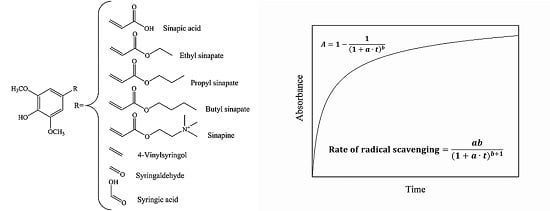
A Kinetic Approach in the Evaluation of Radical-Scavenging Efficiency of Sinapic Acid and Its Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. DPPH˙ Radical Scavenging Activity
2.2. Superoxide Anion Scavenging Activity
2.3. Lipid Radical Scavenging Activity
3. Methods
3.1. Materials and Reagents
3.2. Isolation of Sinapine
3.3. Synthesis of 4-Vinylsyringol
3.4. Synthesis of Sinapoyl Esters
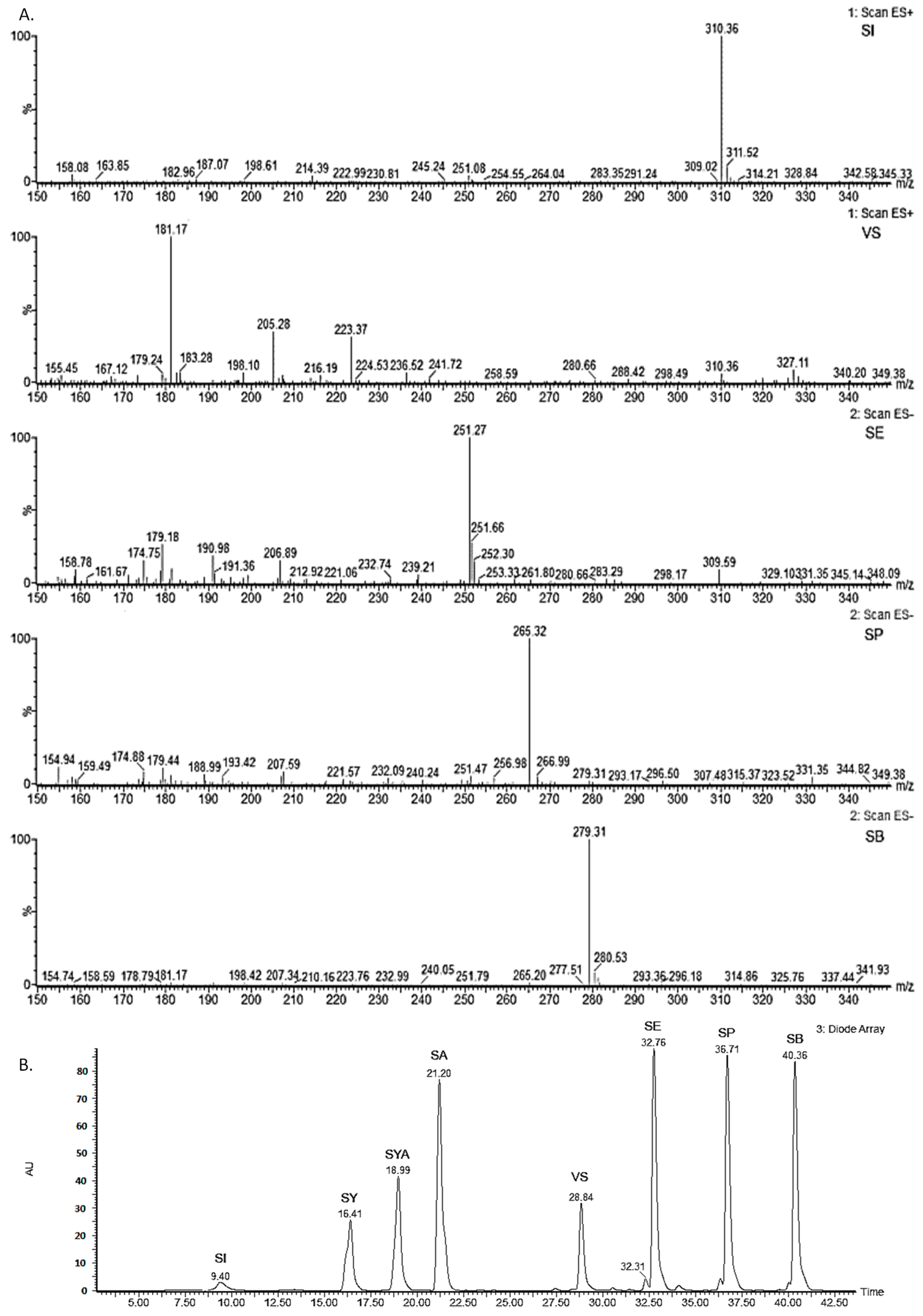
3.5. Liquid Chromatography with Photodiode Array Detection
3.6. Liquid Chromatography–Mass Spectrometry
3.7. Nuclear Magnetic Resonance Spectroscopy
3.8. DPPH˙ Radical Scavenging Activity
3.9. Superoxide Anion Scavenging Activity
3.10. Antioxidant Activity in the β-Carotene-linoleic Acid Emulsion System
3.11. Lipophilicity Determination
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mishra, K.; Ojha, H.; Chaudhury, N.K. Estimation of antiradical properties of antioxidants using DPPH assay: A critical review and results. Food Chem. 2012, 130, 1036–1043. [Google Scholar] [CrossRef]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyürek, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (IUPAC Technical Report). Pure Appl. Chem. 2013, 85, 957–998. [Google Scholar] [CrossRef]
- Villano, D.; Fernandez-Pachon, M.S.; Moya, M.L.; Troncoso, A.M.; Garcia-Parrilla, M.C. Radical scavenging ability of polyphenolic compounds towards DPPH free radical. Talanta 2007, 71, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Terpinc, P.; Abramovič, H. A kinetic approach for evaluation of the antioxidant activity of selected phenolic acids. Food Chem. 2010, 121, 366–371. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.J.; Liu, B.Y.; Zhao, M.M. Details of the antioxidant mechanism of hydroxycinnamic acids. Czech J. Food Sci. 2015, 33, 210–216. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Demirata, B.; Özyürek, M.; Çelik, S.E.; Bektaşoğlu, B.; Berker, K.I.; Özyurt, D. Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.S.; Johnson, E.R.; Dilabio, G.A. Predicting the activity of phenolic antioxidants: Theoretical method, analysis of substituent effects, and application to major families of antioxidants. J. Am. Chem. Soc. 2001, 123, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Najafi, M. On the antioxidant activity of the Ortho and Meta substituted Daidzein derivatives in the gas phase and solvent environment. J. Mex. Chem. Soc. 2014, 58, 36–45. [Google Scholar]
- Chen, Y.; Xiao, H.; Zheng, J.; Liang, G. Structure-thermodynamics-antioxidant activity relationships of selected natural phenolic acids and derivatives: An experimental and theoretical evaluation. PLoS ONE 2015, 10, e0121276. [Google Scholar] [CrossRef] [PubMed]
- Nenadis, N.; Siskos, D. Radical scavenging activity characterization of synthetic isochroman-derivatives of hydroxytyrosol: A gas-phase DFT approach. Food Res. Int. 2015, 76, 506–510. [Google Scholar] [CrossRef]
- Foti, M.C.; Daquino, C.; Geraci, C. Electron-transfer reaction of cinnamic acids and their methyl esters with the DPPH˙ radical in alcoholic solutions. J. Org. Chem. 2004, 69, 2309–2314. [Google Scholar] [CrossRef] [PubMed]
- Nićiforović, N.; Abramovič, H. Sinapic acid and its derivatives: Natural sources and bioactivity. Compr. Rev. Food Sci. Food Saf. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Teixeira, J.; Gaspar, A.; Garrido, E.M.; Garrido, J.; Borges, F. Hydroxycinnamic acid antioxidants: An electrochemical overview. BioMed Res. Int. 2013, 2013, 251754. [Google Scholar] [CrossRef] [PubMed]
- Ordoudi, S.A.; Tsimidou, M.Z.; Vafiadis, A.P.; Bakalbassis, E.G. Structure-DPPH˙ scavenging activity relationships: Parallel study of catechol and guaiacol acid derivatives. J. Agric. Food Chem. 2006, 54, 5763–5768. [Google Scholar] [CrossRef] [PubMed]
- Foti, M.C.; Daquino, C. Kinetic and thermodynamic parameters for the equilibrium reactions of phenols with the DPPH˙ radical. Chem. Commun. 2006, 14, 3252–3254. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.X.L.; Yang, R.T.; Shang, Y.J.; Dai, F.; Qian, Y.P.; Cheng, L.X.; Zhou, B.; Liu, Z.L. Oxidative coupling of cinnamic acid derivatives and their radical-scavenging activities. Chin. Sci. Bull. 2010, 55, 2885–2890. [Google Scholar] [CrossRef]
