Secondary Sulfonamides as Effective Lactoperoxidase Inhibitors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
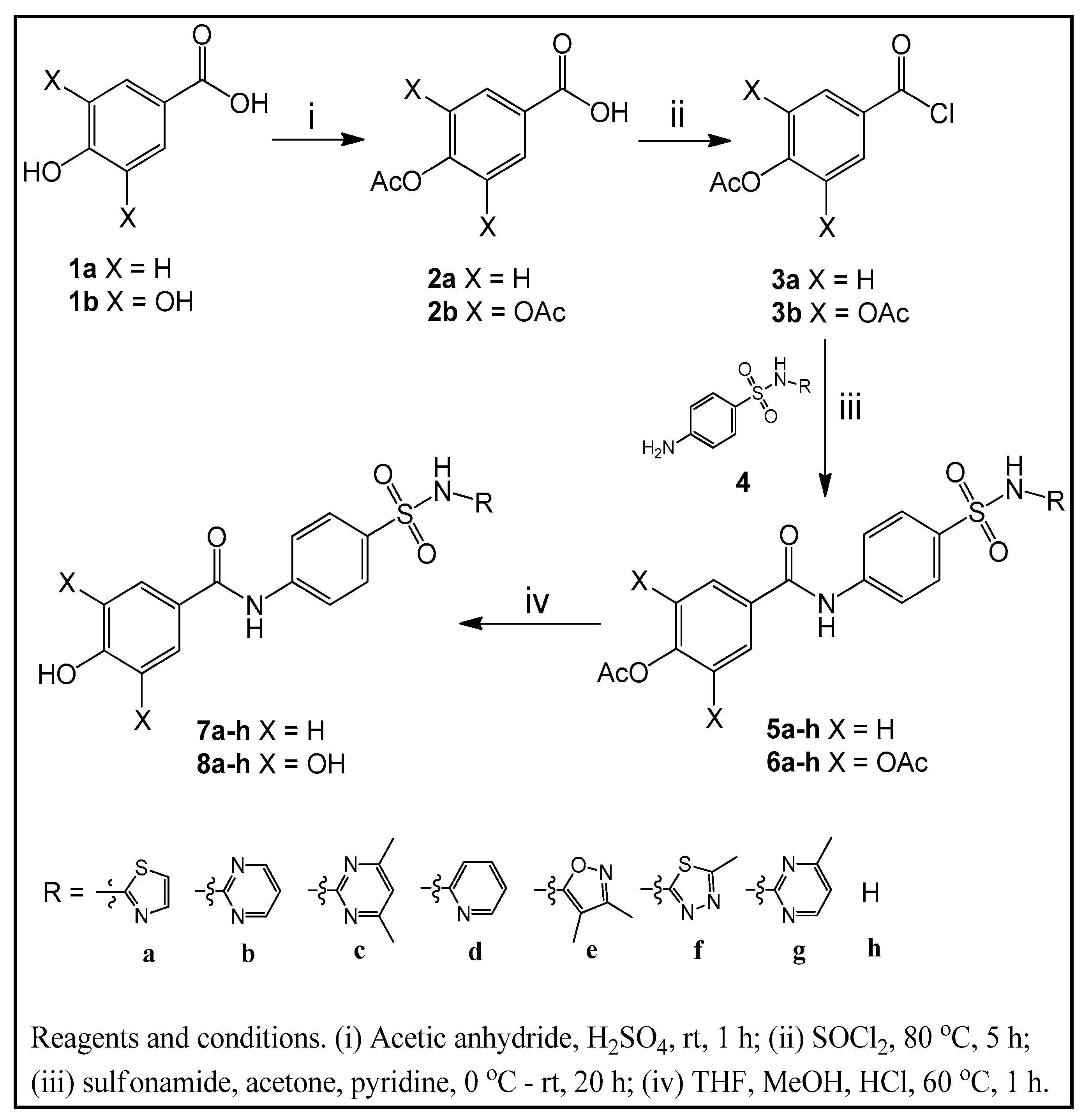
2.2. General Procedure for Sulfonamide Derivatives
2.3. Biochemical Assays
3. Results and Discussion
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Supuran, C.T.; Innocenti, A.; Mastrolorenzo, A.; Scozzafava, A. Antiviral sulfonamide derivatives. Mini Rev. Med. Chem. 2004, 4, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Göçer, H.; Akıncıoğlu, A.; Öztaşkın, N.; Göksu, S.; Gülçin, İ. Synthesis, antioxidant and antiacetylcholinesterase activities of sulfonamide derivatives of dopamine related compounds. Arch. Pharm. 2013, 346, 783–792. [Google Scholar]
- Akbaba, Y.; Akıncıoğlu, A.; Göçer, H.; Göksu, S.; Gülçin, İ.; Supuran, C.T. Carbonic anhydrase inhibitory properties of novel sulfonamide derivatives of aminoindanes and aminotetralins. J. Enzyme Inhib. Med. Chem. 2014, 29, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Akıncıoğlu, A.; Topal, M.; Gülçin, İ.; Göksu, S. Novel sulfamides and sulfonamides incorporating tetralin scaffold as carbonic anhydrase and acetylcholine esterase inhibitors. Arch. Pharm. 2014, 347, 68–76. [Google Scholar]
- Gokcen, T.; Al, M.; Topal, M.; Gülçin, I.; Ozturk, T.; Goren, A.C. Synthesis of some natural sulphonamide derivatives as carbonic anhydrase inhibitors. Org. Commun. 2017, 10, 15–23. [Google Scholar]
- Ivanova, J.; Carta, F.; Vullo, D.; Leitans, J.; Kazaks, A.; Tars, K.; Žalubovskis, R.; Supuran, C.T. N-Substituted and ring opened saccharin derivatives selectively inhibit transmembrane, tumor-associated carbonic anhydrases IX and XII. Bioorg. Med. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Carta, F.; Birkmann, A.; Pfaff, T.; Buschmann, H.; Schwab, W.; Zimmermann, H.; Maresca, A.; Supuran, C.T. Lead development of thiazoylsulfonamides with carbonic anhydrase inhibitory action. J. Med. Chem. 2017, 60, 3154–3164. [Google Scholar] [CrossRef] [PubMed]
- Petrou, A.; Geronikaki, A.; Terzi, E.; Guler, O.O.; Tiziano, T.; Supuran, C.T. Inhibition of carbonic anhydrase isoforms I, II, IX, and XII with secondary sulphonamides incorporating benzothiazole scaffolds. J. Enzyme Inhib. Med. Chem. 2016, 31, 1306–1311. [Google Scholar] [CrossRef] [PubMed]
