Identification, In Vitro Testing and Molecular Docking Studies of Microginins’ Mechanism of Angiotensin-Converting Enzyme Inhibition
Abstract
:1. Introduction
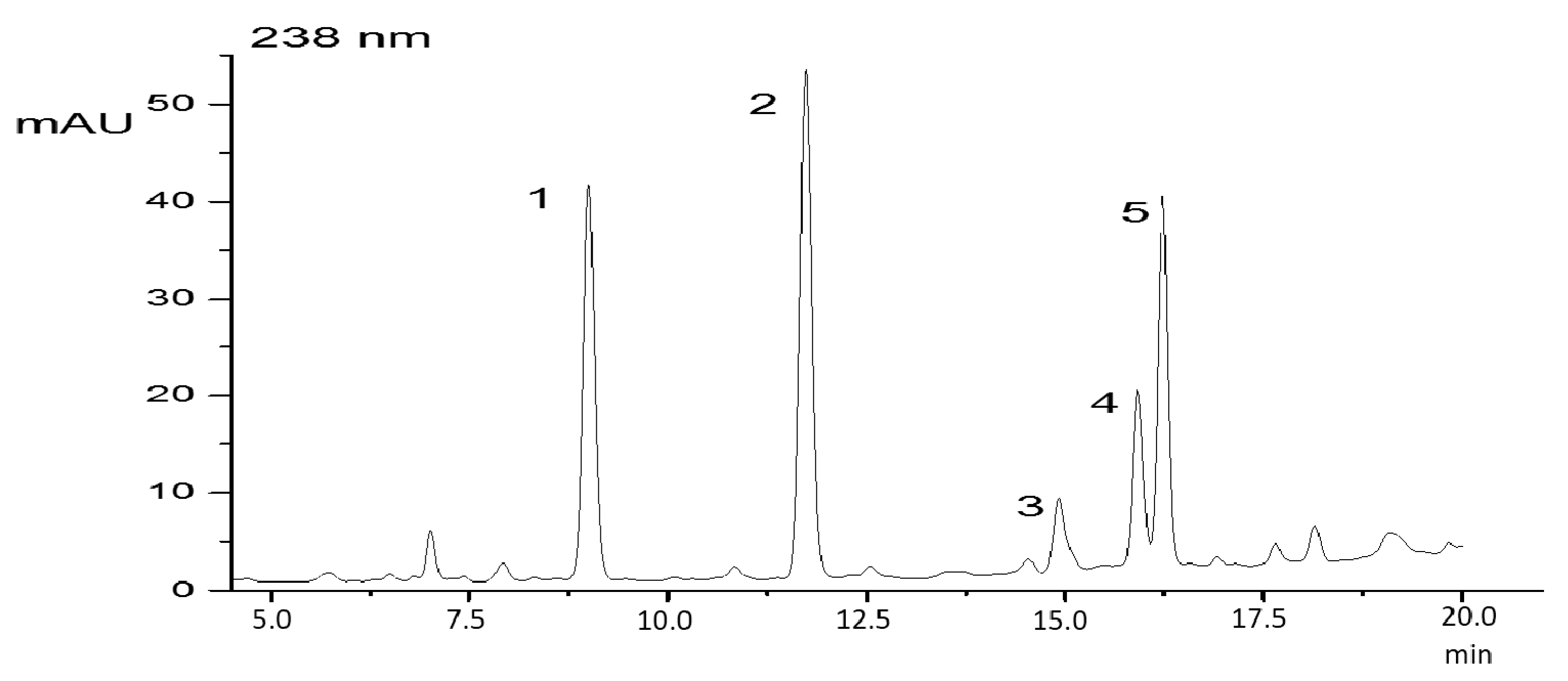
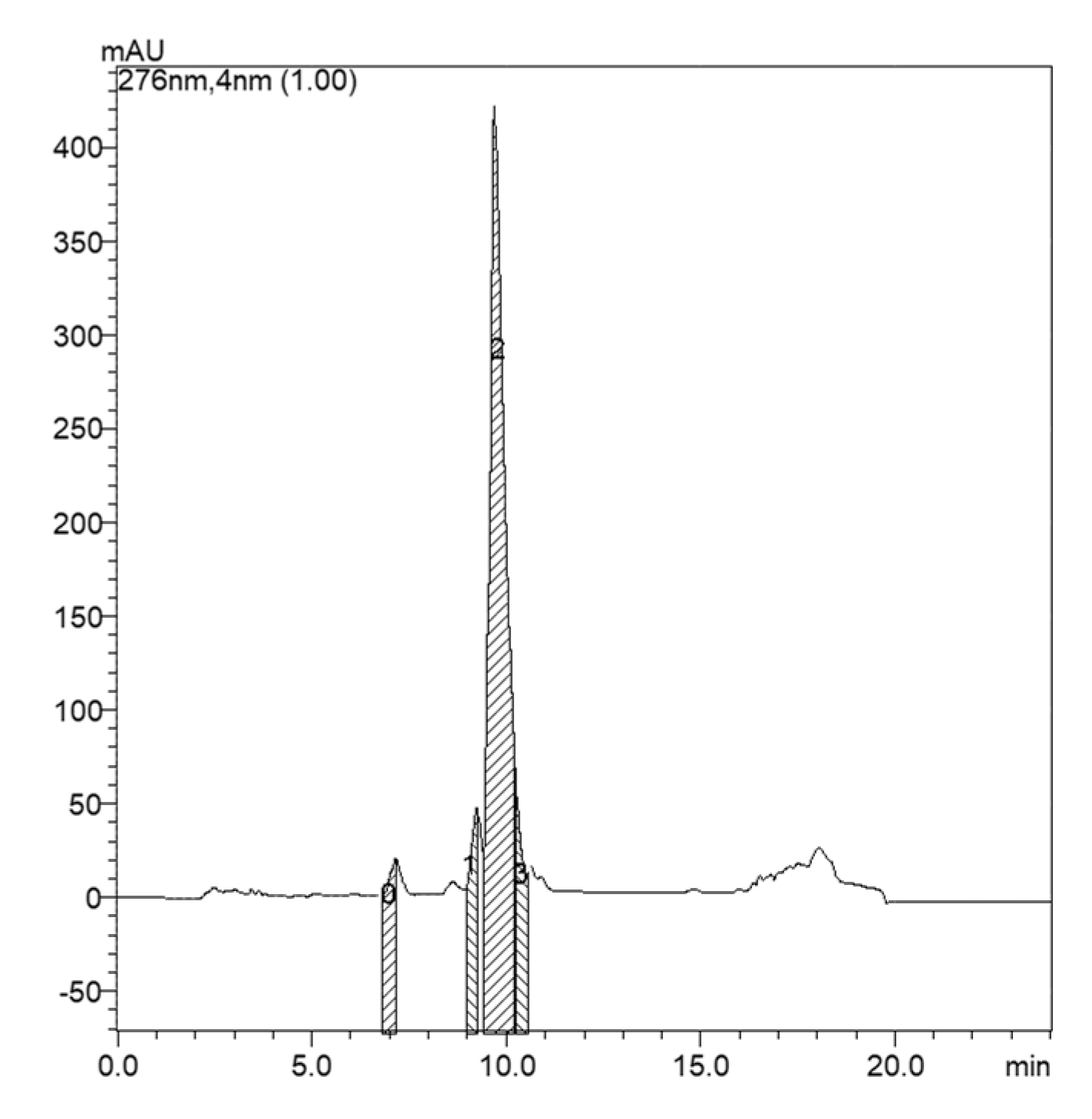
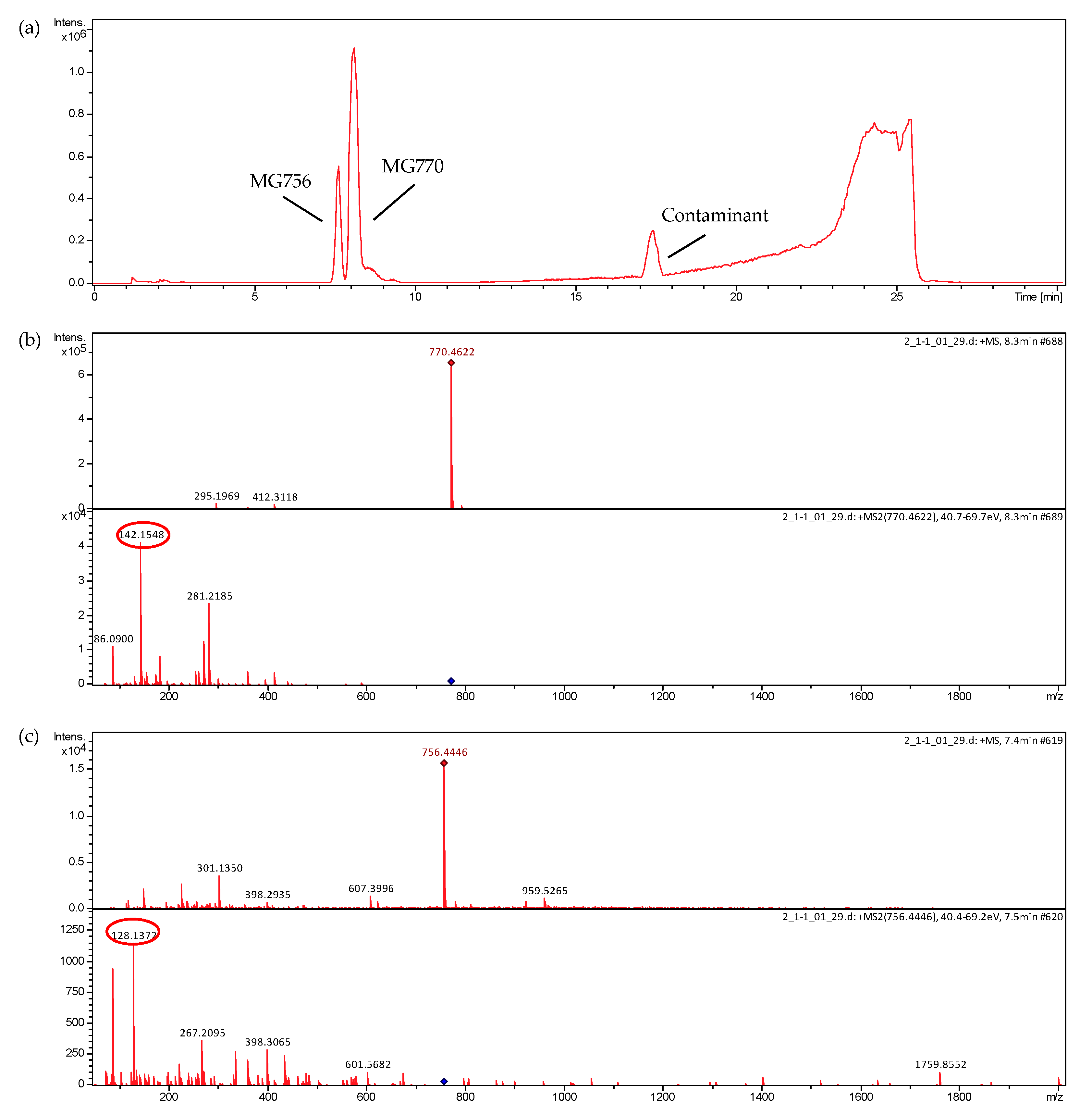
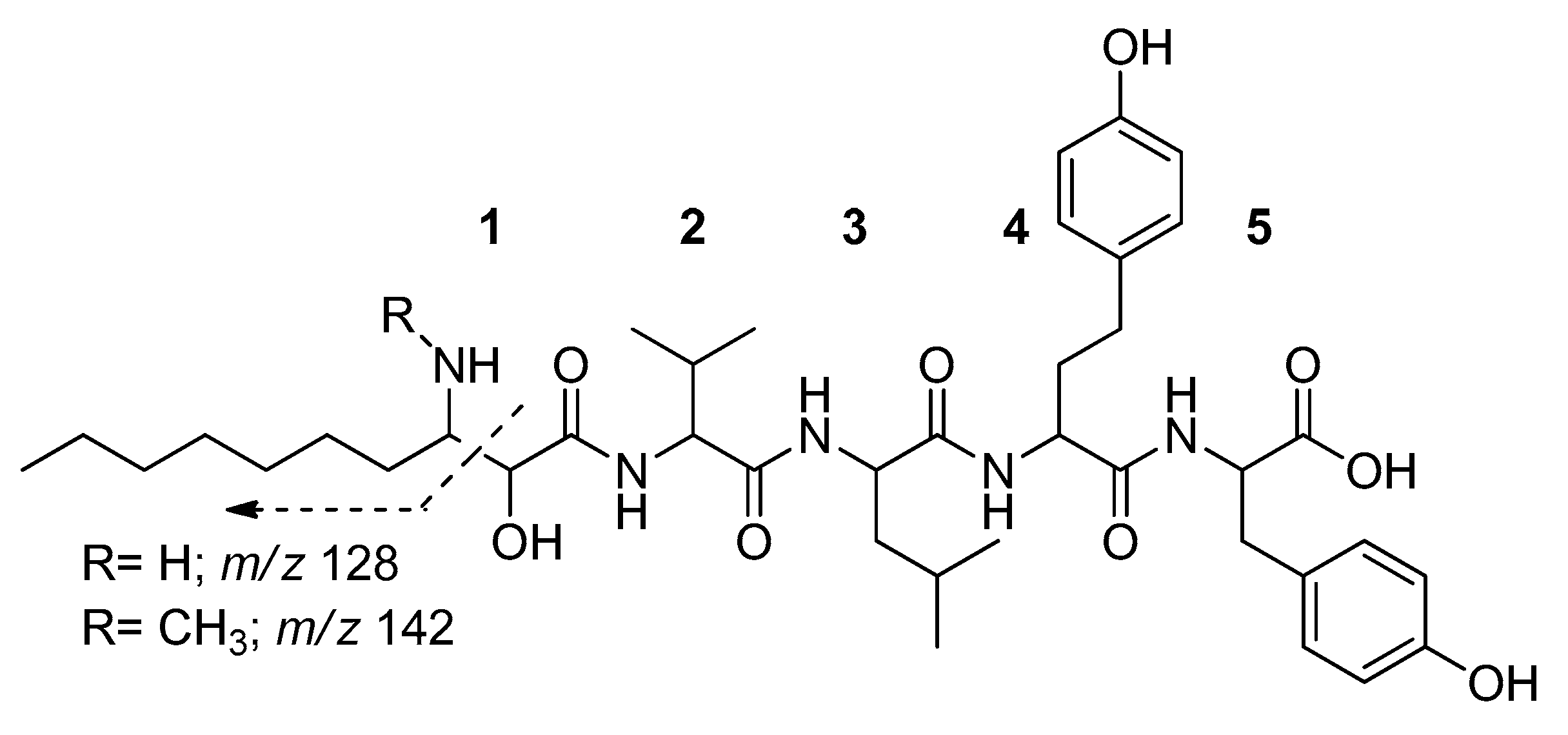
2. Results
3. Discussion
4. Materials and Methods
4.1. Strain Maintenance
4.2. Cyanopeptide Identification
4.3. Isolation and Purification of Microginins
4.4. Indirect Spectrometric Quantification of Microginins with Tyrosine
4.5. LC-MS Analyses
4.6. Enzyme Assay
4.7. Molecular Docking
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tan, L.T. Bioactive natural products from marine cyanobacteria for drug discovery. Phytochemistry 2007, 68, 954–979. [Google Scholar] [CrossRef] [PubMed]
