Synthesis and Antitumor Activity of Novel Arylpiperazine Derivatives Containing the Saccharin Moiety
Abstract
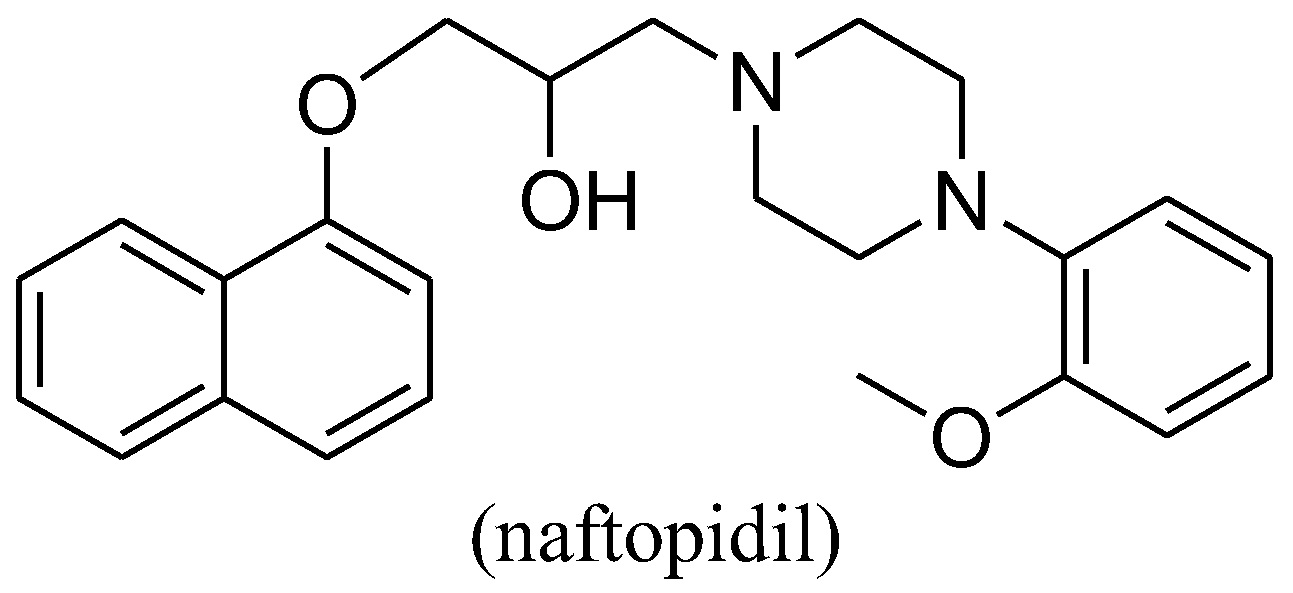
:1. Introduction
2. Results and Discussion
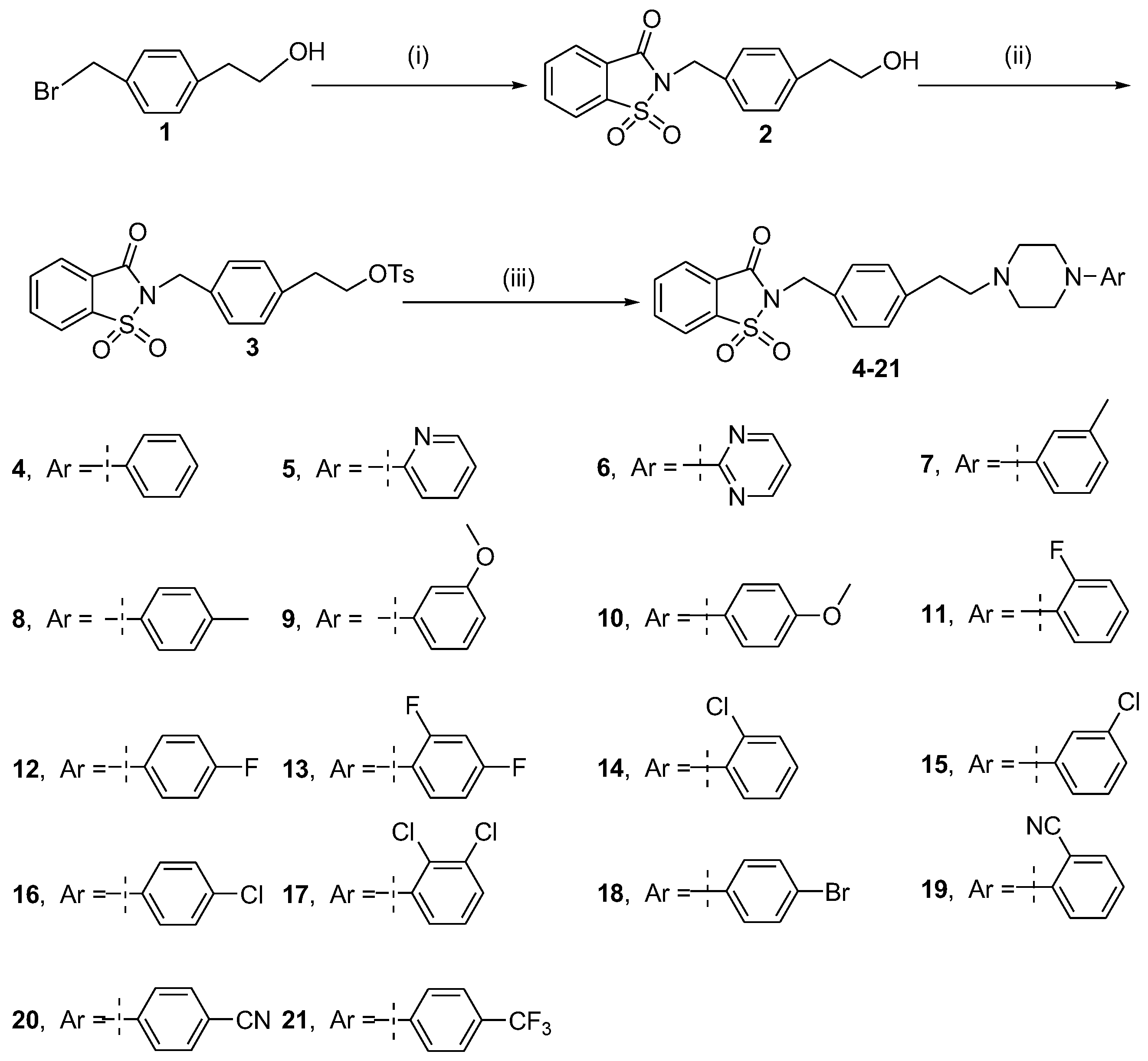
2.1. Chemistry
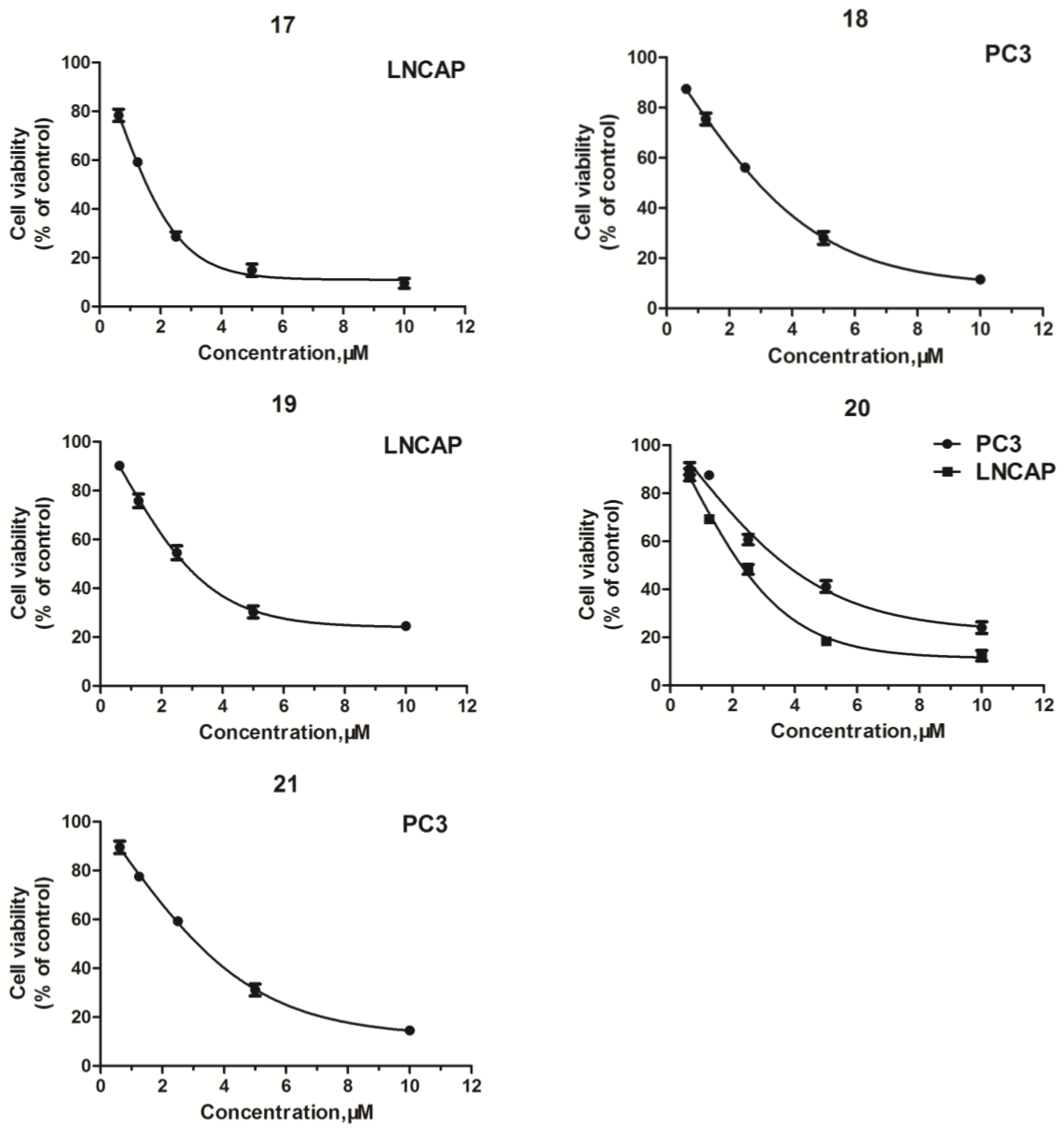
2.2. Structure–Activity Relationship (SAR) Analysis for Antitumor Activity
3. Materials and Methods
3.1. Chemistry
3.1.1. Synthesis of Saccharin N-((2-(4-(methyl)phenyl)ethanol) (2)
3.1.2. Synthesis of Saccharin N-(4-(methyl)phenethyl 4-methylbenzenesulfonate) (3)
3.1.3. General Procedure for the Preparation of Arylpiperazine Derivatives 4–21
3.2. In Vitro Cytotoxic Assay
3.2.1. Cell Culture
3.2.2. Assessment of Antitumor Activity by CCK-8 Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Falciani, C.; Brunetti, J.; Pagliuca, C.; Menichetti, S.; Vitellozzi, L.; Lelli, B.; Pini, A.; Bracci, L. Design and in vitro evaluation of branched peptide conjugates: Turning nonspecific cytotoxic drugs into tumor-selective agents. Chem. Med. Chem. 2010, 5, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Tannock, I. Cell kinetics and chemotherapy: A critical review. Cancer Treat. Rep. 1978, 62, 1117–1133. [Google Scholar] [PubMed]
- Vinaya, K.; Kavitha, C.V.; Chandrappa, S.; Prasanna, D.S.; Raghavan, S.C.; Rangappa, K.S. Synthesis and antileukemic activity of novel 4-(3-(piperidin-4-yl) propyl)piperidine derivatives. Chem. Biol. Drug Des. 2011, 78, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Gelmann, E.P. Molecular biology of the androgen receptor. J. Clin. Oncol. 2002, 20, 3001–3015. [Google Scholar] [CrossRef] [PubMed]