- Moeker, J.; Peat, T.S.; Bornaghi, L.F.; Vullo, D.; Supuran, C.T.; Poulsen, S.A. Cyclic secondary sulfonamides: Unusually good inhibitors of cancer-related carbonic anhydrase enzymes. J. Med. Chem. 2014, 57, 3522–3531. [Google Scholar] [CrossRef] [PubMed]
- Qandil, A.M.; Hassan, M.A.; Al-Shar’i, N.A. Synthesis and anticandidal activity of azole-containing sulfonamides. Arch. Pharm. 2008, 341, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Blank, B.R.; Alayoğlu, P.; Engen, W.; Choi, J.K.; Berkman, C.E.; Anderson, M.O. N-Substituted glutamyl sulphonamides as inhibitors of glutamate carboxypeptidase II (GCP2). Chem. Biol. Drug. Des. 2011, 77, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Burhop, A.; Weck, R.; Atzrodt, J.; Derdau, V. Hydrogen-isotope exchange (HIE) reactions of secondary and tertiary sulfonamides and sulfonylureas with iridium (I) catalysts. Eur. J. Org. Chem. 2017, 11, 1418–1424. [Google Scholar] [CrossRef]
- Ueda, T.; Sakamaki, K.; Kuroki, T.; Yano, I.; Nagata, S. Molecular cloning and characterization of the chromosomal gene for human lactoperoxidase. Eur. J. Biochem. 1976, 243, 32–41. [Google Scholar] [CrossRef]
- Şişecioğlu, M.; Çankaya, M.; Gülçin, İ.; Özdemir, M. The Inhibitory effect of propofol on lactoperoxidase. Protein Pept. Lett. 2009, 16, 46–49. [Google Scholar]
- Şişecioğlu, M.; Çankaya, M.; Gülçin, İ.; Özdemir, M. Interactions of melatonin and serotonin to lactoperoxidase enzyme. J. Enzyme Inhib. Med. Chem. 2010, 25, 779–783. [Google Scholar]
- Elliot, R.M.; McLay, J.C.; Kennedy, M.J.; Simmonds, R.S. Inhibition of foodborne bacteria by the lactoperoxidase system in a beef cube system. Int. J. Food Microbiol. 2004, 91, 73–81. [Google Scholar] [CrossRef]
- Şişecioğlu, M.; Kireçci, E.; Çankaya, M.; Özdemir, H.; Gülçin, İ.; Atasever, A. The prohibitive effect of lactoperoxidase system (LPS) on some pathogen fungi and bacteria. Afr. J. Pharm. Pharmacol. 2010, 4, 671–677. [Google Scholar]
