Phenolic Compounds Present Schinus terebinthifolius Raddi Influence the Lowering of Blood Pressure in Rats
Abstract
:1. Introduction
2. Results and Discussion
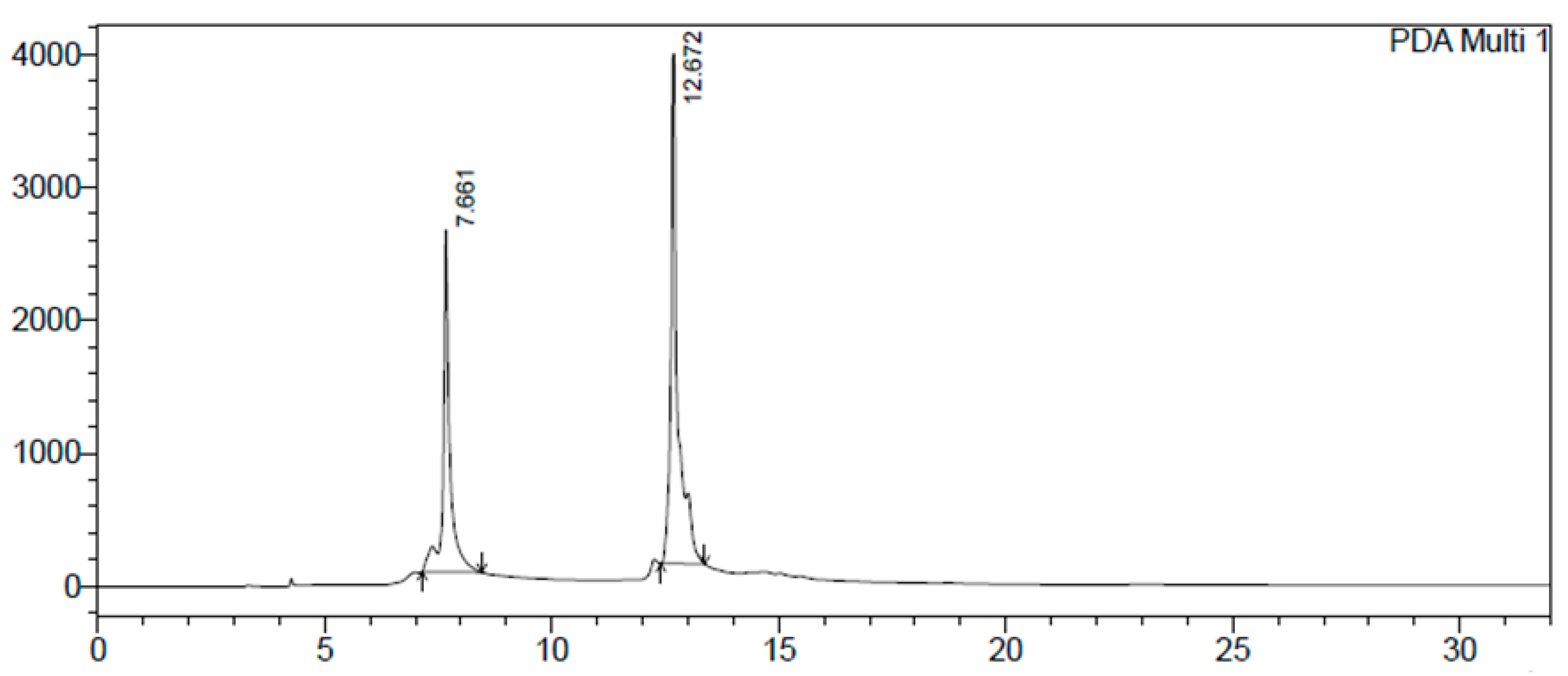
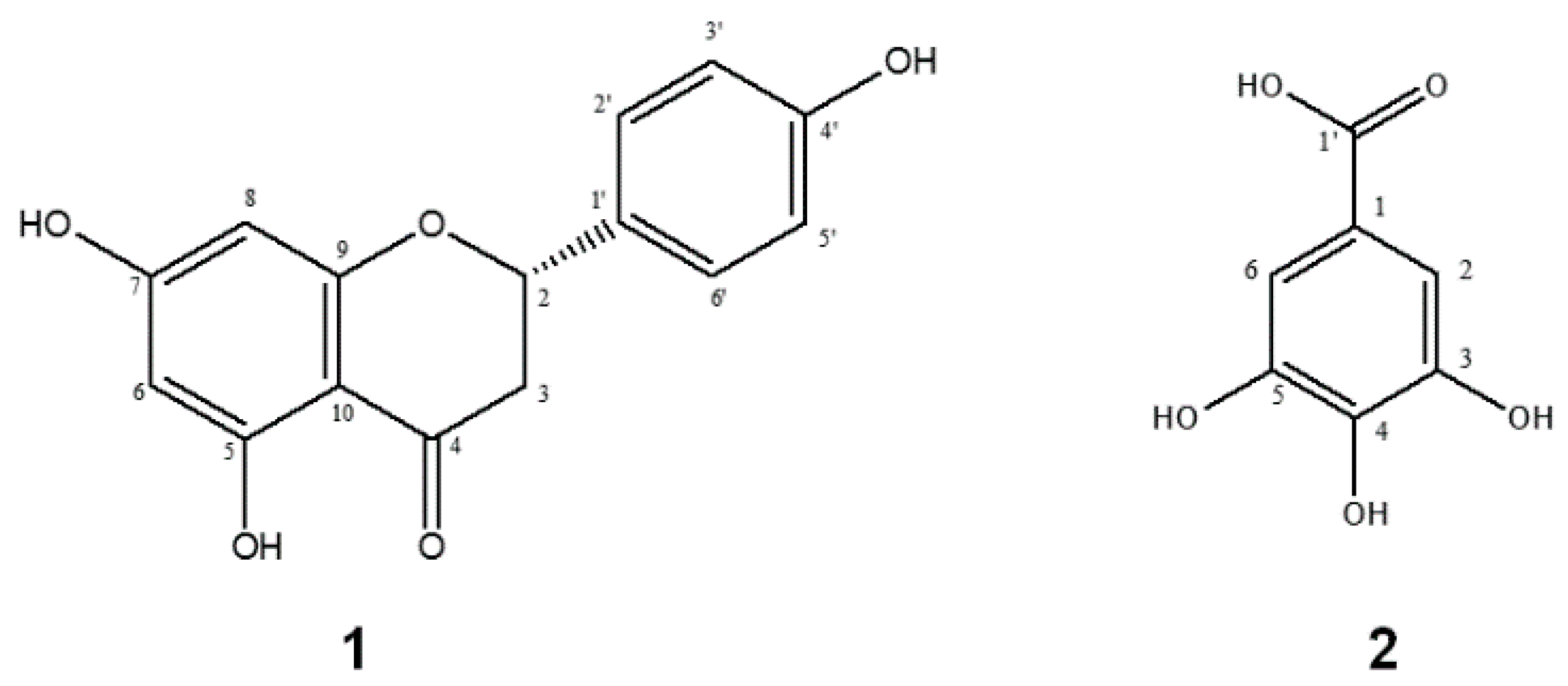
2.1. Identification and Structure Elucidation of the Major Compounds
2.2. Total Phenolic Compounds, Total Flavonoid Content and Antioxidant Activity
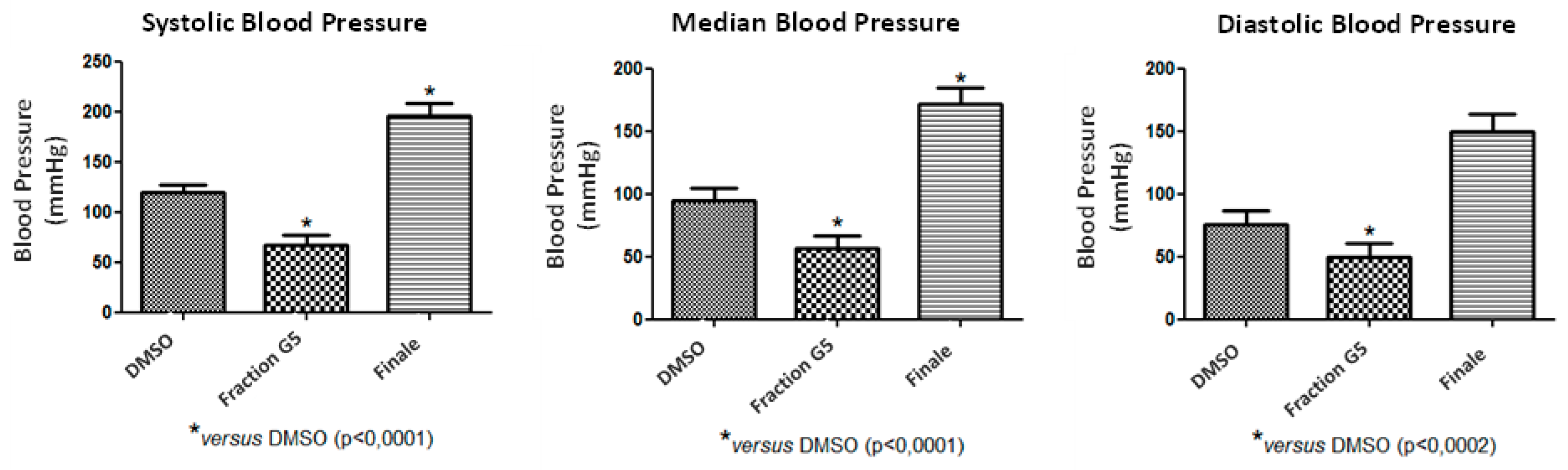
2.3. In Vivo Blood Pressure Assessment
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Chemicals
3.3. Animals
3.4. Plant Material
3.5. Extraction and Isolation
3.6. Total Phenolic Compounds
3.7. Total Flavonoid Content
3.8. Antioxidant Activity
3.9. In Vivo Blood Pressure Assessment
3.10. Rotarod Test
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bae, J.W.; Kim, D.H.; Lee, W.W.; Kim, H.Y.; Son, C.G. Characterizing the human equivalent dose of herbal medicines in animal toxicity studies. J. Ethnopharmacol. 2015, 162, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Holtmann, G.; Talley, N.J. Herbal medicines for the treatment of functional and inflammatorybowel disorders. Clin. Gastroenterol. Hepatol. 2015, 13, 422–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farzaneh, V.; Carvalho, I.S. A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind. Crops Prod. 2015, 65, 247–258. [Google Scholar] [CrossRef]
- Corrêa, M.P. Dicionário de Plantas Úteis do Brasil e das Exóticas Cultivadas; Ministério da Agricultura, Instituto Brasileiro de Desenvolvimento Florestal: Rio de Janeiro, RJ, Brazil, 1984; Volume 6, pp. 170–171.
- DiCiaula, M.C.; Lopes, G.C.; Scarminio, I.S.; De Mello, J.C.P. Optimization of solvent mixtures for extraction from bark of Schinus terebinthifolius by a statistical mixture-design technique and development of a UV-VIS spectrophotometric method for analysis of total polyphenols in the extract. Quim. Nova 2014, 37, 158–163. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Novel Functional Food Ingredients from Marine Sources ScienceDirect Novel functional food ingredients from marine sources. Curr. Opin. Food Sci. 2015, 2, 123–129. [Google Scholar] [CrossRef]
