Unexpected Synthesis of a Bulky Bis-Pocket A3B-Type Meso-Cyano Porphyrin
Abstract
:1. Introduction
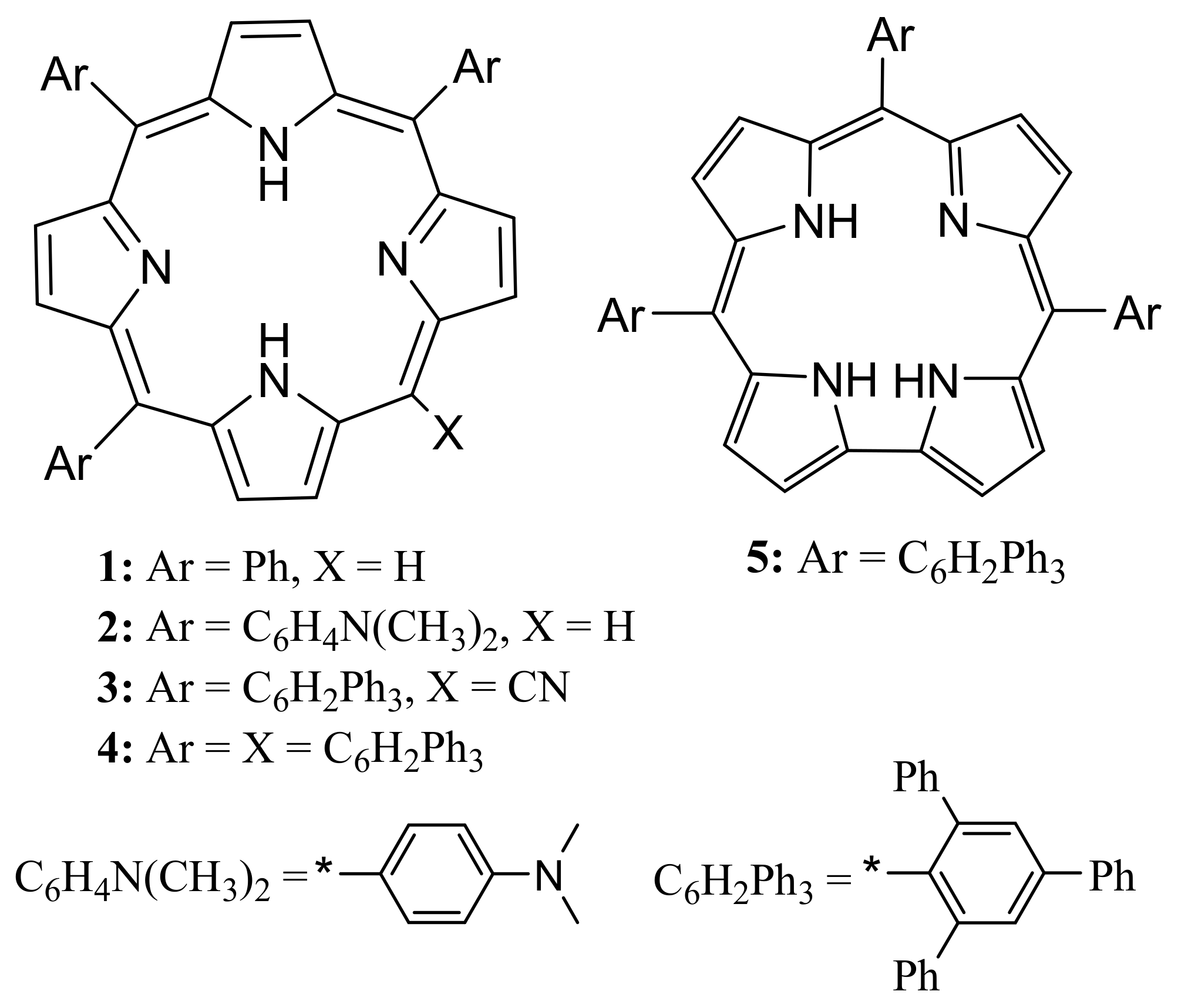
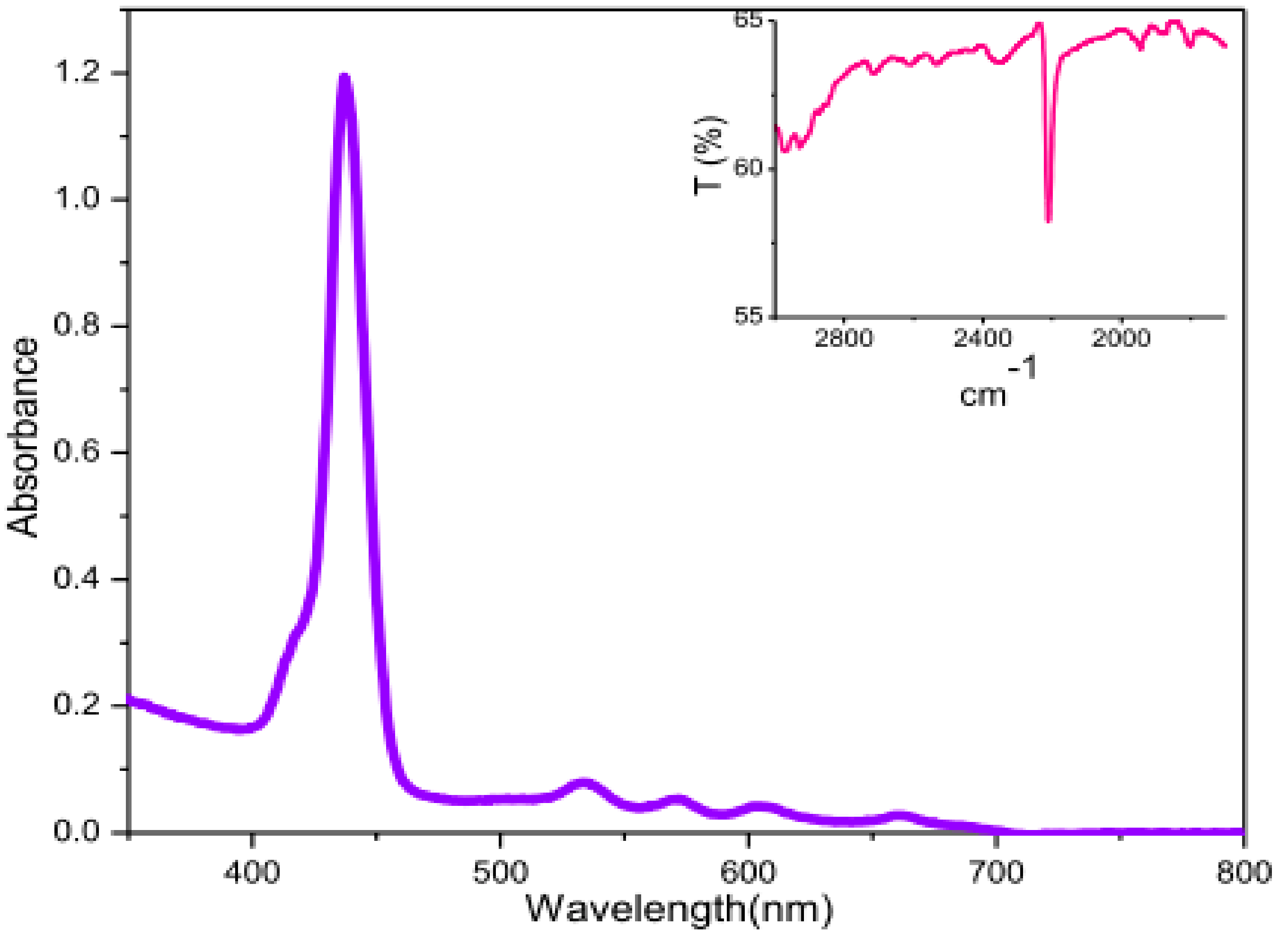
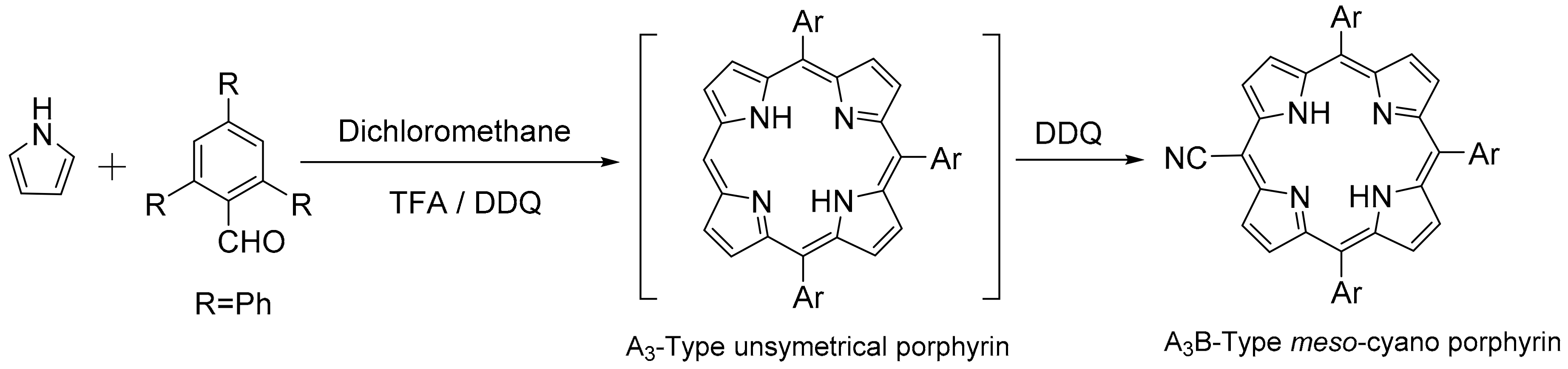
2. Results and Discussion
3. Experimental Section
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kadish, K.M.; Smith, K.M.; Guilard, R. (Eds.) The Porphyrin Handbook; Academic Press: San Diego, CA, USA, 2000; Volumes 1–20. [Google Scholar]
- Smith, K.M. Development of porphyrin syntheses. New J. Chem. 2016, 40, 5644–5649. [Google Scholar] [CrossRef]
- Ghosh, A. A Perspective of One-Pot Pyrrole-Aldehyde Condensations as Versatile Self-Assembly Processes. Angew. Chem. Int. Ed. 2004, 43, 1918–1931. [Google Scholar] [CrossRef] [PubMed]
- Paolesse, R.; Jaquinod, L.; Nurco, D.J.; Mini, S.; Sagone, F.; Boschi, T.; Smith, K.M. 5,10,15-Triphenylcorrole: A Product from a Modified Rothemund Reaction. Chem. Commun. 1999, 14, 1307–1308. [Google Scholar] [CrossRef]
- Gross, Z.; Galili, N.; Saltsman, I. The First Direct Synthesis of Corroles from Pyrrole. Angew. Chem. Int. Ed. 1999, 38, 1427–1429. [Google Scholar] [CrossRef]
- Gryko, D.T. A Simple, Rational Synthesis of Meso-Substituted A2B-Corroles. Chem. Commun. 2000, 22, 2243–2244. [Google Scholar] [CrossRef]
- Orłowski, R.; Gryko, D.; Gryko, D.T. Synthesis of Corroles and Their Heteroanalogs. Chem. Rev. 2017, 117, 3102–3137. [Google Scholar]