- Şişecioğlu, M.; Gülçin, İ.; Çankaya, M.; Atasever, A.; Özdemir, H. The effects of norepinephrine on lactoperoxidase enzyme (LPO). Sci. Res. Essays 2010, 5, 1351–1356. [Google Scholar]
- Jooyandeh, H.; Aberoumand, A.; Nasehi, B. Application of lactoperoxidase system in fish and food products: A review. Am. Eurasian J. Agric. Environ. Sci. 2011, 10, 89–96. [Google Scholar]
- Şişecioğlu, M.; Uguz, M.T.; Çankaya, M.; Özdemir, H.; Gülçin, İ. Effects of ceftazidime pentahydrate, prednisolone, amikacin sulfate, ceftriaxone sodium and teicoplanin on bovine milk lactoperoxidase activity. Int. J. Pharmacol. 2011, 7, 79–83. [Google Scholar]
- Şişecioğlu, M.; Gülçin, İ.; Çankaya, M.; Özdemir, H. The inhibitory effects of L-Adrenaline on lactoperoxidase enzyme (LPO) purified from buffalo milk. Int. J. Food Propert. 2012, 15, 1182–1189. [Google Scholar]
- Munsch-Alatossava, P.; Quintyn, R.; De Man, I.; Alatossava, T.; Gauchi, J.P. Efficiency of N2 gas flushing compared to the lactoperoxidase system at controlling bacterial growth in bovine raw milk stored at mild temperatures. Front. Microbiol. 2016, 7, 839. [Google Scholar] [CrossRef] [PubMed]
- Atasever, A.; Özdemir, H.; Gülçin, İ.; Küfrevioğlu, Ö.İ. One-step purification of lactoperoxidase from bovine milk by affinity chromatography. Food Chem. 2013, 136, 864–870. [Google Scholar] [CrossRef] [PubMed]
- Köksal, Z.; Usanmaz, H.; Özdemir, H.; Gülçin, İ.; Güney, M. Inhibition effects of some phenolic and dimeric phenolic compounds on bovine lactoperoxidase (LPO) enzyme. Int. J. Acad. Res. 2014, 6, 27–32. [Google Scholar]
- Golmohamadi, A.; Morra, M.J.; Popova, I.; Nindo, C.I. Optimizing the use of Sinapis alba seed meal extracts as a source of thiocyanate (SCN−) for the lactoperoxidase system. Lebensm. Wiss. Technol. 2016, 72, 416–422. [Google Scholar] [CrossRef]
- Ozturk Sarikaya, S.B.; Sisecioglu, M.; Cankaya, M.; Gülçin, İ.; Ozdemir, H. Inhibition profile of a series of phenolic acids on bovine lactoperoxidase enzyme. J. Enzyme Inhib. Med. Chem. 2015, 30, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Koksal, Z.; Kalın, R.; Gülçin, İ.; Özdemir, H.; Atasever, A. The impact of some avermectins on lactoperoxidase from bovine milk. Int. J. Food Propert. 2016, 19, 1207–1216. [Google Scholar] [CrossRef]
