Direct Substitution of Alcohols in Pure Water by Brønsted Acid Catalysis
Abstract
:1. Introduction
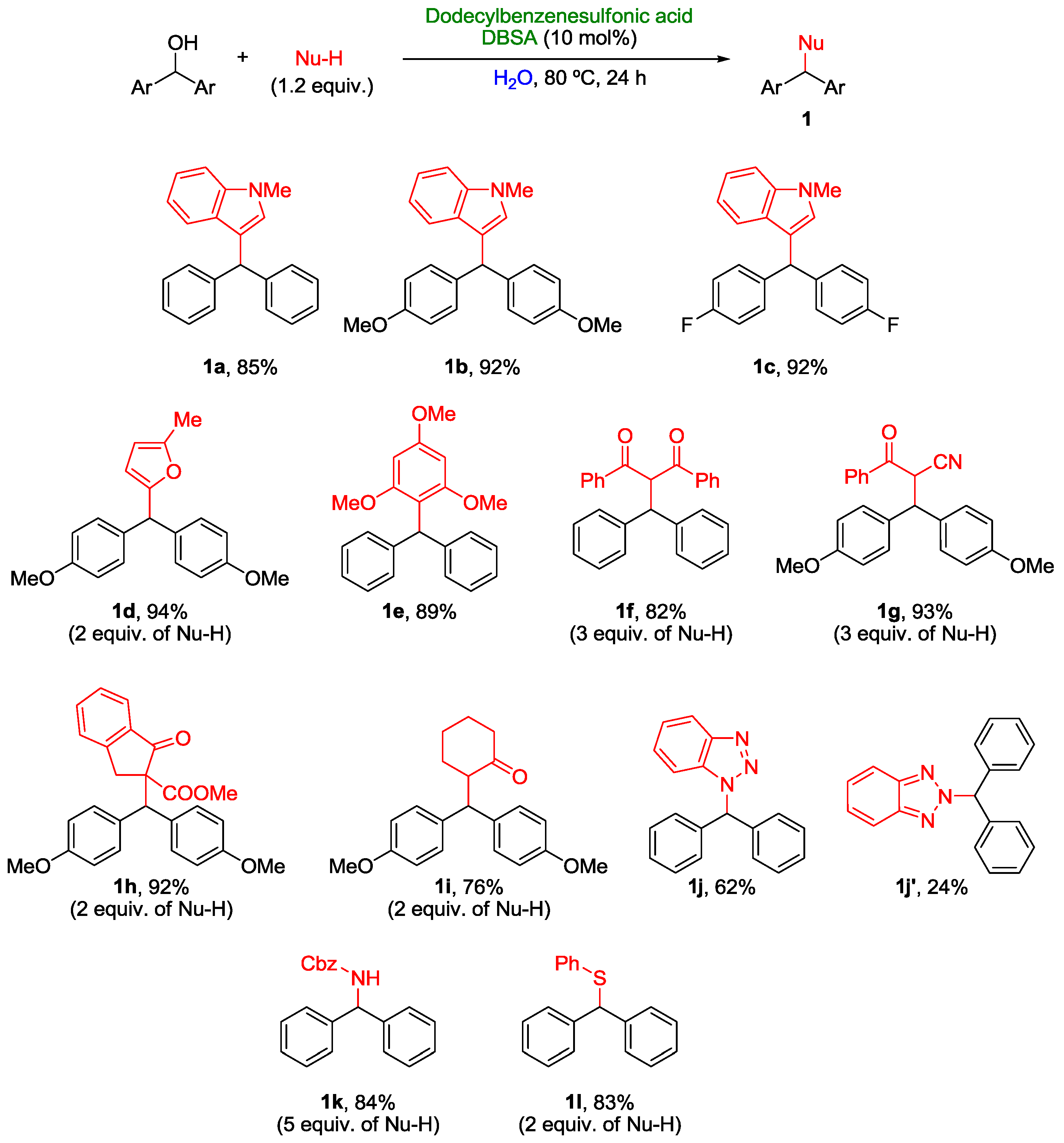
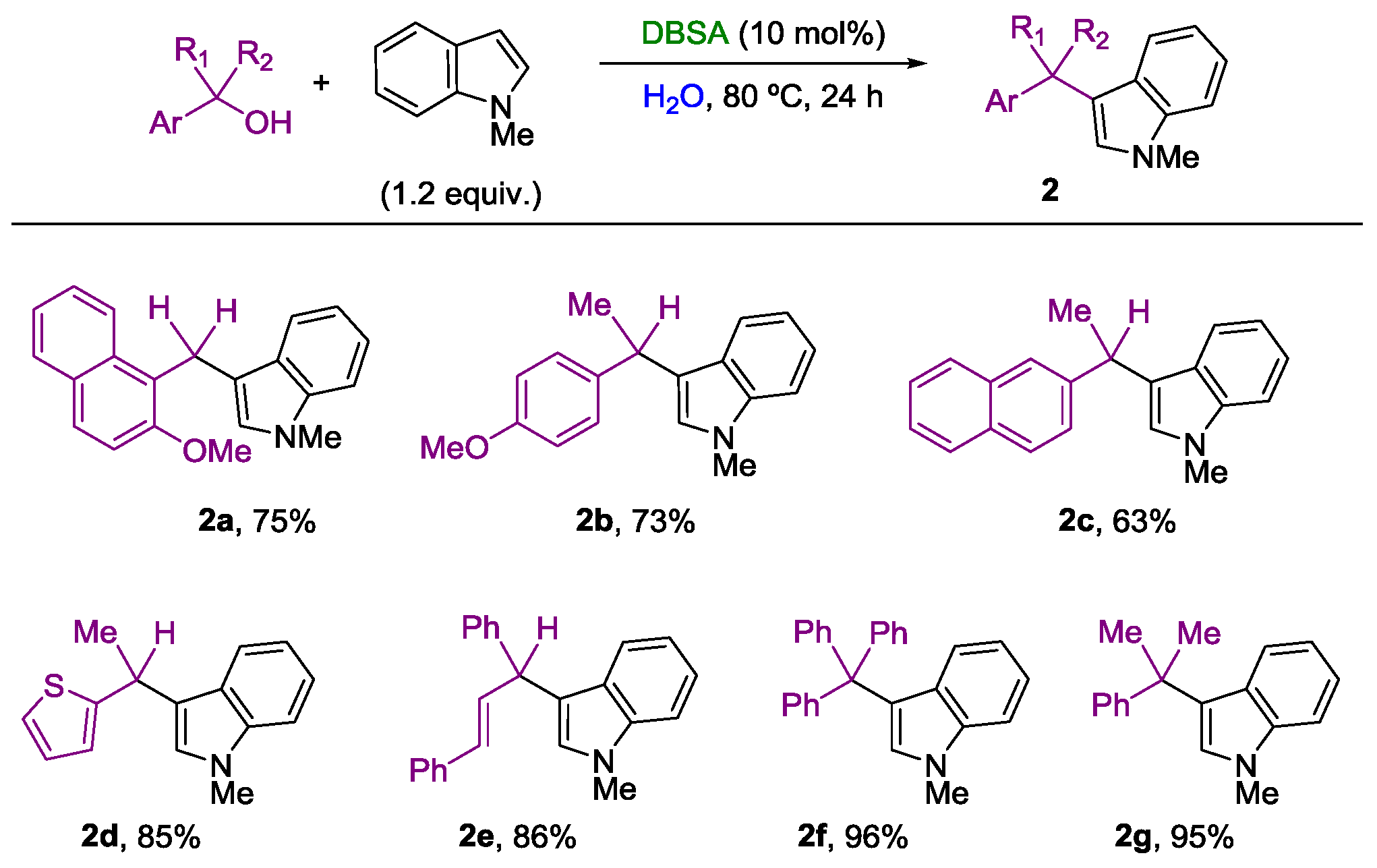
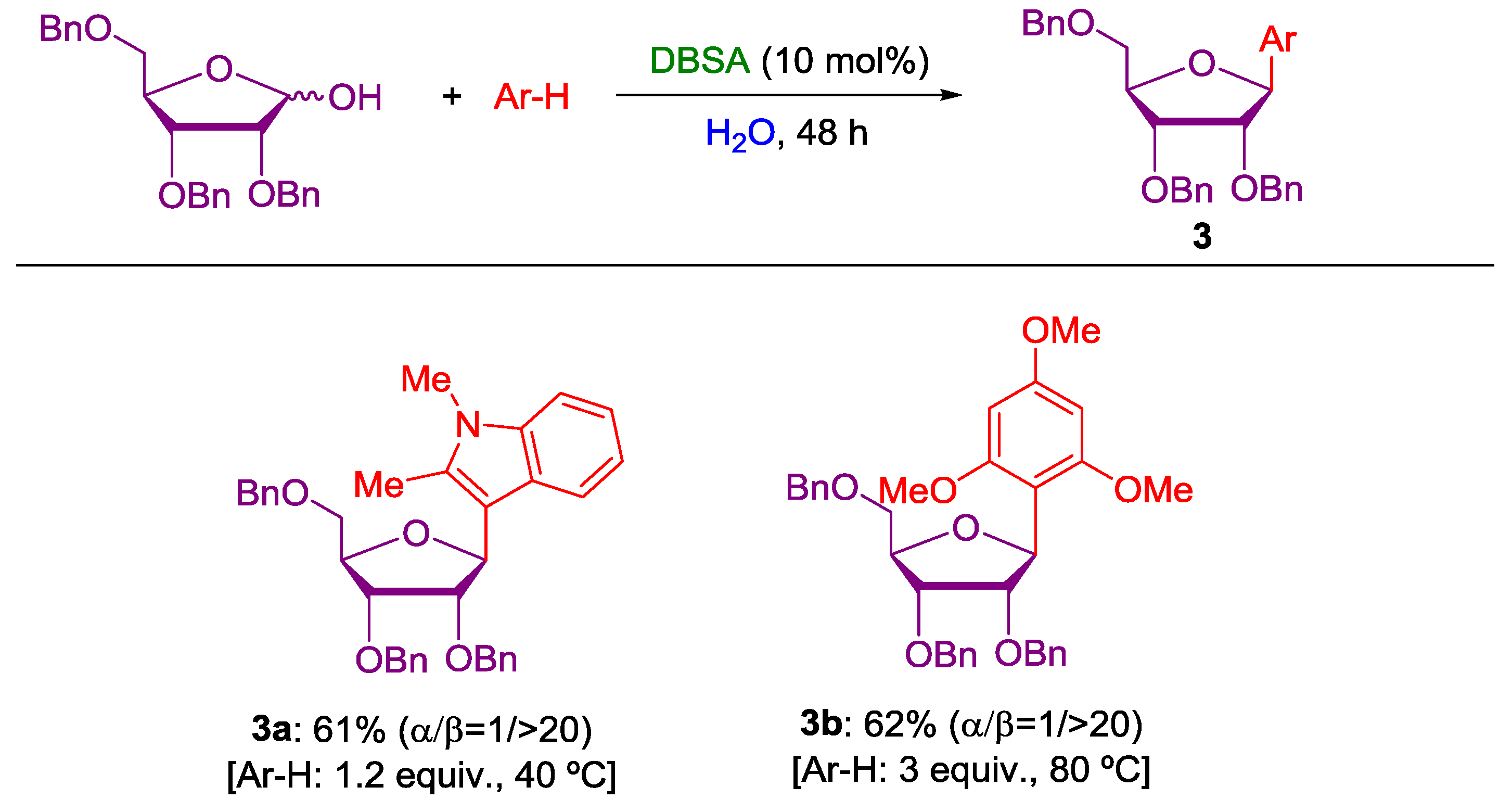
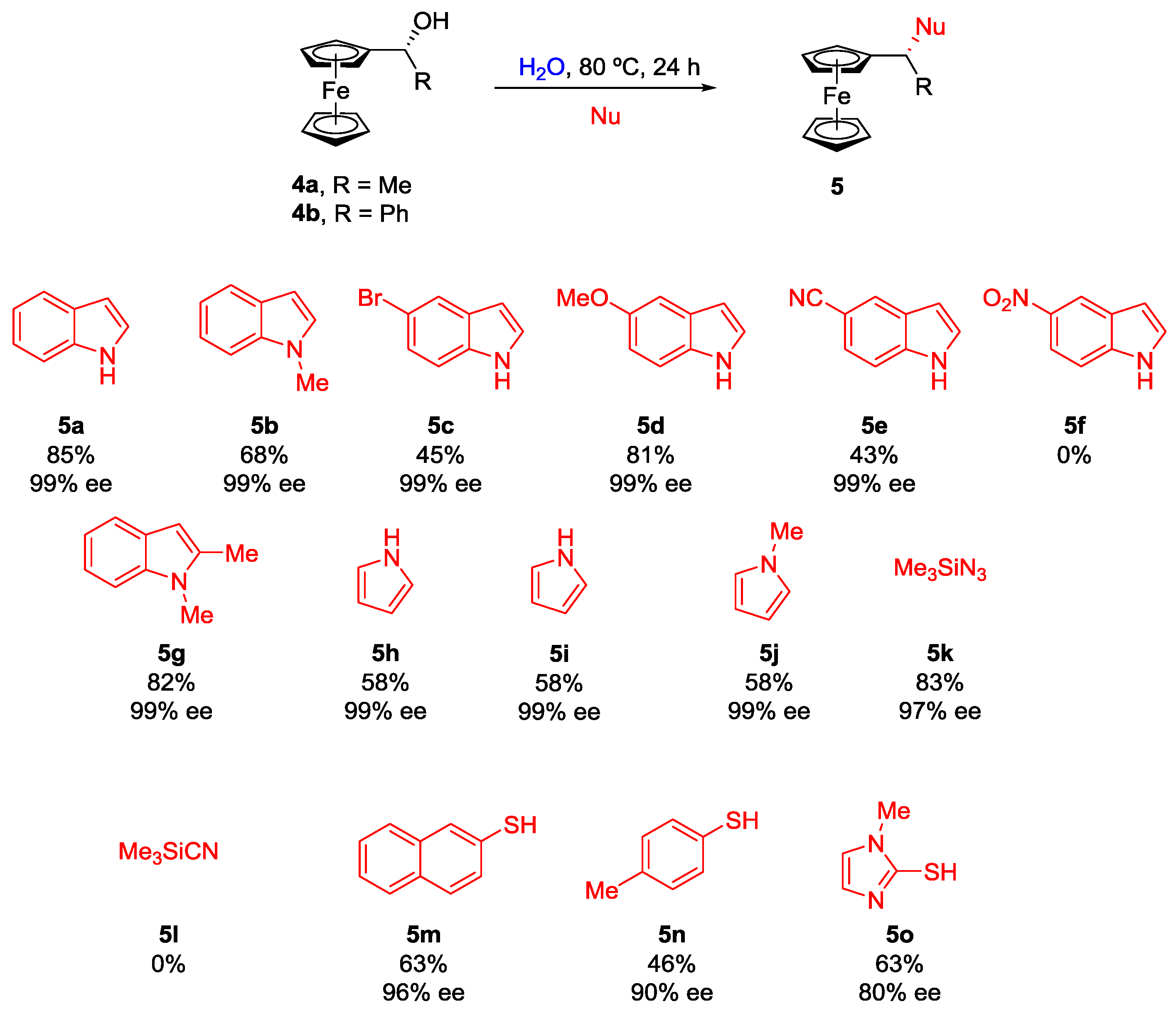
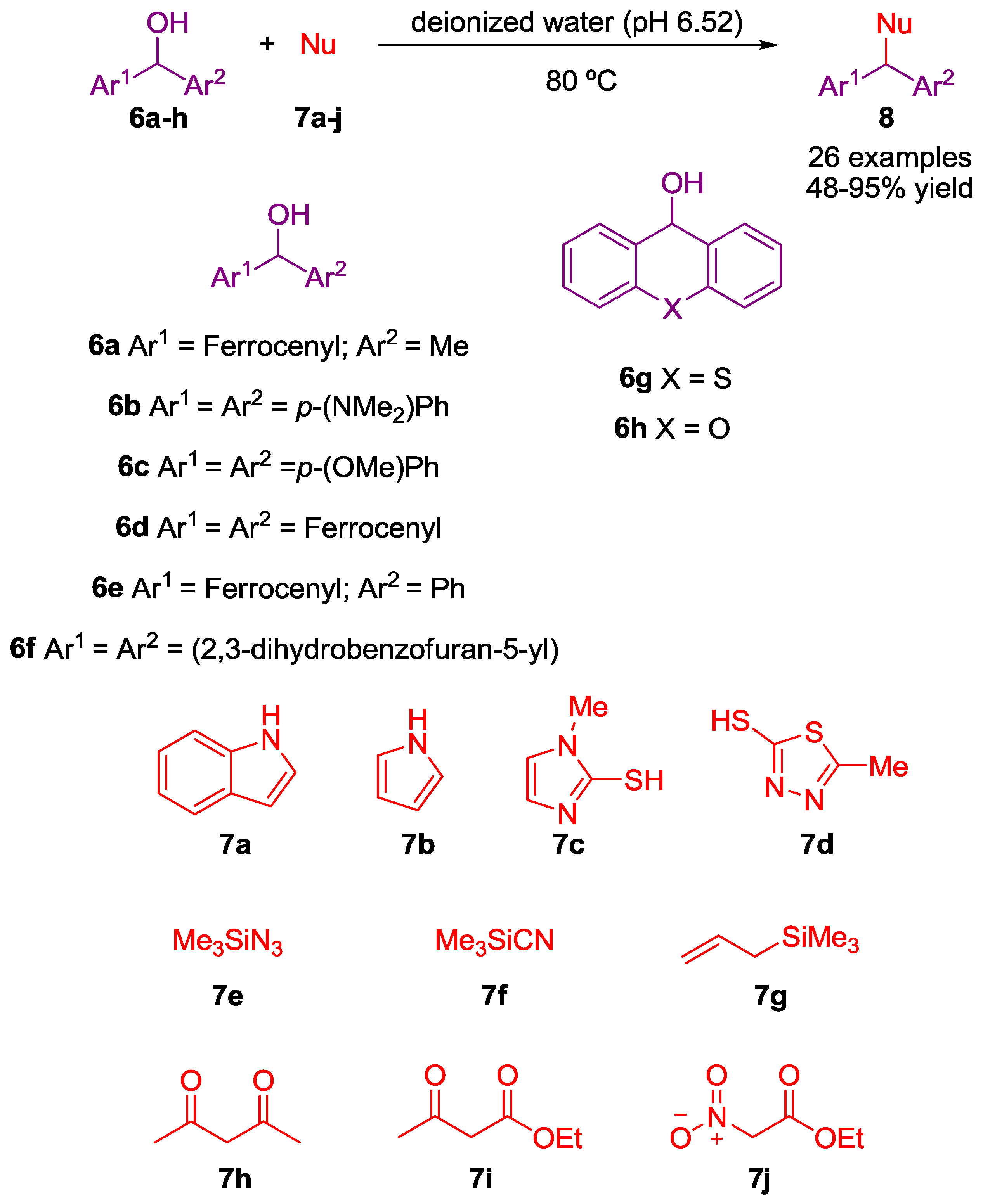
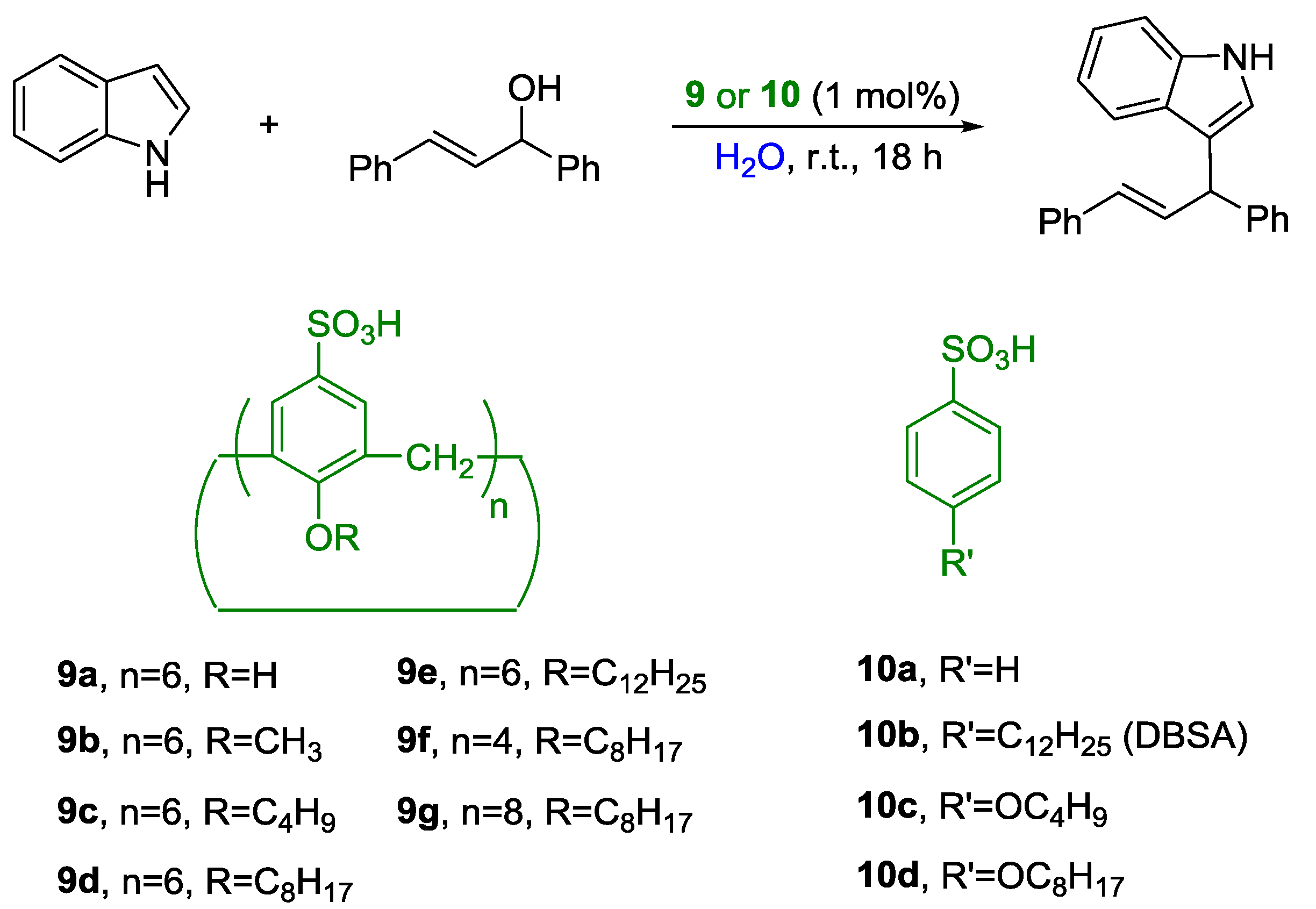
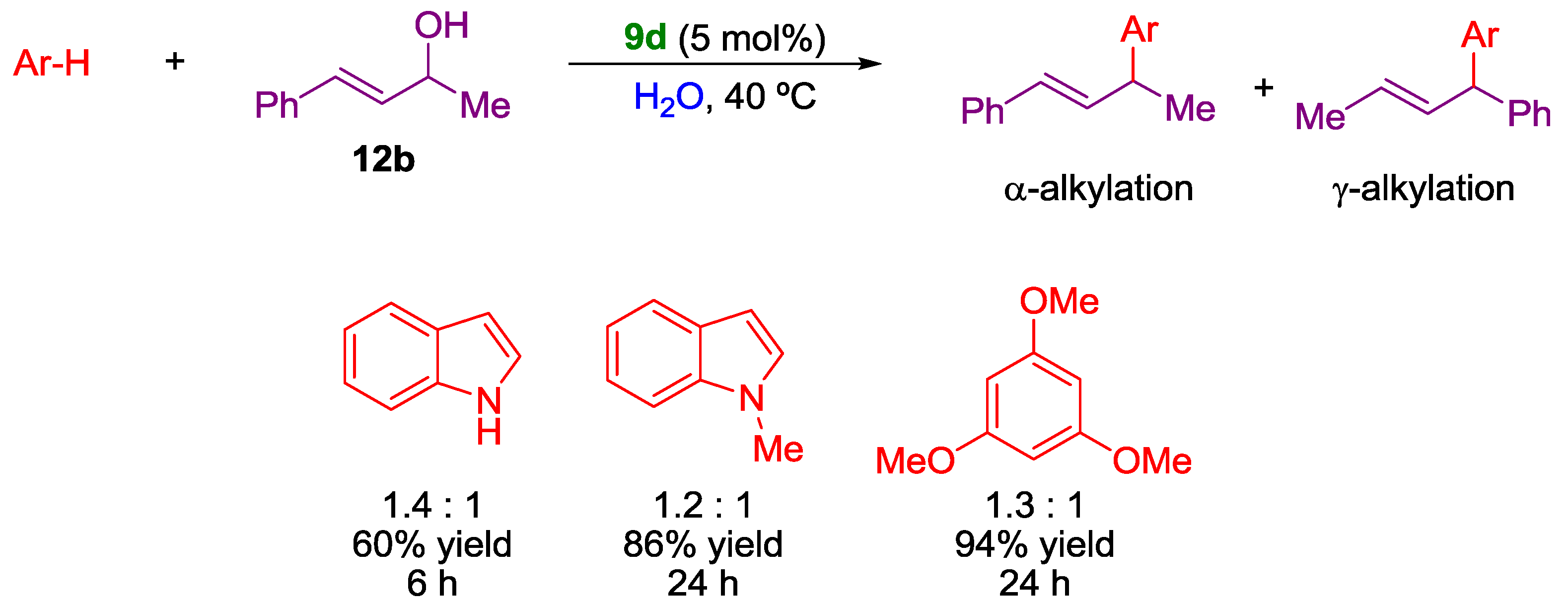
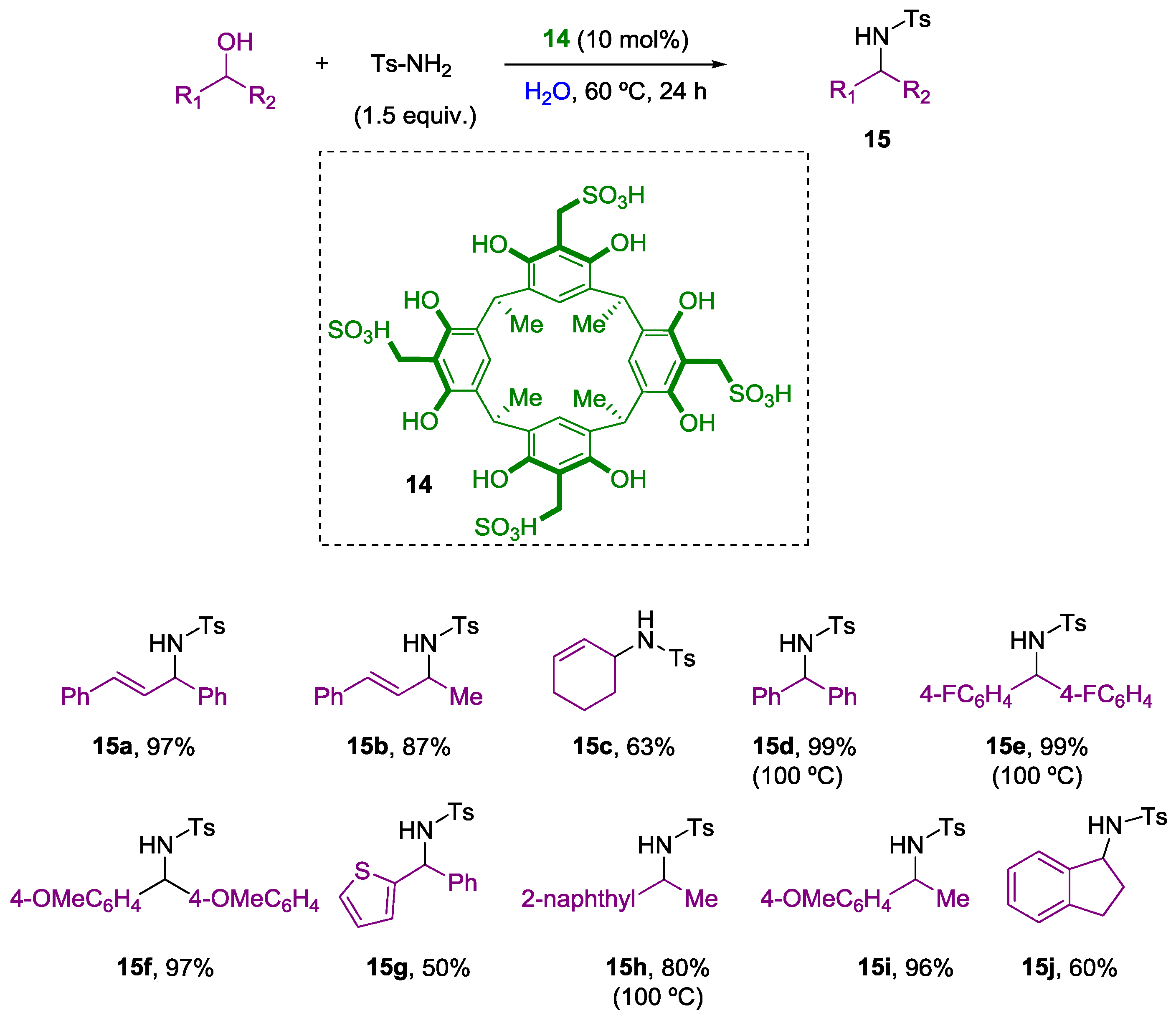
2. Nucleophilic Substitution of Alcohols in Pure Water
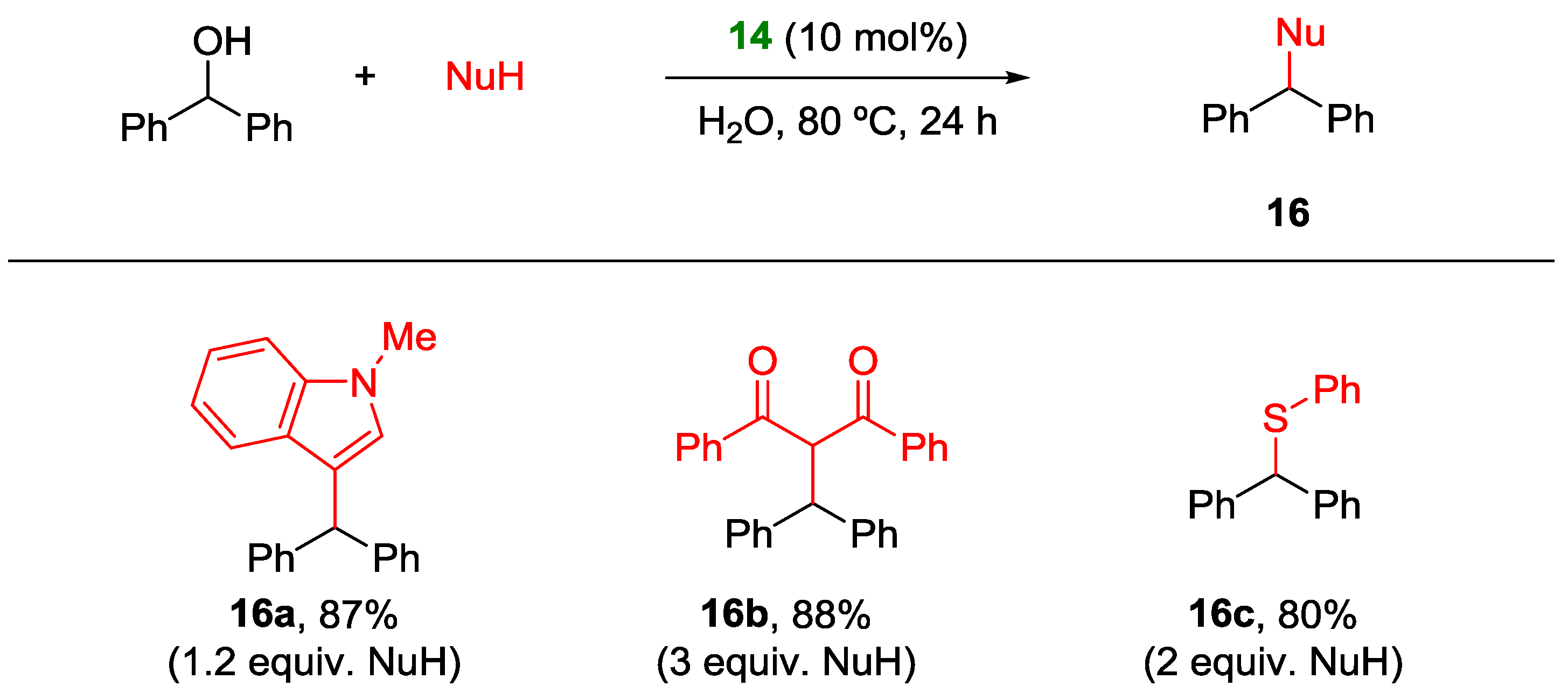
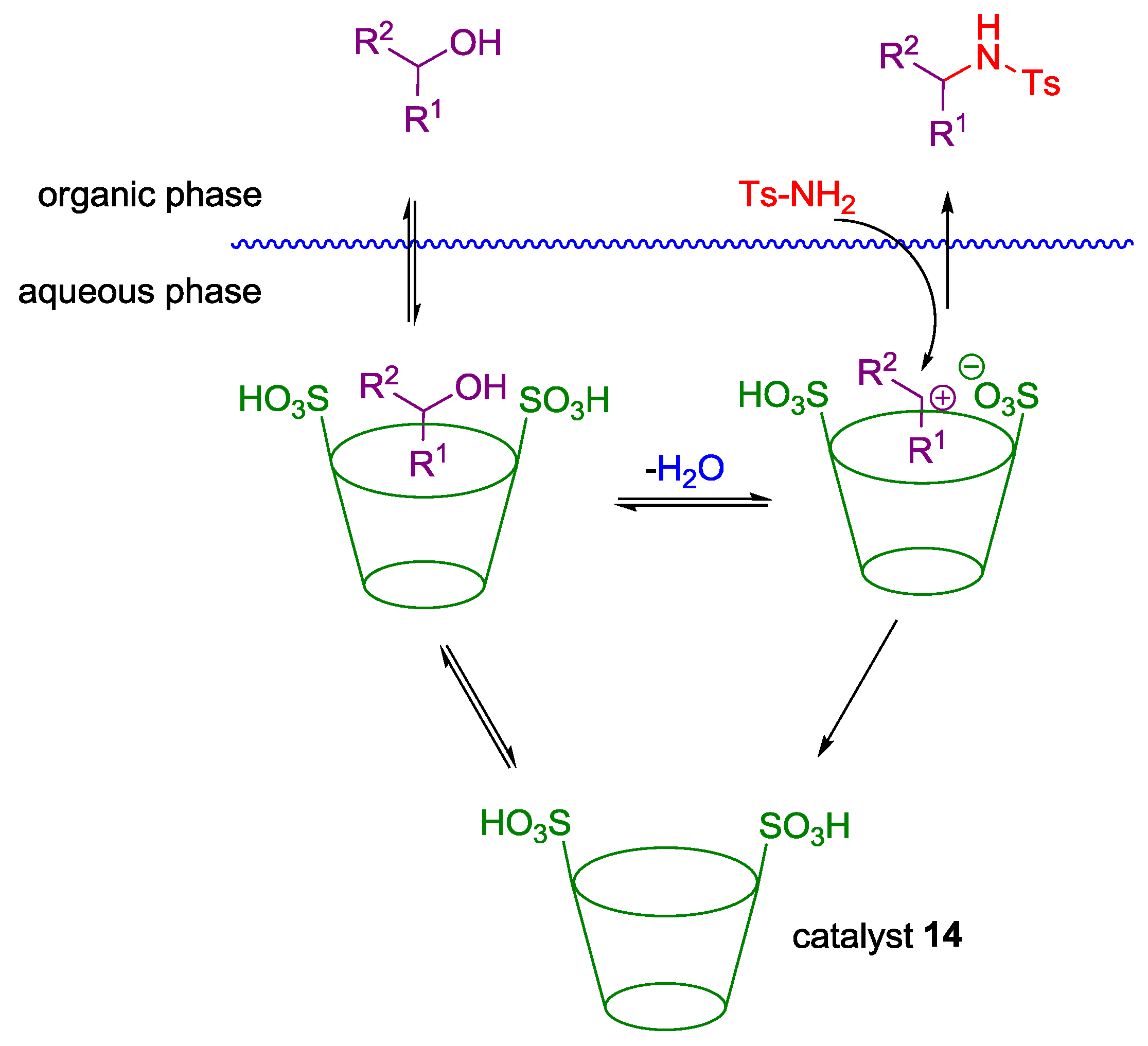
Non Chiral Brønsted Acid Catalysts and Non-Catalyzed Examples
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Emer, E.; Sinisi, R.; Capdevila, M.G.; Petruzziello, D.; De Vincentiis, F.; Cozzi, P.G. Direct nucleophilic SN1-type reactions of alcohols. Eur. J. Org. Chem. 2011, 647–666. [Google Scholar] [CrossRef]
- Muzart, J. Gold-catalysed reactions of alcohols: Isomerisation, inter- and intramolecular reactions leading to C-C and C-heteroatom bonds. Tetrahedron 2008, 64, 5815–5849. [Google Scholar] [CrossRef]
- Muzart, J. Procedures for and possible mechanisms of Pd-catalyzed allylations of primary and secondary amines with allylic alcohols. Eur. J. Org. Chem. 2007, 3077–3089. [Google Scholar] [CrossRef]
- Sundararaju, B.; Achard, M.; Bruneau, C. Transition metal catalyzed nucleophilic allylic substitution: Activation of allylic alcohols via π-allylic species. Chem. Soc. Rev. 2012, 41, 4467–4483. [Google Scholar] [CrossRef] [PubMed]