- Lindsey, J.S.; Hsu, H.C.; Schreiman, I.C. Synthesis of Tetraphenylporphyrins under Very Mild Conditions. Tetrahedron Lett. 1986, 27, 4969–4970. [Google Scholar] [CrossRef]
- Lindsey, J.S.; Schreiman, I.C.; Hsu, H.C.; Kearney, P.C.; Marguettaz, A.M. Rothemund and Adler-Longo Reactions Revisited: Synthesis of Tetraphenylporphyrins under Equilibrium Conditions. J. Org. Chem. 1987, 52, 827–836. [Google Scholar] [CrossRef]
- Saito, S.; Osuka, A. Expanded Porphyrins: Intriguing Structures, Electronic Properties and Reactivities. Angew. Chem. Int. Ed. 2011, 50, 4342–4373. [Google Scholar] [CrossRef] [PubMed]
- Mandoj, F.; Stefanelli, M.; Nardis, S.; Mastroianni, M.; Fronczek, F.R.; Smith, K.M.; Paolesse, R. 6-Azahemiporphycene: A Further Example of Corrole Metamorphosis. Chem. Commun. 2009, 12, 1580–1582. [Google Scholar] [CrossRef] [PubMed]
- Kral, V.; Kralova, J.; Kaplanek, R.; Briza, T.; Martasek, P. Quo Vadis Porphyrin Chemistry? Physiol. Res. 2006, 55 (Suppl. 2), S3–S26. [Google Scholar] [PubMed]
- Lindsey, J.S. Synthetic Routes to meso-Patterned Porphyrins. Acc. Chem. Res. 2010, 43, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Y.; Huang, J.W.; Tian, X.; Jiao, X.D.; Luo, G.T.; Ji, L.N. Chiral Linear Assembly of Amino Acid Bridged Dimeric Porphyrin Hosts. Chem. Commun. 1997, 16, 1575–1576. [Google Scholar] [CrossRef]