- Neilan, B.A.; Dittmann, E.; Rouhiainen, L.; Bass, R.A.; Schaub, V.; Sivonen, K.; Börner, T. Nonribosomal peptide synthesis and toxigenicity of cyanobacteria. J. Bacteriol. 1999, 181, 4089–4097. [Google Scholar] [PubMed]
- Singh, S.; Kate, B.N.; Banerjee, U. Bioactive compounds from cyanobacteria and microalgae: An overview. Crit. Rev. Biotechnol. 2005, 25, 73–95. [Google Scholar] [CrossRef] [PubMed]
- Gademann, K.; Portmann, C. Secondary metabolites from cyanobacteria: Complex structures and powerful bioactivities. Curr. Org. Chem. 2008, 12, 326–341. [Google Scholar] [CrossRef]
- Namikoshi, M.; Rinehart, K. Bioactive compounds produced by cyanobacteria. J. Ind. Microbiol. 1996, 17, 373–384. [Google Scholar] [CrossRef]
- Stenico, M.E.; Lorenzi, A.S.; Silva, C.S.; Rigonato, J.; Fiore, M.F. Inibidores de proteases produzidos por cianobactérias: Uma revisão. Oecol. Aust. 2017, 16, 183–209. [Google Scholar] [CrossRef]
- Ishida, K.; Kato, T.; Murakami, M.; Watanabe, M.; Watanabe, M.F. Microginins, zinc metalloproteases inhibitors from the cyanobacterium microcystis aeruginosa. Tetrahedron 2000, 56, 8643–8656. [Google Scholar] [CrossRef]
- Okino, T.; Matsuda, H.; Murakami, M.; Yamaguchi, K. Microginin, an angiotensin-converting enzyme inhibitor from the blue-green alga microcystis aeruginosa. Tetrahedron Lett. 1993, 34, 501–504. [Google Scholar] [CrossRef]
- Carneiro, R.L.; Dörr, F.A.; Dörr, F.; Bortoli, S.; Delherbe, N.; Vásquez, M.; Pinto, E. Co-occurrence of microcystin and microginin congeners in brazilian strains of microcystis sp. FEMS Microbiol. Ecol. 2012, 82, 692–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Welker, M.; Maršálek, B.; Šejnohová, L.; Von Doehren, H. Detection and identification