Connectivity and Topology Invariance in Self-Assembled and Halogen-Bonded Anionic (6,3)-Networks
Abstract
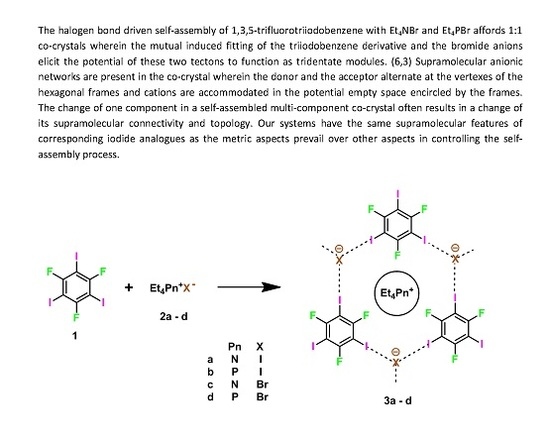
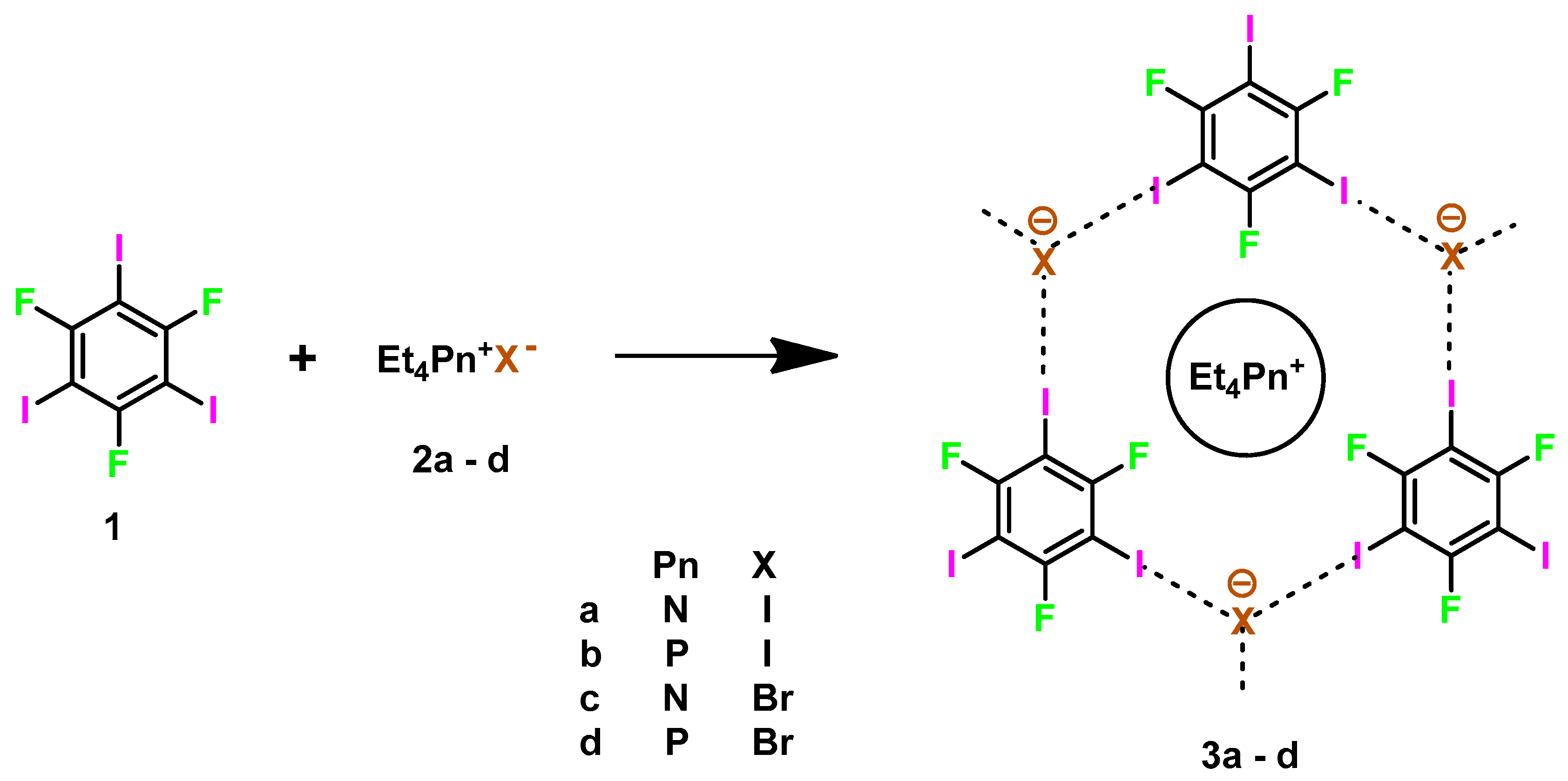
:1. Introduction
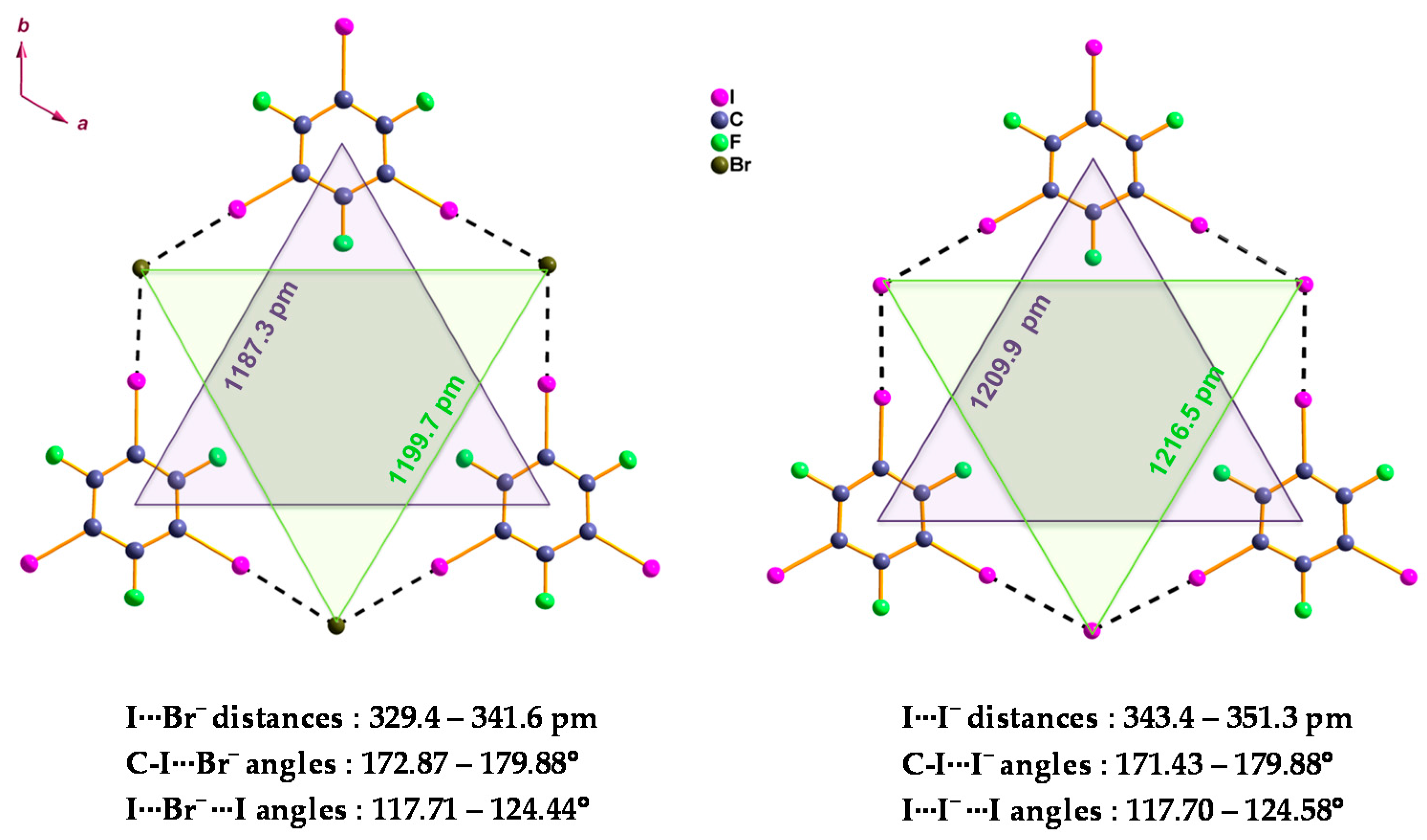
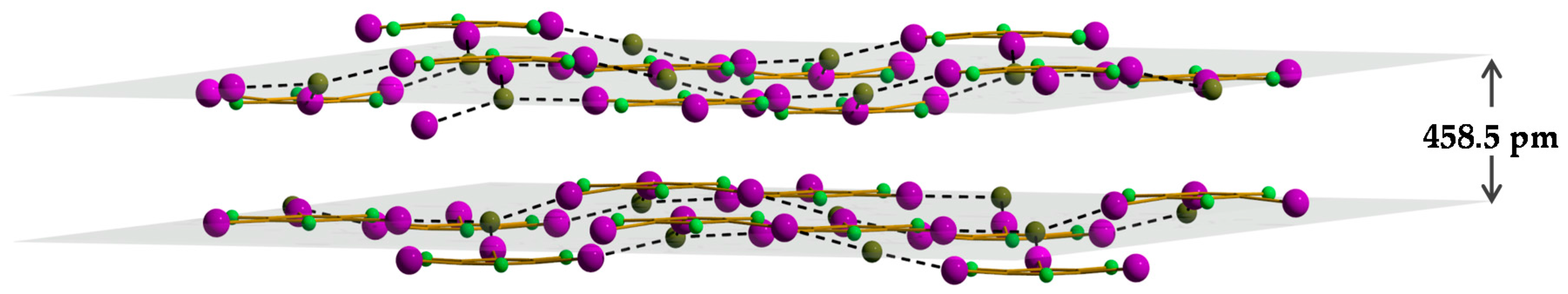
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Procedures and Compound Characterization
3.2.1. Preparation of 1,3,5-Triiodo-2,4,6-trifluorobenzene (1) [36]
3.2.2. Preparation of Co-Crystal 3c
3.2.3. Preparation of Co-Crystal 3d
3.3. Single Crystal Structure Determination
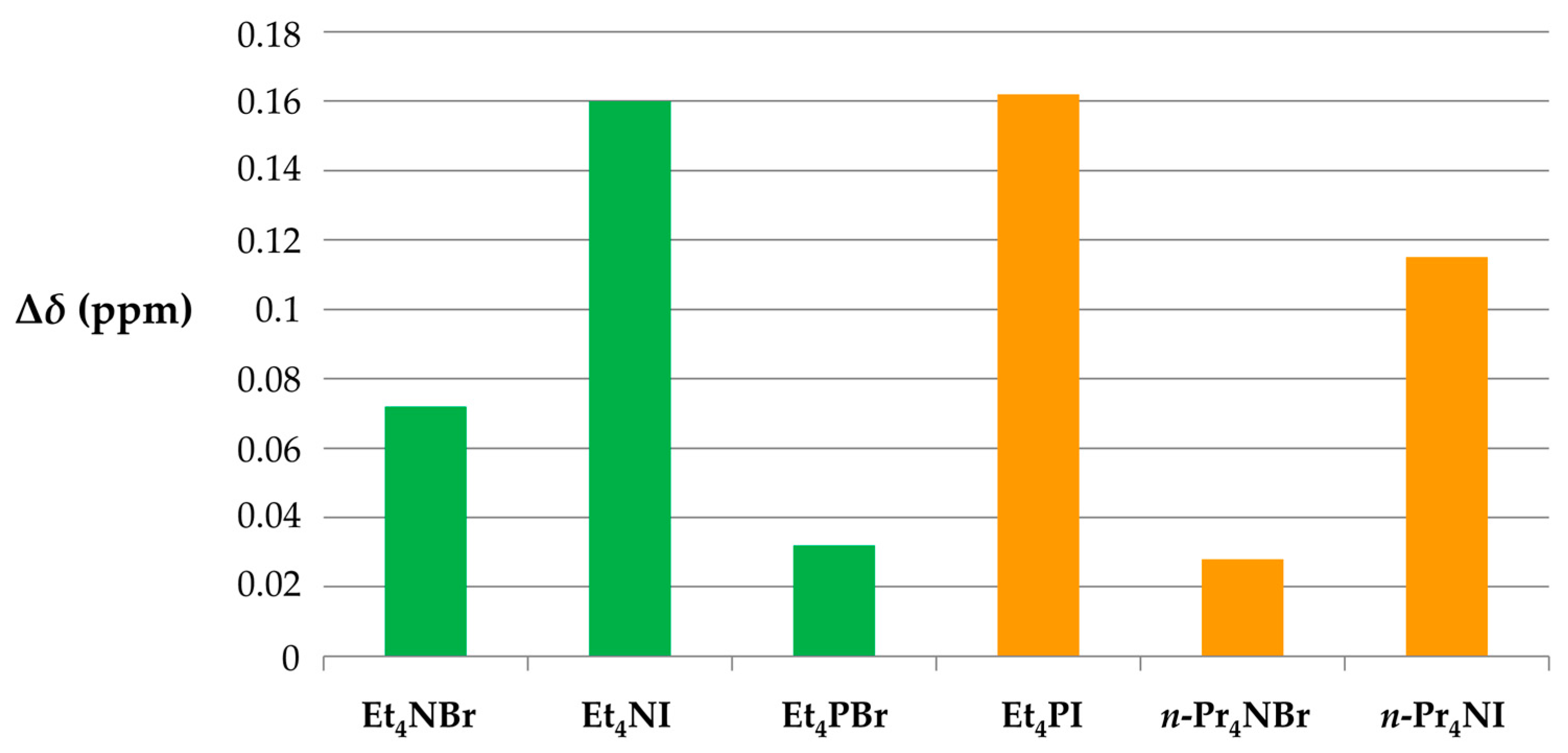
3.4. 19F NMR Experiments
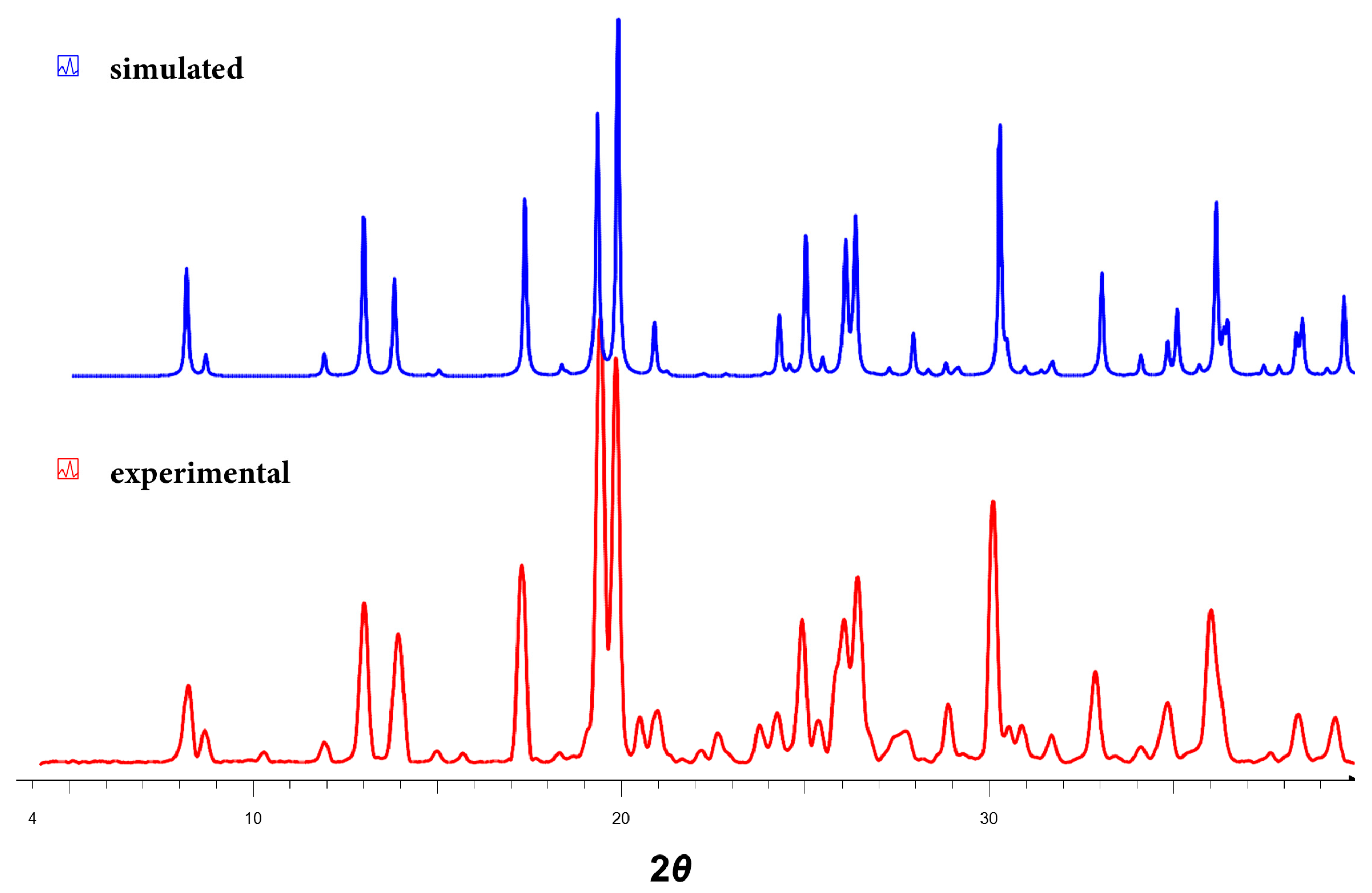
3.5. Powder X-ray Diffraction Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Desiraju, G.R. Crystal engineering: From molecule to crystal. J. Am. Chem. Soc. 2013, 135, 9952–9967. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. Halogen bonding. Chem. Rev. 2016, 115, 2478–2601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, P.S. Biomolecular halogen bonds. Top. Curr. Chem. 2014, 358, 241–276. [Google Scholar]
- Parisini, E.; Metrangolo, P.; Pilati, T.; Resnati, G.; Terraneo, G. Halogen bonding in halocarbon-protein complexes: A structural survey. Chem. Soc. Rev. 2011, 40, 2267–2278. [Google Scholar] [CrossRef] [PubMed]