- Chang, C.J.; Deng, Y.; Heyduk, A.F.; Chang, C.K.; Nocera, D.G. Xanthene-Bridged Cofacial Bisporphyrins. Inorg. Chem. 2000, 39, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Senge, M.O.; Feng, X.J. Regioselective Reaction of 5,15-Disubstituted Porphyrins with Organolithium Reagents-Synthetic Access to 5,10,15-Trisubstituted Porphyrins and Directly meso-meso-Linked Bisporphyrins. Chem. Soc. Perkin Trans. 1 2000, 21, 3615–3621. [Google Scholar] [CrossRef]
- Abada, Z.; Ferrie, L.; Akagah, B.; Lormier, A.T.; Figadere, B. Synthesis of 5,15-Diarylporphyrins via Orthoesters Condensation with Aryldipyrromethanes. Tetrahedron Lett. 2011, 52, 3175–3178. [Google Scholar] [CrossRef]
- Gross, C.P.; Barbe, J.M.; Espinosa, E.; Guilard, R. Room-Temperature Autoconversion of Free-Base Corrole into Free-Base Porphyrin. Angew. Chem. Int. Ed. 2006, 45, 5642–5645. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, M.H.R.; Liu, H.Y.; Wang, H.H.; Jiang, Y.Y.; Chang, C.K. Unexpected One-Pot Synthesis of A3-Type Unsymmetrical Porphyrin. Tetrahedron Lett. 2013, 54, 5853–5856. [Google Scholar] [CrossRef]
- Liu, H.Y.; Mahmood, M.H.R.; Qiu, S.X.; Chang, C.K. Recent Developments in Manganese Corrole Chemistry. Coord. Chem. Rev. 2013, 257, 1306–1333. [Google Scholar] [CrossRef]
- Liu, H.Y.; Lai, T.S.; Yeung, L.L.; Chang, C.K. First Synthesis of Perfluorinated Corrole and Its Mn=O Complex. Org. Lett. 2003, 5, 617–620. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.K.; Yeh, C.Y.; Lai, T.S. Synthesis of Sterically Encumbered Porphyrins as Catalysts for Shape-Selective Oxidations. Macromol. Symp. 2000, 156, 117–124. [Google Scholar] [CrossRef]
