Metal Atom Effect on the Photophysical Properties of Mg(II), Zn(II), Cd(II), and Pd(II) Tetraphenylporphyrin Complexes Proposed as Possible Drugs in Photodynamic Therapy
Abstract
:1. Introduction
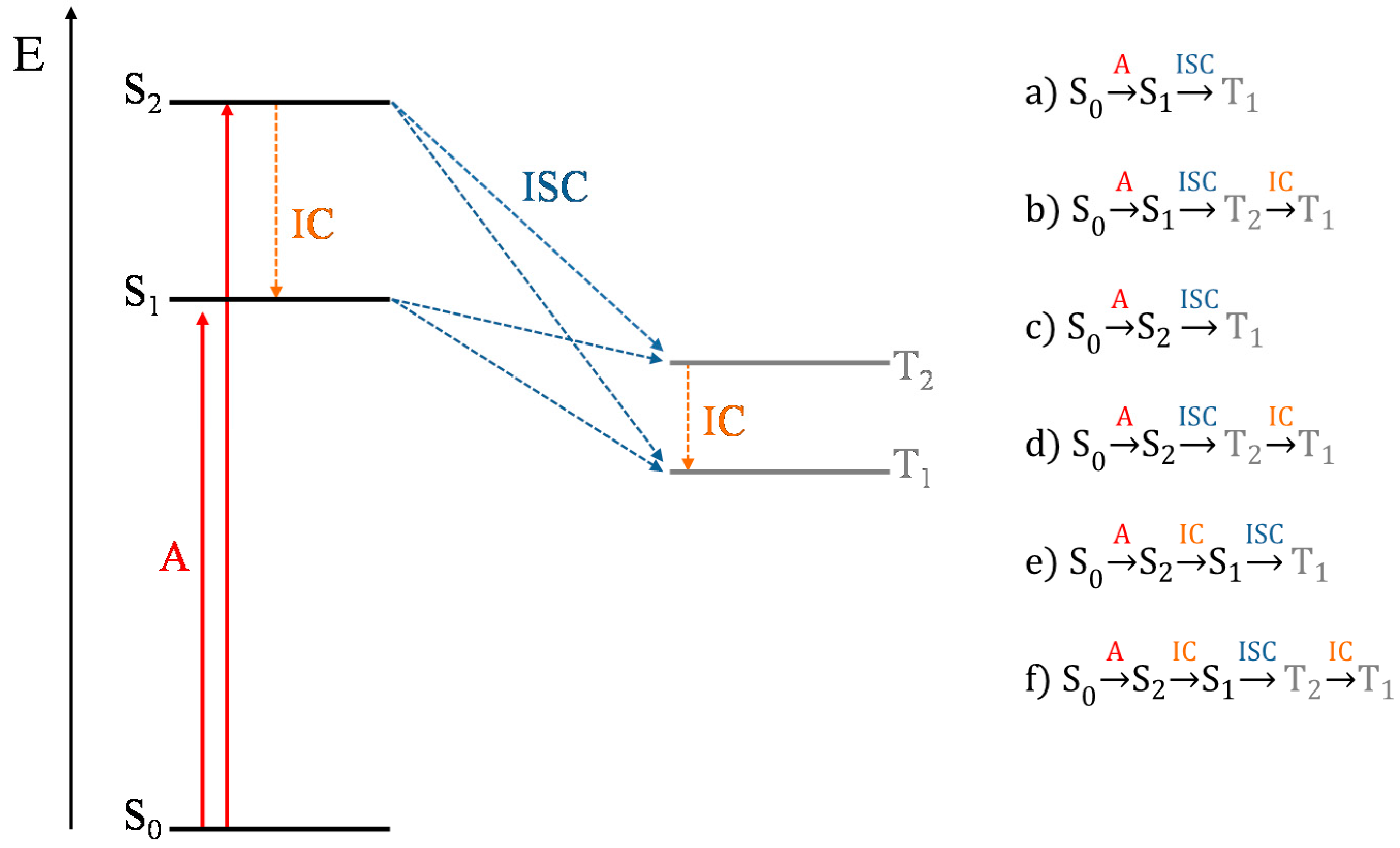
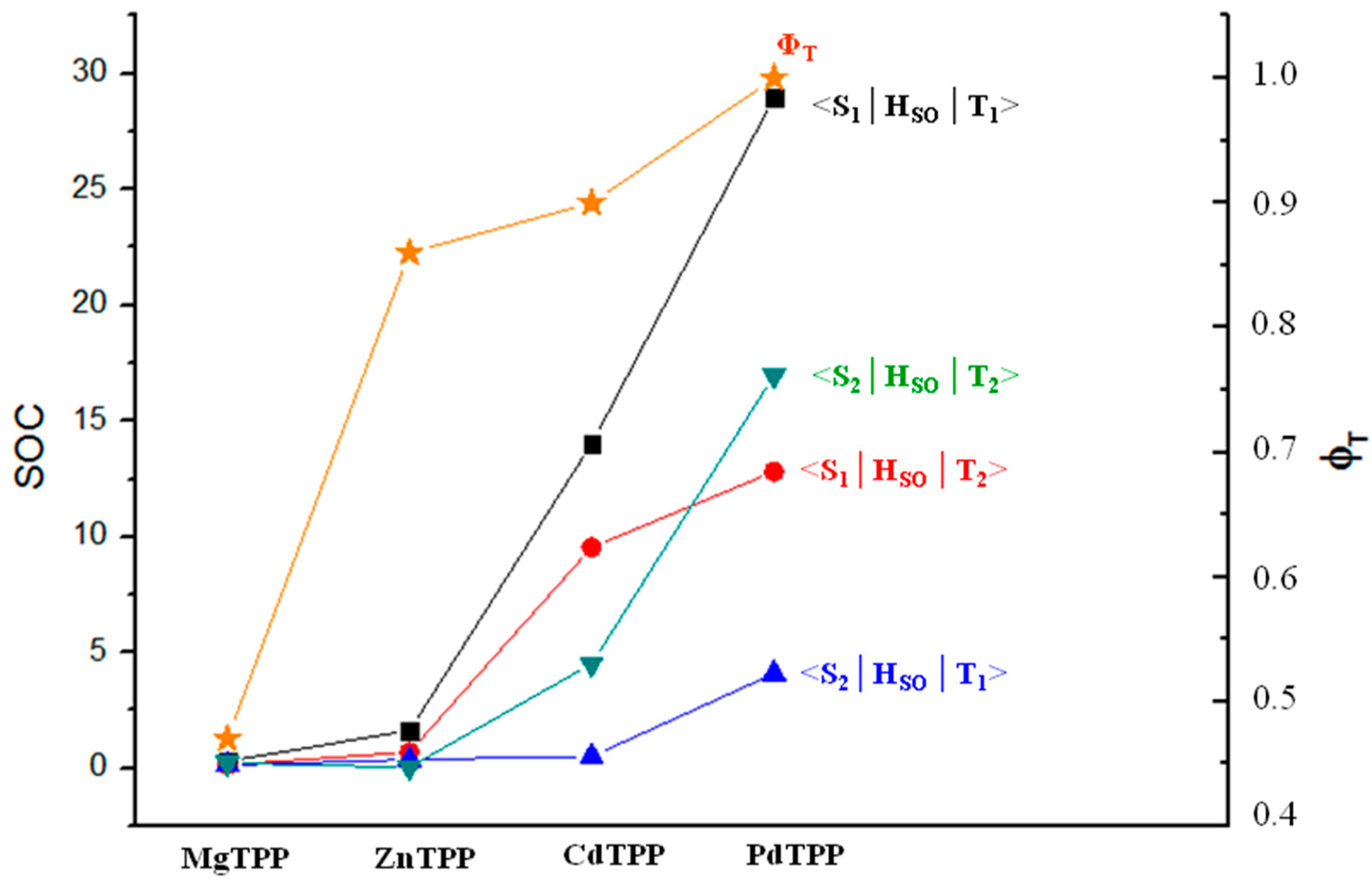
2. Results and Discussion
3. Computational Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Marian, C.M. Spin-orbit coupling and intersystem crossing in molecules. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2012, 2, 187–203. [Google Scholar] [CrossRef]
- Fantacci, S.; De Angelis, F. Impact of Spin-Orbit Coupling on Photocurrent Generation in Ruthenium Dye-Sensitized Solar Cells. J. Phys. Chem. Lett. 2014, 5, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Kannan, U.M.; Muddisetti, V.N.; Kotnana, G.; Kandhadi, J.; Giribabu, L.; Singh, S.P.; Jammalamadaka, S.N. Spin–orbit coupling and Lorentz force enhanced efficiency of TiO2-based dye sensitized solar cells. Phys. Status Solidi A 2017, 214, 1600691. [Google Scholar] [CrossRef]
- McClure, D.S. Spin-Orbit Interaction in Aromatic Molecules. J. Chem. Phys. 1952, 20, 682–688. [Google Scholar] [CrossRef]
- De Simone, B.C.; Mazzone, G.; Pirillo, J.; Russo, N.; Sicilia, E. Halogen atom effect on the photophysical properties of substituted aza-BODIPY derivatives. Phys. Chem. Chem. Phys. 2017, 19, 2530–2536. [Google Scholar] [CrossRef] [PubMed]
- Alberto, M.E.; De Simone, B.C.; Mazzone, G.; Sicilia, E.; Russo, N. The Heavy Atom Effect on Zn(II) Phthalocyanine Derivatives: A Theoretical Exploration of the Photophysical Properties. Phys. Chem. Chem. Phys. 2015, 17, 23595–23601. [Google Scholar] [CrossRef]
- Awuah, S.G.; You, Y. Boron dipyrromethene (BODIPY)-based photosensitizers for photodynamic therapy. RSC Adv. 2012, 2, 11169–11183. [Google Scholar] [CrossRef]
- Dougherty, T.J.; Grindey, G.B.; Fiel, R.; Weishaupt, K.R.; Boyle, D.G.J. Photoradiation therapy. II. Cure of animal tumors with hematoporphyrin and light. J. Natl. Cancer Inst. 1975, 55, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Juzeniene, A.; Peng, Q.; Moan, J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem. Photobiol. Sci. 2007, 6, 1234–1245. [Google Scholar] [CrossRef]
