Characterization of Free, Conjugated, and Bound Phenolic Acids in Seven Commonly Consumed Vegetables
Abstract
:1. Introduction
2. Results and Discussion
2.1. Free Phenolic Acid
2.2. Conjugated Phenolic Acid
2.3. Bound Phenolic Acid
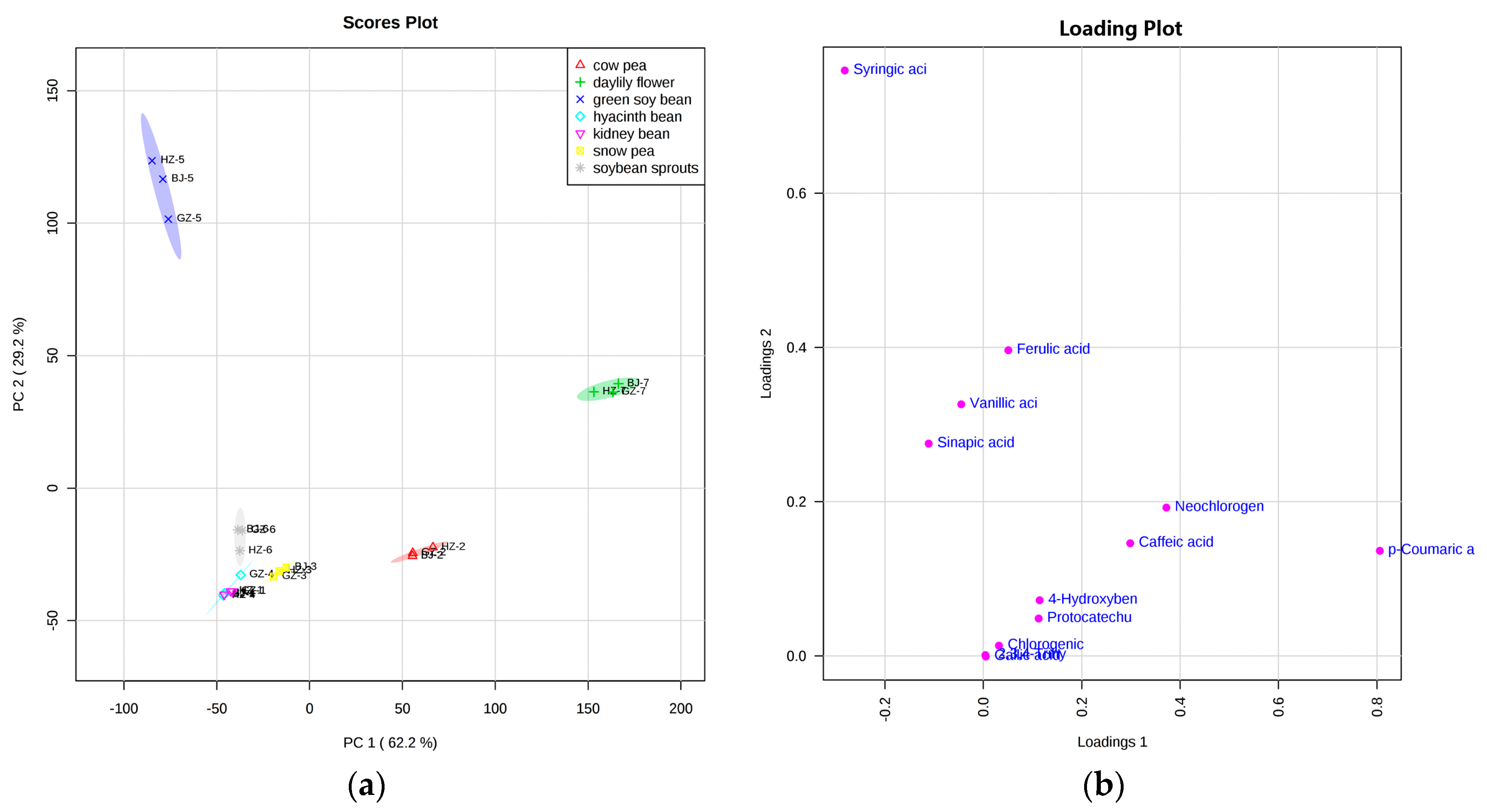
2.4. Principal Component Analysis
3. Materials and Methods
3.1. Sampling and Processing
3.2. Chemicals and Standards
3.3. Free Phenolic Acid Extraction
3.4. Conjugated Phenolic Acid Extraction
3.5. Bound Phenolic Acid Extraction
3.6. UPLC-MS/MS Analysis
3.7. Statistical Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hou, Y.Y. Study of Balance between Supply and Demand on Vegetable in China. Ph.D. Thesis, Northwest A & F University, Yang Ling, China, May 2012. [Google Scholar]
- Liu, X.; Lv, K. Cruciferous vegetables intake is inversely associated with risk of breast cancer: A meta-analysis. Breast 2013, 22, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [PubMed]
- Chandrasekara, A.; Shahidi, F. Antiproliferative potential and DNA scission inhibitory activity of phenolics from whole millet grains. J. Funct. Foods 2011, 3, 159–170. [Google Scholar] [CrossRef]
- Gonzalez, S.; Fernandez, M.; Cuervo, A.; Lasheras, C. Dietary intake of polyphenols and major food sources in an institutionalised elderly population. J. Hum. Nutr. Diet. 2014, 27, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Natella, F.; Maldini, M.; Nardini, M.; Azzini, E.; Foddai, M.S.; Giusti, A.M.; Baima, S.; Morelli, G.; Scaccini, C. Improvement of the nutraceutical quality of broccoli sprouts by elicitation. Food Chem. 2016, 201, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Jiao, W.; Cao, J.; Jiang, W. Effects of chlorogenic acid on capacity of free radicals scavenging and proteomic changes in postharvest fruit of nectarine. PLoS ONE 2017, 12, e0182494. [Google Scholar] [CrossRef] [PubMed]
- Romanazzi, G.; Sanzani, S.M.; Bi, Y.; Tian, S.; Martínez, P.G.; Alkan, N. Induced resistance to control postharvest decay of fruit and vegetables. Postharvest Biol. Technol. 2016, 122, 82–94. [Google Scholar] [CrossRef]
