Protein Tyrosine Phosphatase 1B Inhibitors from the Stems of Akebia quinata
Abstract
:1. Introduction
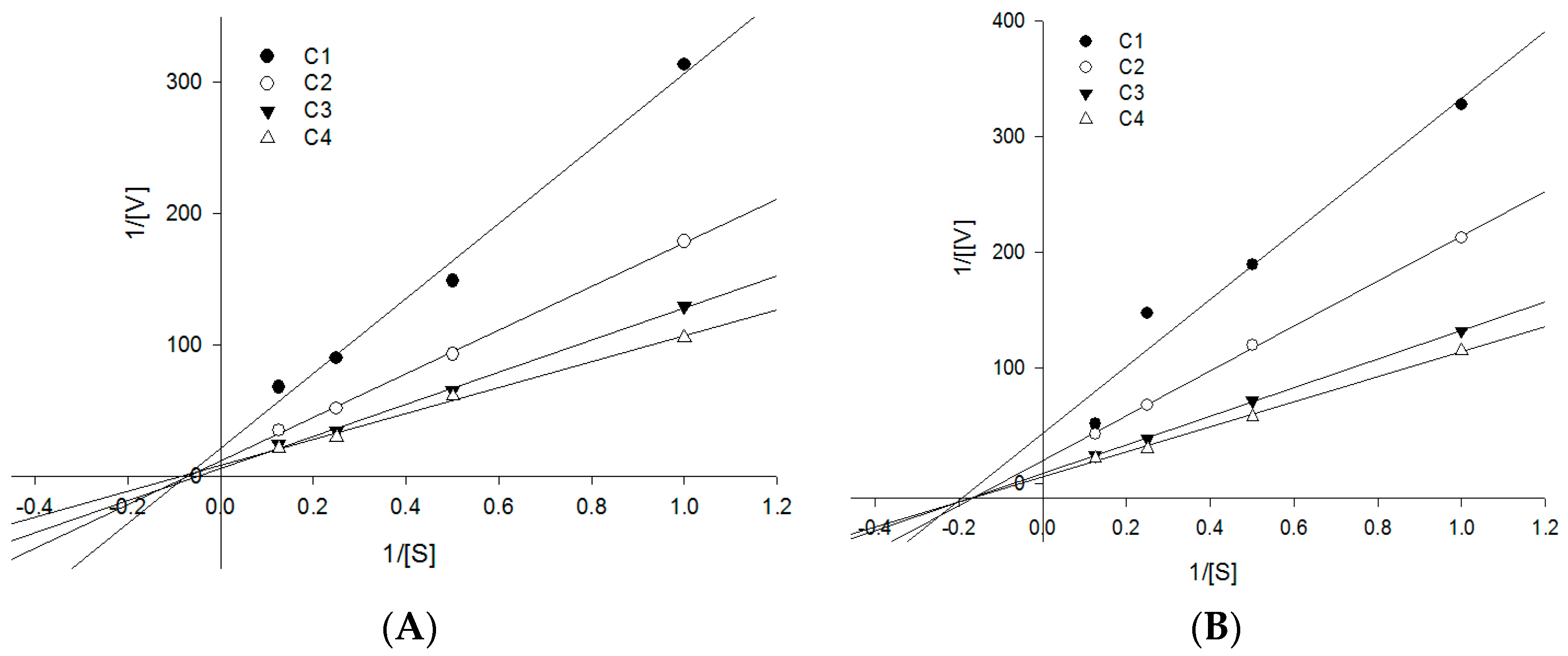
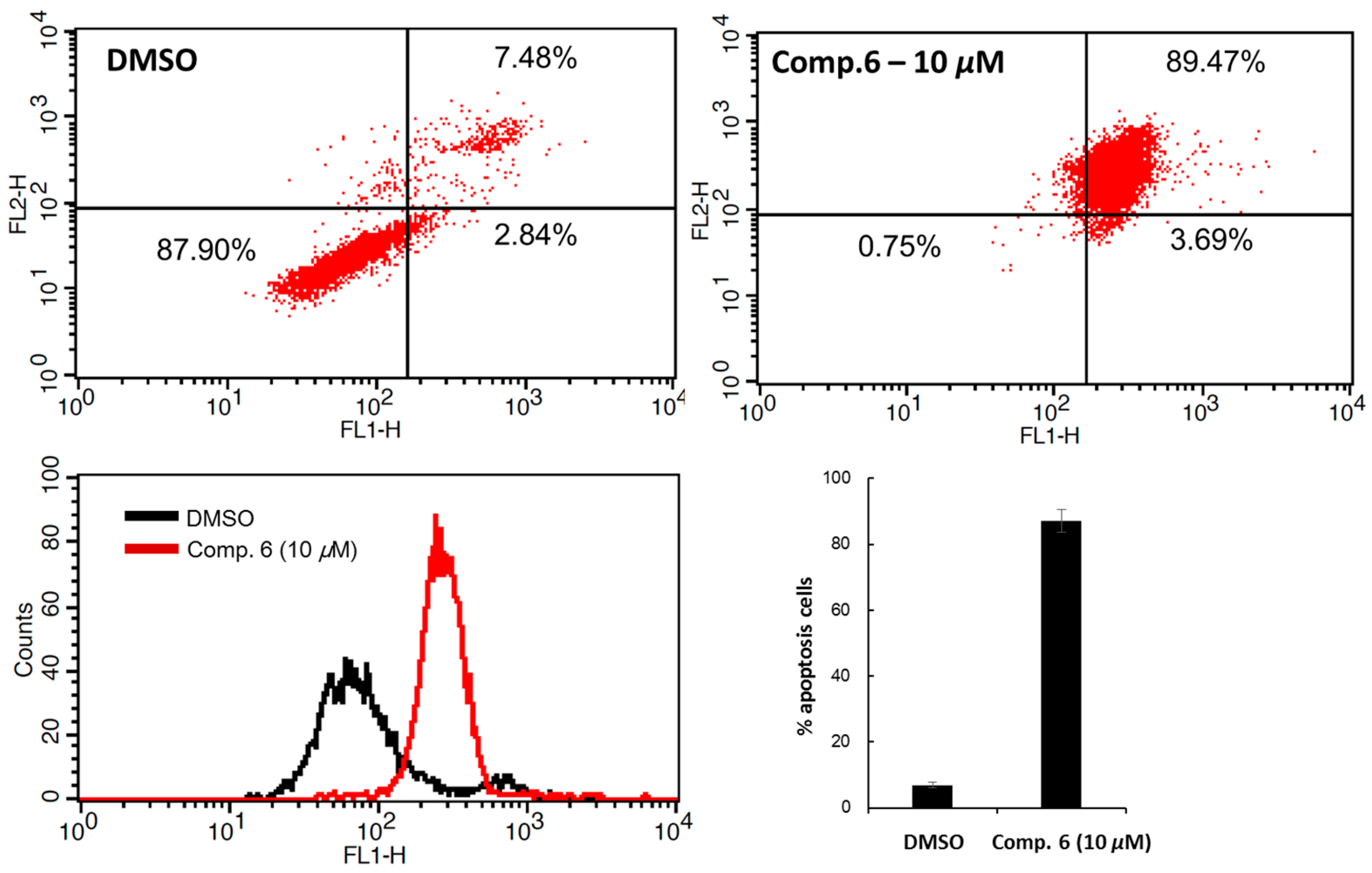
2. Results
3. Experimental Section
3.1. General Information
3.2. Extraction and Isolation
3.3. PTP1B Enzyme Assay
3.4. Flow Cytometric Analysis of the Cell Apoptosis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vasudevan, D.; Jayalakshmy, P.S.; Kumar, S.; Mathew, S. Assessment of pathological response of breast carcinoma in modified radical mastectomy specimens after neoadjuvant chemotherapy. Int. J. Breast Cancer 2015, 2015, 536145–536152. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, A.B.; Yabroff, K.R.; Shao, Y.; Feuer, E.J.; Brown, M.L. Projections of the cost of cancer care in the United States: 2010–2020. J. Natl. Cancer Inst. 2011, 103, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Wiener, J.R.; Kerns, B.J.M.; Harvey, E.L.; Conaway, M.R.; Iglehart, J.D.; Berchuck, A.; Bast, R.C., Jr. Overexpression of the protein-tyrosine-phosphatase 1B in human breast-cancer-association with P185(C-Erbb-2) protein expression. J. Natl. Cancer Inst. 1994, 86, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Blanquart, C.; Karouri, S.E.; Issad, T. Protein tyrosine phosphatase-1B and T-cell protein tyrosine phosphatase regulate IGF-2-induced MCF-7 cell migration. Biochem. Biophys. Res. Commun. 2010, 392, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, B.A.; Benjamin, G.N. Protein-tyrosine phosphatase 1B is required for HER2/Neu–induced breast cancer. Cancer Res. 2007, 67, 2420–2424. [Google Scholar]
- Gonzalez-Rodriguez, A.; Mas Gutierrez, J.A.; Sanz-Gonzalez, S.; Ros, M.; Burks, D.J.; Valverde, A.M. Inhibition of PTP1B restores IRS1-mediated hepatic insulin signaling in IRS2-deficient mice. Diabetes 2010, 59, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.M.; Tao, R.Y.; Liu, Q.; Li, J.; Tian, J.Y.; Zhang, X.L.; Xiao, Z.Y.; Ye, F. PTP1B inhibitor improves both insulin resistance and lipid abnormalities in vivo and in vitro. Mol. Cell. Biochem. 