- Gualandi, A.; Mengozzi, L.; Wilson, C.M.; Cozzi, P.G. Synergistic stereoselective organocatalysis with indium(III) salts. Synthesis 2014, 46, 1321–1328. [Google Scholar] [CrossRef]
- Baeza, A.; Najera, C. Recent advances in the direct nucleophilic substitution of allylic alcohols through SN1-type reactions. Synthesis 2014, 46, 25–34. [Google Scholar] [CrossRef]
- Gualandi, A.; Petruzziello, D.; Emer, E.; Cozzi, P.G. Alpha-alkylation by SN1-type reactions. In Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications, 1st ed.; Dalko, P.I., Ed.; Wiley-VCH: Weinheim, Germany, 2013; pp. 729–755. [Google Scholar]
- Gualandi, A.; Cozzi, P.G. Stereoselective organocatalytic alkylations with carbenium ions. Synlett 2013, 24, 281–296. [Google Scholar] [CrossRef]
- Mayr, H.; Patz, M. Scales of nucleophilicity and electrophilicity: A system for ordering polar organic and organometallic reactions. Angew. Chem. Int. Ed. Engl. 1994, 33, 938–957. [Google Scholar] [CrossRef]
- Lucius, R.; Mayr, H. Constant selectivity relationships of addition reactions of carbanions. Angew. Chem. Int. Ed. 2000, 39, 1995–1997. [Google Scholar] [CrossRef]
- Mayr, H.; Kempf, B.; Ofial, A.R. π-Nucleophilicity in carbon-carbon bond-forming reactions. Acc. Chem. Res. 2003, 36, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Mayr, H.; Ofial, A.R. Kinetics of electrophile-nucleophile combinations: A general approach to polar organic reactivity. Pure Appl. Chem. 2005, 77, 1807–1821. [Google Scholar] [CrossRef]
- Minegishi, S.; Kobayashi, S.; Mayr, H. Solvent nucleophilicity. J. Am. Chem. Soc. 2004, 126, 5174–5181. [Google Scholar] [CrossRef] [PubMed]
