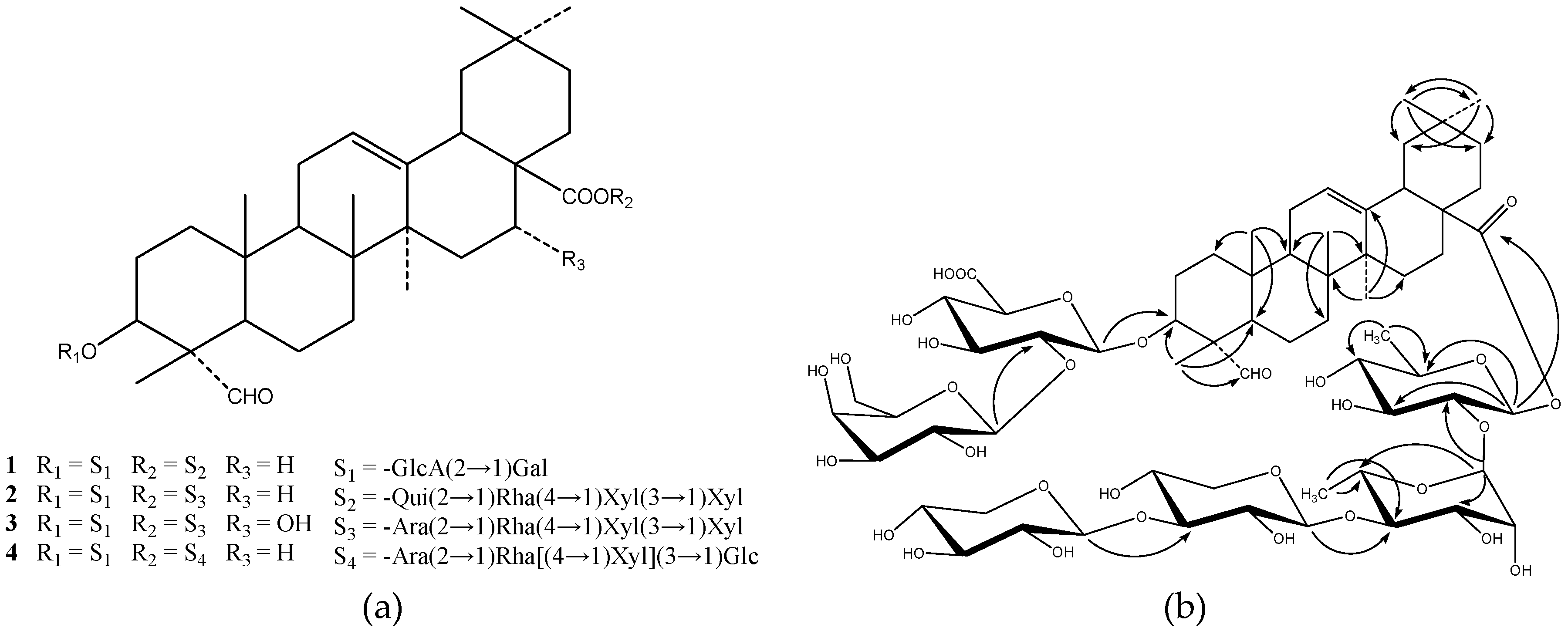
2. Results and Discussion
The crude saponins prepared from the 70% EtOH extract of
H. japonica herbs through a D101 polyporous resin column were subjected to silica gel chromatographies and semi-preparative HPLC to yield two new triterpenoid saponins, named as hylomeconoside A (
1) and B (
2), and two known triterpenoid saponins identified as dubioside C (
3) [
8] and lucyoside P (
4) [
9] on the basis of spectroscopic and chemical evidence.