- Babilas, P.; Schreml, S.; Landthaler, M.; Szeimies, R.M. Photodynamic therapy in dermatology: State-of-the-art. Photodermatol. Photoimmunol. Photomed. 2010, 26, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Kossodo, S.; La Muraglia, G.M. Clinical potential of photodynamic therapy in cardiovascular disorders. Am. J. Cardiovasc. Drugs 2001, 1, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Garrier, J.; Bezdetnaya, L.; Barlier, C.; Gräfe, S.; Guillemin, F.; D’Hallewin, M.A. Foslip®-based photodynamic therapy as a means to improve wound healing. Photodiagn. Photodyn. Ther. 2011, 8, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowski, J.M.; Arnaut, L.G. Photodynamic therapy (PDT) of cancer: From local to systemic treatment. Photochem. Photobiol. Sci. 2015, 14, 1765–1780. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, W.; Mackeyev, Y.; Lee, G.-S.; Kim, H.-J.; Tachikawa, T.; Hong, S.; Lee, S.; Kim, J.; Wilson, L.J.; Majima, T.; Alvarez, P.J.J.; Choi, W.; Lee, J. Selective Oxidative Degradation of Organic Pollutants by Singlet Oxygen-Mediated Photosensitization: Tin Porphyrin versus C60 Aminofullerene Systems. Environ. Sci. Technol. 2012, 46, 9606–9613. [Google Scholar] [CrossRef] [PubMed]
- Han, S.K.; Sik, R.H.; Motten, A.G.; Chignell, C.F.; Bilski, P.J. Photosensitized oxidation of tetrabromobisphenol a by humic acid in aqueous solution. Photochem. Photobiol. 2009, 85, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Kohn, T.; Nelson, K.L. Sunlight-mediated inactivation of MS2 coliphage via exogenous singlet oxygen produced by sensitizers in natural waters. Environ. Sci. Technol. 2007, 41, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Romero, O.C.; Straub, A.P.; Kohn, T.; Nguyen, T.H. Role of temperature and Suwannee River natural organic matter on inactivation kinetics of rotavirus and bacteriophage MS2 by solar irradiation. Environ. Sci. Technol. 2011, 45, 10385–10393. [Google Scholar] [CrossRef] [PubMed]
- Plaetzer, K.; Krammer, B.; Berlanda, J.; Berr, F.; Kiesslich, T. Photophysics and photochemistry of photodynamic therapy: fundamental aspects. Lasers Med. Sci. 2009, 24, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Ethirajan, M.; Chen, Y.; Joshi, P.; Pandey, R.K. The role of porphyrin chemistry in tumor imaging and photodynamic therapy. Chem. Soc. Rev. 2013, 42, 845–856. [Google Scholar] [CrossRef] [PubMed]