- Coote, S.J.; Davies, S.G.; Middlemiss, D.; Naylor, A. Enantiospecific synthesis of (+)-(R)-1-phenyl-3-methyl-1,2,4,5-tetrahydrobenz[d]azepine from (+)-(S)-N-methyl-1-phenyl ethanolamine (halostachine) via arene chromium tricarbonyl methodology. Tetrahedron Lett. 1989, 30, 3581–3584. [Google Scholar] [CrossRef]
- Bisaro, F.; Prestat, G.; Vitale, M.; Poli, G. Alkylation of active methylenes via benzhydryl cations. Synlett 2002, 1823–1826. [Google Scholar] [CrossRef]
- Gullickson, G.C.; Lewis, D.E. Reactions of active methylene compounds with benzhydrol during solvolysis in formic acid. Aust. J. Chem. 2003, 56, 385–388. [Google Scholar] [CrossRef]
- Li, C.-J. Organic reactions in aqueous media with a focus on carbon-carbon bond formations: A decade update. Chem. Rev. 2005, 105, 3095–3165. [Google Scholar] [CrossRef] [PubMed]
- Lindström, U.M.; Andersson, F. Hydrophobically directed organic synthesis. Angew. Chem. Int. Ed. 2006, 45, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Jessop, P.G. Searching for green solvents. Green Chem. 2011, 13, 1391–1398. [Google Scholar] [CrossRef]
- Lindström, U.M. Stereoselective organic reactions in water. Chem. Rev. 2002, 102, 2751–2772. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-J.; Chen, L. Organic chemistry in water. Chem. Soc. Rev. 2006, 35, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Herrerías, C.I.; Yao, X.; Li, Z.; Li, C.-J. Reactions of C-H bonds in water. Chem. Rev. 2007, 107, 2546–2562. [Google Scholar] [CrossRef] [PubMed]
- Chanda, A.; Fokin, V.V. Organic synthesis “on water”. Chem. Rev. 2009, 109, 725–748. [Google Scholar] [CrossRef] [PubMed]