- Gokcen, T.; Gülçin, İ.; Ozturk, T.; Goren, A.C. A Class of Sulfonamides as carbonic anhydrase inhibitor. J. Enzyme Inhib. Med. Chem. 2016, 31, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Shindler, J.S.; Bardsley, W. Steady-state kinetics of lactoperoxidase with ABST as chromogens. Biochem. Biophys. Res. Comm. 1975, 67, 1307–1312. [Google Scholar] [CrossRef]
- Jacob, B.M.; Anthony, K.E.; Sreekumar, B.; Haridas, M. Thiocyanate mediated antifungal and antibacterial property of goat milk lactoperoxidase. Life Sci. 2000, 66, 2433–2439. [Google Scholar] [CrossRef]
- Gülçin, İ.; Scozzafava, A.; Supuran, C.T.; Akıncıoğlu, H.; Koksal, Z.; Turkan, F.; Alwasel, S. The effect of caffeic acid phenethyl ester (CAPE) metabolic enzymes including acetylcholinesterase, butyrylcholinesterase, glutathione s-transferase, lactoperoxidase and carbonic anhydrase ısoenzymes I, II, IX and XII. J. Enzyme Inhib. Med. Chem. 2016, 31, 1095–1101. [Google Scholar]
- Gülçin, İ.; Scozzafava, A.; Supuran, C.T.; Koksal, Z.; Turkan, F.; Çetinkaya, S.; Bingöl, Z.; Huyut, Z.; Alwasel, S.H. Rosmarinic acid inhibits some metabolic enzymes including glutathione S-transferase, lactoperoxidase, acetylcholinesterase, butyrylcholinesterase, and carbonic anhydrase isoenzymes. J. Enzyme Inhib. Med. Chem. 2016, 31, 1698–1702. [Google Scholar]
- Cuatrecasa, P. Protein purification by affinity chromatography. J. Biol. Chem. 1970, 245, 3059–3065. [Google Scholar]
- Arslan, O.; Nalbantoglu, B.; Demir, N.; Ozdemir, H.; Kufrevioglu, O.I. A new method for the purification of carbonic anhydrase isozymes by affinity chromatography. Turk. J. Med. Sci. 1996, 26, 163–166. [Google Scholar]
- Boztaş, M.; Çetinkaya, Y.; Topal, M.; Gülçin, İ.; Menzek, A.; Şahin, E.; Tanc, M.; Supuran, C.T. Synthesis and carbonic anhydrase isoenzymes I, II, IX, and XII inhibitory effects of dimethoxy-bromophenol derivatives incorporating cyclopropane moieties. J. Med. Chem. 2015, 58, 640–650. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–251. [Google Scholar] [CrossRef]