- Duizer, L.M.; Langfried, A. Sensory characterization during repeated ingestion of small-molecular-weight phenolic acids. J. Sci. Food Agric. 2016, 96, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.X.; Tang, Y.; Marcone, M.F.; Pauls, P.K.; Zhang, B.; Liu, R.; Tsao, R. Characterization of free, conjugated and bound phenolics and lipophilic antioxidants in regular-and non-darkening cranberry beans (Phaseolus vulgaris L.). Food Chem. 2015, 185, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Akyol, H.; Riciputi, Y.; Capanoglu, E.; Caboni, M.F.; Verardo, V. Phenolic compounds in the potato and its byproducts: An overview. Int. J. Mol. Sci. 2016, 17, 835. [Google Scholar] [CrossRef] [PubMed]
- Michiels, J.A.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Extraction conditions can greatly influence antioxidant capacity assays in plant food matrices. Food Chem. 2012, 130, 986–993. [Google Scholar] [CrossRef]
- Kaushik, P.; Andújar, I.; Vilanova, S.; Plazas, M.; Gramazio, P.; Herraiz, F.J.; Brar, N.S.; Prohens, J. Breeding vegetables with increased content in bioactive phenolic acids. Molecules 2015, 20, 18464–18481. [Google Scholar] [CrossRef] [PubMed]
- Kacjan Maršić, N.; Mikulič-Petkovšek, M.; Štampar, F. Grafting influences phenolic profile and carpometric traits of fruits of greenhouse-grown eggplant (Solanum melongena L.). J. Agric. Food Chem. 2014, 62, 10504–10514. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Simon, P.W.; Tanumihardjo, S.A. Antioxidant phytochemicals and antioxidant capacity of biofortified carrots (Daucus carota L.) of various colors. J. Agric. Food Chem. 2009, 57, 4142–4147. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Kim, Y.B.; Li, X.; Choi, S.R.; Park, S.; Park, J.S.; Lim, Y.P.; Park, S.U. Accumulation of phenylpropanoids by white, blue, and red light irradiation and their organ-specific distribution in Chinese cabbage (Brassica rapa ssp. pekinensis). J. Agric. Food Chem. 2015, 63, 6772–6778. [Google Scholar] [CrossRef] [PubMed]
- Seong, G.U.; Hwang, I.W.; Chung, S.K. Antioxidant capacities and polyphenolics of Chinese cabbage (Brassica rapa L. ssp. pekinensis) leaves. Food Chem. 2016, 199, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, H.Y.; Seo, W.T.; Lee, J.H.; Cho, K.M. Roasting enhances antioxidant effect of bitter melon (Momordica charantia L.) increasing in flavan-3-ol and phenolic acid contents. Food Sci. Biotechnol. 2012, 21, 19–26. [Google Scholar] [CrossRef]
- Nicoletto, C.; Santagata, S.; Pino, S.; Sambo, P. Antioxidant characterization of different Italian broccoli landraces. Hortic. Bras. 2016, 34, 74–79. [Google Scholar] [CrossRef]