- Goupy, P.; Dufour, C.; Loonis, M.; Dangles, O. Quantitative kinetic analysis of hydrogen transfer reactions from dietary polyphenols to the DPPH radical. J. Agric. Food Chem. 2003, 51, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH) assay for antioxidant activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef] [PubMed]
- Valdez, L.B.; Lores Arnaiz, S.; Bustamante, J.; Alvarez, S.; Costa, L.E.; Boveris, A. Free radical chemistry in biological systems. Biol. Res. 2000, 33, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, J.; Arnér, E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 31, 1287–1312. [Google Scholar] [CrossRef]
- Ponti, V.; Dianzani, M.U.; Cheeseman, K.; Slater, T.F. Studies on the reduction of nitroblue tetrazolium chloride mediated through the action of NADH and phenazine methosulphate. Chem. Biol. Interact. 1978, 23, 281–291. [Google Scholar] [CrossRef]
- Balk, J.M.; Bast, A.; Haenen, G.R.M.M. Evaluation of the accuracy of antioxidant competition assays: Incorrect assumptions with major impact. Free Radic. Biol. Med. 2009, 47, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 3rd ed.; Oxford University Press: New York, NY, USA, 1999; p. 67. [Google Scholar]
- Lee, J.; Koo, N.; Min, D.B. Reactive oxygen species, aging, and antioxidative nutraceuticals. Compr. Rev. Food Sci. Food Saf. 2004, 3, 21–33. [Google Scholar] [CrossRef]
- Frankel, E.N.; Meyer, A.S. The problems of using one-dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Frankel, E.N.; Huang, S.W.; Kanner, J.; German, J.B. Interfacial phenomena in the evaluation of antioxidants: Bulk oils vs. emulsions. J. Agric. Food Chem. 1994, 42, 1054–1059. [Google Scholar] [CrossRef]
- Asnaashari, M.; Farhoosh, R.; Sharif, A. Antioxidant activity of gallic acid and methyl gallate in triacylglycerols of Kilka fish oil and its oil-in-water emulsion. Food Chem. 2014, 159, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Moure, A.; Franco, D.; Sineiro, J.; Dominguez, H.; Nunez, M.J.; Lema, J.M. Evaluation of extracts from Gevuina avellana hulls as antioxidants. J. Agric. Food Chem. 2000, 48, 3890–3897. [Google Scholar] [CrossRef] [PubMed]