- Hardegger, L.A.; Kuhn, B.; Spinnler, B.; Anselm, L.; Ecabert, R.; Stihle, M.; Gsell, B.; Thoma, R.; Diez, J.; Benz, J.; et al. Systematic investigation of halogen bonding in protein-ligand interactions. Angew. Chem. Int. Ed. 2011, 50, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Zhang, L.; Cui, D.; Yao, Z.; Gao, B.; Lin, J.; Wei, D. The Important Role of Halogen Bond in Substrate Selectivity of Enzymatic Catalysis. Sci. Rep. 2016, 6, 34750. [Google Scholar] [CrossRef] [PubMed]
- Bulfield, D.; Huber, S.M. Halogen Bonding in Organic Synthesis and Organocatalysis. Chem. Eur. J. 2016, 22, 14434–14450. [Google Scholar] [CrossRef] [PubMed]
- Gazzera, L.; Milani, R.; Pirrie, L.; Schmutz, M.; Blanck, C.; Resnati, G.; Metrangolo, P.; Krafft, M.P. Design of highly stable echogenic microbubbles through controlled assembly of their hydrophobin shell. Angew. Chem. Int. Ed. 2016, 55, 10263–10267. [Google Scholar] [CrossRef] [PubMed]
- Bolton, O.; Lee, K.; Kim, H.-J.; Lin, K.Y.; Kim, J. Activating efficient phosphorescence from purely organic materials by crystal design. Nat. Chem. 2011, 3, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.; Soubhye, J.; Meyer, F. Halogen bonding in polymer science: From crystal engineering to functional supramolecular polymers and materials. Polym. Chem. 2015, 6, 3559–3580. [Google Scholar] [CrossRef]