- Culig, Z.; Klocker, H.; Bartsch, G.; Hobisch, A. Androgen receptors in prostate cancer. Endocr. Relat. Cancer 2002, 9, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Bentel, J.M.; Tilley, W.D. Androgen receptors in prostate cancer. J. Endocrinol. 1996, 151, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dehm, S.M.; Tindall, D.J. Androgen receptor structural and functional elements: Role and regulation in prostate cancer. J. Mol. Endocrinol. 2007, 21, 2855–2863. [Google Scholar] [CrossRef] [PubMed]
- Heinlein, C.A.; Chang, C. Androgen receptor in prostate cancer. Endocr. Rev. 2004, 25, 276–308. [Google Scholar] [CrossRef] [PubMed]
- Frydenberg, M.; Stricker, P.D.; Kaye, K.W. Prostate cancer diagnosis and management. Lancet 1997, 349, 1681–1687. [Google Scholar] [CrossRef]
- DeFrancesco, L. Prostate cancer prevention trial launched. Nat. Med. 2001, 7, 1076. [Google Scholar] [CrossRef] [PubMed]
- Nishino, Y.; Masue, T.; Miwa, K.; Takahashi, Y.; Ishihara, S.; Deguchi, T. Comparison of two α1-adrenoceptor antagonists, naftopidil and tamsulosin hydrochloride, in the treatment of lower urinary tract symptoms with benign prostatic hyperplasia: A randomized crossover study. BJU Int. 2006, 97, 747–751. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Sasaki, S.; Kubota, Y.; Hayase, M.; Hayashi, Y.; Shinoura, H.; Tsujimoto, G.; Kohri, K. Expression of alpha1-adrenoceptor subtype mRNA as a predictor of the efficacy of subtype selective alpha1-adrenoceptor antagonists in the management of benign prostatic hyperplasia. J. Urol. 2008, 179, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y.; Ishii, K.; Kanda, H.; Iwamoto, Y.; Nishikawa, K.; Soga, N.; Kise, H.; Arima, K.; Sugimura, Y. Naftopidil, a selective α1-adrenoceptor antagonist, suppresses human prostate tumor growth by altering interactions between tumor cells and stroma. Cancer Prev. Res. (Phila.) 2011, 4, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Kanda, H.; Ishii, K.; Ogura, Y.; Imamura, T.; Kanai, M.; Arima, K.; Sugimura, Y. Naftopidil, a selective alpha-1 adrenoceptor antagonist, inhibits growth of human prostate cancer cells by G1 cell cycle arrest. Int. J. Cancer 2008, 122, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Masachika, E.; Kanno, T.; Nakano, T.; Gotoh, A.; Nishizaki, T. Naftopidil induces apoptosis in malignant mesothelioma cell lines independently of α1-adrenoceptor blocking. Anticancer Res. 2013, 33, 887–894. [Google Scholar] [PubMed]
- Patane, M.A.; DiPardo, R.M.; Price, R.P.; Chang, R.S.; Ransom, R.W.; O’Malley, S.S.; Di Salvo, J.; Bock, M.G. Selective α-1A adrenergic receptor antagonists. Effects of pharmacophore regio- and stereochemistry on potency and selectivity. Bioorg. Med. Chem. Lett. 1998, 8, 2495–2500. [Google Scholar] [CrossRef]
- Romero, A.G.; Darlington, W.H.; Piercey, M.F.; Lahtib, R.A. Synthesis of metabolically stable arylpiperazine 5-HT1A receptor agonists. Bioorg. Med. Chem. Lett. 1992, 2, 1703–1706. [Google Scholar] [CrossRef]