- Craft, B.D.; Kerrihard, A.L.; Amarowicz, R.; Pegg, R.B. Phenol-based antioxidants and the in vitro methods used for their assessment. Compr. Rev. Food Sci. Food Saf. 2012, 11, 148–173. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. Vitamin E: Application of the principles of physical organic chemistry to the exploration of its structure and function. Acc. Chem. Res. 1986, 19, 194–201. [Google Scholar] [CrossRef]
- Thiyam, U.; Stöckmann, H.; Zum Felde, T.; Schwarz, K. Antioxidative effect of the main sinapic acid derivatives from rapeseed and mustard oil by-products. Eur. J. Lipid Sci. Technol. 2006, 108, 239–248. [Google Scholar] [CrossRef]
- Obied, H.K.; Song, Y.; Foley, S.; Loughlin, M.; Rehman, A.; Mailer, R.; Masud, T.; Agboola, S. Biophenols and antioxidant properties of Australian canola meal. J. Agric. Food Chem. 2013, 61, 9176–9184. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, A.D.M.; Friel, J.; Winkler-Moser, J.K.; Jacobsen, C.; Huidrom, D.; Reddy, N.; Thiyam-Holländer, U. Impact of endogenous canola phenolics on the oxidative stability of oil-in-water emulsions. Eur. J. Lipid Sci. Technol. 2013, 115, 501–512. [Google Scholar] [CrossRef]
- Schmidt, Š.; Pokorný, J. Potential application of oilseeds as sources of antioxidants for food lipids—A review. Czech J. Food Sci. 2005, 23, 93–102. [Google Scholar]
- Terpinc, P.; Polak, T.; Makuc, D.; Poklar Ulrih, N.; Abramovič, H. The occurrence and characterisation of phenolic compounds in Camelina sativa seed, cake and oil. Food Chem. 2012, 131, 580–589. [Google Scholar] [CrossRef]
- Terpinc, P.; Polak, T.; Šegatin, N.; Hanzlowsky, A.; Poklar Ulrih, N.; Abramovič, H. Antioxidant properties of 4-vinyl derivatives of hydroxycinnamic acids. Food Chem. 2011, 128, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, A.; Martins, M.; Silva, P.; Garrid, E.M.; Garrido, J.; Firuzi, O.; Miri, R.; Saso, L.; Borges, F. Dietary phenolic acids and derivatives. Evaluation of the antioxidant activity of sinapic acid and its alkyl esters. J. Agric. Food Chem. 2010, 58, 11273–11280. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Roback, J.; Gryglewski, R.J. Flavonoids are scavengers of superoxides anions. Biochem. Pharmacol. 1988, 37, 837–841. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds sinapine, 4-vinylsyringol, and ethyl, propyl and butyl sinapate are available from the authors.