- Saccone, M.; Cavallo, G.; Metrangolo, P.; Pace, A.; Pibiri, I.; Pilati, T.; Resnati, G.; Terraneo, G. Halogen bond directionality translates tecton geometry into self-assembled architecture geometry. CrystEngComm 2013, 15, 3102–3105. [Google Scholar] [CrossRef] [Green Version]
- De Santis, A.; Forni, A.; Liantonio, R.; Metrangolo, P.; Pilati, T.; Resnati, G. N···Br halogen bonding: 1D infinite chains through the self-assembly of dibromotetrafluorobenzenes with dipyridyl derivatives. Chem. Eur. J. 2003, 9, 3974–3983. [Google Scholar] [CrossRef] [PubMed]
- Cinĉić, D.; Friŝĉić, T.; Jones, W. Experimental and database studies of three-centered halogen bonds with bifurcated acceptors present in molecular crystals, cocrystals and salts. CrystEngComm 2011, 13, 3224–3231. [Google Scholar] [CrossRef]
- Liantonio, R.; Metrangolo, P.; Pilati, T.; Resnati, G. 4,4′-bipyridine 1,2-diiodo-3,4,5,6-tetrafluorobenzene. Acta Cryst. 2002, E58, o575–o577. [Google Scholar] [CrossRef]
- Metrangolo, P.; Meyer, F.; Pilati, T.; Resnati, G.; Terraneo, G. 4,4′-bipyridine-2,4,5,6-tetrafluoro-1,3-diiodobenzene (1/1). Acta Cryst. 2007, E63, o4243. [Google Scholar] [CrossRef]
