. Inflammation and the intestinal barrier: Leukocyte-epithelial cell interactions, cell junction remodeling, and mucosal repair. Gastroenterology 2016, 151, 616–632. [Google Scholar] [CrossRef] [PubMed]
- Haines, R.J.; Beard, R.S., Jr.; Chen, L.; Eitnier, R.A.; Wu, M.H. Interleukin-1beta mediates beta-catenin-driven downregulation of claudin-3 and barrier dysfunction in caco2 cells. Dig. Dis. Sci. 2016, 61, 2252–2261. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.M.; Macedo, G.A.; Macedo, J.A.; Roberto, B.S.; Chen, Q.; Blumberg, J.B.; Chen, C.-Y.O. Tannase enhances the anti-inflammatory effect of grape pomace in caco-2 cells treated with il-1β. J. Funct. Foods 2017, 29, 69–76. [Google Scholar] [CrossRef]
- Ruiz, P.A.; Moron, B.; Becker, H.M.; Lang, S.; Atrott, K.; Spalinger, M.R.; Scharl, M.; Wojtal, K.A.; Fischbeck-Terhalle, A.; Frey-Wagner, I.; et al. Titanium dioxide nanoparticles exacerbate dss-induced colitis: Role of the nlrp3 inflammasome. Gut 2017, 66, 1216–1224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez-Palacios, A.; Kodani, T.; Kaydo, L.; Pietropaoli, D.; Corridoni, D.; Howell, S.; Katz, J.; Xin, W.; Pizarro, T.T.; Cominelli, F. Stereomicroscopic 3d-pattern profiling of murine and human intestinal inflammation reveals unique structural phenotypes. Nat. Commun. 2015, 6, 7577. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Liu, N.; Wang, Z.; Shi, T.; Gan, J.; Wang, Z.; Zhang, J. Quantitative analysis of gold and carbon nanoparticles in mammalian cells by flow cytometry light scattering. J. Nanopart. Res. 2017, 19, 78. [Google Scholar] [CrossRef]
- Suzuki, H.; Toyooka, T.; Ibuki, Y. Simple and easy method to evaluate uptake potential of nanoparticles in mammalian cells using a flow cytometric light scatter analysis. Environ. Sci. Technol. 2007, 41, 3018–3024. [Google Scholar] [CrossRef] [PubMed]
- Salatin, S.; Yari Khosroushahi, A. Overviews on the cellular uptake mechanism of polysaccharide colloidal nanoparticles. J. Cell. Mol. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Berezhkovskii, A.; Nossal, R. Kinetics of cellular uptake of viruses and nanoparticles via clathrin-mediated endocytosis. Phys. Biol. 2016, 13, 016005. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, J.M.; Seo, Y.-E.; Saltzman, W.M. Cell penetrating peptide-modified poly (lactic-co-glycolic acid) nanoparticles with enhanced cell internalization. Acta Biomater. 2016, 30, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Lagarce, F.; Roger, E. Transport of therapeutics across gastrointestinal epithelium. In Drug Delivery Across Physiological Barriers; Pan Stanford: Singapore, 2016; pp. 181–208. [Google Scholar]
- Arana, M.R.; Tocchetti, G.N.; Rigalli, J.P.; Mottino, A.D.; Villanueva, S.S. Physiological and pathophysiological factors affecting the expression and activity of the drug transporter mrp2 in intestine. Impact on its function as membrane barrier. Pharmacol. Res. 2016, 109, 32–44. [Google Scholar] [CrossRef] [PubMed]
- Chithrani, B.D.; Ghazani, A.A.; Chan, W.C. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006, 6, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.M.; Boyles, M.S.P.; Falagan-Lotsch, P.; Pereira, M.R.; Tutumi, H.R.; Santos, E.O.; Martins, N.B.; Himly, M.; Sommer, A.; Foissner, I. Poly-lactic acid nanoparticles (pla-np) promote physiological modifications in lung epithelial cells and are internalized by clathrin-coated pits and lipid rafts. J. Nanobiotechnol. 2017, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Xia, D.; Zhu, Q.; Hu, L.; Gan, Y. Intracellular transport of nanocarriers across the intestinal epithelium. Drug Discov. Today 2016, 21, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wu, J.; Shan, W.; Zhou, Z.; Liu, M.; Huang, Y. Sub-50 nm nanoparticles with biomimetic surfaces to sequentially overcome the mucosal diffusion barrier and the epithelial absorption barrier. Adv. Funct. Mater. 2016, 26, 2728–2738. [Google Scholar] [CrossRef]
- Li, H.; Ye, X.; Guo, X.; Geng, Z.; Wang, G. Effects of surface ligands on the uptake and transport of gold nanoparticles in rice and tomato. J. Hazard. Mater. 2016, 314, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-H.; Bendris, N.; Hsiao, Y.-J.; Reis, C.R.; Mettlen, M.; Chen, H.-Y.; Yu, S.-L.; Schmid, S.L. Crosstalk between clcb/dyn1-mediated adaptive clathrin-mediated endocytosis and epidermal growth factor receptor signaling increases metastasis. Dev. Cell 2017, 40, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Boucrot, E.; Watanabe, S. Fast and ultrafast endocytosis. Curr. Opin. Cell Biol. 2017, 47, 64–71. [Google Scholar]
- Chithrani, B.D.; Chan, W.C. Elucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapes. Nano Lett. 2007, 7, 1542–1550. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |



| Inhibitors | Functions | Concentrations |
|---|---|---|
| EIPA | Inhibitor of endocytosis pathway through macropinocytosis | 20 μM |
| Methyl-beta-cyclodextrin | Inhibitor of lipid raft/caveolae dependent endocytosis | 10 mM |
| Chloropromazine | Inhibitor of clathrin-related route | 30 μM |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, G.; Zhang, J.; Pan, C.; Liu, N.; Wang, Z.; Zhang, J. Enhanced Uptake of Fe3O4 Nanoparticles by Intestinal Epithelial Cells in a State of Inflammation. Molecules 2017, 22, 1240. https://doi.org/10.3390/molecules22081240
Zhou G, Zhang J, Pan C, Liu N, Wang Z, Zhang J. Enhanced Uptake of Fe3O4 Nanoparticles by Intestinal Epithelial Cells in a State of Inflammation. Molecules. 2017; 22(8):1240. https://doi.org/10.3390/molecules22081240
Chicago/Turabian StyleZhou, Gang, Jin Zhang, Chun Pan, Naicheng Liu, Zhenheng Wang, and Junfeng Zhang. 2017. "Enhanced Uptake of Fe3O4 Nanoparticles by Intestinal Epithelial Cells in a State of Inflammation" Molecules 22, no. 8: 1240. https://doi.org/10.3390/molecules22081240




