Design, Synthesis, and Evaluation of Novel Phenolic Acid/Dipeptide/Borneol Hybrids as Potent Angiotensin Converting Enzyme (ACE) Inhibitors with Anti-hypertension Activity
Abstract
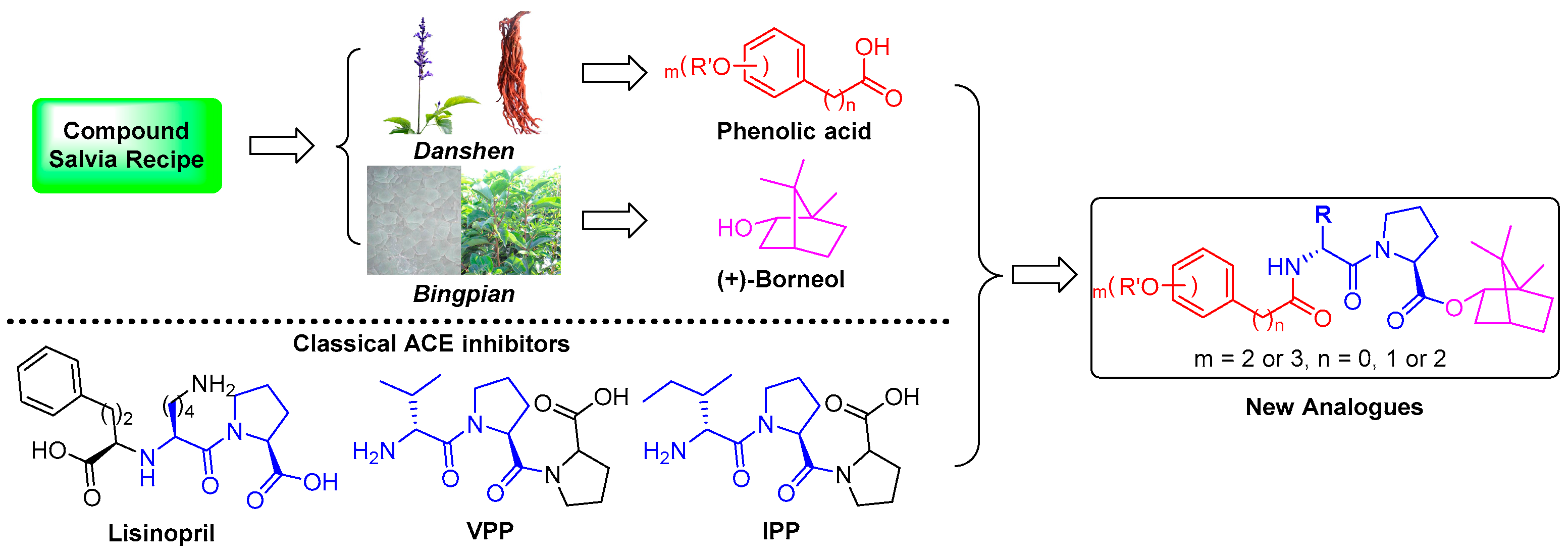
:1. Introduction
2. Materials and Methods
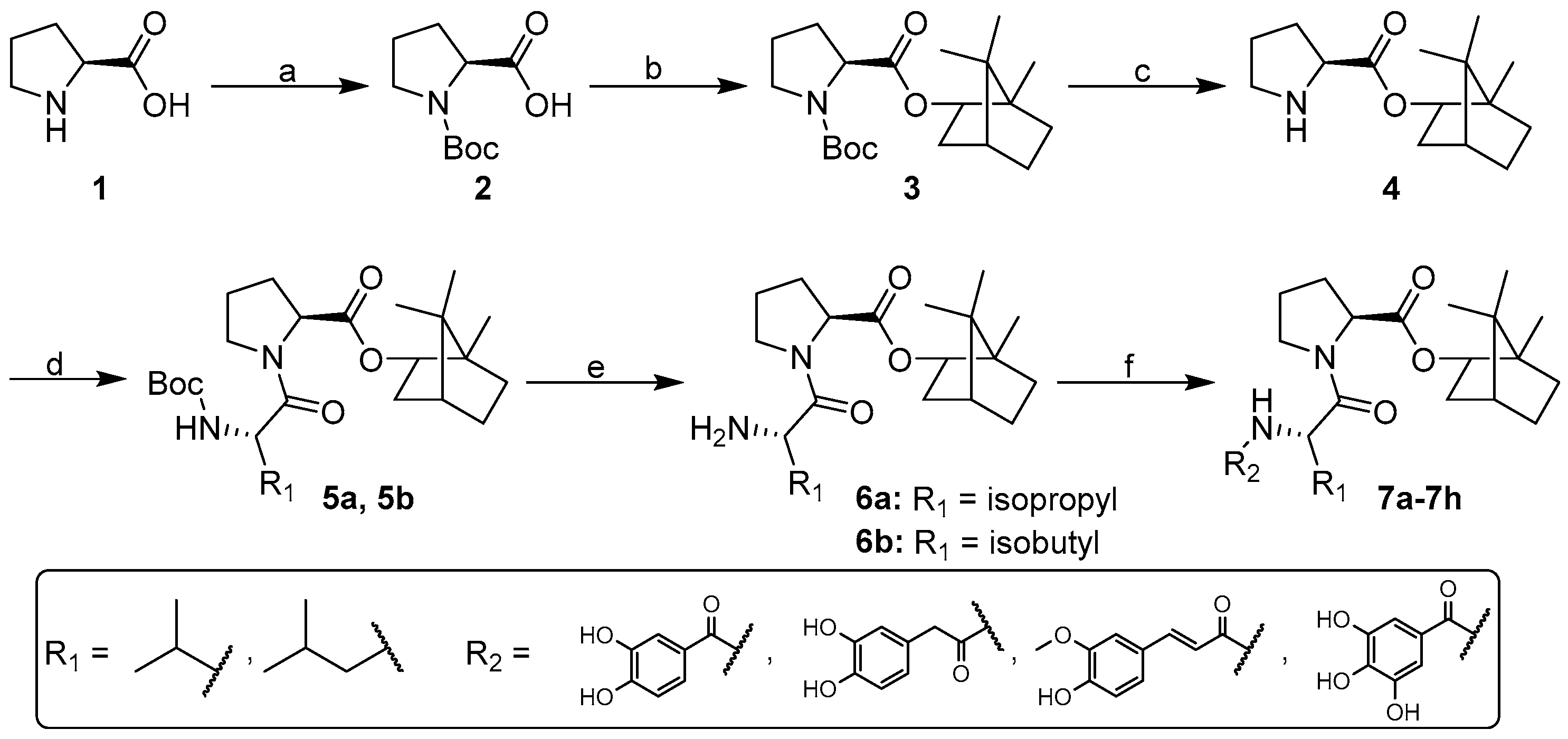
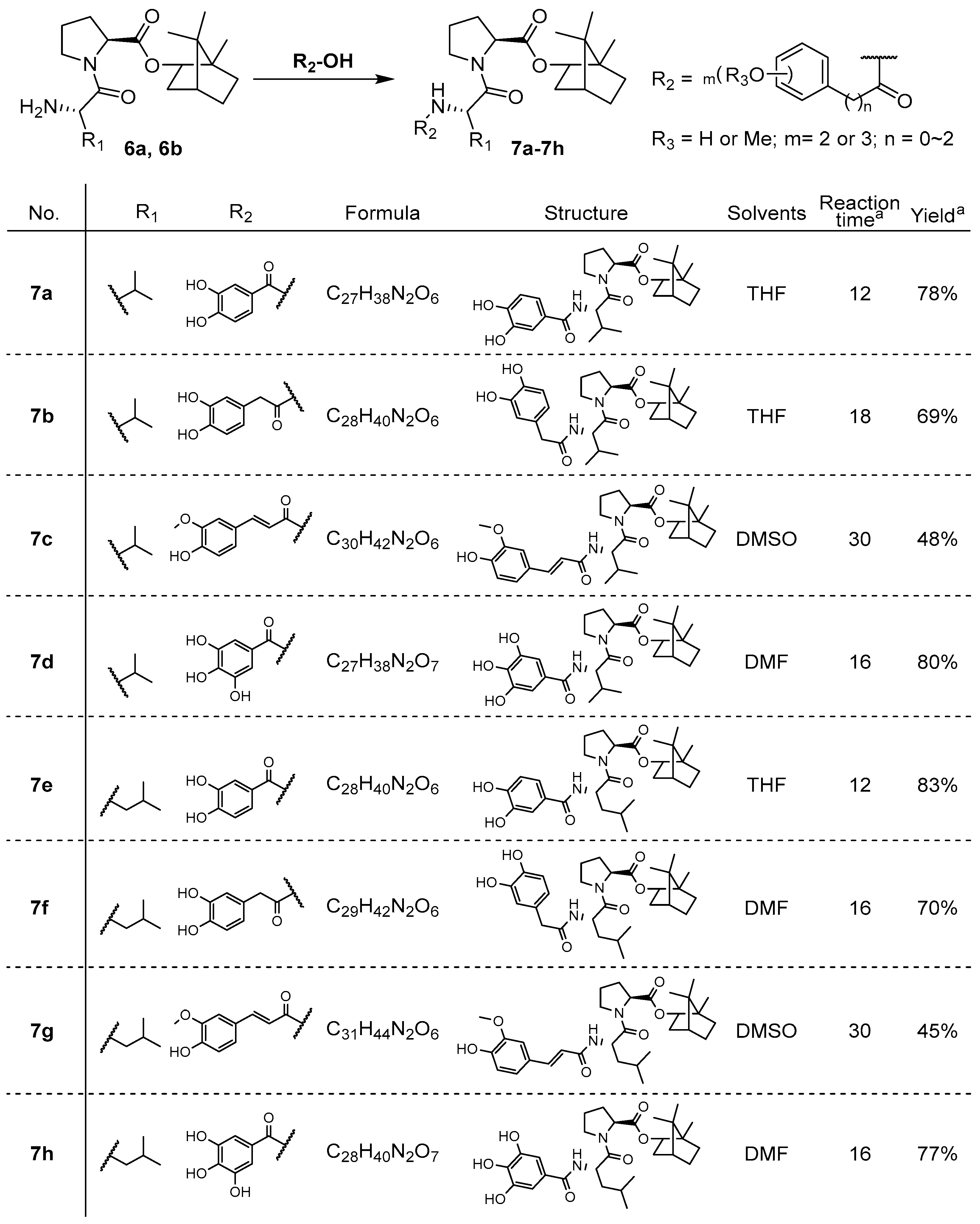
2.1. Synthesis and Preparation of 7a–7h
2.2. Biological Investigations
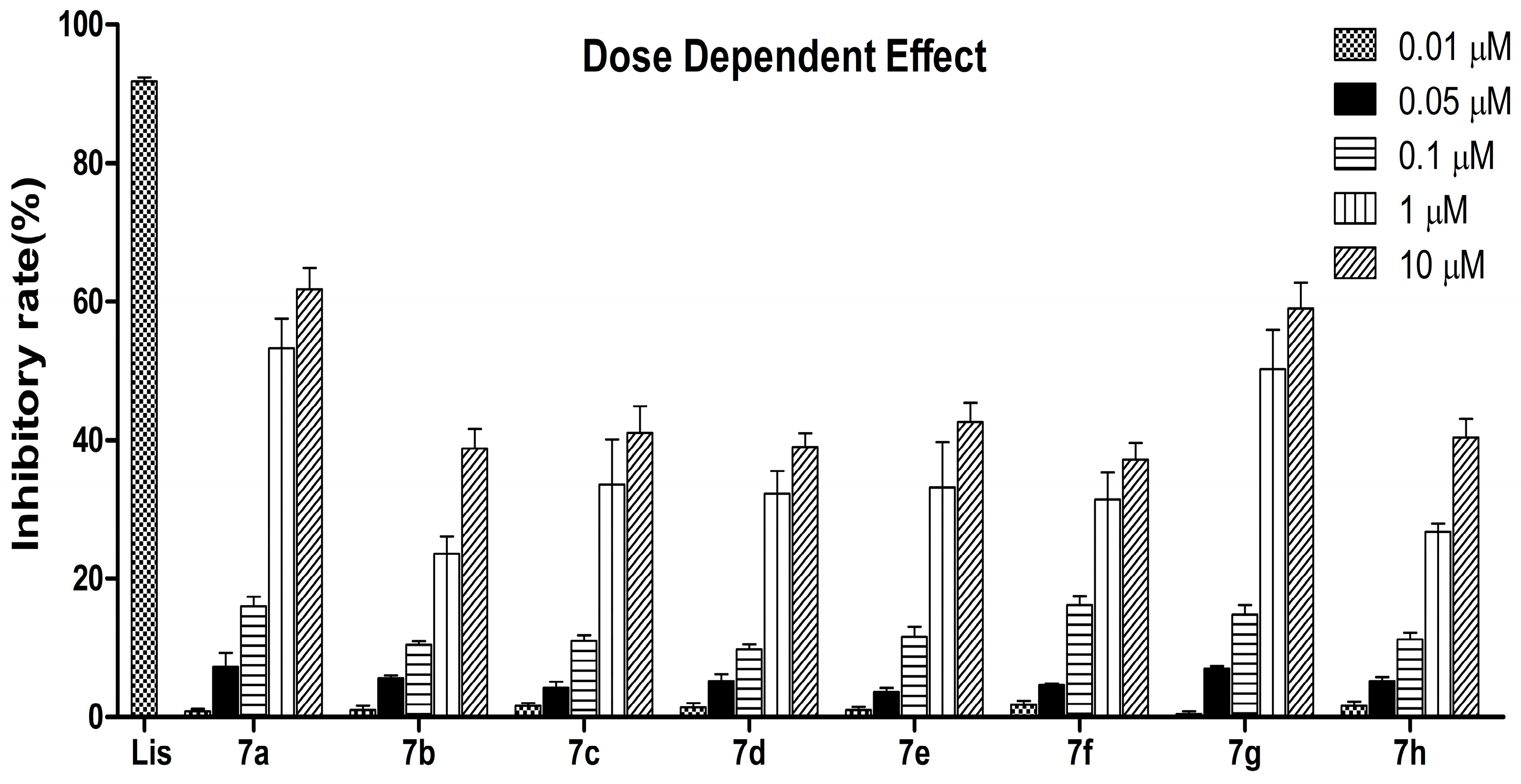
2.2.1. In Vitro ACE-Inhibitory Activity
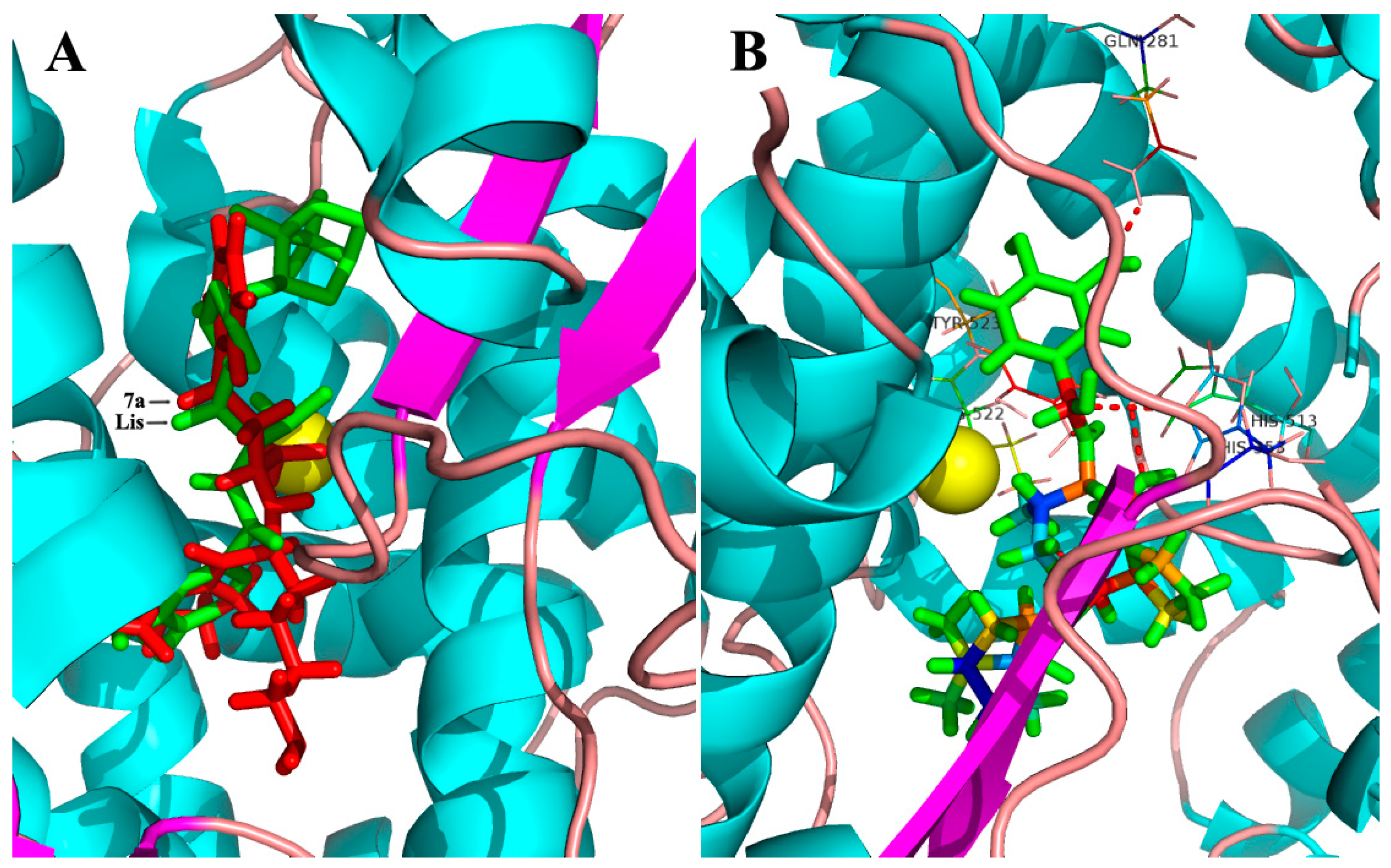
2.2.2. Molecular Docking Study
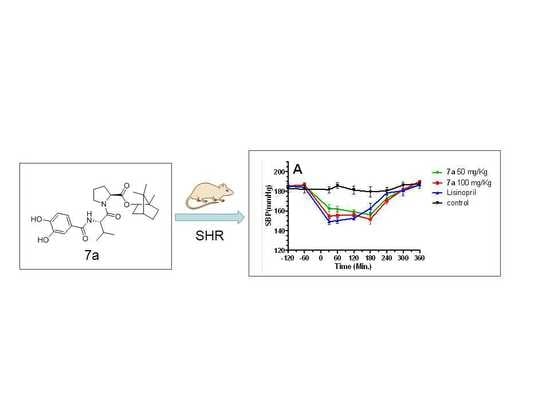
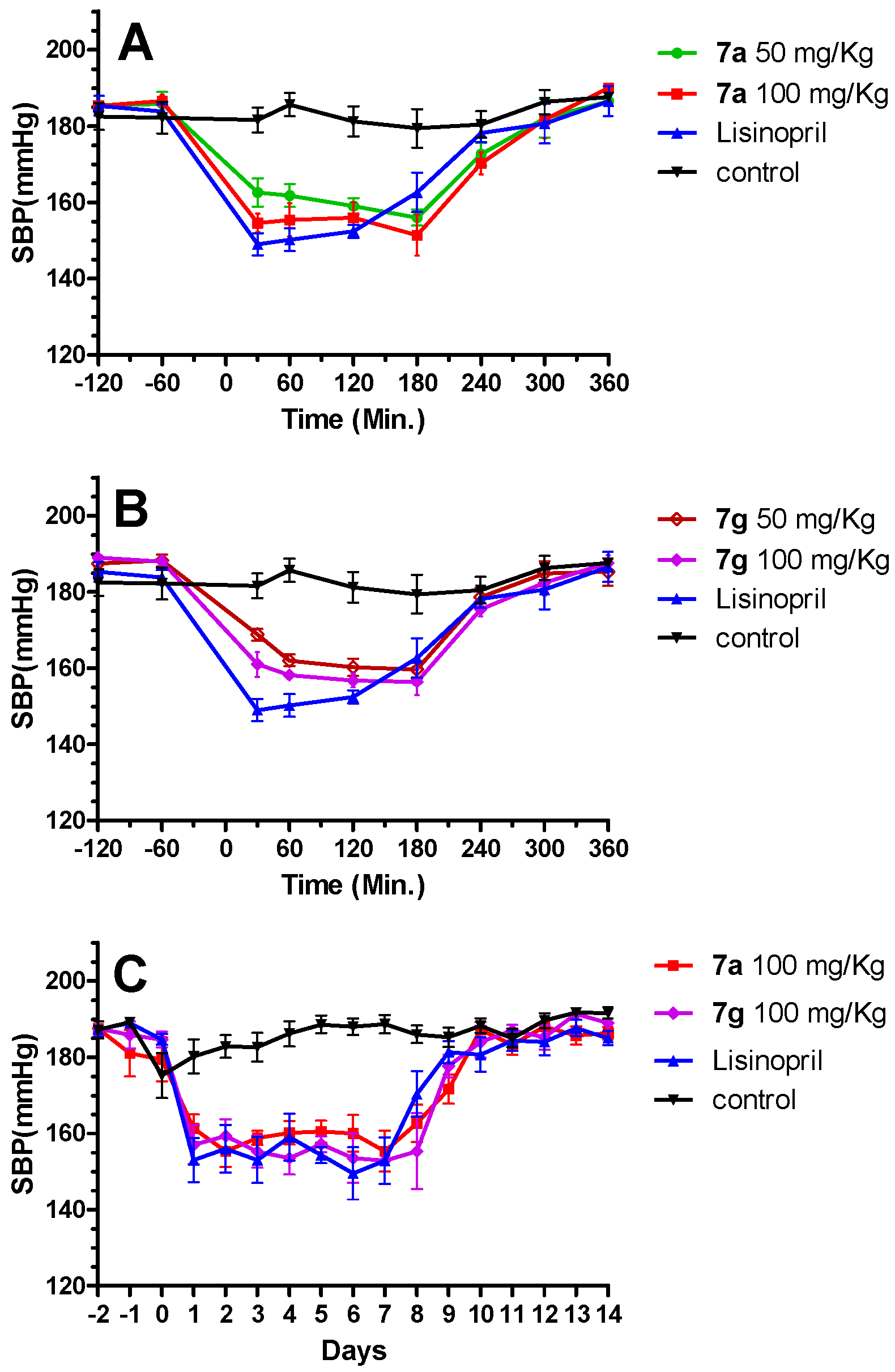
2.2.3. Antihypertensive Tests In Vivo
3. Results and Discussion
3.1. Chemistry
Compound Identification (7a–7h)
3.2. ACE-Inhibitory Activity In Vitro
3.3. ACE Molecular Docking