- Haytowitz, D.B.; Bhagwat, S.; Holden, J.M. Sources of variability in the flavonoid content of foods. Procedia Food Sci. 2013, 2, 46–51. [Google Scholar] [CrossRef]
- Kassem, M.E.S.; El-Desoky, S.K.; Sharaf, M. Biphenyl esters and biflavonoids from the fruits of Schinus terebinthifolius. Chem. Nat. Compd. 2004, 40, 447–450. [Google Scholar] [CrossRef]
- Penso, J.; Cordeiro, K.C.F.A.; Da Cunha, C.R.M.; Da Silva Castro, P.F.; Martins, D.R.; Lião, L.M.; Rocha, M.L.; De Oliveira, V. Vasorelaxant activity of 7-β-O-glycosides biosynthesized from flavonoids. Eur. J. Pharmacol. 2014, 733, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.S.; Kasoju, N.; Luthra, A.; Singh, A.; Sharanabasava, H.; Sahu, A.; Bora, U. Indian medicinal herbs as sources of antioxidants. Food Res. Int. 2008, 41, 1–15. [Google Scholar] [CrossRef]
- Kanaze, F.I.; Kokkalou, E.; Georgarakis, M.; Niopas, I. A validated solid-phase extraction HPLC method for the simultaneous determination of the citrus flavanone aglycones hesperetin and naringenin in urine. J. Pharm. Biomed. Anal. 2004, 36, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Cheng, Y.; Tian, Y.; Zhang, Z.J. A validated solid-phase extraction HPLC method for the simultaneous determination of gallic acid, catechin and epicatechin in rhubarb decoction. Chin. J. Nat. Med. 2012, 10, 275–278. [Google Scholar] [CrossRef]
- Du, Q.Z.; Jerz, G.; Winterhalter, P. Preparation of three flavonoids from the bark of Salix alba by high-speed countercurrent chromatographic separation. J. Liq. Chromatogr. Relat. Technol. 2004, 27, 3257–3264. [Google Scholar] [CrossRef]
- Santana, J.S.; Sartorelli, P.; Lago, J.H.G.; Matsuo, A.L. Isolamento e avaliação do potencial citotóxico de derivados fenólicos de Schinus terebinthifolius raddi (anacardiaceae). Quim. Nova 2012, 35, 2245–2248. [Google Scholar] [CrossRef]