- Martyn, D.C.; Moore, M.J.; Abell, A.D. Succinimide and saccharinbased enzyme-activated inhibitors of serine proteases. Curr. Pharm. Des. 1999, 5, 405–415. [Google Scholar] [PubMed]
- Groutas, W.C.; Epp, J.B.; Venkataraman, R.; Kuang, R.; Truong, T.M.; McClenahan, J.J.; Prakash, O. Design, synthesis, and in vitro inhibitory activity toward human leukocyte elastase, cathepsin G, and proteinase 3 of saccharin-derived sulfones and congeners. Bioorg. Med. Chem. 1996, 4, 1393–1400. [Google Scholar] [CrossRef]
- Hlasta, D.J.; Bell, M.R.; Boaz, N.W.; Court, J.J.; Desai, R.C. A benzisothiazolone class of potent, selective mechanism-based inhibitors of human leukocyte elastase. Bioorg. Med. Chem. Lett. 1994, 4, 1801–1806. [Google Scholar] [CrossRef]
- Hlasta, D.J.; Bell, M.R.; Court, J.J.; Cundy, K.C.; Desai, R.C.; Ferguson, E.W.; Gordon, R.J.; Kumar, V.; Maycock, A.L.; Subramanyam, C.; et al. The design of potent and stable benzisothiazolone inhibitors of human leukocyte elastase. Bioorg. Med. Chem. Lett. 1995, 5, 331–336. [Google Scholar] [CrossRef]
- Hlasta, D.J.; Subramanyam, C.; Bell, M.R.; Carabateas, P.M.; Court, J.J.; Desai, R.C.; Drozd, M.L.; Eickhoff, W.M.; Ferguson, E.W.; Gordon, R.J. Orally bioavailable benzisothiazolone inhibitors of human leukocyte elastase. J. Med. Chem. 1995, 38, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Martin, G.; Lyndon, C.; Sunkel, C. Hepatic kinetics of SCP-1(N-[α-(1,2-benzisothiazol-3(2H)-ona-1,1-dioxide-2-yl)-acetyl]-p-aminophenol) compared with acetaminophen in isolated rat liver. Eur. J. Pharm. Biopharm. 1998, 46, 293–297. [Google Scholar] [CrossRef]