3.4. Antihypertensive Tests In Vivo
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Os, I.; Hjermann, I.; Nordby, G. Hypertension as a part of metabolic cardiovascular syndrome. Tidsskr. Nor. Laegeforen. 1991, 111, 3062–3064. [Google Scholar] [PubMed]
- Kearney, P.M.; Whelton, M.; Reynolds, K.; Muntner, P.; Whelton, P.K.; He, J. Global burden of hypertension: Analysis of worldwide data. Lancet 2005, 365, 217–223. [Google Scholar] [CrossRef]
- Wong, T.Y.; Wong, T.; Mitchell, P. The eye in hypertension. Lancet 2007, 369, 425–435. [Google Scholar] [CrossRef]
- Messerli, F.H.; Williams, B.; Ritz, E. Essential hypertension. Lancet 2007, 370, 591–603. [Google Scholar] [CrossRef]
- Anonymous. Angiotensin II receptor antagonists and heart failure: Angiotensin-converting-enzyme inhibitors remain the first-line option. Prescrire Int. 2005, 14, 180–186. [Google Scholar]
- Thurston, H. Angiotensin-converting enzyme inhibition as first-line treatment for hypertension. Clin. Exp. Pharmacol. Physiol. Suppl. 1992, 19, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Antonios, T.F.T.; MacGregor, G.A. Angiotensin converting enzyme inhibitors in hypertension: Potential problems. J. Hypertens. 1995, 13, S11–S16. [Google Scholar] [CrossRef]
- Israili, Z.H.; Hall, W.D. Cough and angioneurotic edema associated with angiotensin-converting enzyme inhibitor therapy. A review of the literature and pathophysiology. Ann. Intern. Med. 1992, 117, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Galardy, R.E. Inhibition of angiotensin-converting enzyme by phosphoramidates and polyphosphates. Biochemistry 1982, 21, 5777–5781. [Google Scholar] [CrossRef] [PubMed]