Hylomeconoside A (
1) was obtained as a white amorphous solid. The molecular formula of
1 was determined as C
64H
100O
31 by high-resolution HRESI-MS which indicated a [M + H]
+ ion at
m/
z 1365.6318. Acid hydrolysis of
1 afforded an aglycone which was identified as gypsogenin on the basis of the
1H- and
13C-NMR data [
10]. The sugars obtained from the saponin hydrolysate were identified as
d-xylose,
l-rhamnose,
d-quinovose,
d-glucuronic acid and
d-galactose based on GC analysis of their chiral derivatives. The
1H-NMR spectrum revealed signals due to six quaternary methyls at δ
H 0.68, 0.76, 0.79, 0.90, 1.14, 1.28, an olefinic proton at δ
H 5.30 (br. s), an aldehyde proton at δ
H 9.83 (s), and six anomeric protons at δ
H 6.34 (br s), 5.98 (d,
J = 8.0 Hz), 5.11 (d,
J = 7.5 Hz) , 5.08 (d,
J = 7.4 Hz), 4.94 (d,
J = 6.8 Hz), and 4.72 (d,
J = 6.3 Hz). The
13C-NMR spectrum displayed signals due to six quaternary carbon at δ
C 30.9, 36.4, 40.4, 42.4, 47.3, and 55.2, an oxygen-bearing methine carbon at δ
C 83.5, a set of olefinic carbons at δ
C 122.7 and 144.2, an ester carbonyl carbon at δ
C 176.7, an aldehyde carbon at δ
C 210.2, and six anomeric carbons at δ
C 94.5, 101.4, 103.4, 105.9, 106.2, and 106.9. All the above spectral information suggested that the aglycone moiety of
1 was gypsogenin and the chemical shift of the carbonyl C-28 (δ
C 176.7) and the deshielded carbon C-3 (δ
C 83.5) of gypsogenin indicated that
1 was the 3,28-bisdesmoside of gypsogenin, having six monosaccharide units.
The six anomeric proton signals at δH 6.34 (br s), 5.98 (d, J = 8.0 Hz), 5.11 (d, J = 7.5 Hz), 5.08 (d, J = 7.4 Hz), 4.94 (d, J = 6.8 Hz), and 4.72 (d, J = 6.3 Hz) were correlated with anomeric carbon signals at δC 101.4, 94.5, 106.2,105.9, 106.9, 103.4, respectively, and the analysis of 1H-1H COSY, HMQC, HMBC, DEPT NMR and spin-spin couplings in 1H-NMR of 1 allowed the identification of one α-l-rhamnopyranosyl (Rha), one β-d-quinovopyranosyl (Qui), one β-d-glucuronopyranosyl (GlcA), one β-d-galactopyranosyl (Gal) and two β-d-xylopyranosyl (Xyl) moieties. The β-anomeric configurations for d-xylopyranosyl, d-quinovopyranosyl, d-glucuronopyranosyl and d-galactopyranosyl moieties were determined by their large 3JH1-H2 coupling constants of 6–8 Hz and the α-anomeric configuration for l-rhamnopyranosyl unit was determined by its small 3JH1-H2 coupling constant.
One Xyl unit, Xyla, identified starting from anomeric signals at δH 5.08 (H-1 of Xyla) and δC 105.9 (C-1 of Xyla), was identified to be in the terminal position, as observed by its 13C-NMR chemical shifts. Another Xyl unit, Xylb, identified starting from anomeric signals at δH 4.94 (H-1 of Xylb) and δC 106.9 (C-1 of Xylb), was substituted at the position of C-3 of Xylb based on the deshielding of C-3 of Xylb (δC 87.2), and the Xyla unit was attached to this position based on the long-range correlation observed in the HMBC experiment between signals at δH 5.08 (H-1 of Xyla) and δC 87.2 (C-3 of Xylb). The methyl doublet at δH 1.66 (3H, d, J = 6.4 Hz) and the typical broad single of anomeric proton at δH 6.34 (br s) were characteristic of Rha unit, and the deshielding of C-4 of Rha (δC 85.3) indicated a substitution at the position of C-4 of Rha, and the long-range correlation between signals at δH 4.94 (H-1 of Xylb) and δC 85.3 (C-4 of Rha) indicated that Xylb was attached to the position of C-4 of Rha. The Qui unit was identified starting from the deshielded anomeric proton at δH 5.98 (J = 8.0 Hz) and characterized by its methyl doublet at δH 1.42 (3H, d, J = 6.0 Hz). The deshielding of anomeric proton and the chemical shift of anomeric carbon at δC 94.5 suggested that Qui was attached by an ester linkage to the C-28 carboxylic group of the aglycone, which was further confirmed by the long-range correlation observed in the HMBC experiment between signals at δH 5.98 (H-1 of Qui) and δC 176.7 (C-28 of gypsogenin). Qui was substituted at the position of C-2 of Qui as observed by its deshielded H-2 of of Qui (δH 4.23) and the long-range correlation observed in the HMBC experiment between signals of the Rha anomeric proton (δH 6.34) and C-2 of Qui (δC 76.6). Therefore, the sequencing of the ester chain was obtained by analysis of HMBC experiment which showed cross-peaks between H-1 of Rha (δH 6.34) and C-2 of Qui (δC 76.6), between H-1 of Xylb (inner xylose) (δH 4.94) and C-4 of Rha (δC 85.3), and between H-1 of Xyla (the terminal xylose) (δH 5.08) and C-3 of Xylb (δC 87.2), and thus the ester chain was a tetrasaccharide, β-d-xylopyranosyl-(1→3)-β-d-xylopyranosyl-(1→4)-α-l-rhamnopyranosyl-(1→2)-β-d-quinovopyranosyl unit.