2011, 357, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Mimaki, Y.; Doi, S.; Kuroda, M.; Yokosuka, A. Triterpene glycosides from the stems of Akebia quinata. Chem. Pharm. Bull. 2007, 55, 1319–1324. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.J.; Sung, Y.Y.; Kim, H.K. Inhibitory effects of Akebia quinata ethanol extract on TNF-α-mediated vascular inflammation in human aortic smooth muscle cells. Mol. Med. Rep. 2013, 7, 379–383. [Google Scholar] [PubMed]
- Tian, X.; Min, Z.; Xie, N.; Lei, Y.; Tian, Z.; Zheng, Q.; Xu, R.; Tanaka, T.; Iinuma, M.; Mizuno, M. Abietane diterpenes from Clerodendron cyrtophyllum. Chem. Pharm. Bull. 1993, 41, 1415–1417. [Google Scholar] [CrossRef]
- Ulubelen, A.; Topcu, G.; Olcal, S. Rearranged abietane diterpenes from Teucrium divaricatum Subsp. Villosum. Phytochemistry 1994, 37, 1371–1375. [Google Scholar] [CrossRef]
- Morgan, A.M.A.; Kim, J.H.; Lee, H.W.; Lee, S.H.; Lim, C.; Jang, H.; Kim, Y.H. Phytochemical constituents from the aerial part of Ducrosia ismaelis Asch. Nat. Prod. Sci. 2015, 21, 6–13. [Google Scholar]
- Martin, P.K.C.; Laurence, V.N. Synthesis of l-arabinopyranose containing hederagenin saponins. Tetrahedron 2005, 61, 4347–4362. [Google Scholar]
- Grishkovets, V.I.; Sobolev, E.A.; Shashkov, A.S.; Chirva, V.Y. Triterpenoid glycosides of Fatsia japonica. II. Isolation and structure of glycosides from the leaves. Chem. Nat. Compd. 2000, 36, 501–505. [Google Scholar] [CrossRef]
- Shao, C.J.; Kasai, R.; Xu, J.D.; Tanaka, O. Saponins from leaves of Acanthopanax senticosus Harms., ciwujia. II.: Structures of ciwujianosides A1, A2, A3, A4 and D3. Chem. Pharm. Bull. 1989, 37, 42–45. [Google Scholar] [CrossRef]
- Shao, C.J.; Kasai, R.; Xu, J.D.; Tanaka, O. Saponins from leaves of Acanthopanax senticosus HARMS., ciwujia: Structures of ciwujianosides B, C1, C2, C3, C4, D1, D2 and E. Chem. Pharm. Bull. 1988, 36, 601–608. [Google Scholar] [CrossRef]