- Thorsett, E.D.; Harris, E.E.; Peterson, E.R.; Greenlee, W.J.; Patchett, A.A.; Ulm, E.H.; Vassil, T.C. Phosphorus-containing inhibitors of angiotensin-converting enzyme. Proc. Natl. Acad. Sci. USA 1982, 79, 2176–2180. [Google Scholar] [CrossRef] [PubMed]
- Galardy, R.E. Inhibition of angiotensin-converting enzyme with Nα-phosphoryl-l-alanyl-l-proline and Nα-phosphoryl-l-valyl-l-tryptophan. Biochem. Biophys. Res. Commun. 1980, 97, 94–99. [Google Scholar] [CrossRef]
- Galardy, R.E.; Kontoyiannidou-Ostrem, V.; Kortylewicz, Z.P. Inhibition of angiotensin converting enzyme by phosphonic amides and phosphonic acids. Biochemistry 1983, 22, 1990–1995. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-B.; Ni, Z.-Y.; Shi, Q.-W.; Dong, M.; Kiyota, H.; Gu, Y.-C.; Cong, B. Constituents from Salvia Species and Their Biological Activities. Chem. Rev. 2012, 112, 5967–6026. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Bi, H.; Xie, X.; Guo, J.; Wang, X.; Liu, D. Natural borneol enhances geniposide ophthalmic absorption in rabbits. Int. J. Pharm. 2013, 445, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fu, S.; Wei, N.; Hou, Y.; Zhang, X.; Cui, H. The effects of combined menthol and borneol on fluconazole permeation through the cornea ex vivo. Eur. J. Pharmacol. 2012, 688, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bala, M.; Qadar Pasha, M.A.; Bhardwaj, D.K.; Pasha, S. Novel peptidomimics as angiotensin-converting enzyme inhibitors: A combinatorial approach. Bioorg. Med. Chem. 2002, 10, 3685–3691. [Google Scholar] [CrossRef]
- Chen, C.H.; Lin, J.Y.; Lin, C.N.; Hsu, S.Y. Inhibition of Angiotensin-I-Converting Enzyme by Tetrahydroxyxanthones Isolated from Tripterospermum lanceolatum. J. Nat. Prod. 1992, 55, 691–695. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.A.; Sernia, C.; Brown, L. Ferulic Acid Improves Cardiovascular and Kidney Structure and Function in Hypertensive Rats. J. Cardiovasc. Pharmacol. 2013, 61, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Actis-Goretta, L.; Ottaviani, J.I.; Fraga, C.G. Inhibition of Angiotensin Converting Enzyme Activity by Flavanol-Rich Foods. J. Agric. Food Chem. 2005, 54, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Siltari, A.; Viitanen, R.; Kukkurainen, S.; Vapaatalo, H.; Valjakka, J. Does the cis/trans configuration of peptide bonds in bioactive tripeptides play a role in ACE-1 enzyme inhibition? Biol. Targets Ther. 2014, 8, 59–65. [Google Scholar]
- Vermeirssen, V.; van Camp, J.; Verstraete, W. Bioavailability of angiotensin I converting enzyme inhibitory peptides. Br. J. Nutr. 2004, 92, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.-G.; Kim, S.-K. Structure and activity of angiotensin I converting enzyme inhibitory peptides derived from Alaskan pollack skin. J. Biochem. Mol. Biol. 2002, 35, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 7a–7h are available from the author Yajun Bai. |
| Compound | Compound Structure | IC50 (μM) |
|---|---|---|
| Lisinopril |  | <0.01 |
| 7a |  | 6.76 ± 0.96 |
| 7b |  | 13.94 ± 1.61 |
| 7c |  | 11.88 ± 1.82 |
| 7d |  | 13.46 ± 2.02 |
| 7e |  | 10.58 ± 1.72 |
| 7f |  | 13.97 ± 1.84 |
| 7g |  | 6.88 ± 0.85 |
| 7h |  | 13.25 ± 2.47 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Bai, Y.; He, X.; Bai, Y.; Liu, P.; Zhao, Z.; Chen, X.; Zheng, X. Design, Synthesis, and Evaluation of Novel Phenolic Acid/Dipeptide/Borneol Hybrids as Potent Angiotensin Converting Enzyme (ACE) Inhibitors with Anti-hypertension Activity. Molecules 2017, 22, 1739. https://doi.org/10.3390/molecules22111739
Sun Y, Bai Y, He X, Bai Y, Liu P, Zhao Z, Chen X, Zheng X. Design, Synthesis, and Evaluation of Novel Phenolic Acid/Dipeptide/Borneol Hybrids as Potent Angiotensin Converting Enzyme (ACE) Inhibitors with Anti-hypertension Activity. Molecules. 2017; 22(11):1739. https://doi.org/10.3390/molecules22111739
Chicago/Turabian StyleSun, Ying, Yujun Bai, Xirui He, Yajun Bai, Pei Liu, Zefeng Zhao, Xufei Chen, and Xiaohui Zheng. 2017. "Design, Synthesis, and Evaluation of Novel Phenolic Acid/Dipeptide/Borneol Hybrids as Potent Angiotensin Converting Enzyme (ACE) Inhibitors with Anti-hypertension Activity" Molecules 22, no. 11: 1739. https://doi.org/10.3390/molecules22111739