- Yıldırım, A.; Atmaca, U.; Keskin, A.; Topal, M.; Çelik, M.; Gülçin, İ.; Supuran, C.T. N-Acylsulfonamides strongly inhibit human carbonic anhydrase isoenzymes I and II. Bioorg. Med. Chem. 2015, 23, 2598–2605. [Google Scholar]
- Aydin, B.; Gülcin, I.; Alwasel, S.H. Purification and characterization of polyphenol oxidase from Hemşin apple (Malus communis L.). Int. J. Food Propert. 2015, 18, 2735–2745. [Google Scholar] [CrossRef]
- Arabaci, B.; Gülçin, İ.; Alwasel, S. Capsaicin: A potent inhibitor of carbonic anhydrase isoenzymes. Molecules 2014, 19, 10103–10114. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, D.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1975, 227, 680–685. [Google Scholar] [CrossRef]
- Göksu, S.; Naderi, A.; Akbaba, Y.; Kalın, P.; Akıncıoğlu, A.; Gulcin, İ.; Durdaği, S.; Salmas, R.E. Carbonic anhydrase inhibitory properties of novel benzylsulfamides using molecular modeling and experimental studies. Bioorg. Chem. 2014, 56, 75–82. [Google Scholar]
- Güney, M.; Coşkun, A.; Topal, F.; Daştan, A.; Gülçin, İ.; Supuran, C.T. Oxidation of cyanobenzocycloheptatrienes: Synthesis, photooxygenation reaction and carbonic anhydrase isoenzymes inhibition properties of some new benzotropone derivatives. Bioorg. Med. Chem. 2014, 22, 3537–3543. [Google Scholar]
- Topal, M.; Gülçin, İ. Rosmarinic acid: A potent carbonic anhydrase isoenzymes inhibitor. Turk. J. Chem. 2014, 38, 894–902. [Google Scholar] [CrossRef]
- Lineweaver, H.; Burk, D. The determination of enzyme dissociation constants. J. Am. Chem. Soc. 1934, 56, 658–666. [Google Scholar] [CrossRef]
- Aksu, K.; Nar, M.; Tanç, M.; Vullo, D.; Gülçin, İ.; Göksu, S.; Tümer, F.; Supuran, C.T. The synthesis of sulfamide analogues of dopamine related compounds and their carbonic anhydrase inhibitory properties. Bioorg. Med. Chem. 2013, 21, 2925–2931. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, İ.; Beydemir, S. Phenolic compounds as antioxidants: Carbonic anhydrase isoenzymes inhibitors. Mini Rev. Med. Chem. 2013, 13, 408–430. [Google Scholar]
- Akıncıoğlu, A.; Akbaba, Y.; Göçer, H.; Göksu, S.; Gülçin, İ.; Supuran, C.T. Novel sulfamides as potential carbonic anhydrase isoenzymes inhibitors. Bioorg. Med. Chem. 2013, 21, 1379–1385. [Google Scholar]