- Gruttadauria, M.; Giacalone, F.; Noto, R. Water in stereoselective organocatalytic reactions. Adv. Synth. Catal. 2009, 351, 33–57. [Google Scholar] [CrossRef]
- Paradowska, J.; Stodulski, M.; Mlynarski, J. Catalysts based on amino acids for asymmetric reactions in water. Angew. Chem. Int. Ed. 2009, 48, 4288–4297. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.N.; Coyne, A.G. Water: Nature’s reaction enforcers comparative effects for organic synthesis “in-water” and “on-water”. Chem. Rev. 2010, 110, 6302–6337. [Google Scholar] [CrossRef] [PubMed]
- Bhowmick, S.; Bhowmick, K.C. Catalytic asymmetric carbon-carbon bond-forming reactions in aqueous media. Tetrahedron: Asymmetry 2011, 22, 1945–1979. [Google Scholar] [CrossRef]
- Gawande, M.B.; Bonifácio, V.D.B.; Luque, R.; Branco, P.S.; Varma, R.S. Benign by design: Catalyst-free in-water, on-water green chemical methodologies in organic synthesis. Chem. Soc. Rev. 2013, 42, 5522–5551. [Google Scholar] [CrossRef] [PubMed]
- Narayan, S.; Muldoon, J.; Finn, M.G.; Fokin, V.V.; Kolb, H.C.; Sharpless, K.B. “On water”: Unique reactivity of organic compounds in aqueous suspension. Angew. Chem. Int. Ed. 2005, 44, 3275–3279. [Google Scholar] [CrossRef] [PubMed]
- Pirrung, M.C.; Das Sarma, K. Multicomponent reactions are accelerated in water. J. Am. Chem. Soc. 2004, 126, 444–445. [Google Scholar] [CrossRef] [PubMed]
- Price, B.K.; Tour, J. Functionalization of single-walled carbon nanotubes “on water”. J. Am. Chem. Soc. 2006, 128, 12899–12904. [Google Scholar] [CrossRef] [PubMed]