The Gal unit, identified starting from anomeric signals at δH 5.11 (H-1 of Gal) and δC 106.2 (C-1 of Gal), was identified to be in terminal position, as observed by its 13C-NMR chemical shifts. Starting from the anomeric proton at δH 4.72 (d, J = 6.3 Hz), a GlcA unit was identified with its carbonyl C-6 at δC 173.7. The deshielding of C-2 of GlcA (δC 82.1) indicated a substitution of glucuronic acid. Observation of long-range proton-carbon correlations in the HMBC spectrum between the anomeric proton of GlcA (δH 4.49) and C-3 of gypsogenin (δC 83.5) and between the anomeric proton of Gal (δH 5.11) and C-2 of GlcA (δC 82.1) indicated a disaccharide chain attached at C-3 of gypsogenin, β-d-galactopyranosyl-(1→2)-β-d-glucuronopyranosyl unit.
The complete assignment of the signals of
1 was based on DEPT
13C-NMR and 2D NMR of
1H-
1H COSY, HMQC and HMBC. All the data of
1H-,
13C-NMR and 2D-NMR of
1 see
Table 1, and the key correlations in HMBC NMR and the structure of
1 see
Figure 1. In conclusion, compound
1 was identified as 3-
O-β-
d-galactopyranosyl-(1→2)-β-
d-glucuronopyranosyl-gypsogenin-28-
O-β-
d-xylo-pyranosyl-(1→3)-β-
d-xylopyranosyl-(1→4)-α-
l-rhamnopyranosyl-(1→2)-β-
d-quinovopyranoside.
Hylomeconoside B (
2) was obtained as a white amorphous solid. The molecular formula of
2 was determined as C
63H
98O
31 by high-resolution HRESI-MS which showed a [M + H]
+ ion at
m/
z 1351.6151. Acid hydrolysis of
2 afforded an aglycone which was identified as gypsogenin on the basis of the
1H- and
13C-NMR spectra [
10]. The sugars obtained from the saponin hydrolysate were identified as
l-arabinose,
d-xylose,
l-rhamnose,
d-glucuronic acid and
d-galactose based on GC analysis of their chiral derivatives. In comparative analysis of the
13C-NMR data of
2 with those of
1 and
3, it was found that the spectra data of the aglycone moiety of
2 are same as those of
1, and the spectra data of the sugar moiety of
2 are consistent with those of
3 (
Table 2), which suggested that the aglycone of
2 is gypsogenin and the structure of the sugar moiety is same as those of
3. By further analysis of spectral data with the similar method as above,
2 was identified as 3-
O-β-
d-galactopyranosyl-(1→2)-β-
d-glucuronopyranosyl-gypsogenin-28-
O-β-
d-xylopyranosyl-(1→3)-β-
d-xylopyranosyl-(1→4)-α-
l-rhamnopyranosyl-(1→2)-α-
l-arabinopyranoside.
Dubioside C (
3), one of active constituents of
Thladianthae dubiae roots, has been reported to possess analgesic and anti-inflammatory effects [
11,
12,
13] and lucyoside P (
4), one of active constituntes of
Luffa cylindrica fructus, has beneficial effects on intelligence [
14], therefore we have only carried out an activity assay on the new saponins
1 and
2. The cytotoxic activities of compounds
1 and
2 on MGC-803 (human gastric cancer), HL-60 (human promyelocytic leukemia), BEL-7404 (humanhepatoma carcinoma), MCF-7 (human breast cancer), and SPC-A1 (lung adenocarcinoma) cells were assessed by the XTT (2,3-Bis-(2-methoxy-4-nitro-5-sulfophenyl)-2
H-tetrazolium-5-carboxanilide) reduction method.