- Banfi, S.; Caruso, E.; Caprioli, S.; Mazzagatti, L.; Canti, G.; Ravizza, R.; Gariboldi, M.; Monti, E. Photodynamic effects of porphyrin and chlorin photosensitizers in human colon adenocarcinoma cells. Bioorg. Med. Chem. 2004, 12, 4853–4860. [Google Scholar] [CrossRef] [PubMed]
- Garcıa-Dıaz, M.; Sanchez-Garcıa, D.; Soriano, J.; Sagrist, M.L.; Mora, M.; Villanueva, A.; Stockert, J.C.; Canete, M.; Nonell, S. Temocene: The porphycene analogue of temoporfin (Foscan®). MedChemComm 2011, 2, 616–619. [Google Scholar] [CrossRef]
- Alberto, M.E.; Marino, T.; Quartarolo, A.D.; Russo, N. Photophysical origin of the reduced photodynamic therapy activity of temocene compared to Foscan®: Insights from theory. Phys. Chem. Chem. Phys. 2013, 15, 16167–16171. [Google Scholar] [CrossRef] [PubMed]
- Alberto, M.E.; De Simone, B.C.; Mazzone, G.; Marino, T.; Russo, N. Photophysical properties of free and metallated meso-substituted tetrabenzotriazaporphyrin from density functional theory investigation. Dyes Pigm. 2015, 120, 335–339. [Google Scholar] [CrossRef]
- Mazzone, G.; Alberto, M.E.; De Simone, B.C.; Marino, T.; Russo, N. Can Expanded Bacteriochlorins Act as Photosensitizers in Photodynamic Therapy? Good News from Density Functional Theory Computations. Molecules 2016, 21, 288. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, G.; Russo, N.; Sicilia, E. Theoretical investigation of the absorption spectra and singlet-triplet energy gap of positively charged tetraphenylporphyrins as potential photodynamic therapy photosensitizers. Can. J. Chem. 2013, 91, 902–906. [Google Scholar] [CrossRef]
- Lawetz, V.; Orlandi, G.; Siebrand, J.W. Theory of Intersystem Crossing in Aromatic Hydrocarbons. J. Chem. Phys. 1972, 56, 4058–4072. [Google Scholar] [CrossRef]
- Robinson, G.W.; Frosch, R.P.J. Electronic Excitation Transfer and Relaxation. J. Chem. Phys. 1963, 38, 1187–1203. [Google Scholar] [CrossRef]
- El-Sayed, M.A. Triplet state. Its radiative and nonradiative properties. Acc. Chem. Res. 1968, 1, 8–16. [Google Scholar] [CrossRef]