- González-Cruz, D.; Tejedor, D.; de Armas, P.; García-Tellado, F. Dual reactivity pattern of allenolates “on water”: The chemical basis for efficient allenolate-driven organocatalytic systems. Chem. Eur. J. 2007, 13, 4823–4832. [Google Scholar] [CrossRef] [PubMed]
- Marqués-López, E.; Herrera, R.P.; Fernández, R.; Lassaletta, J.M. Uncatalyzed Strecker-type reaction of N,N-dialkylhydrazones in pure water. Eur. J. Org. Chem. 2008, 3457–3460. [Google Scholar] [CrossRef]
- Xiao, J.; Wen, H.; Wang, L.; Xu, L.; Hao, Z.; Shao, C.-L.; Wang, C.-Y. Catalyst-free dehydrative SN1-type reaction of indolyl alcohols with diverse nucleophiles “on water”. Green Chem. 2016, 18, 1032–1037. [Google Scholar] [CrossRef]
- Butler, R.N.; Coyne, A.G. Organic synthesis reactions on-water at the organic–liquid water interface. Org. Biomol. Chem. 2016, 14, 9945–9960. [Google Scholar] [CrossRef] [PubMed]
- Hikawa, H.; Suzuki, H.; Yokoyama, Y.; Azumaya, I. Chemoselective benzylation of unprotected anthranilic acids with benzhydryl alcohols by water-soluble Au(III)/TPPMS in water. J. Org. Chem. 2013, 78, 6714–6720. [Google Scholar] [CrossRef] [PubMed]
- Hikawa, H.; Suzuki, H.; Azumaya, I. Au(III)/TPPMS-catalyzed benzylation of indoles with benzylic alcohols in water. J. Org. Chem. 2013, 78, 12128–12135. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Jiang, R.; Yang, J.-M.; Wang, S.-Y.; Ji, S.-J. In(OTf)3 catalyzed C3-benzylation of indoles with benzyl alcohols in water. RSC Adv. 2013, 3, 5459–5464. [Google Scholar] [CrossRef]
- Yang, S.-C.; Hsu, Y.-C.; Gan, K.-H. Direct palladium/carboxylic acid-catalyzed allylation of anilines with allylic alcohols in water. Tetrahedron 2006, 62, 3949–3958. [Google Scholar] [CrossRef]