- Darmadi-Blackberry, I.; Wahlqvist, M.L.; Kouris-Blazos, A.; Steen, B.; Lukito, W.; Horie, Y.; Horie, K. Legumes: The most important dietary predictor of survival in older people of different ethnicities. Asia Pac. J. Clin. Nutr. 2004, 13, 217–220. [Google Scholar] [PubMed]
- García-Lafuente, A.; Moro, C.; Manchón, N.; Gonzalo-Ruiz, A.; Villares, A.; Guillamón, E.; Rostagno, M.; Mateo-Vivaracho, L. In vitro anti-inflammatory activity of phenolic rich extracts from white and red common beans. Food Chem. 2014, 161, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Wen, T.N.; Prasad, K.N.; Yang, B.; Ismail, A. Bioactive substance contents and antioxidant capacity of raw and blanched vegetables. Innov. Food Sci. Emerg. 2010, 11, 464–469. [Google Scholar] [CrossRef]
- Bor, J.Y.; Chen, H.Y.; Yen, G.C. Evaluation of antioxidant activity and inhibitory effect on nitric oxide production of some common vegetables. J. Agric. Food Chem. 2006, 54, 1680–1686. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.Y.; Jeon, B.S.; Bock, J.Y. Free, esterified, and insoluble-bound phenolic acids in white and red Korean ginsengs (Panax ginseng CA Meyer). Food Chem. 2002, 79, 105–111. [Google Scholar] [CrossRef]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef] [PubMed]
- Madhujith, T.; Shahidi, F. Antioxidant potential of barley as affected by alkaline hydrolysis and release of insoluble-bound phenolics. Food Chem. 2009, 117, 615–620. [Google Scholar] [CrossRef]
- Dos Santos, M.D.; Almeida, M.C.; Lopes, N.P.; De Souza, G.E.P. Evaluation of the anti-inflammatory, analgesic and antipyretic activities of the natural polyphenol chlorogenic acid. Biol. Pharm. Bull. 2006, 29, 2236–2240. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharmaceut. 2011, 403, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Stommel, J.R.; Whitaker, B.D.; Haynes, K.G.; Prohens, J. Genotype × environment interactions in eggplant for fruit phenolic acid content. Euphytica 2015, 205, 823–836. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, N.; Wang, Y.; Jiang, D.; Feng, X. Characterization of phenolic compounds from early and late ripening sweet cherries and their antioxidant and antifungal activities. J. Agric. Food Chem. 2017, 65, 5413–5420. [Google Scholar] [CrossRef] [PubMed]