- Kasha, M. Characterization of Electronic Transitions in Complex Molecules. Discuss. Faraday Soc. 1950, 9, 14–19. [Google Scholar] [CrossRef]
- Scheidt, W.R.; Mondal, J.U.; Eigenbrodt, C.W.; Adler, A.; Radonvich, L.J.; Hoard, J.L. Crystal and Molecular Structure of the Silver(II) and Zinc(II) Derivatives of meso-Tetraphenylporphyrin. An Exploration of Crystal-Packing Effects on Bond Distance. Inorg. Chem. 1986, 25, 795–799. [Google Scholar] [CrossRef]
- Nguyen, K.A.; Day, P.N.; Pachter, R.; Tretiak, S.; Chernyak, V.; Mukamel, S. Analysis of Absorption Spectra of Zinc Porphyrin, Zinc meso-Tetraphenylporphyrin, and Halogenated Derivatives. J. Phys. Chem. A 2002, 106, 10285–10293. [Google Scholar] [CrossRef]
- Liu, X.; Yeow, E.K.L.; Velate, S.; Steer, R.P. Photophysics and spectroscopy of the higher electronic states of zinc metalloporphyrins: A theoretical and experimental study. Phys. Chem. Chem. Phys. 2006, 8, 1298–1309. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, U.; Kowalska, D.; Liu, X.; Velate, S.; Steer, R.P. Photophysics of Soret-Excited Tetrapyrroles in Solution. I. Metalloporphyrins: MgTPP, ZnTPP, and CdTPP. J. Phys. Chem. A 2008, 112, 5824–5833. [Google Scholar] [CrossRef] [PubMed]
- Obata, M.; Hirohara, S.; Tanaka, R.; Kinoshita, I.; Ohkubo, K.; Fukuzumi, S.; Tanihara, M.; Yano, S. In Vitro Heavy-Atom Effect of Palladium(II) and Platinum(II) Complexes of Pyrrolidine-Fused Chlorin in Photodynamic Therapy. J. Med. Chem. 2009, 52, 2747–2753. [Google Scholar] [CrossRef] [PubMed]
- Azenha, E.G.; Serra, A.C.; Pineiro, M.; Pereira, M.M.; Seixas de Melo, J.; Arnauta, L.G.; Formosinho, S.J.; Rocha Gonsalves, A.M.d’A. Heavy-atom effects on metalloporphyrins and polyhalogenated porphyrins. Chem. Phys. 2002, 280, 177–190. [Google Scholar] [CrossRef]
- Yu, H.Z.; Baskin, J.S.; Zewail, A.H. Ultrafast Dynamics of Porphyrins in the Condensed Phase: II. Zinc Tetraphenylporphyrin. J. Phys. Chem. A 2002, 106, 9845–9854. [Google Scholar] [CrossRef]