- Hikawa, H.; Ino, Y.; Suzuki, H.; Yokoyama, Y. Pd-catalyzed benzylic C-H amidation with benzyl alcohols in water: A strategy to construct quinazolinones. J. Org. Chem. 2012, 77, 7046–7051. [Google Scholar] [CrossRef] [PubMed]
- Hikawa, H.; Yokoyama, Y. Palladium-catalyzed S-benzylation of unprotected mercaptobenzoic acid with benzyl alcohols in water. Org. Biomol. Chem. 2012, 10, 2942–2945. [Google Scholar] [CrossRef] [PubMed]
- Hikawa, H.; Suzuki, H.; Yokoyama, Y.; Azumaya, I. Mechanistic studies for synthesis of bis(indolyl)methanes: Pd-catalyzed C-H activation of indole-carboxylic acids with benzyl alcohols in water. Catalysts 2013, 3, 486–500. [Google Scholar] [CrossRef]
- Hikawa, H.; Yokoyama, Y. Pd-catalyzed C-H activation in water: Synthesis of bis(indolyl)methanes from indoles and benzyl alcohols. RSC Adv. 2013, 3, 1061–1064. [Google Scholar] [CrossRef]
- Shirakawa, S.; Kobayashi, S. Surfactant-type Brønsted acid catalyzed dehydrative nucleophilic substitutions of alcohols in water. Org. Lett. 2007, 9, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Manabe, K.; Sun, X.-M.; Kobayashi, S. Dehydration reactions in water. Surfactant-type Brønsted acid-catalyzed direct esterification of carboxylic acids with alcohols in an emulsion system. J. Am. Chem. Soc. 2001, 123, 10101–10102. [Google Scholar] [CrossRef]