- Saura-Calixto, F. Dietary fiber as a carrier of dietary antioxidants: An essential physiological function. J. Agric. Food Chem. 2010, 59, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Mazzeo, T.; Paciulli, M.; Chiavaro, E.; Visconti, A.; Fogliano, V.; Ganino, T.; Pellegrini, N. Impact of the industrial freezing process on selected vegetables–Part II. Colour and bioactive compounds. Food Res. Int. 2015, 75, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Luthria, D.L.; Pastor-Corrales, M.A. Phenolic acids content of fifteen dry edible bean (Phaseolus vulgaris L.) varieties. J. Food Compos. Anal. 2006, 19, 205–211. [Google Scholar] [CrossRef]
- Stratil, P.; Klejdus, B.; Kubáň, V. Determination of total content of phenolic compounds and their antioxidant activity in vegetables evaluation of spectrophotometric methods. J. Agric. Food Chem. 2006, 54, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.R.; Pereira, M.J.; Azevedo, J.; Gonçalves, R.F.; Valentão, P.; de Pinho, P.G.; Andrade, P.B. Glycine max (L.) Merr., Vigna radiata L. and Medicago sativa L. sprouts: A natural source of bioactive compounds. Food Res. Int. 2013, 50, 167–175. [Google Scholar] [CrossRef]
- Häkkinen, S.H.; Törrönen, A.R. Content of flavonols and selected phenolic acids in strawberries and Vaccinium species: Influence of cultivar, cultivation site and technique. Food Res. Int. 2000, 33, 517–524. [Google Scholar] [CrossRef]
- Xu, G.; Ye, X.; Chen, J.; Liu, D. Effect of heat treatment on the phenolic compounds and antioxidant capacity of citrus peel extract. J. Agric. Food Chem. 2007, 55, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Alonso, L.G.; Lygin, A.; Widholm, J.M.; Valverde, M.E.; Paredes-Lopez, O. Polyphenols in wild and weedy Mexican common beans (Phaseolus vulgaris L.). J. Agric. Food Chem. 2006, 54, 4436–4444. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chang, S.K.C. Total phenolic, phenolic acid, anthocyanin, flavan-3-ol, and flavonol profiles and antioxidant properties of pinto and black beans (Phaseolus vulgaris L.) as affected by thermal processing. J. Agric. Food Chem. 2009, 57, 4754–4764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Monk, J.M.; Lu, J.T.; Zarepoor, L.; Wu, W.; Liu, R.; Pauls, K.P.; Wood, G.A.; Robinson, L.; Tsao, R.; et al. Cooked navy and black bean diets improve biomarkers of colon health and reduce inflammation during colitis. Br. J. Nutr. 2014, 111, 1549–1563. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Shewry, P.R.; Ward, J.L. Phenolic acids in wheat varieties in the HEALTHGRAIN diversity screen. J. Agric. Food Chem. 2008, 56, 9732–9739. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the seven vegetables are available from the authors. |