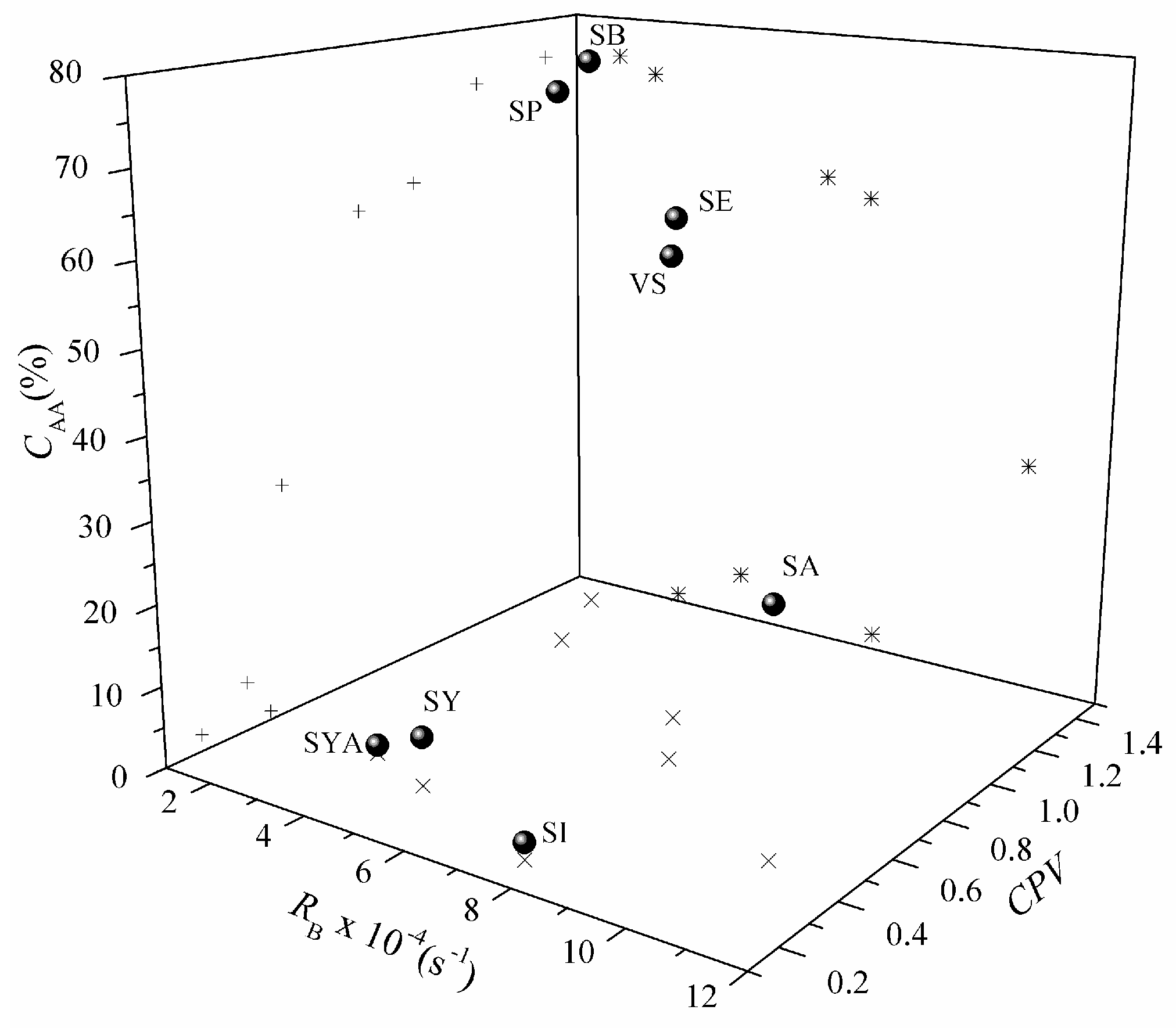
| Selected Compound | RS × 10−2 (s−1) | RFF × 10−3 (s−1) | RB × 10−4 (s−1) | r2 (DPPH˙) | r2 (O2˙−) | r2 (β-Carotene) | IDPPH (%) a | CSASA (%) a | CAA (%) a | CPV |
|---|---|---|---|---|---|---|---|---|---|---|
| Sinapic acid (SA) | 15.40 | 1.57 | 10.54 | 0.946 | 0.967 | 0.978 | 86 ± 0.4 | 66 ± 4 | 29 ± 2 | 0.46 |
| Sinapine (SI) | 0.27 | 14.21 | 7.62 | 0.967 | 0.996 | 0.967 | 23 ± 0.5 | 0 | 2 ± 0.5 | 0.20 |
| Vinyl syringol (VS) | 34.83 | 2.31 | 7.33 | 0.967 | 0.965 | 0.984 | 74 ± 0.6 | 26 ± 1 | 60 ± 1 | 0.73 |
| Syringic acid (SY) | 1.21 | 0.92 | 4.78 | 0.962 | 0.953 | 0.975 | 80 ± 1 | 75 ± 0.1 | 6 ± 1 | 0.34 |
| Syringaldehyde (SYA) | 0.05 | 6.37 | 3.37 | 0.447 | 0.981 | 0.968 | 6 ± 0.6 | 23 ± 3 | 1 ± 0.2 | 0.40 |
| Ethyl sinapate (SE) | 46.27 | 9.88 | 6.46 | 0.901 | 0.988 | 0.987 | 69 ± 1 | 5 ± 1 | 62 ± 2 | 0.90 |
| Propyl sinapate (SP) | 81.44 | ND | 2.79 | 0.969 | ND | 0.984 | 67 ± 1 | ND | 73 ± 1 | 1.13 |
| Butyl sinapate (SB) | 35.93 | ND | 1.99 | 0.875 | ND | 0.980 | 67 ± 0.8 | ND | 75 ± 1 | 1.38 |
| Compound | 1H- and 13C-NMR Spectroscopic Data |
|---|---|
| Sinapine (SI) | δH (297.80 MHz, DMSO-d6): 7.61 (d, J = 15.9, 1H, –CH=), 7.03 (s, 2H, Ar–H), 6.55 (d, J = 15.9, 1H, =CH–), 4.58 (s, 2H, –OCH2), 3.80 (s, 6H, –OCH3), 3.72 (m, 2H, CH2) 3.17 (s, 9H, –NCH3); δC (74.89 MHz, DMSO-d6): 165.93 (C=O), 148.07 (C(3), C(5)), 146.25 (C(β)), 138.82 (C–OH), 124.05 (C(1)), 113.97 (C(α)), 106.45 (C(2), C(6)), 63.97 (CH2), 57.65 (CH2), 56.13 (2× OCH3), 52.99 (N(CH3)3) |
| 4-Vinylsyringol (VS) | δH (297.80 MHz, DMSO-d6): 8.44 (s, 1H, OH), 6.73 (s, 2H, Ar–H), 6.60 (d, J = 17.6, 10.8, 1H, –CH=), 5.67 (d, J = 17.6, 1.1, 1H, =CH2), 5.08 (d, J = 10.8, 1.1, 1H, =CH2), 3.77 (s, 6H, –OCH3); δC (74.89 MHz, DMSO-d6): C(6)), 147.97 (C(3), C(5)), 136.94 (C–OH), 135.73 (=CH), 127.66 (C(1)), 111.34 (=CH2), 103.77 (C(2), C(6)), 55.94 (2× OCH3) |