- Ullah, F.; Hussain, H.; Hussain, J.; Bukhari, I.A.; Khan, M.T.; Choudhary, M.I.; Gilani, A.H.; Ahmad, V.U. Tyrosinase inhibitory pentacyclic triterpenes and analgesic and spasmolytic activities of methanol extracts of Rhododendron collettianum. Phytother. Res. 2007, 21, 1076–1081. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.N.; Yang, G.E.; Li, J.K.; Du, H.J.; Li, Q.S.; Zhang, Z.M. Cytotoxic constituents from Viscum coloratum. Chem. Nat. Compd. 2009, 45, 547–549. [Google Scholar] [CrossRef]
- Tsukamoto, H.; Hisada, S.; Nishibe, S. Lignans from bark of Fraxinus mandshurica var. japonica and F. japonica. Chem. Pharm. Bull. 1984, 32, 4482–4489. [Google Scholar] [CrossRef]
- Miyazawa, M.; Kasahara, H.; Kameoka, H. Phenolic lignans from flower buds of Magnolia fargesii. Phytochemistry 1992, 31, 3666–3668. [Google Scholar] [CrossRef]
- Wu, J. Apoptosis and angiogenesis: Two promising tumor markers in breast cancer (review). Anticancer Res. 1996, 16, 2233–2239. [Google Scholar] [PubMed]
- Na, M.; Cui, L.; Min, B.S.; Bae, K.; Yoo, J.K.; Kim, B.Y.; Oh, W.K.; Ahn, J.S. Protein tyrosine phosphatase 1B inhibitory activity of triterpenes isolated from Astilbe koreana. Bioorg. Med. Chem. Lett. 2006, 16, 3273–3276. [Google Scholar] [CrossRef] [PubMed]
- Na, M.; Oh, W.K.; Kim, Y.H.; Cai, X.F.; Kim, S.H.; Kim, B.Y.; Ahn, J.S. Inhibition of protein tyrosine phosphatase 1B by diterpenoids isolated from Acanthopanax koreanum. Bioorg. Med. Chem. Lett. 2006, 16, 3061–3064. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.R., Jr.; Ye, B.; Yan, X.; Wang, S.; Jia, Z.; Chen, L.; Zhang, Z.Y.; Barford, D. Small molecule interactions with protein-tyrosine phosphatase PTP1B and their use in inhibitor design. Biochemistry 1996, 35, 15989–15996. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors. A voucher specimen (No. 2014-04) has been deposited at the Herbarium of the Korea Bioactive Natural Material Bank, Seoul, Korea.

