Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines
Abstract
:1. Introduction
2. Results
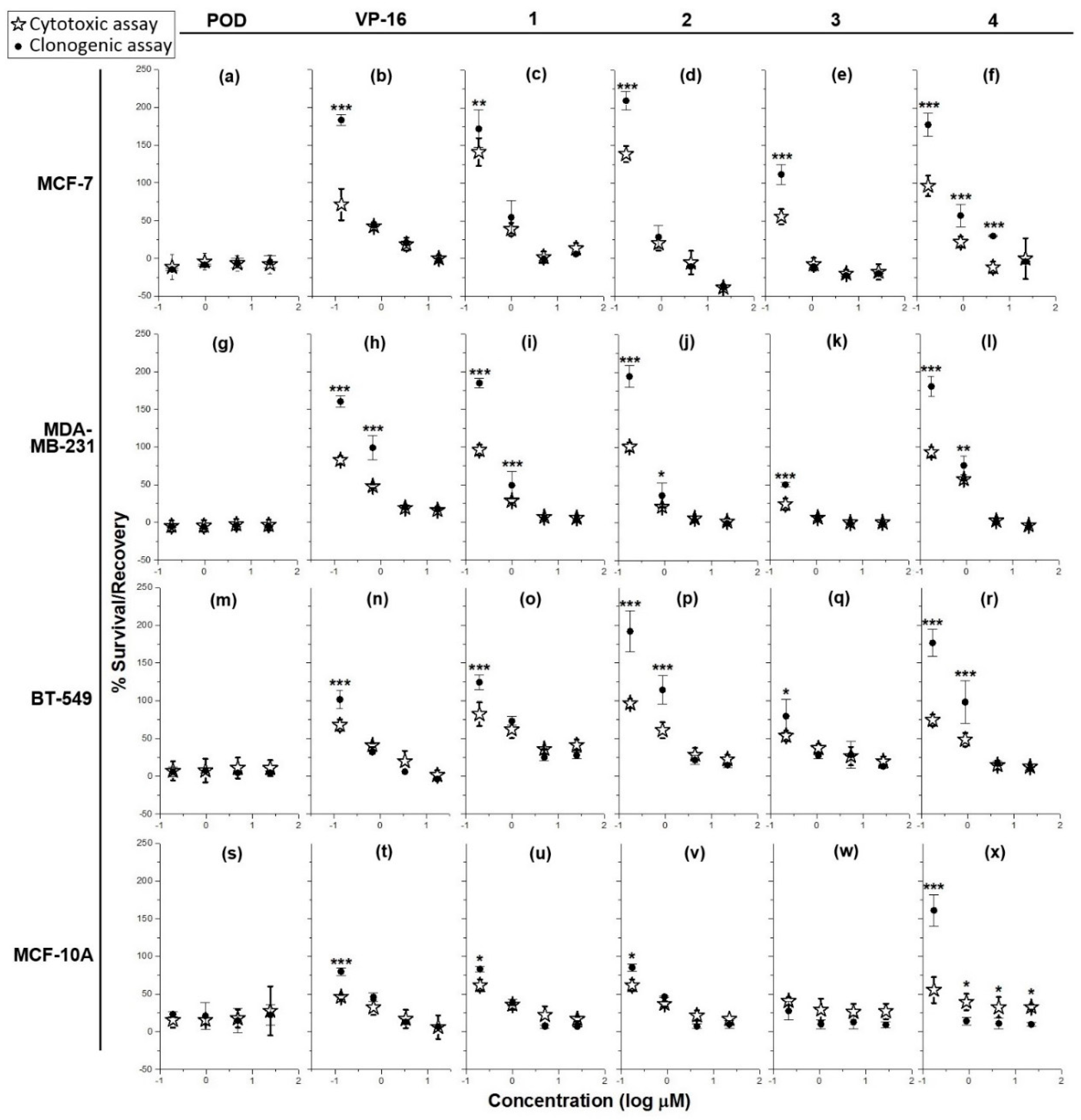
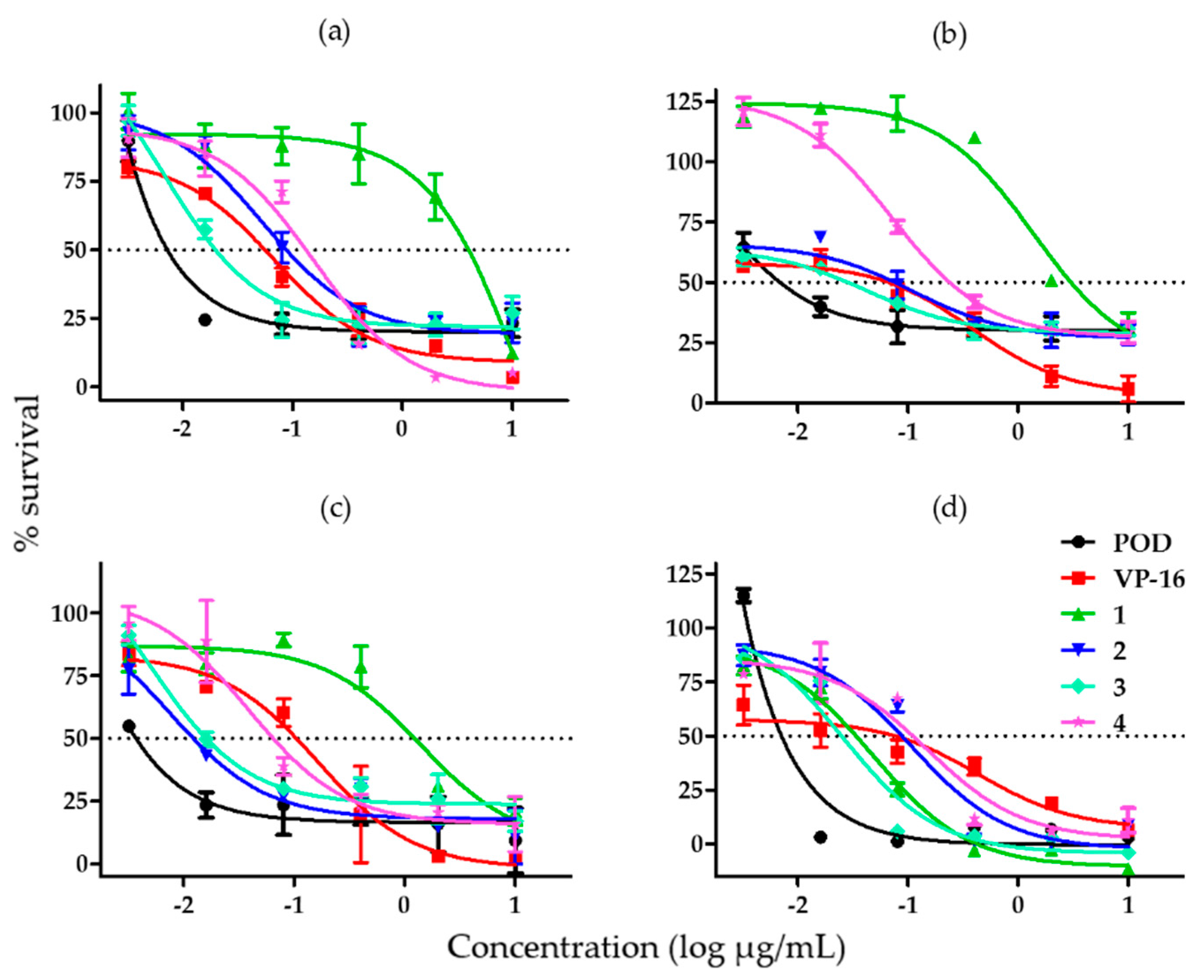
2.1. Cytotoxicity and Clonogenicity Assays, and the Selctive-index
2.2 Similarity Analysis of Compounds
3. Discussion
3.1. Cytotoxicity and Clonogenicity Assays, and the Selective-Index
3.2 Similarity Analysis of Compounds
4. Materials and Methods
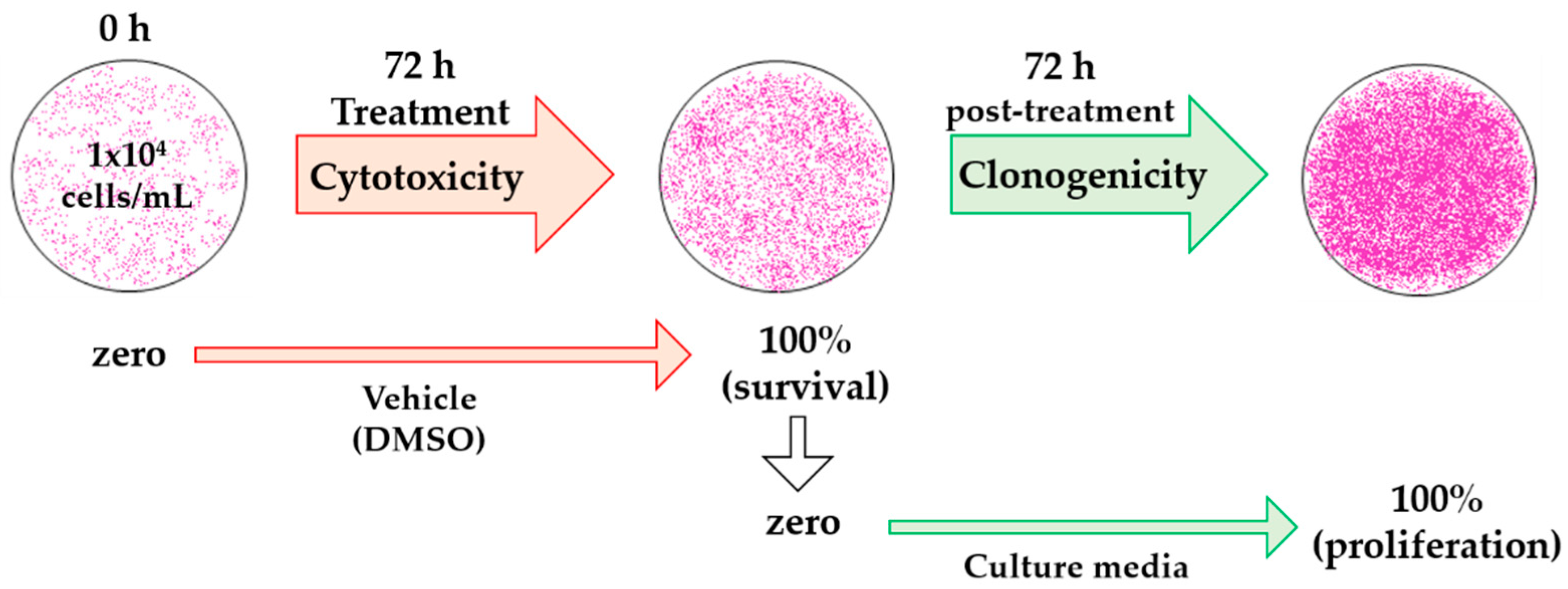
4.1. Cytotoxicity and Clonogenicity Assays
4.2. Selective-Index
4.3. Similarity Analysis of Compounds
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SI | Selectivity index |
| WHO | World Health Organization |
| ER | Estrogen receptor |
| PgR | Progesterone receptor |
| Her2/neu | Human epidermal growth factor receptor 2 |
| IUPAC | International Union of Pure and Applied Chemistry |
| NMR | Nuclear Magnetic Resonance |
| FAB-MS | Fast Bombardment-mass spectrometry |
| DSMO | Dimethyl sulfoxide |
| SRB | Sulforhodamine B |
| qHTS | quantitative High Throughput Screening |
| TDP1 | Tyrosil-DNA-Phosphodiesterase 1 |
| NIH | National Institutes of Health |
| NCI | National Cancer Institute |
| PBS | Phosphate Buffered Saline |
| ANOVA | Analysis Of Variance |
| MEGM | Mammary Epithelial Cell Growth Medium |
| RPMI | Roswell Park Memorial Institute medium |
References
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: IARC cancer base No. 11. IARC. 2013. Available online: http://globocan.iarc.fr (accessed on 7 April 2016).