- Sereniki, A.; Linard-Medeiros, C.F.B.; Silva, S.N.; Silva, J.B.R.; Peixoto Sobrinho, T.J.S.; da Silva, J.R.; Alvesa, L.D.S.; Smailic, S.S.; Wanderley, A.G.; Lafayette, S.S.L. Schinus terebinthifolius administration prevented behavioral and biochemical alterations in a rotenone model of Parkinson’s disease. Braz. J. Pharmacogn. 2016, 26, 240–245. [Google Scholar] [CrossRef]
- Feuereisen, M.M.; Gamero Barraza, M.; Zimmermann, B.F.; Schieber, A.; Schulze-Kaysers, N. Pressurized liquid extraction of anthocyanins and biflavonoids from Schinus terebinthifolius Raddi: A multivariate optimization. Food Chem. 2017, 214, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Skopp, G.; Schwenker, G. Biflavonoids from Schinus terebinthifolius Raddi (Anacardiaceae). Z. Naturforsch. B 1986, 41, 1479–1482. [Google Scholar] [CrossRef]
- Ono, M.; Yamashita, M.; Mori, K.; Masuoka, C.; Eto, M.; Kinjo, J.; Ikeda, T.; Yoshimitsu, H.; Nohara, T. Sesquiterpenoids, triterpenoids, and flavonoids from the fruits of Schinus molle. Food Sci. Technol. Res. 2008, 14, 499–508. [Google Scholar] [CrossRef]
- Yueqin, Z.; Recio, M.C.; Máñez, S.; Giner, R.M.; Cerdá-Nicolás, M.; Ríos, J.L. Isolation of two triterpenoids and a biflavanone with anti-inflammatory activity from Schinus molle fruits. Planta Med. 2003, 69, 893–898. [Google Scholar] [PubMed]
- Uliana, M.P.; Fronza, M.; da Silva, A.G.; Vargas, T.S.; de Andrade, T.U.; Scherer, R. Composition and biological activity of Brazilian rose pepper (Schinus terebinthifolius Raddi) leaves. Ind. Crops Prod. 2016, 83, 235–240. [Google Scholar] [CrossRef]
- Ribeiro, P.; Fernandez, L.; Loureiro, M.; Simões, R.; de Castro, R.; D′Sousa′ Costa, C. Phytochemical screening, antioxidant and antibacterial activities of extracts prepared from different tissues of Schinus terebinthifolius Raddi that occurs in the coast of Bahia, Brazil. Pharmacogn. Mag. 2015, 11, 607. [Google Scholar] [CrossRef] [PubMed]