- Öztürk, Sarı; kaya, S.B.; Topal, F.; Şentürk, M.; Gülçin, İ.; Supuran, C.T. In vitro inhibition of α-carbonic anhydrase isozymes by some phenolic compounds. Bioorg. Med. Chem. Lett. 2011, 21, 4259–4262. [Google Scholar]
- Temperini, C.; Scozzafava, A.; Supuran, C.T. Carbonic anhydrase inhibitors. X-ray crystal studies of the carbonic anhydrase II-trithiocarbonate adduct-An inhibitor mimicking the sulfonamide and urea binding to the enzyme. Bioorg. Med. Chem. Lett. 2010, 20, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Thiry, A.; Dogné, J.M.; Supuran, C.T.; Masereel, B. Anticonvulsant sulfonamides/sulfamates/sulfamides with carbonic anhydrase inhibitory activity: Drug design and mechanism of action. Curr. Pharm. Des. 2008, 14, 661–671. [Google Scholar] [PubMed]
- Alterio, V.; Di Fiore, A.; D’Ambrosio, K.; Supuran, C.T.; De Simone, G. Multiple binding modes of inhibitors to carbonic anhydrases: How to design specific drugs targeting 15 different isoforms? Chem. Rev. 2012, 112, 4421–4468. [Google Scholar] [CrossRef] [PubMed]
- Topal, F.; Gülçin, İ.; Dastan, A; Guney, M. Novel eugenol derivatives: Potent acetylcholinesterase and carbonic anhydrase inhibitors. Int. J. Biol. Macromol. 2017, 94, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Kirthana, M.V.; Khan, F.N.; Sivakumar, P.M.; Doble, M.; Manivel, P.; Prabakaran, K.; Krishnakumar, V. Antithyroid agents and QSAR studies: Inhibition of lactoperoxidase-catalyzed iodination reaction by isochromene-1-thiones. Med. Chem. Res. 2013, 22, 4810–4817. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
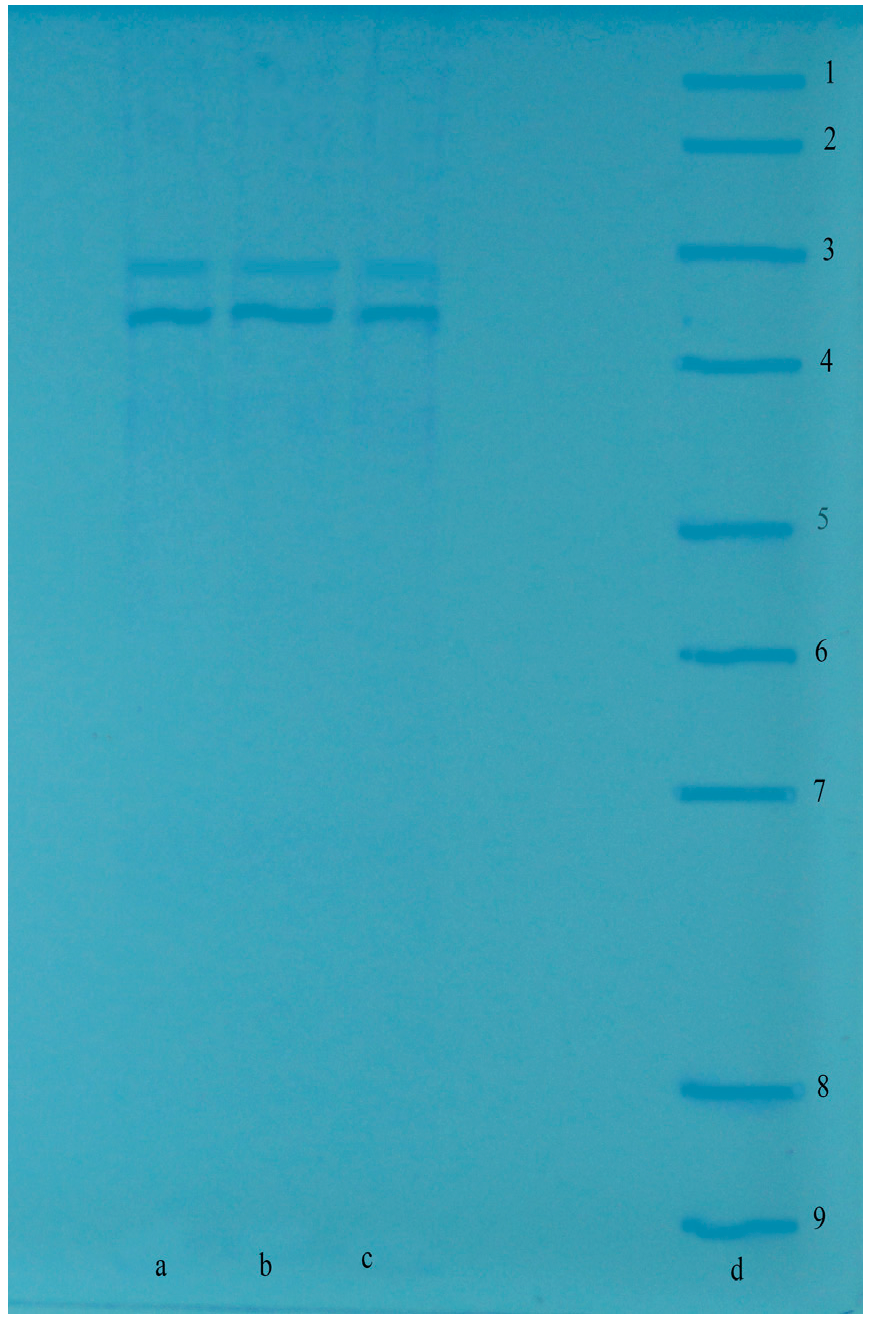
| Purification Steps | Total Volume (mL) | Enzyme Activity (EU/mL) | Total Enzyme Activity (EU/mL·min) | Protein (mg/mL) | Total Protein (mg) | Specific Activity (EU/mg) | Yield (%) | Purification Fold |
|---|---|---|---|---|---|---|---|---|
| Homogenate | 60.0 | 1.0 | 60.0 | 15.0 | 900.0 | 0.06 | 100 | 1.00 |
| Sepharose 4B-l-tyrosine-sulfonamide | 10.0 | 4.5 | 45.0 | 0.184 | 1.84 | 24.45 | 75.6 | 407.0 |