| Vegetables | Region | Chlorogenic Acid | Neochlorogenic Acid | p-Coumaric Acid | p-Hydroxybenzoic Acid | Protocatechuic Acid | Vanillic Acid | Total Free Phenolic Acid |
|---|---|---|---|---|---|---|---|---|
| kidney bean | BJ | 1.95 ± 0.24 b | n.d. | tr | n.d. | n.d. | n.d. | 1.95 ± 0.26 b |
| HZ | 1.24 ± 0.07 c | n.d. | 0.81 ± 0.03 a | n.d. | n.d. | n.d. | 2.05 ± 0.11 b | |
| GZ | 2.47 ± 0.27 a | n.d. | 0.37 ± 0.02 b | n.d. | n.d. | n.d. | 2.84 ± 0.35 a | |
| cow pea | BJ | 0.22 ± 0.01 a | n.d. | 0.65 ± 0.06 c | n.d. | n.d. | n.d. | 0.87 ± 0.06 b |
| HZ | 0.11 ± 0.00 c | n.d. | 1.28 ± 0.17 a | n.d. | n.d. | n.d. | 1.39 ± 0.17 a | |
| GZ | 0.15 ± 0.01 b | n.d. | 0.79 ± 0.03 b | n.d. | n.d. | n.d. | 0.94 ± 0.03 b | |
| snow pea | BJ | n.d. | n.d. | 0.09 ± 0.07 c | n.d. | n.d. | n.d. | 0.09 ± 0.04 b |
| HZ | n.d. | n.d. | 0.22 ± 0.01 a | n.d. | n.d. | n.d. | 0.22 ± 0.01 a | |
| GZ | n.d. | n.d. | 0.11 ± 0.01 b | n.d. | n.d. | n.d. | 0.11 ± 0.01 b | |
| hyacinth bean | BJ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| HZ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | |
| GZ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | |
| green soy bean | BJ | n.d. | n.d. | n.d. | 0.25 ± 0.01 a | n.d. | n.d. | 0.25 ± 0.01 a |
| HZ | n.d. | n.d. | n.d. | 0.13 ± 0.01 b | n.d. | n.d. | 0.13 ± 0.01 b | |
| GZ | n.d. | n.d. | n.d. | 0.12 ± 0.01 b | n.d. | n.d. | 0.12 ± 0.01 b | |
| soybean sprouts | BJ | n.d. | n.d. | n.d. | 2.26 ± 0.11 | n.d. | n.d. | 2.26 ± 0.11 a |
| HZ | n.d. | n.d. | n.d. | 1.29 ± 0.09 | n.d. | n.d. | 1.29 ± 0.09 b | |
| GZ | n.d. | n.d. | n.d. | 1.07 ± 0.04 | n.d. | n.d. | 1.07 ± 0.04 c | |
| daylily | BJ | 8.11 ± 0.44 b | 103.10 ± 5.21 a | 5.11 ± 0.27 a | 12.85 ± 1.07 a | 8.56 ± 0.45 a | 5.55 ± 0.29 b | 143.28 ± 3.51 a |
| HZ | 9.84 ± 0.57 a | 84.04 ± 7.22 c | 4.03 ± 0.33 b | 11.89 ± 0.98 ab | 8.33 ± 0.36 a | 6.87 ± 0.41 a | 125.00 ± 4.22 b | |
| GZ | 7.98 ± 0.35 b | 98.86 ± 3.21 b | 4.44 ± 0.56 ab | 10.64 ± 0.79 b | 5.22 ± 0.83 b | 4.90 ± 0.28 c | 132.04 ± 2.84 b |