- Ghosh, M.; Mora, A.K.; Nath, S.; Chandra, A.K.; Hajra, A.; Sinha, S. Photophysics of Soret-excited free base tetraphenylporphyrin and its zinc analog in solution. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 116, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Carvalho, A.L.; Pereira, M.M.; Rocha Gonsalves, A.M.d’A.; Arnaut, L.G.; Formosinho, S.J. Photoacoustic Measurements of Porphyrin Triplet-State Quantum Yields and Singlet-Oxygen Efficiencies. Chem. Eur. J. 1998, 4, 2299–2307. [Google Scholar] [CrossRef]
- Dabrowski, J.M.; Pucelik, B.; Regiel-Futyra, A.; Brindell, M.; Mazuryk, O.; Kyzioł, A.; Stochel, G.; Macyk, W.; Arnaut, L.G. Engineering of relevant photodynamic processes through structural modifications of metallotetrapyrrolic photosensitizers. Coordinat. Chem. Rev. 2016, 325, 67–101. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. The M06 Suite of Density Functionals for Main Group Thermochemistry, Thermochemical Kinetics, Noncovalent Interactions, Excited States, and Transition Elements: Two New Functionals and Systematic Testing of Four M06-Class Functionals and 12 Other Functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar]
- Pirillo, J.; De Simone, B.C.; Russo, N. Photophysical Properties Prediction of Selenium- and Tellurium-Substituted Thymidine as Potential UVA Chemotherapeutic Agents. Theor. Chem. Acc. 2016, 135, 8. [Google Scholar] [CrossRef]
- Pirillo, J.; Mazzone, G.; Russo, N.; Bertini, L. Photophysical Properties of S, Se and Te-Substituted Deoxyguanosines: Insight into Their Ability to Act as Chemotherapeutic Agents. J. Chem. Inf. Model. 2017, 57, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, G.; Quartarolo, A.D.; Russo, N. PDT-Correlated Photophysical Properties of Thienopyrrole BODIPY Derivatives. Theoretical Insights. Dyes Pigm. 2016, 130, 9–15. [Google Scholar] [CrossRef]
- Alberto, M.E.; Mazzone, G.; Quartarolo, A.D.; Sousa, F.F.R.; Sicilia, E.; Russo, N. Electronic Spectra and Intersystem Spin-Orbit Coupling in 1,2- and 1,3-Squaraines. J. Comput. Chem. 2014, 35, 2107–2113. [Google Scholar] [CrossRef] [PubMed]
- Jacquemin, D.; Perpète, E.A.; Ciofini, I.; Adamo, C. Accurate Simulation of Optical Properties in Dyes. Acc. Chem. Res. 2009, 42, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, E.S.E.; Eriksson, L.A. Predictive Power of Long- Range Corrected Functionals on the Spectroscopic Properties of Tetrapyrrole Derivatives for Photodynamic Therapy. Phys. Chem. Chem. Phys. 2011, 13, 7207–7217. [Google Scholar] [CrossRef] [PubMed]