The results (
Table 3) showed that compound
1 exhibited moderate cytotoxicity on MGC-803 and HL-60, with IC
50 values of 43.8 and 32.4 μg·mL
−1, respectively. Therefore, compound
1 is considered to be one of the antitumor principles in this plant.
3. Materials and Methods
3.1. General Information
NMR spectra were recorded on an AV-400 spectrometer (Bruker Corporation, Faellanden, Switzerland). HRESI-MS were recorded on a Bruker microOTOF-Q II mass spectrometer (Bruker Corporation, Bremen, Germany). Optical rotations were measured with a HORIBA SEPA-300 high-sensitive polarimeter (Horiba Ltd, Kyoto, Japan). HPLC was performed on a Shimadzu LC-10A system equipped with a SPD-10A detector (Shimadzu Corporation, Kyoto, Japan) and a Gemini 5 μm C18 110A column (250 mm × 10.00 mm, 5 μm, Phenomenex, Torrance, CA, USA). GC was performed an Agilent 7820A gas chromatograph with a quartz capillary column (30 mm × 0.32 mm × 0.25 μm, Agilent Technologies Inc., Santa Clara, CA, USA); detection, FID. Column chromatography was performed on silica gel (200–300 mesh, Qingdao Marine Chemical Inc., Qingdao, China), D101 polyporous resin (Tianjin Pesticide Co., LTD., Resin Branch, Tianjin, China). TLC was performed on glass precoated silica gel GF254 plates (Qingdao Haiyang Chemical Co., Ltd, Qingdao, China), detection under UV light or by heating after spraying with 10% H2SO4 in 95% EtOH. Distilled water was purchased from Hangzhou Wahaha Group Co., Ltd. (Hongzhou, China). Acetonitrile of chromatographic grade for HPLC was purchased from Fisher Scientific (Fair Lawn, NJ, USA). Other chemicals and reagents of analytical grade were from Beijing Chemical Works (Beijing, China).
The bioactivities were measured on a DNM-9602 enzyme immunoassay spectrophotometer (Beijing, China). RPMI 1640 medium was purchased from HyClone (Logan, UT, USA), PBS from Gibco company (Carlsbad, CA, USA), fetal bovine serum (FBS) from Zhejiang Tianhang Biotechnology Co., Ltd. (Hangzhou, China), XTT (2,3-Bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H- tetrazolium-5-carboxanilide) and phenazine methosulphate from Sigma-Aldrich Shanghai Trading Co. Ltd. (Shanghai, China).
The Hylomecon japonica herb was collected in the Changchun District of Jilin Province, China. They were identified by Jing-min Zhang of the School of Pharmaceutical Sciences, Jilin University. A voucher specimen (No. 2015062001) is deposited at the Herbarium of the School of Pharmaceutical Sciences, Jilin University.
3.2. Extraction and Isolation
Two kg of air-dried whole Hylomecon japonica herbs were extracted twice with 20 L of 70% aqueous ethanol solution (v/v) at room temperature. The extraction solution was concentrated under reduced pressure to remove ethanol, and the water concentrate was filtered and then passed through a D101 polyporous resin column eluting successively with H2O, 30% EtOH, 50% EtOH, 70% EtOH, and 95% EtOH. The crude saponin extracts were obtained from 50% aqueous ethanol eluate by vacuum distillation recovery and used for the next experiments. The crude saponin extracts were chromatographed on silica gel columns repeatedly eluted with EtOAc–MeOH–H2O (7:3:0.5) and futher purified by semi-preparative HPLC using acetonitrile and 0.1% formic acid solution in water as the mobile phase and the eluate was monitored at 207 nm, to yield compounds 1 (60 mg), 2 (50 mg), 3 (30 mg), and 4 (30 mg).
3.3. Characterization
Compound
1: White amorphous solid,
+5.0°(c 0.5, pyridine). HRESIMS,
m/
z: 1365.6318 [M + H]
+; calcd for C
64H
101O
31, 1365.6327.
1H-NMR (400 MHz, C
5D
5N) and
13C-NMR (100 MHz, C
5D
5N): see
Table 1.