| Vegetables | Region | Caffeic Acid | Ferulic Acid | p-Coumaric Acid | Sinapic Acid | Gallic Acid | p-Hydroxybenzoic Acid | Protocatechuic Acid | Syringic Acid | 2,3,4-Trihydroxybenzoic Acid | Vanillic Acid | Total Conjugated Phenolic Acid |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| kidney bean | BJ | 1.42 ± 0.05 a | 1.17 ± 0.05 c | 1.17 ± 0.32 c | 0.32 ± 0.01 b | 2.35 ± 0.08 c | n.d. | n.d. | n.d. | 0.54 ± 0.04 a | n.d. | 6.97 ± 0.37 b |
| HZ | 0.79 ± 0.03 b | 2.69 ± 0.08 b | 4.99 ± 0.70 b | 1.42 ± 0.06 a | 3.77 ± 0.17 a | n.d. | n.d. | n.d. | 0.54 ± 0.03 a | n.d. | 14.20 ± 0.72 a | |
| GZ | 0.64 ± 0.02 c | 3.00 ± 0.12 a | 6.70 ± 0.05 a | 0.29 ± 0.02 b | 2.59 ± 0.14 b | n.d. | n.d. | n.d. | 0.56 ± 0.02 a | n.d. | 13.78 ± 0.55 a | |
| cow pea | BJ | 0.97 ± 0.04 c | 4.55 ± 0.21 c | 119.69 ± 3.47 c | 0.26 ± 0.02 b | 0.78 ± 0.02 a | n.d. | 1.71 ± 0.05 c | n.d. | n.d. | n.d. | 127.96 ± 4.27 c |
| HZ | 1.4 ± 0.03 a | 8.93 ± 0.37 a | 134.31 ± 2.99 a | 0.31 ± 0.02 a | 0.43 ± 0.02 c | n.d. | 3.25 ± 0.16 a | n.d. | n.d. | n.d. | 148.63 ± 3.44 a | |
| GZ | 1.28 ± 0.02 b | 7.28 ± 0.28 b | 124.49 ± 4.22 b | 0.28 ± 0.02 ab | 0.62 ± 0.03 b | n.d. | 2.16 ± 0.16 b | n.d. | n.d. | n.d. | 136.11 ± 3.68 b | |
| snow pea | BJ | n.d. | 2.60 ± 0.12 b | 44.36 ± 1.77 a | 17.82 ± 0.60 a | 0.28 ± 0.01 b | n.d. | 2.51 ± 0.12 a | n.d. | n.d. | n.d. | 67.57 ± 2.33 a |
| HZ | n.d. | 3.23 ± 0.13 a | 38.84 ± 2.05 b | 14.17 ± 0.13 b | 0.34 ± 0.03 a | n.d. | 2.41 ± 0.03 a | n.d. | n.d. | n.d. | 58.99 ± 2.07 b | |
| GZ | n.d. | 2.22 ± 0.07 c | 35.85 ± 2.62 b | 10.01 ± 0.46 c | 0.32 ± 0.02 a | n.d. | 1.83 ± 0.05 b | n.d. | n.d. | n.d. | 50.23 ± 2.78 c | |
| hyacinth bean | BJ | 0.20 ± 0.00 a | 2.12 ± 0.05 a | 1.10 ± 0.04 a | n.d. | 0.09 ± 0.03 b | n.d. | 1.30 ± 0.04 b | n.d. | n.d. | n.d. | 4.81 ± 0.12 a |
| HZ | 0.12 ± 0.01 b | 0.50 ± 0.01 c | 0.72 ± 0.03 c | n.d. | 0.06 ± 0.00 b | n.d. | 1.36 ± 0.12 ab | n.d. | n.d. | n.d. | 2.76 ± 0.11 b | |
| GZ | 0.12 ± 0.04 b | 1.30 ± 0.44 b | 0.94 ± 1.24 ac | n.d. | 0.17 ± 0.01 a | n.d. | 1.57 ± 0.18 a | n.d. | n.d. | n.d. | 4.10 ± 1.05 a | |
| green soy bean | BJ | n.d. | 46.61 ± 2.09 a | 2.82 ± 0.13 c | 42.01 ± 0.79 a | 0.48 ± 0.02 a | 1.82 ± 0.05 a | 1.00 ± 0.07 b | 102.46 ± 3.58 a | n.d. | 41.59 ± 0.79 a | 238.79 ± 6.88 a |
| HZ | n.d. | 39.92 ± 1.28 b | 5.74 ± 0.13 a | 32.93 ± 1.27 c | 0.46 ± 0.02 a | 1.01 ± 0.03 b | 1.14 ± 0.03 a | 87.45 ± 3.15 b | n.d. | 32.82 ± 1.26 b | 201.47 ± 5.33 b | |