- Meunier, B. Metalloporphyrin-Catalyzed Oxygenation of Hydrocarbons. Bull. Soc. Chim. Fr. 1986, 2, 578–594. [Google Scholar]
- Suslick, K.S.; Fox, M.M. A Bis-Pocket Porphyrin. J. Am. Chem. Soc. 1983, 105, 3507–3510. [Google Scholar] [CrossRef]
- Liu, H.Y.; Yam, F.; Xie, Y.T.; Li, X.Y.; Chang, C.K. A Bulky Bis-Pocket Manganese (V)-Oxo Corrole Complex: Observation of Oxygen Atom Transfer between Triply Bonded MnV≡O and Alkene. J. Am. Chem. Soc. 2009, 131, 12890–12891. [Google Scholar] [CrossRef] [PubMed]
- Suslick, K.S.; Cook, B.; Fox, M. Shape-Selective Alkane Hydroxylation. J. Chem. Soc. Chem. Commun. 1985, 9, 580–582. [Google Scholar] [CrossRef]
- Van der Made, A.W.; Hoppenbrouwer, E.J.H.; Nolte, R.J.M.; Drenth, W. An Improved Synthesis of Tetraarylporphyrins. Recl. Trav. Chim. Pays-Bas. 1988, 107, 15–16. [Google Scholar] [CrossRef]
- Mahmood, M.H.R.; Liu, Z.Y.; Liu, H.Y.; Zou, H.B.; Chang, C.K. Improved Synthesis of Sterically Encumbered Multibrominated Corroles. Chin. Chem. Lett. 2014, 25, 1349–1353. [Google Scholar] [CrossRef]
- Garai, A.; Nandy, P.; Sinha, W.; Purohit, C.S.; Kar, S. A New Synthetic Protocol for the Preparation of 5-Cyano-10,15,20-Tris (Alkoxyphenyl) Porphyrins. Polyhedron 2013, 56, 18–23. [Google Scholar] [CrossRef]
- Takanami, T.; Hayashi, M.; Chijimatsu, H.; Inoue, W.; Suda, K. Palladium-Catalyzed Cyanation of Porphyrins Utilizing Cyanoethylzinc Bromide as an Efficient Cyanide Ion Source. Org. Lett. 2005, 7, 3937–3940. [Google Scholar] [CrossRef] [PubMed]