- Manabe, K.; Iimura, S.; Sun, X.-M.; Kobayashi, S. Dehydration reactions in water. Brønsted acid-surfactant-combined catalyst for ester, ether, thioether, and dithioacetal formation in water. J. Am. Chem. Soc. 2002, 124, 11971–11978. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Iimura, S.; Manabe, K. Dehydration reactions in water. Surfactant-type Brønsted acid-catalyzed dehydrative etherification, thioetherification, and dithioacetalization in water. Chem. Lett. 2002, 10–11. [Google Scholar] [CrossRef]
- Vicennati, P.; Cozzi, P.G. Facile access to optically active ferrocenyl derivatives with direct substitution of the hydroxy group catalyzed by indium tribromide. Eur. J. Org. Chem. 2007, 2248–2253. [Google Scholar] [CrossRef]
- Cozzi, P.G.; Zoli, L. Nucleophilic substitution of ferrocenyl alcohols “on water”. Green Chem. 2007, 9, 1292–1295. [Google Scholar] [CrossRef]
- Lakhdar, S.; Westermaier, M.; Terrier, F.; Goumont, R.; Boubaker, T.; Ofial, A.R.; Mayr, H. Nucleophilic reactivities of indoles. J. Org. Chem. 2006, 71, 9088–9095. [Google Scholar] [CrossRef] [PubMed]
- Van Staveren, D.R.; Metzler-Nolte, N. Bioorganometallic chemistry of ferrocene. Chem. Rev. 2004, 104, 5931–5985. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, P.G.; Zoli, L. A rational approach towards the nucleophilic substitutions of alcohols “on water”. Angew. Chem. Int. Ed. 2008, 47, 4162–4166. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; Marcus, R.A. On the theory of organic catalysis “on water”. J. Am. Chem. Soc. 2007, 129, 5492–5502. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-L.; Liu, L.; Wang, Y.-L.; Han, Y.-C.; Wang, D.; Chen, Y.-J. Calix[n]arene sulfonic acids bearing pendant aliphatic chains as recyclable surfactant-type Brønsted acid catalysts for allylic alkylation with allyl alcohols in water. Green Chem. 2008, 10, 635–640. [Google Scholar] [CrossRef]