Compound
2: White amorphous solid,
−15.0°(c 0.5, pyridine). HRESIMS,
m/
z: 1351.6151 [M + H]
+; calcd for C
63H
99O
31, 1351.6165.
1H-NMR (400 MHz, C
5D
5N) δ: 0.67 (3H, s, H-25), 0.80 (3H, s, H-26), 0.87 (6H, s, H-30, H-29), 1.12 (3H, s, H-27), 1.29 (3H, s, H-24), 1.61 (3H, d,
J = 4.8, Rha-6), 3.13 (1H, m, H-18), 3.34 (1H, t-like,
J = 10.0 Hz, Xyl(inner)-5α), 3.55 (1H, t-like,
J = 10.0 Hz, Xyl(terminal)-5α), 4.71 (1H, d,
J = 6.8 Hz, GluA-1), 5.01 (1H, d,
J = 6.8 Hz, Xyl(inner)-1), 5.08 (1H, d,
J = 7.2 Hz, Gal-1), 5.10 (1H, d,
J = 7.6 Hz, Xyl(terminal)-1), 5.29 (1H, br. s, H-12), 5.66 (1H, br. s, Rha-1), 6.34 (1H, br. s, Ara-1), 9.82 (1H, s, H-23).
13C-NMR (100 MHz, C
5D
5N): see
Table 2.
Compound
3: White amorphous solid,
−29.2°(c 0.5, pyridine). HRESIMS,
m/
z: 1367.6101 [M + H]
+; calcd for C
63H
99O
32, 1367.6114.
1H-NMR (400 MHz, C
5D
5N) δ: 0.70 (3H, s, H-25), 0.90 (6H, s, H-26, H-29), 1.02 (3H, s, H-30), 1.28 (3H, s, H-27), 1.57 (3H, s, H-24), 1.65 (3H, d,
J = 4.8, Rha-6), 3.44 (1H, m, H-18), 3.31 (1H, t-like,
J = 10.0 Hz, Xyl(inner)-5α), 3.54 (1H, t-like,
J = 10.0 Hz, Xyl(terminal)-5α), 4.68 (1H, d,
J = 6.8 Hz, GluA-1), 4.98 (1H, d,
J = 6.8 Hz, Xyl(inner)-1), 5.05 (1H, d,
J = 7.2 Hz, Gal-1), 5.10 (1H, d,
J = 7.6 Hz, Xyl(terminal)-1), 5.47 (1H, br. s, H-12), 5.62 (1H, br. s, Rha-1), 6.35 (1H, br. s, Ara-1), 9.76 (1H, s, H-23).
13C-NMR (100 MHz, C
5D
5N): see
Table 2.
Compound
4: White amorphous solid,
−10.0°(c 0.5, pyridine). HRESIMS,
m/
z: 1381.6258 [M + H]
+; calcd for C
64H
101O
32, 1381.6270.
1H-NMR (400 MHz, D
2O-C
5D
5N) δ: 0.90 (3H, s, H-25), 0.98 (3H, s, H-26), 1.03 (3H, s, H-29), 1.04 (3H, s, H-30), 1.26 (3H, s, H-27), 1.45 (3H, s, H-24), 1.67 (3H, d,
J = 4.8, Rha-6), 3.17 (1H, m, H-18), 3.57 (1H, t-like,
J = 10.0 Hz, Xyl(terminal)-5α), 4.82 (1H, d,
J = 7.2 Hz, GluA-1), 5.09 (1H, d,
J = 8.0 Hz, Glu-1), 5.18 (1H, d,
J = 7.6 Hz, Gal-1), 5.29 (1H, d,
J = 8.0 Hz, Xyl-1), 5.45 (1H, br. s, H-12), 5.57 (1H, br. s, Rha-1), 6.23 z, Ara-1), 9.78 (1H, s, H-23).
13C-NMR (100 MHz, D
2O-C
5D
5N): See
Table 2.