| No. | Compound Names | IC50 (µM) | Ki (µM) | Inhibition Type |
|---|---|---|---|---|
| 4a | Sulfathiazole | 231.00 | 38.43 ± 6.06 | Non-competitive |
| 4b | Sulfadiazine | 92.24 | 90.66 ± 2.52 | Non-competitive |
| 4c | Sulfamethazine | 346.50 | 198.00 ± 24.46 | Non-competitive |
| 4d | Sulfapyridine | 227.00 | 65.00 ± 3.61 | Competitive |
| 4e | Sulfisoxazole | 221.70 | 182.66 ± 34.07 | Non-competitive |
| 4f | Sulfamethizole | 115.50 | 21.18 ± 5.66 | Non-competitive |
| 4g | Sulfamerazine | 18.73 | 20.52 ± 2.14 | Non-competitive |
| 4h | Sulfanilamide | 8.48 | 35.70 ± 4.88 | Competitive |
| 5a | N-(sulfathiazole)-p-acetoxybenzamide | 99.00 | 56.06 ± 23.56 | Non-competitive |
| 5b | N-(sulfadiazine)-p-acetoxybenzamide | 0.015 | 0.026 ± 0.004 | Non-competitive |
| 5c | N-(sulfamethazine)-p-acetoxybenzamide | 0.010 | 0.008 ± 0.003 | Competitive |
| 5d | N-(sulfapyridine)-p-acetoxybenzamide | 10.04 | 49.15 ± 19.99 | Competitive |
| 5e | N-(sulfisoxazole)-p-acetoxybenzamide | 3.03 | 0.934 ± 0.357 | Competitive |
| 5f | N-(sulfamethizole)-p-acetoxybenzamide | 0.238 | 0.324 ± 0.115 | Non-competitive |
| 5g | N-(sulfamerazine)-p-acetoxybenzamide | 3.94 | 13.39 ± 6.19 | Competitive |
| 5h | N-(sulfanilamide)-p-acetoxybenzamide | 3.053 | 2.29 ± 1.02 | Non-competitive |
| 6a | N-(sulfathiazole)-3,4,5-triacetoxybenzamide | 0.656 × 10−3 | 1.096 × 10−3 ± 0.471 × 10−3 | Non-competitive |
| 6b | N-(sulfadiazine)-3,4,5-triacetoxybenzamide | 13.86 | 2.90 ± 1.13 | Competitive |
| 6c | N-(sulfamethazine)-3,4,5-triacetoxybenzamide | 138.60 | 152.03 ± 48.69 | Non-competitive |
| 6d | N-(sulfapyridine)-3,4,5-triacetoxybenzamide | 1.56 | 1.39 ± 0.15 | Non-competitive |
| 6e | N-(sulfisoxazole)-3,4,5-triacetoxybenzamide | 2.03 | 1.07 ± 0.02 | Competitive |
| 6f | N-(sulfamethizole)-3,4,5-triacetoxybenzamide | 6.66 | 8.55 ± 1.45 | Non-competitive |
| 6g | N-(sulfamerazine)-3,4,5-triacetoxybenzamide | 1.28 | 1.49 ± 0.28 | Non-competitive |
| 6h | N-(sulfanilamide)-3,4,5-triacetoxybenzamide | 3.05 | 2.29 ± 0.98 | Non-competitive |
| 7a | N-(sulfathiazole)-p-hydroxybenzamide | 15.06 | 6.85 ± 1.81 | Non-competitive |