- Adamo, C.; Jacquemin, D. The Calculations of Excited-State Properties with Time-Dependent Density Functional Theory. Chem. Soc. Rev. 2013, 42, 845–856. [Google Scholar] [CrossRef] [PubMed]
- Cossi, M.; Barone, V. Solvent Effect on Vertical Electronic Transitions by the Polarizable Continuum Model. J. Chem. Phys. 2000, 112, 2427–2435. [Google Scholar] [CrossRef]
- Tomasi, J.; Menucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09; Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Aidas, K.; Angeli, C.; Bak, K.L.; Bakken, V.; Bast, R.; Boman, L.; Christiansen, O.; Cimiraglia, R.; Coriani, S.; Dahle, P.; et al. The Dalton quantum chemistry program system. WIREs Comput. Mol. Sci. 2014, 4, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Koseki, S.; Schmidt, M.W.; Gordon, M.S. Effective Nuclear Charges for the First- Through Third-Row Transition Metal Elements in Spin-Orbit Calculations. J. Phys. Chem. A 1998, 102, 10430–10436. [Google Scholar]
- Ruud, K.; Schimmelpfennig, B.; Ågren, H. Internal and external heavy-atom effects on phosphorescence radiative lifetimes calculated using a mean-field spin-orbit Hamiltonian. Chem. Phys. Lett. 1999, 310, 215–221. [Google Scholar] [CrossRef]
- Becke, A.D. Density-Functional Thermochemistry. III. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron Density. Phys. Rev. B: Condens. Matter Mater. Phys. 1988, 37, 785–789. [Google Scholar] [CrossRef]


| Compound | Excited State | λtheo (nm) | ΔE (eV) | f | λexp (nm) | Transitions d |
|---|---|---|---|---|---|---|
| MgTPP | S1 (S’1) | 559 | 2.22 | 0.008 | 607 a | H → L, 54% (H → L + 1, 54%) |
| S2 (S’2) | 404 | 3.07 | 1.739 | 428 a | H − 1 → L, 44% (H − 1 → L + 1, 44%) | |
| T1 (T’1) | 831 | 1.49 | 0 | 794 c | H → L, 83% (H → L + 1, 83%) | |
| T2 (T’2) | 630 | 1.97 | 0 | H − 1→L, 84% (H − 1 → L + 1, 84%) | ||
| ZnTPP | S1 (S’1) | 561 | 2.21 | 0.038 | 593 a | H → L, 50% (H → L + 1, 50%) |
| S2 (S’2) | 412 | 3.01 | 1.699 | 424 a | H − 1 → L + 1, 47% (H − 1 → L, 47%) | |
| T1 (T’1) | 812 | 1.53 | 0 | 778 c | H → L, 70% (H → L + 1, 70%) | |
| T2 (T’2) | 626 | 1.98 | 0 | H − 1 → L, 82% (H − 1 → L + 1, 82%) | ||