- Cinĉić, D.; Friŝĉić, T.; Jones, W. Isostructural materials achieved by using structurally equivalent donors and acceptors in halogen-bonded cocrystals. Chem. Eur. J. 2008, 14, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.B.; Padgett, C.W.; Metrangolo, P.; Resnati, G.; Hanks, T.W.; Pennington, W.T. Crystal engineering through halogen bonding: Complexes of nitrogen heterocycles with organic iodides. Cryst. Growth Des. 2001, 1, 165–175. [Google Scholar] [CrossRef]
- Cavallo, G.; Biella, S.; Lü, J.; Metrangolo, P.; Pilati, T.; Resnati, G.; Terraneo, G. Halide anion-templated assembly of di- and triiodoperfluorobenzenes into 2D and 3D supramolecular networks. J. Fluorine Chem. 2010, 131, 1165–1172. [Google Scholar] [CrossRef]
- Roper, L.C.; Präsang, C.; Kozhevnikov, V.N.; Whitwood, A.C.; Karadakov, P.B.; Bruce, D.W. Experimental and theoretical study of halogen-bonded complexes of DMAP with di- and triiodofluorobenzenes. A complex with a very short N∙∙∙I halogen bond. Cryst. Growth Des. 2010, 10, 3710–3720. [Google Scholar] [CrossRef]