of oligopeptides in microcystis (cyanobacteria) colonies: Toward an understanding of metabolic diversity. Peptides 2006, 27, 2090–2103. [Google Scholar] [CrossRef] [PubMed]
- Chorus, I.; Niesel, V. Toxic and Bioactive Peptides in Cyanobacteria; PEPCY Final Report; EU Contract Number Contract Number: QLK4-CT-2002–02634; PEPCY: Berlin, Germany, 2007. [Google Scholar]
- Neumann, U.; Forchert, A.; Flury, T.; Weckesser, J. Microginin fr1, a linear peptide from a water bloom of microcystis species. FEMS Microbiol. Lett. 1997, 153, 475–478. [Google Scholar] [CrossRef]
- Ishida, K.; Matsuda, H.; Murakami, M.; Yamaguchi, K. Microginins 299-a and-b, leucine aminopeptidase inhibitors from the cyanobacterium microcystis aeruginosa (nies-299). Tetrahedron 1997, 53, 10281–10288. [Google Scholar] [CrossRef]
- Wang, W.; Wang, N.; Zhang, Y.; Cai, Z.; Chen, Q.; He, G. A convenient rp-hplc method for assay bioactivities of angiotensin i-converting enzyme inhibitory peptides. ISRN Biotechnol. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Kodani, S.; Suzuki, S.; Ishida, K.; Murakami, M. Five new cyanobacterial peptides from water bloom materials of lake teganuma (Japan). FEMS Microbiol. Lett. 1999, 178, 343–348. [Google Scholar] [CrossRef]
- Sano, T.; Takagi, H.; Morrison, L.F.; Metcalf, J.S.; Codd, G.A.; Kaya, K. Leucine aminopeptidase m inhibitors, cyanostatin a and b, isolated from cyanobacterial water blooms in scotland. Phytochemistry 2005, 66, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.C.; Abdullah, N.; Shuib, A.S.; Aminudin, N. Novel angiotensin i-converting enzyme inhibitory peptides derived from edible mushroom Agaricus bisporus (je lange) imbach identified