- Bulla, M.K.; Hernandes, L.; Baesso, M.L.; Nogueira, A.C.; Bento, A.C.; Bortoluzzi, B.B.; Serra, L.Z.; Cortez, D.A.G. Evaluation of photoprotective potential and percutaneous penetration by photoacoustic spectroscopy of the Schinus terebinthifolius Raddi extract. Photochem. Photobiol. 2015, 91, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Tabaldi, L.A.; Vieira, M.C.; Zárate, N.A.H.; Formagio, A.S.N.; Pilecco, M.; Silva, L.R.; Santos, K.P.; Santos, L.A.C.; Cardoso, C.A.L. Biomass yield and flavonoid and phenol content of Schinus terebinthifolius cultivated in single or double row with poultry litter. Ciênc. Florest. 2016, 26, 787–796. [Google Scholar] [CrossRef]
- Fedel-Miyasato, L.E.S.; Kassuya, C.A.L.; Auharek, S.A.; Formagio, A.S.N.; Cardoso, C.A.L.; Mauro, M.O.; Cunha-Laura, A.L.; Monreal, A.C.D.; Vieira, M.C.; Oliveira, R.J. Evaluation of anti-inflammatory, immunomodulatory, chemopreventive and wound healing potentials from Schinus terebinthifolius methanolic extract. Braz. J. Pharmacogn. 2014, 24, 565–575. [Google Scholar] [CrossRef]
- El-Massry, K.F.; El-Ghorab, A.H.; Shaaban, H.A.; Shibamoto, T. Chemical compositions and antioxidant/antimicrobial activities of various samples prepared from Schinus terebinthifolius leaves cultivated in Egypt. J. Agric. Food Chem. 2009, 57, 5265–5270. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Bernardes, N.R.; Heggdorne-Araújo, M.; Borges, I.F.J.C.; Almeida, F.M.; Amaral, E.P.; Lasunskaia, E.B.; Muzitano, M.F.; Oliveira, D.B. Nitric oxide production, inhibitory, antioxidant and antimycobacterial activities of the fruits extract and flavonoid content of Schinus terebinthifolius. Braz. J. Pharmacogn. 2014, 24, 644–650. [Google Scholar] [CrossRef]
- Šamec, D.; Piljac-Žegarac, J. Postharvest stability of antioxidant compounds in hawthorn and cornelian cherries at room and refrigerator temperatures-Comparison with blackberries, white and red grapes. Sci. Hortic. (Amst.) 2011, 131, 15–21. [Google Scholar] [CrossRef]
- Torres-Piedra, M.; Figueroa, M.; Hernández-Abreu, O.; Ibarra-Barajas, M.; Navarrete-Vázquez, G.; Estrada-Soto, S. Vasorelaxant effect of flavonoids through calmodulin inhibition: Ex vivo, in vitro, and in silico approaches. Bioorg. Med. Chem. 2011, 19, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Lobo De