- Lucassen, A.C.B.; Karton, A.; Leitus, G.; Shimon, L.J.W.; Martin, J.M.L.; van der Boom, M.E. Co-crystallization of sym-triiodo-trifluorobenzene with bipyridyl donors: Consistent formation of two instead of anticipated three N∙∙∙I halogen bonds. Cryst. Growth Des. 2007, 7, 386–392. [Google Scholar] [CrossRef]
- Metrangolo, P.; Meyer, F.; Pilati, T.; Resnati, G.; Terraneo, G. Mutual induced coordination in halogen-bonded anionic assemblies with (6,3) cation-templated topologies. Chem. Commun. 2008, 1635–1637. [Google Scholar] [CrossRef] [PubMed]
- Cauliez, P.; Polo, V.; Roisnel, T.; Llusar, R.; Fourmigué, M. The thiocyanate anion as a polydentate halogen bond acceptor. CrystEngComm 2010, 12, 558–566. [Google Scholar] [CrossRef] [Green Version]
- Jeon, I.-R.; Mathonière, C.; Clérac, R.; Rouzières, M.; Jeannin, O.; Trzop, E.; Collet, E.; Fourmigué, M. Photoinduced reversible spin-state switching of an Fe(III) complex assisted by a halogen-bonded supramolecular network. Chem. Commun. 2017, 53, 10283–10286. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.; Roeben, K.; Soubhye, J.; Wintjens, R.; Meyer, F. Halogen bonding in multi-connected 1,2,2-triiodo-alkene involving geminal and/or vicinal iodines: A crystallographic and DFT study. CrystEngComm 2016, 18, 683–690. [Google Scholar] [CrossRef]