- Advani, P.; Moreno-Aspitia, A. Current strategies for the prevention of breast cancer. Breast Cancer 2014, 6, 59–71. [Google Scholar] [PubMed]
- Tate, C.; Rhodes, L.; Segar, C.; Driver, J.; Pounder, N.; Burow, M.; Collins-Burow, B. Targeting triple-negative breast cancer cells with the histone deacetylase inhibitor panobinostat. Breast Cancer Res. 2012, 14. [Google Scholar] [CrossRef] [PubMed]
- Podwyssotzki, V. Pharmakologische studien über Podophyllum peltatum. Arch. Exp. Pathol. Pharmakol. 1880, 13, 29–52. [Google Scholar] [CrossRef]
- Xu, H.; Lv, M.; Tian, X. A review on hemisynthesis, biosynthesis, biological activities, mode of action, and structure-activity relationship of podophyllotoxins: 2003–2007. Curr. Med. Chem. 2009, 16, 327–349. [Google Scholar] [CrossRef] [PubMed]
- Gordaliza, M.; Castro, M.A.; Miguel del Corral, J.M.; San Feliciano, A. Antitumor properties of podophyllotoxin and related compounds. Curr. Pharm. Des. 2000, 6, 1811–1839. [Google Scholar] [CrossRef] [PubMed]
- Damayanthi, Y.; Lown, W. Podophyllotoxins: Current status and recent developments. Curr. Med. Chem. 1998, 5, 205–252. [Google Scholar] [PubMed]
- Stähelin, H.; von Wartburg, A. The chemical and biological route from podophyllotoxin glucoside to etoposide: Ninth Cain memorial award lecture. Cancer Res. 1991, 51, 5–15. [Google Scholar] [PubMed]
- Hande, K.R. Etoposide: Four decades of development of a topoisomerase II inhibitor. Eur. J. Cancer 1998, 34, 1514–1521. [Google Scholar] [CrossRef]
- Guerram, M.; Jiang, Z.-Z.; Zhang, L.-Y. Podophyllotoxin, a medicinal agent of plant origin: Past, present and future. Chin. J. Nat. Med. 2012, 10, 161–169. [Google Scholar] [CrossRef]
- Ravelli, R.; Gigant, B.; Curmi, A.; Jourdain, I.; Lachkar, S.; Sobel, A.; Knossow, M. Insight into tubulin regulation from a complex with colchicines and a stathmin-like domain. Nature 2004, 428, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.J.; Wang, Y.J.; Liang, Y.C.; Jeng, J.H.; Lee, W.S.; Lin, J.K.; Chen, C.H.; Liu, I.C.; Ho, Y.S. Microtubule damaging agents induce apoptosis in HL 60 cells and G2/M cell cycle arrest in HT 29 cells. Toxicology 2002, 175, 123–142. [Google Scholar] [CrossRef]
- Wu, C.C.; Li, T.K.; Farh, L.; Lin, L.Y.; Lin, T.S.; Yu, Y.J.; Yen, T.J.; Chiang, C.W.; Chan, N.L. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science 2011, 333, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Montecucco, A.; Biamonti, G. Cellular response to etoposide treatment. Cancer Lett. 2007, 252, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Canel, C.; Moraes, R.; Dayan, F.; Ferreira, D. Molecules of interest: Podophyllotoxin. Phytochemistry 2000, 54, 115–120. [Google Scholar] [CrossRef]
- Lv, M.; Xu, H. Recent advances in semisynthesis, biosynthesis, biological activities, mode of action, and structure-activity relationship of podophyllotoxins: An update (2008–2010). Mini Rev. Med. Chem. 2011, 11, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Q.; Tian, K.; Zhao, X.B.; Morris-Natschke, S.; Yang, L.; Nan, X.; Tian, X.; Lee, K.H. Recent progress on C-4 modified podophyllotoxin analogs as potent antitumor agents. Med. Res. Rev. 2015, 35. [Google Scholar] [CrossRef] [PubMed]
- Holliday, D.; Speirs, V. Choosing the right cell line for breast cancer research. Breast Cancer Res. 2011, 13. [Google Scholar] [CrossRef] [PubMed]