Andrade, D.M.; Reis, C.D.F.; Castro, P.F.D.S.; Borges, L.L.; Amaral, N.O.; Torres, I.M.S.; Rezende, S.G.; Gil, E.D.S.; Cardoso Da Conceicao, E.; Pedrino, G.R.; et al. Vasorelaxant and hypotensive effects of jaboticaba fruit (Myrciaria cauliflora) extract in rats. Evid. Based Complement. Altern. Med. 2015, 2015, 696135. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Li, Q.; Bi, K. Bioactive flavonoids in medicinal plants: Structure, activity and biological fate. Asian J. Pharm. Sci. 2017, in press. [Google Scholar] [CrossRef]
- Tchikaya, F.O.; Bantsielé, G.B.; Kouakou-Siransy, G.; Datté, J.Y.; Yapo, P.A.; Zirihi, N.G.; Offoumou, M.A. Anacardium occidentale Linn. (Anacardiaceae) stem bark extract induces hypotensive and cardio-inhibitory effects in experimental animal models. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Ojewole, J.A. Hypoglycaemic and hypotensive effects of Harpephyllum caffrum Bernh ex CF Krauss (Anacardiaceae) stem—Bark aqueous extract in rats. Cardiovasc. J. S. Afr. 2006, 17, 67–72. [Google Scholar] [PubMed]
- Belemtougri, R.G.; Constantin, B.; Cognard, C.; Raymond, G.; Sawadogo, L. Effects of Sclerocarya birrea (A. rich) hochst (Anacardiaceae) leaf extracts on calcium signalling in cultured rat skeletal muscle cells. J. Ethnopharmacol. 2001, 76, 247–252. [Google Scholar] [CrossRef]
- Lopes, S.C.; Da Silva, A.V.L.; Arruda, B.R.; Morais, T.C.; Rios, J.B.; Trevisan, M.T.S.; Rao, V.S.; Santos, F.A. Peripheral antinociceptive action of mangiferin in mouse models of experimental pain: Role of endogenous opioids, KATP-channels and adenosine. Pharmacol. Biochem. Behav. 2013, 110, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Seaman, R.L. Effects of acute systemic 3-nitropropionic acid administration on rat activity and acoustic startle. Neurosci. Lett. 2000, 280, 183–186. [Google Scholar] [CrossRef]
- Association of Official Analysis Chemists (AOAC). Official Methods of Analysis of the Association of Official Analysis Chemists; AOAC: Gaithersburg, MD, USA, 1995; p. 957. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enziymol. 1999, 299, 152–178. [Google Scholar]