by lc–ms/ms. Food Chem. 2014, 148, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Ishida, K.; Matsuda, H.; Murakami, M. Four new microginins, linear peptides from the cyanobacterium microcystis aeruginosa. Tetrahedron 1998, 54, 13475–13484. [Google Scholar] [CrossRef]
- Goodford, P.J. A computational procedure for determining energetically favorable binding sites on biologically important macromolecules. J. Med. Chem. 1985, 28, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Gorham, P.; McLachlan, J.; Hammer, U.; Kim, W. Isolation and Culture of Toxic Strains of Anabaena Flos-aquae(lyngb.) de Breb; Schweizerbart Science Publisher: Stuttgart, Germany, 1964. [Google Scholar]
- Lawton, L.A.; Edwards, C.; Codd, G.A. Extraction and high-performance liquid chromatographic method for the determination of microcystins in raw and treated waters. Analyst 1994, 119, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.N.; Vajdos, F.; Fee, L.; Grimsley, G.; Gray, T. How to measure and predict the molar absorption coefficient of a protein. Protein Sci. 1995, 4, 2411–2423. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Fractions containing microginins can be available from authors. |
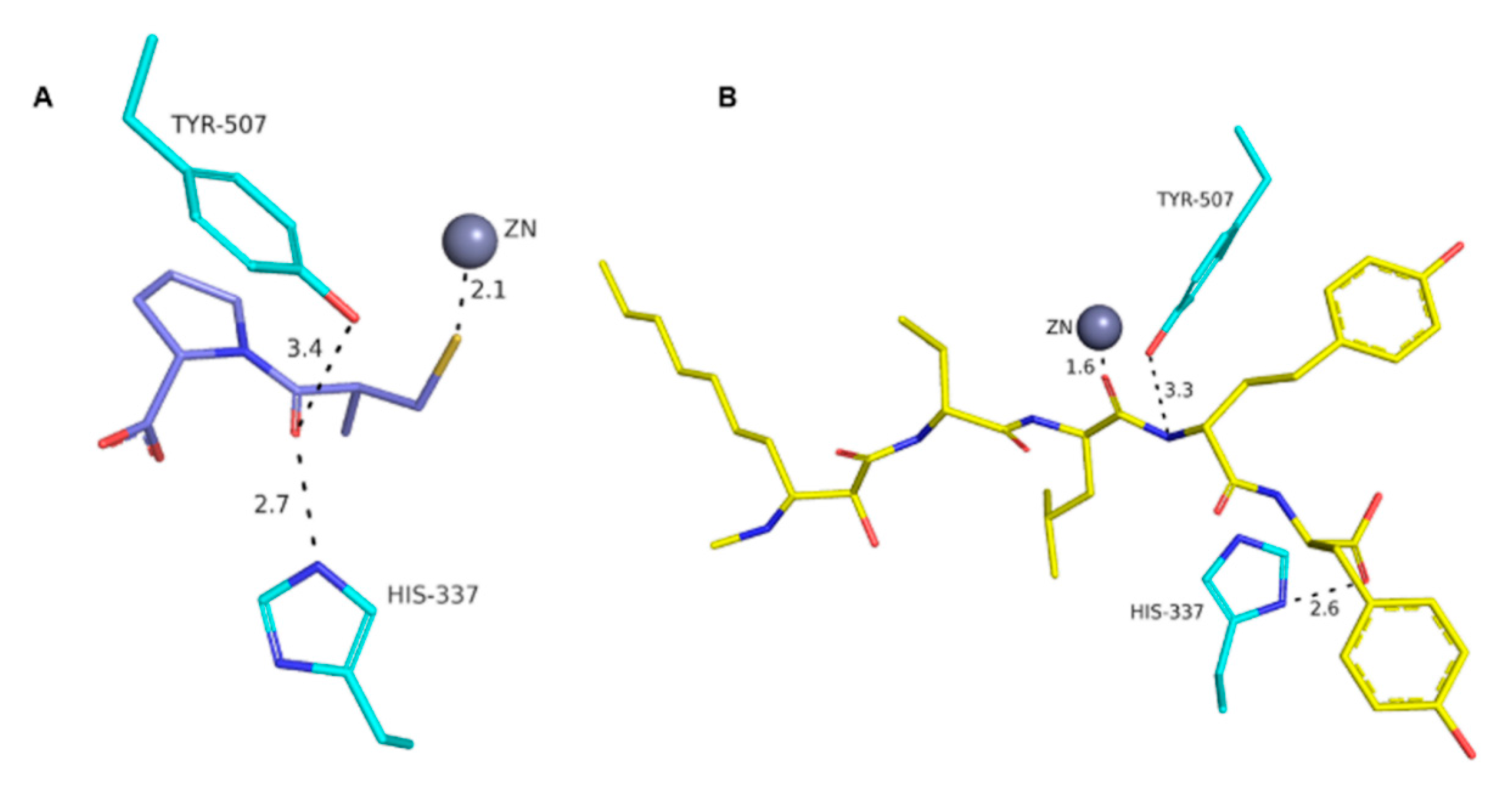
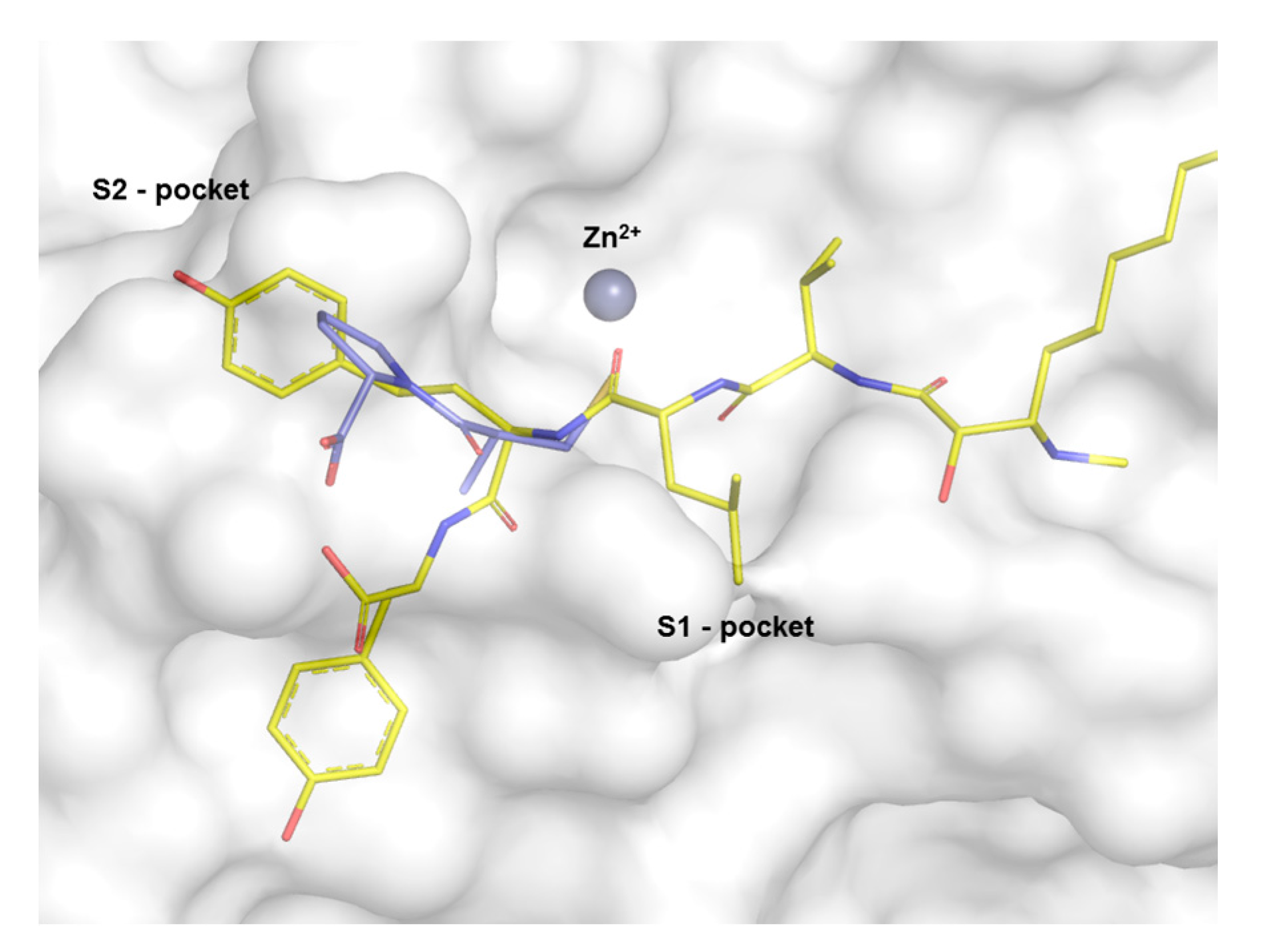
| Peptide | IC50 (µg·mL−1) | Reference |
|---|---|---|
| MG 478 | 10 | [7] |
| MG 51-A | >100 | [7] |
| MG 51-B | >100 | [7] |
| MG 91-A | >100 | [7] |
| MG 91-B | >100 | [7] |
| MG | 7 | [8] |
| MG T1 | 5 | [15] |
| MG T2 | 7 | [15] |
| A-Mg 478 | 700 | [7] |
| MG 770 | 3.33 | Current study |
| Cyanostatins A * | 110 | [16] |
| Cyanostatins B * | 130 | [16] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paiva, F.C.R.; Ferreira, G.M.; Trossini, G.H.G.; Pinto, E. Identification, In Vitro Testing and Molecular Docking Studies of Microginins’ Mechanism of Angiotensin-Converting Enzyme Inhibition. Molecules 2017, 22, 1884. https://doi.org/10.3390/molecules22121884
Paiva FCR, Ferreira GM, Trossini GHG, Pinto E. Identification, In Vitro Testing and Molecular Docking Studies of Microginins’ Mechanism of Angiotensin-Converting Enzyme Inhibition. Molecules. 2017; 22(12):1884. https://doi.org/10.3390/molecules22121884
Chicago/Turabian StylePaiva, Fernanda C. R., Glaucio Monteiro Ferreira, Gustavo H. G. Trossini, and Ernani Pinto. 2017. "Identification, In Vitro Testing and Molecular Docking Studies of Microginins’ Mechanism of Angiotensin-Converting Enzyme Inhibition" Molecules 22, no. 12: 1884. https://doi.org/10.3390/molecules22121884