- Pfrunder, M.C.; Micallef, A.S.; Rintoul, L.; Arnold, D.P.; Davy, K.J.P.; McMurtrie, J. Exploitation of the Menshutkin reaction for the controlled assembly of halogen bonded architectures incorporating 1,2-diiodotetrafluorobenzene and 1,3,5-triiodotrifluorobenzene. Cryst. Growth Des. 2012, 12, 714–724. [Google Scholar] [CrossRef]
- Jeon, I.-R.; Jeannin, O.; Clerac, R.; Rouzieres, M.; Fourmigue, M. Spin-state modulation of molecular Fe(III) complexes via inclusion in halogen-bonded supramolecular networks. Chem. Commun. 2017, 53, 4989–4992. [Google Scholar] [CrossRef] [PubMed]
- Aakeröy, C.B.; Wijethunga, T.K.; Desper, J. Practical crystal engineering using halogen bonding: A hierarchy based on calculated molecular electrostatic potential surfaces. J. Mol. Struct. 2014, 1072, 20–27. [Google Scholar] [CrossRef]
- Raffo, P.A.; Cukiernik, F.D.; Baggio, R.F. The three-component cocrystal 1,3,5-trifluoro-2,4,6-triiodobenzene-pyridine N-oxide-water (1/2/1) built up by halogen bonds, hydrogen bonds and π-π interactions. Acta Cryst. 2015, C71, 84–88. [Google Scholar]
- Aakeröy, C.B.; Wijethunga, T.K.; Desper, J. Constructing molecular polygons using halogen bonding and bifurcated N-oxides. CrystEngComm 2014, 16, 28–31. [Google Scholar] [CrossRef]