- Woisky, R.G.; Salatino, A. Analysis of propolis: Some parameters and procedures for chemical quality control. J. Apic. Res. 1998, 37, 99–105. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1 and 2 are available from the authors. |

| Total Phenolic Compounds (mg GAE */g) | Total Flavonoid Content (mg RE **/g) | Antioxidant Activity (%) | |||
|---|---|---|---|---|---|
| 0.01 mg/mL | 0.01 mg/mL | 0.01 mg/mL | |||
| Fraction G5 | 659.21 ± 6.05 | 140.69 ± 9.44 | 77.5 ± 2.57 | 75.0 ± 6.78 | 69.2 ± 6.47 |
| Quercetin | 1214.71 ± 9.51 | 1009.27 ± 7.85 | 77.2 ± 0.73 | 74.1 ± 3.82 | 71.0 ± 3.63 |
| BHT *** | 76.4 ± 1.98 | 75.0 ± 4.94 | 66.3 ± 4.20 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Lima Glória, L.; Barreto de Souza Arantes, M.; Menezes de Faria Pereira, S.; De Souza Vieira, G.; Xavier Martins, C.; Ribeiro de Carvalho Junior, A.; Antunes, F.; Braz-Filho, R.; José Curcino Vieira, I.; Leandro da Cruz, L.; et al. Phenolic Compounds Present Schinus terebinthifolius Raddi Influence the Lowering of Blood Pressure in Rats. Molecules 2017, 22, 1792. https://doi.org/10.3390/molecules22101792
De Lima Glória L, Barreto de Souza Arantes M, Menezes de Faria Pereira S, De Souza Vieira G, Xavier Martins C, Ribeiro de Carvalho Junior A, Antunes F, Braz-Filho R, José Curcino Vieira I, Leandro da Cruz L, et al. Phenolic Compounds Present Schinus terebinthifolius Raddi Influence the Lowering of Blood Pressure in Rats. Molecules. 2017; 22(10):1792. https://doi.org/10.3390/molecules22101792
Chicago/Turabian StyleDe Lima Glória, Lorena, Mariana Barreto de Souza Arantes, Silvia Menezes de Faria Pereira, Guilherme De Souza Vieira, Camilla Xavier Martins, Almir Ribeiro de Carvalho Junior, Fernanda Antunes, Raimundo Braz-Filho, Ivo José Curcino Vieira, Larissa Leandro da Cruz, and et al. 2017. "Phenolic Compounds Present Schinus terebinthifolius Raddi Influence the Lowering of Blood Pressure in Rats" Molecules 22, no. 10: 1792. https://doi.org/10.3390/molecules22101792