- Shinkai, S.; Mori, S.; Koreishi, H.; Tsubaki, T.; Manabe, O. Hexasulfonated calix[6]arene derivatives: A new class of catalysts, surfactants, and host molecules. J. Am. Chem. Soc. 1986, 108, 2409–2416. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Shimada, N.; Sasaki, Y. Mannich-type reactions in water using anionic water-soluble calixarenes as recoverable and reusable catalysts. Green Chem. 2006, 8, 608–614. [Google Scholar] [CrossRef]
- Shirakawa, S.; Shimizu, S. Dehydrative amination of alcohols in water using a water-soluble calix[4]resorcinarene sulfonic acid. Synlett 2008, 1539–1542. [Google Scholar] [CrossRef]
- Hirashita, T.; Kuwahara, S.; Okochi, S.; Tsuji, M.; Araki, S. Direct benzylation and allylic alkylation in high-temperature water without added catalysts. Tetrahedron Lett. 2010, 51, 1847–1851. [Google Scholar] [CrossRef]
- Hayashi, Y. In water or in the presence of water? Angew. Chem. Int. Ed. 2006, 45, 8103–8104. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz, R.; Herrera, R.P. Direct Substitution of Alcohols in Pure Water by Brønsted Acid Catalysis. Molecules 2017, 22, 574. https://doi.org/10.3390/molecules22040574
Ortiz R, Herrera RP. Direct Substitution of Alcohols in Pure Water by Brønsted Acid Catalysis. Molecules. 2017; 22(4):574. https://doi.org/10.3390/molecules22040574
Chicago/Turabian StyleOrtiz, Rosa, and Raquel P. Herrera. 2017. "Direct Substitution of Alcohols in Pure Water by Brønsted Acid Catalysis" Molecules 22, no. 4: 574. https://doi.org/10.3390/molecules22040574