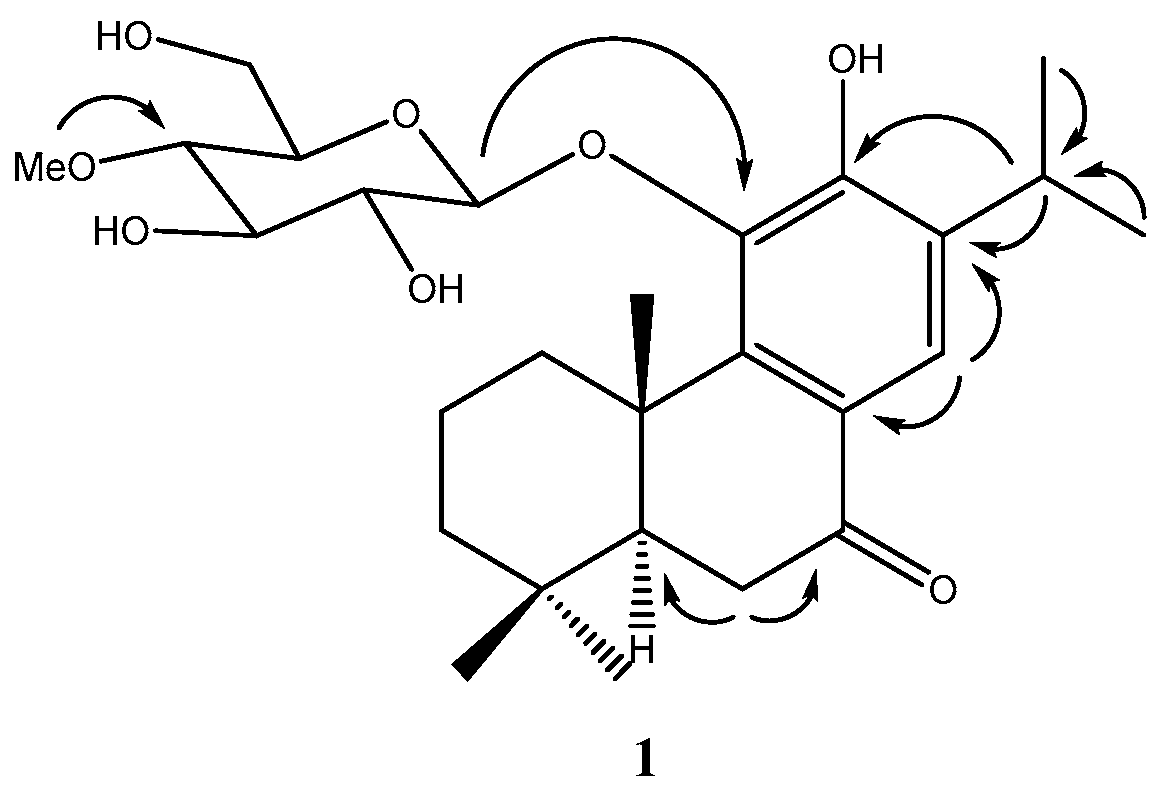
| No. | δH J (Hz) | δC | HMBC | No. | δH J (Hz) | δC | HMBC |
|---|---|---|---|---|---|---|---|
| 1 | 3.47 (1H, d, J = 13.5 Hz) | 38.3 | C-2, C-3, C-5 | 15 | 3.27 (1H, m) | 28.2 | C-12, C-13, C-14, C-17 |
| 1.40 (1H, m) | |||||||
| 2 | 1.81 (1H, dt, J = 3.7, 16.6 Hz) | 20.1 | C-3, C-5 | 16 | 1.20 (3H, d, J = 6.9 Hz) | 22.8 | C-15, C-17 |
| 1.56 (1H, m) | |||||||
| 3 | 1.48 (1H, d, J = 15.8 Hz) | 42.4 | C-2, C-4, C-5 | 17 | 1.23 (3H, d, J = 6.9 Hz) | 22.5 | C-15,C-16 |
| 1.29 (1H, dd, J = 4.6, 16.1 Hz) | |||||||
| 4 | 34.5 | 18 | 0.93 (3H, s) | 33.5 | C-3, C-4, C-5, C-19 | ||
| 5 | 1.76 (1H, dd, J = 3.9, 13.9 Hz) | 51.7 | C-3, C-4, C-6, C-7 | 19 | 1.02 (3H, s) | 22.0 | C-3, C-4, C-5, C-18 |
| 6 | 2.61 (1H, m) | 36.3 | C-5, C-7, C-8 | 20 | 1.57 (3H, s) | 20.5 | C-1, C-3, C-5, C-9 |
| 2.58 (1H, m) | |||||||
| 7 | 201.7 | 1′ | 4.54 (1H, d, J = 7.7 Hz) | 106.8 | C-2′, C-11 | ||
| 8 | 124.8 | 2′ | 3.54 (1H, m) | 75.9 | C-1′, C-3′ | ||
| 9 | 148.5 | 3′ | 3.55 (1H, m) | 78.1 | C-2’, C-4’ | ||
| 10 | 40.4 | 4′ | 3.22 (1H, m) | 80.3 | C-3’, C-5’ | ||
| 11 | 143.8 | 5′ | 3.22 (1H, m) | 77.9 | C-4’, C-6’ | ||
| 12 | 154.6 | 6′ | 3.81 (1H, d, J = 11.3 Hz) | 61.9 | C-4’, C-5’ | ||
| 3.71 (1H, d, J = 11.3 Hz) | |||||||
| 13 | 136.1 | 4′-OCH3 | 3.58 (3H, s) | 60.8 | C-4’ | ||
| 14 | 7.73 (1H, s) | 123.7 | C-7, C-9, C-11, C-12 |
| Compounds | PTP1B (IC50, μM) a | Cytotoxic Activities (IC50, μM) a | ||