| Ethyl sinapate (SE) | δH (297.80 MHz, DMSO-d6): 8.94 (s, 1H, OH), 7.55 (d, J = 15.9, 1H, –CH=), 7.03 (s, 2H, Ar–H), 6.53 (d, J = 15.9, 1H, =CH–), 4.16 (q, J = 7.1, 2H, –OCH2), 3.80 (s, 6H, –OCH3), 1.25 (t, J = 7.1, 3H, CH3); δC (74.89 MHz, DMSO-d6): 166.55 (C=O), 147.98 (C(3), C(5)), 145.19 (C(β)), 138.23 (C–OH), 124.36 (C(1)), 114.96 (C(α)), 106.19 (C(2), C(6)), 59.66 (CH2), 56.06 (2× OCH3), 14.23 (CH3) |
| Propyl sinapate (SP) | δH (297.80 MHz, DMSO-d6): 8.93 (s, 1H, OH), 7.55 (d, J = 15.9, 1H, –CH=), 7.03 (s, 2H, Ar–H), 6.53 (d, J = 15.9, 1H, =CH–), 4.08 (t, J = 6.6, 2H, –OCH2), 3.80 (s, 6H, –OCH3), 1.65 (t, J = 7.4, 6.6, 2H, CH2), 0.93 (t, J = 7.4, 3H, CH3); δC (74.89 MHz, DMSO-d6): 166.65 (C=O), 147.98 (C(3), C(5)), 145.22 (C(β)), 138.23 (C–OH), 124.36 (C(1)), 114.91 (C(α)), 106.20 (C(2), C(6)), 65.18 (CH2), 56.06 (2× OCH3), 21.66 (CH2), 10.33(CH3) |
| Butyl sinapate (SB) | δH (297.80 MHz, DMSO-d6): 8.94 (s, 1H, OH), 7.54 (d, J = 15.9, 1H, –CH=), 7.03 (s, 2H, Ar–H), 6.53 (d, J = 15.9, 1H, =CH–), 4.12 (t, J = 6.6, 2H, –OCH2), 3.80 (s, 6H, –OCH3), 1.61 (m, 2H, CH2), 1.38 (m, 2H, CH2), 0.92 (t, J = 7.3, 3H, CH3); δC (74.89 MHz, DMSO-d6): 166.65 (C=O), 147.98 (C(3), C(5)), 145.21 (C(β)), 138.23 (C–OH), 124.36 (C(1)), 114.92 (C(α)), 106.20 (C(2), C(6)), 63.38 (CH2), 56.06 (2× OCH3), 30.33 (CH2), 18.67 (CH2), 13.57 (CH3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nićiforović, N.; Polak, T.; Makuc, D.; Poklar Ulrih, N.; Abramovič, H. A Kinetic Approach in the Evaluation of Radical-Scavenging Efficiency of Sinapic Acid and Its Derivatives. Molecules 2017, 22, 375. https://doi.org/10.3390/molecules22030375
Nićiforović N, Polak T, Makuc D, Poklar Ulrih N, Abramovič H. A Kinetic Approach in the Evaluation of Radical-Scavenging Efficiency of Sinapic Acid and Its Derivatives. Molecules. 2017; 22(3):375. https://doi.org/10.3390/molecules22030375
Chicago/Turabian StyleNićiforović, Neda, Tomaž Polak, Damjan Makuc, Nataša Poklar Ulrih, and Helena Abramovič. 2017. "A Kinetic Approach in the Evaluation of Radical-Scavenging Efficiency of Sinapic Acid and Its Derivatives" Molecules 22, no. 3: 375. https://doi.org/10.3390/molecules22030375