- Kennya, P.A.; Leea, G.Y.; Myersa, C.A.; Nevea, R.M.; Semeiksa, J.R.; Spellmana, P.T.; Lorenza, K.; Leea, E.H.; Barcellos-Hoff, M.H.; Petersenb, O.W.; et al. The morphologies of breast cancer cell lines in three-dimensional assays correlate with their profiles of gene expression. Mol. Oncol. 2007, 1, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.K.; Mittal, M.K.; Misra, S.; Chaudhuri, G. High motility of triple-negative breast cancer cells is due to repression of plakoglobin gene by metastasis modulator protein SLUG. J. Biol. Chem. 2012, 287, 19472–19486. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, M.; Leclercq, G. Relevance of breast cancer cell lines as models for breast tumours: An update. Breast Cancer Res. Treat 2004, 83, 249–249. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sepúlveda, A.M.; Mendieta-Serrano, M.; Antúnez Mojica, M.Y.; Salas-Vidal, E.; Marquina, S.; Villarreal, M.L.; Puebla, A.M.; Delgado, J.; Alvarez, L. Cytotoxic podophyllotoxin type-lignans from the steam bark of Bursera fagaroides var. fagaroides. Molecules 2012, 17, 9506–9519. [Google Scholar] [CrossRef] [PubMed]
- Antúnez, M.M.; León, A.; Rojas-Sepúlveda, M.; Marquina, S.; Mendieta-Serrano, M.A.; Salas-Vidal, E.; Villarreal, M.L.; Alvarez, L. Aryldihydronaphthalene-type lignans from Bursera fagaroides var. fagaroides and their antimitotic mechanism of action. RSC Adv. 2016, 6, 4950–4959. [Google Scholar]
- Liu, S.; Lin, Y.C. Transformation of MCF-10A human breast epithelial cells by zeranol and estradiol-17β. Breast J. 2004, 10, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Jänicke, R.U. MCF-7 breast carcinoma cells do not express caspase-3. Breast Cancer Res. Treat 2009, 117, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Bartek, J.; Iggo, R.; Gannon, J.; Lane, D.P. Genetic and immunochemical analysis of mutant p53 in human breast cancer cell lines. Oncogene 1990, 5, 893–899. [Google Scholar] [PubMed]
- Portugal, J.; Bataller, M.; Mansilla, S. Cell death pathways in response to antitumor therapy. Tumori 2009, 95, 409–421. [Google Scholar] [PubMed]
- NCBI, National Center for Biotechnology Information. PubChem BioAssay Database. 2013; AID=686981. Available online: https://pubchem.ncbi.nlm.nih.gov/bioassay/686981 (accessed on 30 March 2016). [Google Scholar]
- NCBI, National Center for Biotechnology Information. PubChem BioAssay Database. 2007; AID=942. Available online: https://pubchem.ncbi.nlm.nih.gov/bioassay/942 (accessed on 30 March 2016). [Google Scholar]
- NCBI, National Center for Biotechnology Information. PubChem BioAssay Database. 2007; AID=192, 256 and 270. Available online: https://pubchem.ncbi.nlm.nih.gov/bioassay/192, 256, and 270 (accessed on 30 March 2016). [Google Scholar]
- Martin, Y.; Kofron, J.; Traphagen, L. Do structurally similar molecules have similar biological activity? J. Med. Chem. 2002, 45, 4350–4358. [Google Scholar] [CrossRef] [PubMed]
- Magedov, I.; Evdokimov, N.; Karki, M.; Peretti, A.; Lima, D.; Frolova, L.; Reisenauer, M.; Romero, A.; Tongwa, P.; Fonari, A.; et al. Reengineered epipodophyllotoxin. Chem. Commun. 2012, 48, 10416–10418. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.L.; Liu, Y.; Rowe, T.C.; Halligan, D.; Tewey, K.M.; Liu, L.F. Nonintercalative antitumor drugs interfere with the breakage-reunion reaction of mammalian DNA topoisomerase II. J. Biol. Chem. 1984, 259, 13560–13566. [Google Scholar] [PubMed]