- Combrink, K.D.; Gülgeze, H.B.; Meanwell, N.A.; Pearce, B.C.; Zulan, P.; Bisacchi, G.S.; Roberts, D.G.; Stanley, P.; Seiler, S.M. 1,2-Benzisothiazol-3-one 1,1-dioxide Inhibitors of human mast cell tryptase. J. Med. Chem. 1998, 41, 4854–4860. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, H.T.; Kawle, S.P.; Elberling, J.A.; DeMaster, E.G.; Fukuto, J.M. Prodrugs of nitroxyl as potential aldehyde dehydrogenase inhibitors vis-a-vis vascular smooth muscle relaxants. J. Med. Chem. 1995, 38, 1865–1871. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Liang, X.; Xu, F.; Xu, B.B.; He, X.L.; Huang, B.Y.; Yuan, M. Synthesis and cytotoxic activity evaluation of novel arylpiperazine derivatives on human prostate cancer cell lines. Molecules 2014, 19, 12048–12064. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xu, F.; Liang, X.; Xu, B.B.; Yang, Z.L.; He, X.L.; Huang, B.Y.; Yuan, M. Design, synthesis and biological evaluation of novel arylpiperazine derivatives on human prostate cancer cell lines. Bioorg. Med. Chem. Lett. 2015, 25, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xu, F.; Xu, B.B.; Xu, J.Y.; Shao, B.H.; Huang, B.Y.; Yuan, M. Design, synthesis and biological evaluation of novel arylpiperazine derivatives on human prostate cancer cell lines. Chin. Chem. Lett. 2016, 27, 277–282. [Google Scholar] [CrossRef]