- Triguero, S.; Llusar, R.; Polo, V.; Fourmigué, M. Halogen bonding interactions of sym-triiodotrifluorobenzene with halide anions: A combined structural and theoretical study. Cryst. Growth Des. 2008, 8, 2241–2247. [Google Scholar] [CrossRef]
- Kumar, V.; Pilati, T.; Terraneo, G.; Meyer, F.; Metrangolo, P.; Resnati, G. Halogen bonded Borromean networks by design: Topology invariance and metric tuning in a library of multi component systems. Chem. Sci. 2017, 8, 1801–1810. [Google Scholar] [CrossRef] [PubMed]
- Cardillo, P.; Corradi, E.; Lunghi, A.; Meille, S.V.; Messina, M.T.; Metrangolo, P.; Resnati, G. The N···I intermolecular interaction as a general protocol in the formation of perfluorocarbon-hydrocarbon supramolecular architectures. Tetrahedron 2000, 56, 5535–5550. [Google Scholar] [CrossRef]
- The normalized contact Nc is defined as the ratio DXY/(rX + rY), where DXY is the experimental distance between the halogen bonded iodine atoms X and halide anions Y and rX and rY are the van der Waals radius for iodine and the Pauling ionic radius of the halide anion Y, respectively. Nc is a useful indicator of the relative interaction strength, more useful than the XB distance itself, because it allows distances between different interacting sites to be meaningfully compared.