- Beers, S.; Imakura, Y.; Dai, H.-J.; Li, D.-H.; Cheng, Y.-C.; Lee, K.-H. Antitumor agents, 99. Synthetic ring C aromatized podophyllotoxin analogues as potential inhibitors of human DNA topoisomerase II. J. Nat. Prod. 1988, 51, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Y.; Lehuédé, C.; Laurent, V.; Dirat, B.; Dauvillier, S.; Bochet, L.; Le Gonidec, S.; Escourrou, G.; Valet, P.; Muller, C. Adipose tissue and breast epithelial cells: A dangerous dynamic duo in breast cancer. Cancer Lett. 2012, 324, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sepúlveda, A.M. Búsqueda de metabolitos con actividad citotóxica y antitumoral en Bursera fagaroides var. fagaroides y Bursera morelensis, y evaluación de su efecto como inhibidor de ciclo celular en el modelo de pez cebra. Ph.D. Thesis, Centro de Investigaciones Químicas. Universidad Autónoma del Estado de Morelos, Cuernavaca, Morelos, Mexico, 2012. [Google Scholar]
- Da Silva, R.; Gomes, H.V.C.; Albuquerque, S.; Kenupp, B.J.; Andrade, S.M.L.; Marcos, D.P.; da Silva, G.V.J. Complete assignment of 1H- and 13C-NMR data for three aryltretalin lignan lactones. Magn. Reson. Chem. 2004, 42, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Jolad, S.D.; Wiedhopf, R.M.; Cole, J.R. Cytotoxic agents from Bursera morelensis (Burseraceae): Deoxypodophyllotoxin and a new lignan, 5′-desmethoxydeoxypodophyllotoxin. J. Pharm. Sci. 1977, 66, 892–893. [Google Scholar] [CrossRef] [PubMed]
- Geran, R.I.; Greenberg, H.M.; McDonald, M.; Abbott, B.J. Protocols for Screening Chemical Agents and Natural Products against Animal Tumors and Other Biological Systems. Cancer Chemother. Rep. 1972, 3, 51–61. [Google Scholar]
- Shekan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.; Bokesch, H.; Kenney, S.; Boyd, M. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar]
- Houghton, P.J.; Morton, C.L.; Tucker, C.; Payne, D.; Favours, E.; Cole, C.; Gorlick, R.; Kolb, E.A.; Zhang, W.; Lock, R.; et al. The pediatric preclinical testing program: Description of models and early testing results. Pediatr Blood Cancer 2007, 49, 928–940. [Google Scholar] [CrossRef] [PubMed]
- Suffness, M.; Pezzuto, J.M. Methods in Plant Biochemistry; Academic Press: San Diego, CA, USA, 1991; pp. 71–134. [Google Scholar]
- Quispe, A.; Zavala, C.; Rojas, J.; Posso, M.; Vaisberg, A. Efecto citotóxico selectivo in vitro de murcin H (acetogenina de Annona muricata) en cultivos celulares de cáncer de pulmón. Rev. Peru. Med. Exp. Salud Pública 2006, 23, 265–269. [Google Scholar]
- Valdés-García, S.; Del barrio-Alonso, G.; Gutiérrez-Gaitén, Y.; Morier-Díaz, L. Evaluación preliminar de la actividad antiviral del extracto acuoso de Phyllathus orbicularis frente al virus VHS-1. Rev. Cuba. Med. Trop. 2003, 55, 169–173. [Google Scholar]
- Megantara, S.; Iwo, M.; Levita, J.; Ibrahim, S. Determination of ligand position in aspartic proteases by correlating tanimoto coefficient and binding affinity with root mean square deviation. J. Appl. Pharm. Sci. 2016, 6, 125–129. [Google Scholar] [CrossRef]
- Radifar, M.; Yuniarti, N.; Istyastono, P. PyPLIF: Python-based protein-ligand interaction fingerprinting. Bioinformation 2013, 9, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1–4 are available from the authors.