- Kaspers, G.J.; Veerman, A.J.; Pieters, R.; Van Zantwijk, C.H.; Smets, L.A.; Van Wering, E.R.; Van Der Does-Van Den Berg, A. In vitro cellular drug resistance and prognosis in newly diagnosed childhood acute lymphoblastic leukemia. Blood 1997, 90, 2723–2729. [Google Scholar] [PubMed]
- Kaspers, G.J.; Pieters, R.; Van Zantwijk, C.H.; VanWering, E.R.; Van Der Does-Van Den Berg, A.; Veerman, A.J. Prednisolone resistance in childhood acute lymphoblastic leukemia: Vitro-vivo correlations and cross-resistance to other drugs. Blood 1998, 92, 259–266. [Google Scholar] [PubMed]
- Ding, J.; Huang, S.L.; Wu, S.Q.; Zhao, Y.J.; Liang, L.H.; Yan, M.X.; Ge, C.; Yao, J.; Chen, T.Y.; Wan, D.F.; et al. Gain of miR-151 on chromosome 8q24.3 facilitates tumour cell migration and spreading through downregulating RhoGDIA. Nat. Cell Biol. 2010, 12, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Banday, A.H.; Giri, A.K.; Parveen, R.; Bashir, N. Design and synthesis of D-ring steroidal isoxazolines and oxazolines as potential antiproliferative agents against LNCaP, PC-3 and DU-145 cells. Steroids 2014, 87, 93–98. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
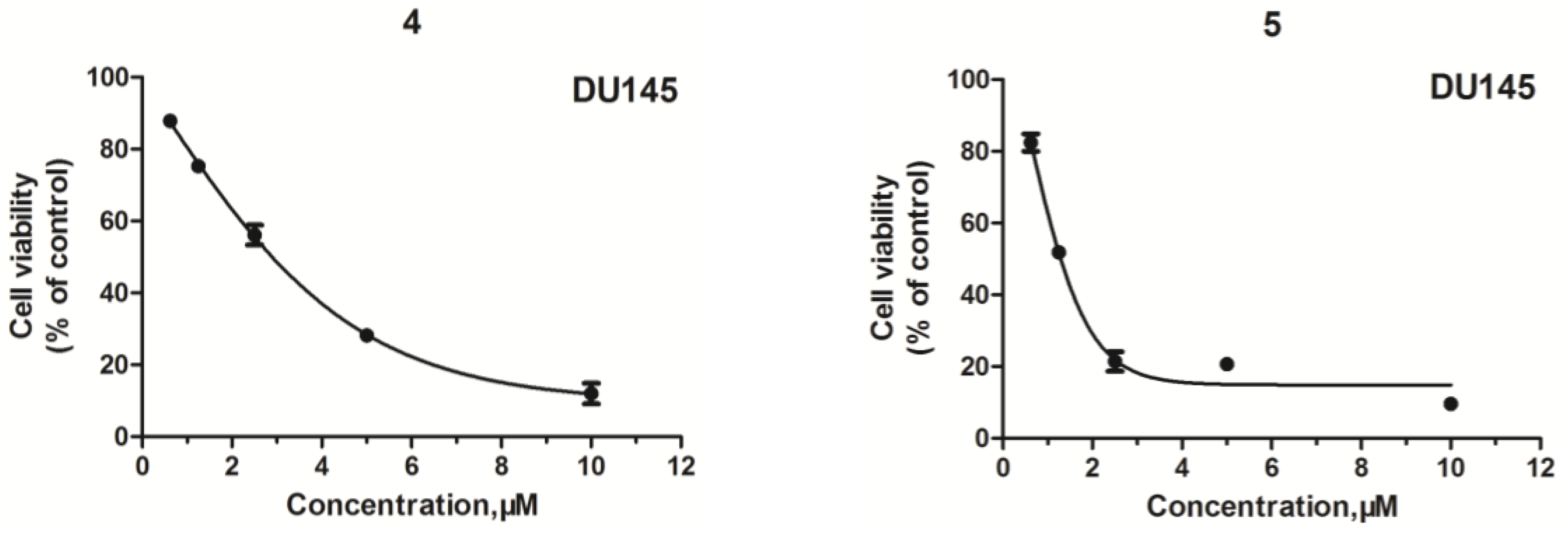
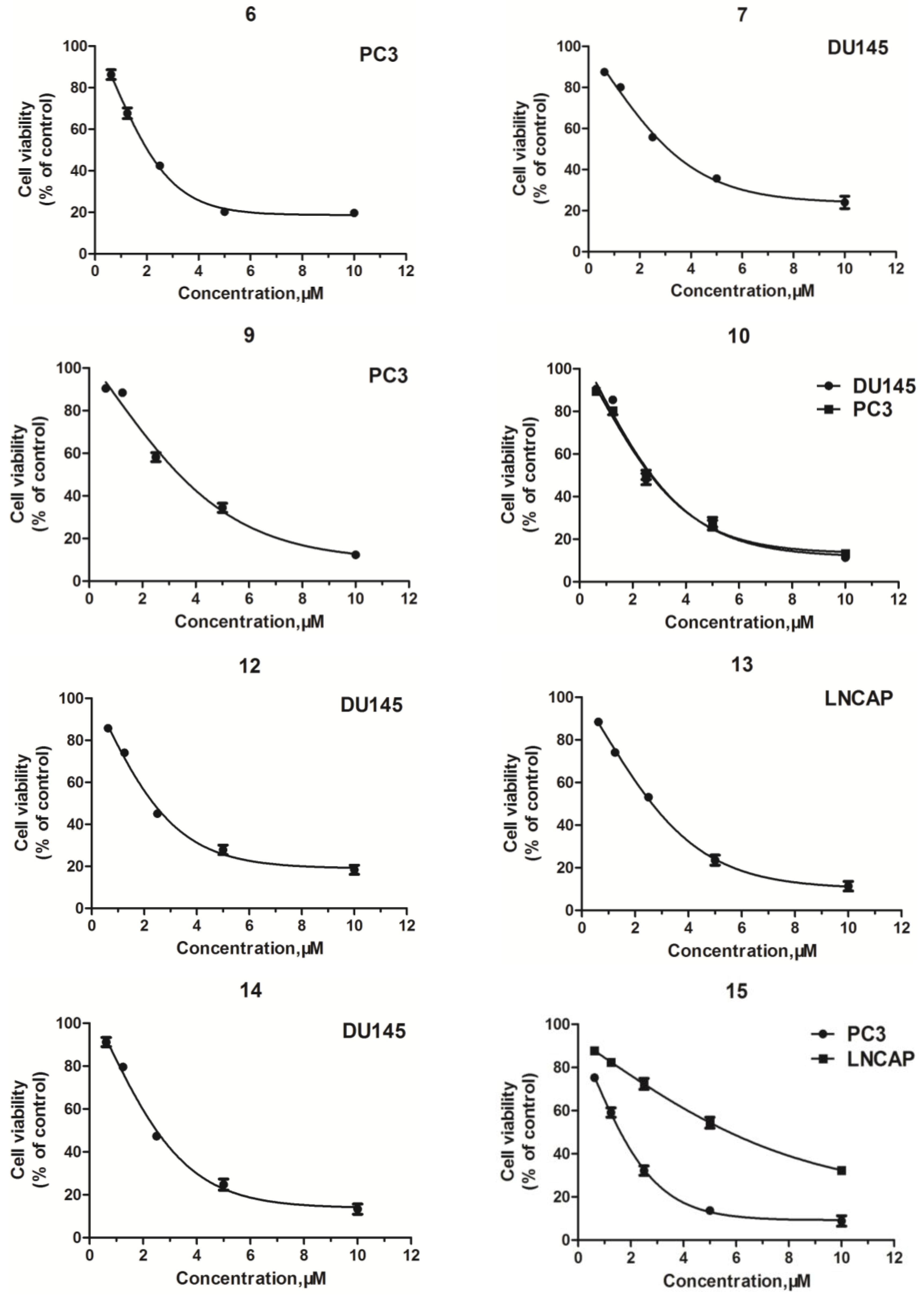
| Compd. | IC50 (μM) a | |||
|---|---|---|---|---|
| PC-3 b | LNCaP b | DU145 b | WPMY-1 b | |
| 4 | >50 | >50 | 1.28 ± 0.04 | >50 |
| 5 | >50 | >50 | 3.57 ± 0.08 | >50 |
| 6 | 4.84 ± 0.17 | >50 | >50 | 48.27 ± 0.56 |