| CdTPP | S1 (S’1) | 549 | 2.26 | 0.013 | 617 a | H → L, 54% (H → L + 1, 54%) |
| S2 (S’2) | 397 | 3.12 | 1.670 | 437 a | H − 1 → L+1, 59% (H − 1 → L, 59%) | |
| T1 (T’1) | 846 | 1.46 | 0 | 814 c | H → L, 52% (H → L + 1, 52%) | |
| T2 (T’2) | 619 | 2.00 | 0 | H − 1 → L, 85% (H – 1 → L + 1, 85%) | ||
| PdTPP | S1 (S’1) | 537 | 2.31 | 0.032 | 553 b | H → L, 48% (H → L + 1, 48%) |
| S2 (S’2) | 409 | 3.03 | 1.519 | 407 b | H − 1 → L, 59% (H − 1 → L + 1, 59%) | |
| T1 (T’1) | 758 | 1.63 | 0 | H → L, 78% (H → L + 1, 78%) | ||
| T2 (T’2) | 597 | 2.07 | 0 | H − 1 → L, 84% ( H − 1 → L + 1, 84%) |
| MgTPP | ZnTPP | CdTPP | PdTPP | |
|---|---|---|---|---|
| 0.31 0.73 | 1.64 0.68 | 14.01 0.80 | 28.99 0.68 | |
| 0.15 0.25 | 0.70 0.23 | 9.55 0.26 | 12.81 0.24 | |
| 0.14 1.58 | 0.38 1.48 | 0.52 1.66 | 4.10 1.40 | |
| 0.22 1.10 | 0.05 1.03 | 4.52 1.12 | 16.97 0.96 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simone, B.C.D.; Mazzone, G.; Russo, N.; Sicilia, E.; Toscano, M. Metal Atom Effect on the Photophysical Properties of Mg(II), Zn(II), Cd(II), and Pd(II) Tetraphenylporphyrin Complexes Proposed as Possible Drugs in Photodynamic Therapy. Molecules 2017, 22, 1093. https://doi.org/10.3390/molecules22071093
Simone BCD, Mazzone G, Russo N, Sicilia E, Toscano M. Metal Atom Effect on the Photophysical Properties of Mg(II), Zn(II), Cd(II), and Pd(II) Tetraphenylporphyrin Complexes Proposed as Possible Drugs in Photodynamic Therapy. Molecules. 2017; 22(7):1093. https://doi.org/10.3390/molecules22071093
Chicago/Turabian StyleSimone, Bruna Clara De, Gloria Mazzone, Nino Russo, Emilia Sicilia, and Marirosa Toscano. 2017. "Metal Atom Effect on the Photophysical Properties of Mg(II), Zn(II), Cd(II), and Pd(II) Tetraphenylporphyrin Complexes Proposed as Possible Drugs in Photodynamic Therapy" Molecules 22, no. 7: 1093. https://doi.org/10.3390/molecules22071093
APA StyleSimone, B. C. D., Mazzone, G., Russo, N., Sicilia, E., & Toscano, M. (2017). Metal Atom Effect on the Photophysical Properties of Mg(II), Zn(II), Cd(II), and Pd(II) Tetraphenylporphyrin Complexes Proposed as Possible Drugs in Photodynamic Therapy. Molecules, 22(7), 1093. https://doi.org/10.3390/molecules22071093