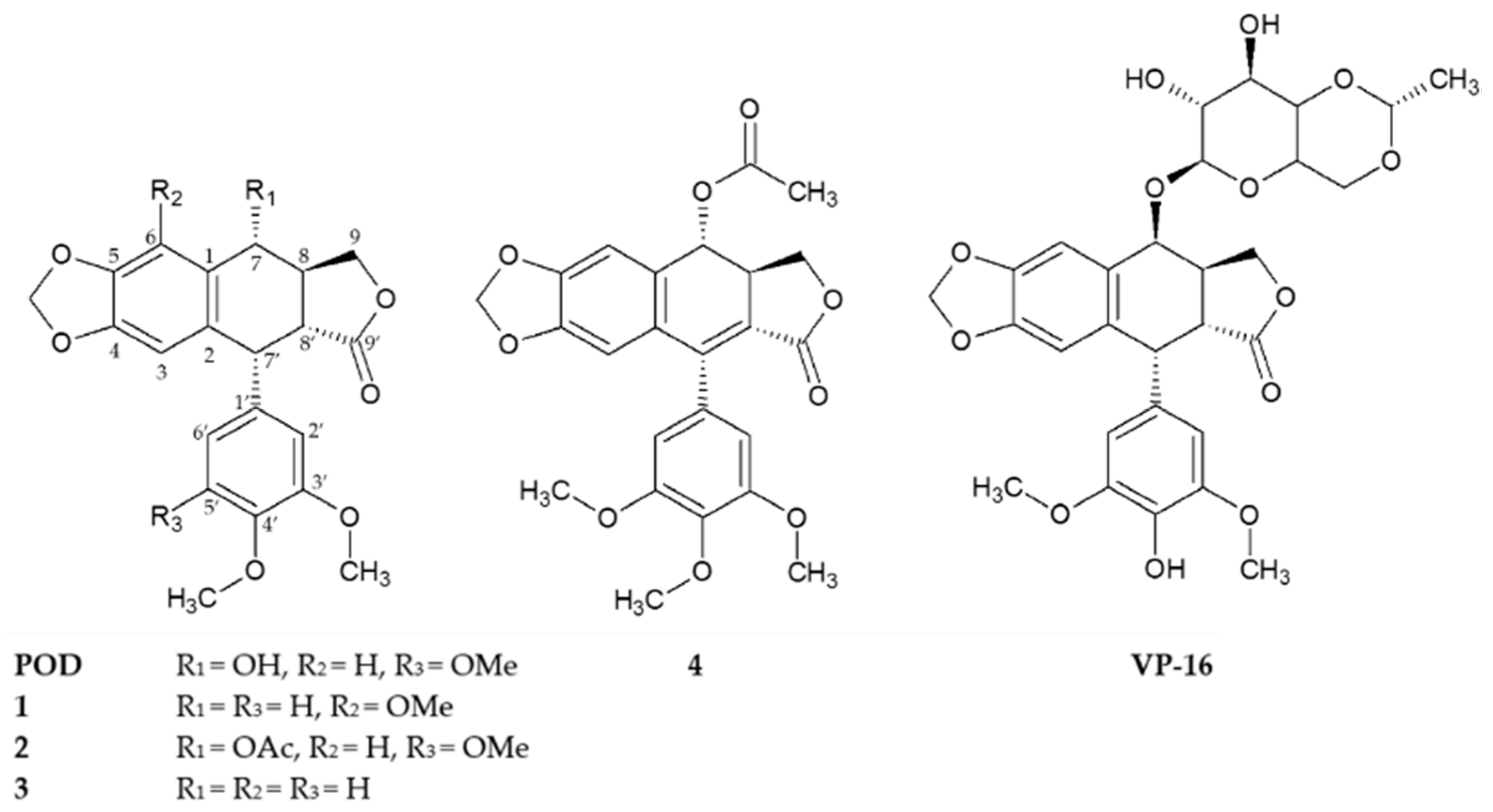
| Compounds | MCF-7 | MDA-MB-231 | BT-549 | MCF-10A |
|---|---|---|---|---|
| 1 | 7.222 ± 0.098 | 2.444 ± 0.087 | 1.269 ± 0.087 | 0.137 ± 0.015 |
| 2 | 0.132 ± 0.017 | 0.180 ± 0.003 | 0.011 ± 0.004 | 0.318 ± 0.012 |
| 3 | 0.040 ± 0.011 | 0.145 ± 0.045 | 0.021 ± 0.003 | 0.092 ± 0.009 |
| 4 | 0.353 ± 0.088 | 0.161 ± 0.024 | 0.061 ± 0.012 | 0.468 ± 0.002 |
| POD | 0.018 ± 0.001 | 0.024 ± 0.006 | 0.005 ± 0.001 | 0.020 ± 0.008 |
| VP-16 | 0.124 ± 0.026 | 0.038 ± 0.004 | 0.160 ± 0.010 | 0.047 ± 0.009 |
| Cell Line | Controls | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| MCF-7 | POD | 0.002 | 0.135 | 0.449 | 0.050 |
| VP-16 | 0.017 | 0.942 | 3.124 | 0.351 | |
| MDA-MB-231 | POD | 0.010 | 0.136 | 0.168 | 0.151 |
| VP-16 | 0.016 | 0.217 | 0.268 | 0.242 | |
| BT-549 | POD | 0.004 | 0.465 | 0.252 | 0.086 |
| VP-16 | 0.126 | 14.151 | 7.661 | 2.604 | |
| MCF-10A | POD | 0.144 | 0.062 | 0.214 | 0.042 |
| VP-16 | 0.341 | 0.147 | 0.507 | 0.100 |
| Compounds | MCF-7 | MDA-MB-231 | BT-549 |
|---|---|---|---|
| 1 | 0.02 | 0.06 | 0.11 |