|---|---|---|---|---|
| MCF7 | MDA-MB-231 | MCF7/TAMR | ||
| 2 | 6.77 ± 1.28 | 7.91 ± 0.39 | 4.04 ± 0.80 | 13.42 ± 1.26 |
| 3 | 5.41 ± 0.68 | 6.02 ± 2.32 | 5.14 ± 1.55 | 7.73 ± 1.02 |
| 6 | 4.08 ± 1.09 | 1.11 ± 0.04 | 0.84 ± 0.17 | 1.39 ± 0.18 |
| 8 | 21.80 ± 4.74 | 1.29 ± 0.09 | 1.55 ± 0.21 | 1.78 ± 0.13 |
| 11 | 7.78 ± 1.43 | >40 | NT | NT |
| Ursolic acid b | 4.25 ± 0.62 | NT | NT | NT |
| Tamoxifen b | NT | 8.01 ± 1.81 | 10.09 ± 1.51 | 25.42 ± 1.79 |
| 4-OH tamoxifen b | NT | 2.11 ± 0.39 | 3.10 ± 0.31 | 13.58 ± 1.25 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
An, J.-P.; Ha, T.K.Q.; Kim, J.; Cho, T.O.; Oh, W.K. Protein Tyrosine Phosphatase 1B Inhibitors from the Stems of Akebia quinata. Molecules 2016, 21, 1091. https://doi.org/10.3390/molecules21081091
An J-P, Ha TKQ, Kim J, Cho TO, Oh WK. Protein Tyrosine Phosphatase 1B Inhibitors from the Stems of Akebia quinata. Molecules. 2016; 21(8):1091. https://doi.org/10.3390/molecules21081091
Chicago/Turabian StyleAn, Jin-Pyo, Thi Kim Quy Ha, Jinwoong Kim, Tae Oh Cho, and Won Keun Oh. 2016. "Protein Tyrosine Phosphatase 1B Inhibitors from the Stems of Akebia quinata" Molecules 21, no. 8: 1091. https://doi.org/10.3390/molecules21081091
APA StyleAn, J.-P., Ha, T. K. Q., Kim, J., Cho, T. O., & Oh, W. K. (2016). Protein Tyrosine Phosphatase 1B Inhibitors from the Stems of Akebia quinata. Molecules, 21(8), 1091. https://doi.org/10.3390/molecules21081091