3.4. Acid Hydrolysis and Sterochemistry of Sugars of 1 and 2
Compounds 1 and 2 (each 20 mg) were hydrolyzed with 2 M HCl (5 mL) at 90 °C for 4 h. The reaction mixture was extracted with EtOAc (3 × 5 mL), washed with H2O, and evaporated to dryness. The EtOAc extract was purified by silica gel column using a gradient of CHCl3-MeOH (1:0–95:5) to the aglycones of 1 and 2 which were determined to be gypsogenin by comparison of their spectral data with those of an authentic sample. Aglycone: White amorphous solid; 13C-NMR (100 MHz, C5D5N) δ: 207.7 (C-23), 180.0 (C-28), 144.8 (C-13), 122.2 (C-12), 71.6 (C-3), 56.2(C-4), 48.0 (C-5), 47.9 (C-9), 46.6 (C-17), 46.5 (C-19), 42.2 (C-14), 41.9 (C-18), 40.0 (C-8), 38.4 (C-1), 36.2 (C-10), 34.2 (C-21), 33.2 (C-29), 32.8 (C-7), 32.5 (C-22), 30.9 (C-20), 28.3 (C-15), 26.1 (C-27), 27.0 (C-2), 23.8 (C-16, C-30), 23.7 (C-11), 21.0 (C-6), 17.3 (C-26), 15.7 (C-25), 9.6 (C-24); 1H-NMR (400 MHz, C5D5N) δ: 0.94 (3H, s), 0.97 (6H, s), 1.12 (6H, s), 1.23 (6H, s), 1.41 (6H, s), 3.02 (1H, m), 3.90(1H, m), 5.29 (1H, brs), 9.66 (1H, m). The H2O layer was concentrated under reduced pressure to dryness, to give a residue of the sugar fraction. The residue was dissolved in pyridine (2 mL), l-cysteine methyl ester hydrochloride (1.5 mg) was added, and the mixture was heated at 60 °C for 1 h. Trimethylsilylimidazole (1.5 mL) was added, and the mixture was heated at 60 °C for another 0.5 h. An aliquot (4 μL) of the supernatant was subjected to GC analysis under the following conditions: column temp 180–280 °C at 3 deg/min, carrier gas N2 (1 mL/min), injector and detector temp 250 °C, split ratio 1:50. The configurations of monosugars for 1 and 2 were determined by comparison of the retentions times (tR) of the corresponding derivatives with standard l-arabinose (tR 10.748 min), d-xylose (tR 11.496 min), l-rhamnose (tR 12.162 min), d-quinovose (tR 13.648 min), d-glucuronic acid (tR 15.814 min) and d-galactose (tR 18.026 min). 1 yielded d-xylose, l-rhamnose, d-quinovose, d-glucuronic acid and d-galactose, and 2 gave l-arabinose, d-xylose, l-rhamnose, d-glucuronic acid and d-galactose.
3.5. Cytotoxicity Assay
The procedure for the cytotoxic assay was performed according to the XTT (2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide) reduction method. In this study, MGC-803 (human gastric cancer), HL-60 (human promyelocytic leukemia), BEL-7402 (humanhepatoma carcinoma), MCF-7 (human breast cancer), and SPC-A1 (lung adenocarcinoma ) cell lines were used. Compounds 1 and 2 was dissolved at a concentration of 10 mg·mL−1 in phosphate-buffered saline (PBS) (as a stock solution), filtered through a filter 0.22 μm (Millipore, Bedford, MA, USA) and stored at 4 °C. The stock solution was diluted to 160, 80, 40, 20, 10 μg·mL−1 before the cytotoxic assay.
Logarithmic-phase growing cells were diluted into 5 × 104 cells·mL−1, seeded in in 96-well plates with 100 μL per well, and incubated in RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 U·mL−1 penicillin, 100 μg·mL−1 streptomycin at 37 °C in a humidified atmosphere with 5% CO2. Blank control wells were added 100 μL of cultrure medium, and maintained under the same conditions. After 24 h, 100 μL of the above culture medium containing different concentrations of 1 and 2 and normal culture medium (untreated control group) were added, and incubated for 72 h. For the analysis of cytotoxicity, 50 μL (1 mg·mL−1) XTT containing 0.15 mg·mL−1 phenazine methosulphate was added to each well, and cells were incubated for 3 h at 37 °C. The absorbances (A) of the produced formazan were measured by a DNM-9602 enzyme immunoassay spectrophotometer at 450 nm. Three replicate well were used for each analysis. The cell inhibitory rate (%) = (Asample − Ablank)/(Auntreated − Ablank) × 100. The concentration of compound producing 50% of cell inhibitory rate (IC50) was calculated by using SPSS version 20.0 (International Business Machines Corporation, Armonk, NY, USA).