| 2 | 2.42 | 1.77 | 28.17 |
| 3 | 2.33 | 0.64 | 4.43 |
| 4 | 1.32 | 2.90 | 7.62 |
| POD | 1.11 | 0.81 | 3.76 |
| VP-16 | 0.38 | 1.20 | 0.29 |
| Compounds | POD | VP-16 |
|---|---|---|
| 1 | 0.43 | 0.33 |
| 2 | 0.50 | 0.27 |
| 3 | 0.50 | 0.38 |
| 4 | 0.38 | 0.18 |
| POD | 1.00 | 0.33 |
| VP-16 | 0.33 | 1.00 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peña-Morán, O.A.; Villarreal, M.L.; Álvarez-Berber, L.; Meneses-Acosta, A.; Rodríguez-López, V. Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines. Molecules 2016, 21, 1013. https://doi.org/10.3390/molecules21081013
Peña-Morán OA, Villarreal ML, Álvarez-Berber L, Meneses-Acosta A, Rodríguez-López V. Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines. Molecules. 2016; 21(8):1013. https://doi.org/10.3390/molecules21081013
Chicago/Turabian StylePeña-Morán, Omar Aristeo, María Luisa Villarreal, Laura Álvarez-Berber, Angélica Meneses-Acosta, and Verónica Rodríguez-López. 2016. "Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines" Molecules 21, no. 8: 1013. https://doi.org/10.3390/molecules21081013