| 7b | N-(sulfadiazine)-p-hydroxybenzamide | 0.040 | 0.044 ± 0.022 | Noncompetitive |
| 7c | N-(sulfamethazine)-p-hydroxybenzamide | 0.426 | 0.484 ± 0.146 | Non-competitive |
| 7d | N-(sulfapyridine)-p-hydroxybenzamide | 2.96 | 1.41 ± 0.13 | Competitive |
| 7e | N-(sulfisoxazole)-p-hydroxybenzamide | 10.83 | 15.08 ± 3.90 | Non-competitive |
| 7f | N-(sulfamethizole)-p-hydroxybenzamide | 0.799 | 0.387 ± 0.150 | Competitive |
| 7g | N-(sulfamerazine)-p-hydroxybenzamide | 1.759 | 3.939 ± 1.098 | Non-competitive |
| 7h | N-(sulfanilamide)-p-hydroxybenzamide | 0.285 | 0.217 ± 0.099 | Non-competitive |
| 8a | N-(sulfathiazole)-3,4,5-trihydroxybenzamide | 0.014 | 0.025 ± 0.010 | Competitive |
| 8b | N-(sulfadiazine)-3,4,5-trihydroxybenzamide | 0.715 | 1.292 ± 0.465 | Non-competitive |
| 8c | N-(sulfamethazine)-3,4,5-trihydroxybenzamide | 2.53 | 2.17 ± 0.88 | Competitive |
| 8d | N-(sulfapyridine)-3,4,5-trihydroxybenzamide | 10.19 | 5.85 ± 1.28 | Competitive |
| 8e | N-(sulfisoxazole)-3,4,5-trihydroxybenzamide | 1.99 | 1.44 ± 0.52 | Competitive |
| 8f | N-(sulfamethizole)-3,4,5-trihydroxybenzamide | 173.25 | 292.78 ± 61.96 | Non-competitive |
| 8g | N-(sulfamerazine)-3,4,5-trihydroxybenzamide | 1.484 | 3.87 ± 0.43 | Non-competitive |
| 8h | N-(sulfanilamide)-3,4,5-trihydroxybenzamide | 1.782 | 3.104 ± 0.578 | Non-competitive |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Köksal, Z.; Kalin, R.; Camadan, Y.; Usanmaz, H.; Almaz, Z.; Gülçin, İ.; Gokcen, T.; Gören, A.C.; Ozdemir, H. Secondary Sulfonamides as Effective Lactoperoxidase Inhibitors. Molecules 2017, 22, 793. https://doi.org/10.3390/molecules22060793
Köksal Z, Kalin R, Camadan Y, Usanmaz H, Almaz Z, Gülçin İ, Gokcen T, Gören AC, Ozdemir H. Secondary Sulfonamides as Effective Lactoperoxidase Inhibitors. Molecules. 2017; 22(6):793. https://doi.org/10.3390/molecules22060793
Chicago/Turabian StyleKöksal, Zeynep, Ramazan Kalin, Yasemin Camadan, Hande Usanmaz, Züleyha Almaz, İlhami Gülçin, Taner Gokcen, Ahmet Ceyhan Gören, and Hasan Ozdemir. 2017. "Secondary Sulfonamides as Effective Lactoperoxidase Inhibitors" Molecules 22, no. 6: 793. https://doi.org/10.3390/molecules22060793