| GZ | n.d. | 32.67 ± 1.79 c | 4.15 ± 0.18 b | 36.99 ± 2.14 b | 0.37 ± 0.02 b | 0.76 ± 0.10 c | 0.92 ± 0.03 b | 89.84 ± 3.24 b | n.d. | 26.88 ± 1.07 c | 192.58 ± 5.22 b | |
| soybean sprouts | BJ | 0.49 ± 0.02 b | 14.58 ± 0.66 a | 18.38 ± 1.04 a | 2.98 ± 0.30 b | 0.38 ± 0.01 b | 2.18 ± 0.10 b | 1.03 ± 0.53 ab | n.d. | 0.60 ± 0.10 b | 10.32 ± 0.52 a | 50.94 ± 1.99 a |
| HZ | 0.57 ± 0.02 a | 7.22 ± 0.27 c | 15.10 ± 0.67 b | 2.33 ± 0.22 c | 0.53 ± 0.02 a | 1.84 ± 0.08 c | 1.38 ± 0.02 a | n.d. | 0.87 ± 0.03 a | 8.53 ± 0.29 b | 38.37 ± 2.09 c | |
| GZ | 0.43 ± 0.01 c | 8.08 ± 0.32 b | 15.98 ± 0.72 b | 3.45 ± 0.12 a | 0.26 ± 0.01 c | 2.42 ± 0.10 a | 1.09 ± 0.04 b | n.d. | 0.69 ± 0.02 b | 11.56 ± 0.76 a | 43.96 ± 1.87 b | |
| daylily | BJ | 69.54 ± 2.42 a | 40.10 ± 1.54 a | 146.31 ± 4.22 a | 0.92 ± 0.04 a | 2.16 ± 0.07 b | 17.39 ± 0.71 b | 22.40 ± 0.78 a | 0.54 ± 0.01 b | 1.38 ± 0.06 ab | 12.89 ± 1.24 a | 313.63 ± 5.61 a |
| HZ | 73.42 ± 2.85 a | 37.86 ± 1.24 a | 134.66 ± 2.79 b | 0.81 ± 0.02 b | 2.68 ± 0.11 a | 14.92 ± 0.44 c | 22.27 ± 1.28 a | 0.67 ± 0.02 a | 1.53 ± 0.10 a | 11.76 ± 0.55 a | 300.58 ± 4.21 b | |
| GZ | 70.7 ± 2.69 a | 39.62 ± 1.56 a | 149.61 ± 5.21 a | 1.02 ± 0.38 ab | 1.77 ± 0.11 c | 20.32 ± 1.25 a | 14.94 ± 0.95 b | 0.55 ± 0.10 b | 1.30 ± 0.04 b | 8.82 ± 0.72 b | 308.65 ± 5.92 ab |
| Vegetables | Region | Caffeic Acid | Ferulic Acid | p-Coumaric Acid | Sinapic Acid | p-Hydroxybenzoic Acid | Protocatechuic Acid | Syringic Acid | Vanillic Acid | Total Bound Phenolic Acid |
|---|---|---|---|---|---|---|---|---|---|---|
| kidney bean | BJ | n.d. | n.d. | 0.54 ± 0.03 a | n.d. | n.d. | n.d. | n.d. | n.d. | 0.54 ± 0.03 a |
| HZ | n.d. | n.d. | 0.59 ± 0.08 a | n.d. | n.d. | n.d. | n.d. | n.d. | 0.59 ± 0.08 a | |
| GZ | n.d. | n.d. | 0.42 ± 0.09 a | n.d. | n.d. | n.d. | n.d. | n.d. | 0.42 ± 0.09 a | |
| cow pea | BJ | n.d. | n.d. | 9.55 ± 0.06 a | n.d. | n.d. | n.d. | n.d. | n.d. | 9.55 ± 0.06 a |
| HZ | n.d. | n.d. | 7.59 ± 0.31 b | n.d. | n.d. | n.d. | n.d. | n.d. | 7.59 ± 0.31 b | |
| GZ | n.d. | n.d. | 4.41 ± 0.18 c | n.d. | n.d. | n.d. | n.d. | n.d. | 4.41 ± 0.18 c | |
| snow pea | BJ | n.d. | 0.14 ± 0.01 a | 1.48 ± 0.03 a | 0.29 ± 0.01 a | n.d. | 4.35 ± 0.25 b | n.d. | n.d. | 6.26 ± 0.28 b |
| HZ | n.d. | 0.14 ± 0.01 a | 1.52 ± 0.07 a | 0.28 ± 0.01 a | n.d. | 5.30 ± 0.10 a | n.d. | n.d. | 7.20 ± 0.14 a | |
| GZ | n.d. | 0.08 ± 0.01 b | 0.56 ± 0.01 b | 0.23 ± 0.02 b | n.d. | 2.86 ± 0.11 c | n.d. | n.d. | 3.73 ± 0.12 c | |