- The interlayer distance is calculated as the distance between the mean square planes passing through the anions of two adjacent layers.
- Liantonio, R.; Metrangolo, P.; Pilati, T.; Resnati, G. Fluorous interpenetrated layers in a three-component crystal matrix. Cryst. Growth Des. 2003, 3, 355–361. [Google Scholar] [CrossRef]
- Wenk, H.H.; Sander, W. Generation of fluorinated m-benzyne derivatives in neon matrices. Eur. J. Org. Chem. 2002, 3927–3935. [Google Scholar] [CrossRef]
- Bruker. SMART, SAINT and SADABS; Bruker AXS Inc.: Madison, WI, USA, 1999. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- White, N.G.; MacLachlan, M.J. Anion-templated hexagonal nanotubes. Chem. Sci. 2015, 6, 6245–6249. [Google Scholar] [CrossRef]
- Mercurio, J.M.; Knighton, R.C.; Cookson, J.; Beer, P.D. Halotriazolium axle functionalized [2]rotaxanes for anion recognition: Investigating the effects of halogen-bond donor and preorganization. Chem. Eur. J. 2014, 20, 11740–11749. [Google Scholar] [CrossRef] [PubMed]
- Langton, M.J.; Robinson, S.W.; Marques, I.; Félix, V.; Beer, P.D. Halogen bonding in water results in enhanced anion recognition in acyclic and rotaxane hosts. Nat. Chem. 2014, 6, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.D.; Smith, J.M.; Hanks, T.W.; Pennington, W.T. Computational and Crystallographic Studies of Pseudo-Polyhalides. Cryst. Growth Des. 2012, 12, 2759–2768. [Google Scholar] [CrossRef]
- Ding, X.H.; Ou, C.J.; Wang, S.; Xie, L.H.; Lin, J.Y.; Wang, J.P.; Huang, W. Co-crystallization of 1,3,5-trifluoro-2,4,6-triiodobenzene (1,3,5-TFTIB) with a variety of Lewis bases through halogen-bonding interactions. CrystEngComm 2017, 19, 5504–5521. [Google Scholar] [CrossRef]
- Metrangolo, P.; Pilati, T.; Terraneo, G.; Biella, S.; Resnati, G. Anion coordination and anion-templated assembly under halogen bonding control. CrystEngComm 2009, 11, 1187–1196. [Google Scholar] [CrossRef]
- Lindeman, S.V.; Hecht, J.; Kochi, J.K. The Charge-transfer motif in crystal engineering. Self-assembly of acentric (diamondoid) networks from halide salts and carbon tetrabromide as electron-donor/acceptor synthons. J. Am. Chem. Soc. 2003, 125, 11597–11606. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyer, F.; Pilati, T.; Konidaris, K.F.; Metrangolo, P.; Resnati, G. Connectivity and Topology Invariance in Self-Assembled and Halogen-Bonded Anionic (6,3)-Networks. Molecules 2017, 22, 2060. https://doi.org/10.3390/molecules22122060
Meyer F, Pilati T, Konidaris KF, Metrangolo P, Resnati G. Connectivity and Topology Invariance in Self-Assembled and Halogen-Bonded Anionic (6,3)-Networks. Molecules. 2017; 22(12):2060. https://doi.org/10.3390/molecules22122060
Chicago/Turabian StyleMeyer, Franck, Tullio Pilati, Konstantis F. Konidaris, Pierangelo Metrangolo, and Giuseppe Resnati. 2017. "Connectivity and Topology Invariance in Self-Assembled and Halogen-Bonded Anionic (6,3)-Networks" Molecules 22, no. 12: 2060. https://doi.org/10.3390/molecules22122060