- Hiroto, S.; Miyake, Y.; Shinokubo, H. Synthesis and functionalization of porphyrins through organometallic methodologies. Chem. Rev. 2017, 117, 2910–3043. [Google Scholar] [CrossRef] [PubMed]
- Mohajer, D.; Dehghani, H. Exclusive 2:1 Molecular Complexation of 2,3-Dichloro-5,6-Dicyano-Benzoquinone and para-Substituted meso-Tetraphenylporphyrins: Spectral Analogues for Diprotonated meso-Tetraphenylporphyrin. J. Chem. Soc. Perkin Trans. 2 2000, 2, 199–205. [Google Scholar] [CrossRef]
- Rapport, Z. (Ed.) The Chemistry of the Cyano Group; Interscience Publishers: London, UK, 1970. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.-Y.; Mahmood, M.H.; Wu, J.-Z.; Yang, S.-B.; Liu, H.-Y. Unexpected Synthesis of a Bulky Bis-Pocket A3B-Type Meso-Cyano Porphyrin. Molecules 2017, 22, 1941. https://doi.org/10.3390/molecules22111941
Liu Z-Y, Mahmood MH, Wu J-Z, Yang S-B, Liu H-Y. Unexpected Synthesis of a Bulky Bis-Pocket A3B-Type Meso-Cyano Porphyrin. Molecules. 2017; 22(11):1941. https://doi.org/10.3390/molecules22111941
Chicago/Turabian StyleLiu, Ze-Yu, Mian HR Mahmood, Jian-Zhong Wu, Shu-Bao Yang, and Hai-Yang Liu. 2017. "Unexpected Synthesis of a Bulky Bis-Pocket A3B-Type Meso-Cyano Porphyrin" Molecules 22, no. 11: 1941. https://doi.org/10.3390/molecules22111941
APA StyleLiu, Z.-Y., Mahmood, M. H., Wu, J.-Z., Yang, S.-B., & Liu, H.-Y. (2017). Unexpected Synthesis of a Bulky Bis-Pocket A3B-Type Meso-Cyano Porphyrin. Molecules, 22(11), 1941. https://doi.org/10.3390/molecules22111941