| hyacinth bean | BJ | 1.74 ± 0.05 b | 0.23 ± 0.01 a | 0.24 ± 0.01 b | n.d. | n.d. | 0.11 ± 0.01 a | n.d. | n.d. | 2.32 ± 0.07 b |
| HZ | 1.34 ± 0.04 c | 0.21 ± 0.01 a | 0.28 ± 0.01 a | n.d. | n.d. | 0.12 ± 0.01 a | n.d. | n.d. | 1.95 ± 0.07 c | |
| GZ | 4.41 ± 0.23 a | 0.10 ± 0.00 b | 0.18 ± 0.01 c | n.d. | n.d. | 0.08 ± 0.01 b | n.d. | n.d. | 4.77 ± 0.25 a | |
| green soy bean | BJ | n.d. | 22.09 ± 1.32 a | 3.77 ± 0.17 b | 16.34 ± 0.69 a | 0.78 ± 0.05 a | n.d. | 45.10 ± 2.18 a | 20.67 ± 1.33 a | 108.75 ± 3.57 a |
| HZ | n.d. | 18.33 ± 0.89 b | 1.32 ± 0.56 c | 13.54 ± 0.52 b | 0.80 ± 0.03 a | n.d. | 33.40 ± 1.97 c | 18.17 ± 0.73 b | 85.56 ± 2.88 b | |
| GZ | n.d. | 12.91 ± 0.61 c | 7.81 ± 0.37 a | 12.85 ± 0.49 b | 0.81 ± 0.03 a | n.d. | 36.87 ± 1.24 b | 12.65 ± 0.45 c | 83.9 ± 2.54 b | |
| soybean sprouts | BJ | n.d. | 0.84 ± 0.03 b | 1.22 ± 0.05 a | n.d. | 0.42 ± 0.01 a | n.d. | 17.63 ± 0.76 a | 4.68 ± 0.16 a | 24.79 ± 1.55 a |
| HZ | n.d. | 0.71 ± 0.04 c | 0.84 ± 0.03 b | n.d. | 0.44 ± 0.02 a | n.d. | 10.22 ± 0.47 c | 3.51 ± 0.12 b | 15.72 ± 0.77 c | |
| GZ | n.d. | 1.13 ± 0.06 a | 0.64 ± 0.02 c | n.d. | 0.44 ± 0.01 a | n.d. | 14.98 ± 0.39 b | 3.41 ± 0.15 b | 20.60 ± 1.02 b | |
| daylily | BJ | 7.98 ± 0.36 a | 8.23 ± 0.37 a | 22.43 ± 1.04 a | n.d. | 2.00 ± 0.05 b | 3.02 ± 0.03 a | 0.07 ± 0.01 a | 2.31 ± 0.10 a | 46.04 ± 2.01 a |
| HZ | 5.88 ± 0.30 b | 5.24 ± 0.39 b | 20.15 ± 1.12 b | n.d. | 2.92 ± 0.14 a | 2.51 ± 0.04 b | 0.05 ± 0.01 ab | 1.87 ± 0.06 b | 38.62 ± 1.28 b | |
| GZ | 5.18 ± 0.31 c | 4.71 ± 0.22 b | 16.31 ± 0.77 c | n.d. | 1.38 ± 0.04 c | 2.38 ± 0.06 c | 0.04 ± 0.01 b | 1.55 ± 0.05 c | 31.55 ± 1.27 c |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Y.; Ma, S.; Wang, M.; Feng, X.-Y. Characterization of Free, Conjugated, and Bound Phenolic Acids in Seven Commonly Consumed Vegetables. Molecules 2017, 22, 1878. https://doi.org/10.3390/molecules22111878
Gao Y, Ma S, Wang M, Feng X-Y. Characterization of Free, Conjugated, and Bound Phenolic Acids in Seven Commonly Consumed Vegetables. Molecules. 2017; 22(11):1878. https://doi.org/10.3390/molecules22111878
Chicago/Turabian StyleGao, Yuan, Shuai Ma, Meng Wang, and Xiao-Yuan Feng. 2017. "Characterization of Free, Conjugated, and Bound Phenolic Acids in Seven Commonly Consumed Vegetables" Molecules 22, no. 11: 1878. https://doi.org/10.3390/molecules22111878