| 7 | >50 | >50 | 3.65 ± 0.10 | >50 |
| 8 | >50 | >50 | >50 | ND c |
| 9 | 5.43 ± 0.18 | >50 | >50 | >50 |
| 10 | 4.38 ± 0.13 | >50 | 2.28 ± 0.05 | >50 |
| 11 | >50 | 47.46 ± 2.17 | >50 | >50 |
| 12 | >50 | >50 | 1.14 ± 0.10 | >50 |
| 13 | >50 | 5.03 ± 0.13 | >50 | >50 |
| 14 | 14.57 ± 1.12 | >50 | 3.39 ± 0.11 | ND c |
| 15 | 2.74 ± 0.11 | 3.43 ± 0.16 | >50 | >50 |
| 16 | >50 | >50 | >50 | ND c |
| 17 | >50 | 4.08 ± 0.15 | >50 | >50 |
| 18 | 2.25 ± 0.07 | >50 | 9.05 ± 0.23 | >50 |
| 19 | >50 | 5.14 ± 0.16 | >50 | 39.15 ± 0.17 |
| 20 | 2.66 ± 0.04 | 3.43 ± 0.10 | >50 | 46.34 ± 0.51 |
| 21 | 3.73 ± 0.08 | >50 | >50 | ND c |
| Naftopidil | 42.10 ± 0.79 | 22.36 ± 0.61 | 34.58 ± 0.31 | >50 |
| Finasteride | 17.83 | 14.53 | 13.53 | − |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Xu, B.-B.; Sun, T.; Zhou, Z.; Ya, H.-Y.; Yuan, M. Synthesis and Antitumor Activity of Novel Arylpiperazine Derivatives Containing the Saccharin Moiety. Molecules 2017, 22, 1857. https://doi.org/10.3390/molecules22111857
Chen H, Xu B-B, Sun T, Zhou Z, Ya H-Y, Yuan M. Synthesis and Antitumor Activity of Novel Arylpiperazine Derivatives Containing the Saccharin Moiety. Molecules. 2017; 22(11):1857. https://doi.org/10.3390/molecules22111857
Chicago/Turabian StyleChen, Hong, Bing-Bing Xu, Tao Sun, Zhan Zhou, Hui-Yuan Ya, and Mu Yuan. 2017. "Synthesis and Antitumor Activity of Novel Arylpiperazine Derivatives Containing the Saccharin Moiety" Molecules 22, no. 11: 1857. https://doi.org/10.3390/molecules22111857




