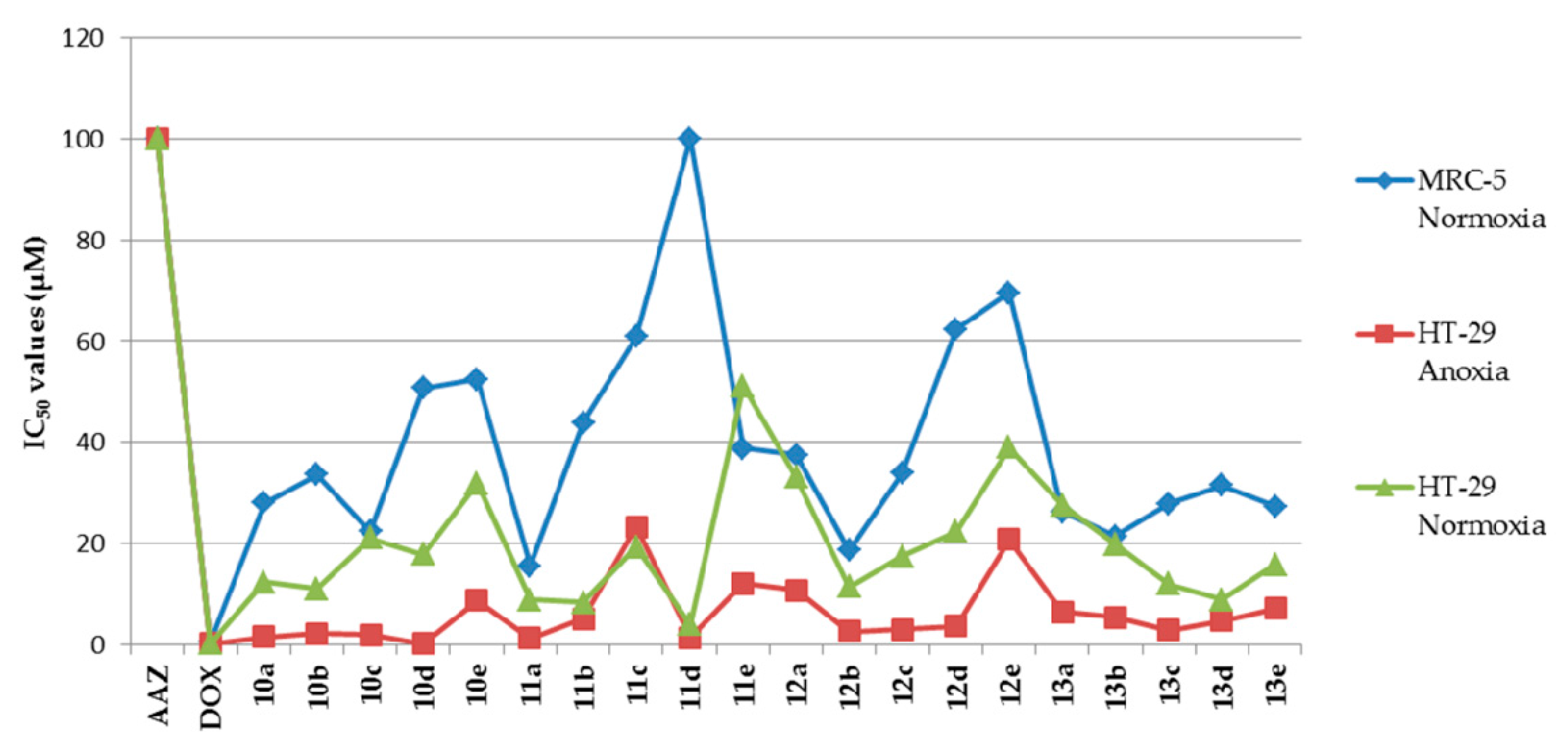
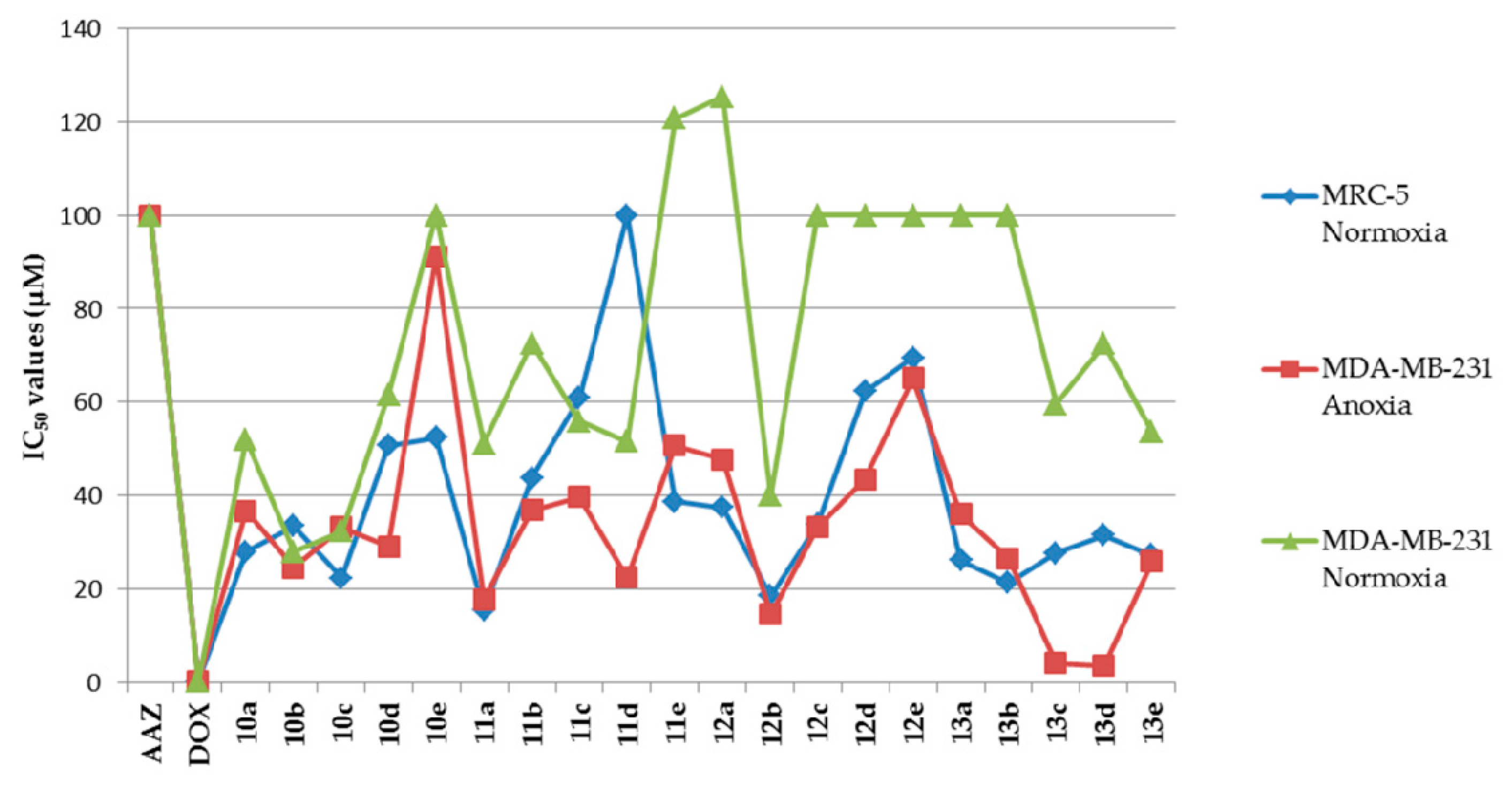
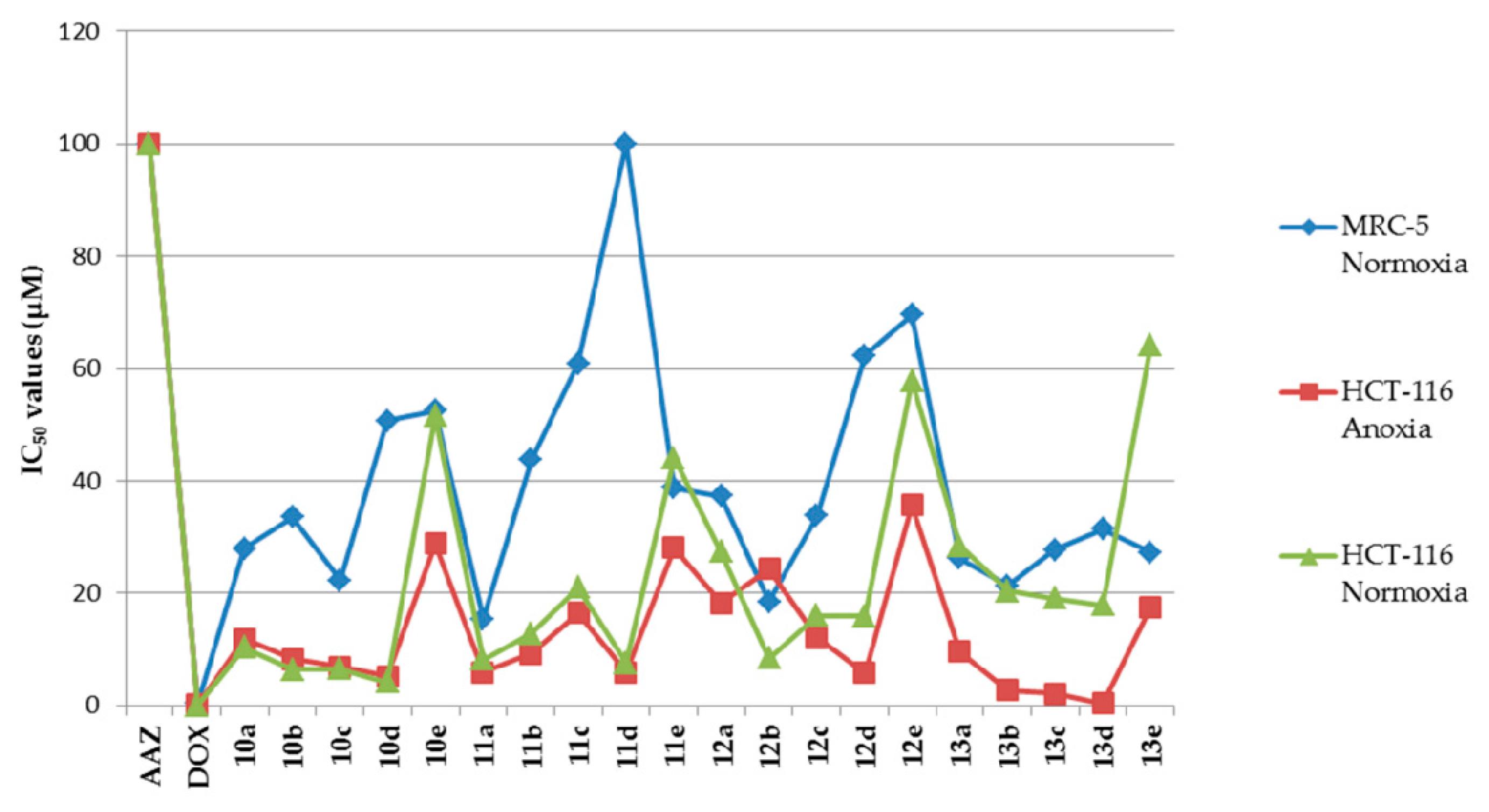
Melting points were recorded on an X-4 microscope melting point apparatus (Beijing Tech Instrument Co., Ltd., Beijing, China) without calibration. The 1H-NMR and 13C-NMR spectra were measured by a Bruker AV-400 spectrometer (Bruker Bioscience, Billerica, MA, USA), with tetramethylsilane as an internal standard. High-resolution mass spectra (HRMS) were measured with an Agilent Accurate-Mass Q-TOF 6530 (Agilent, Santa Clara, CA, USA) in ESI mode. Reaction progress was monitored by TLC on silica gel precoated GF254 plates (Qingdao Haiyang Chemical Co. Ltd., Qingdao, Shandong, China). Preparative flash column chromatography was performed on the 200–300 mesh silica gel (Qingdao Haiyang Chemical Co. Ltd., Qingdao, Shandong, China). Unless otherwise noted, all solvents and reagents were commercially available and used without further purification.
3.1.7. General Procedure for the Synthesis of Compounds 10a–e, 11a–e, 12a–e, 13a–e
To a solution of compound 8a (10 mmol) and 4a (10 mmol) in CH2Cl2 (20 mL), a mixture of CuSO4·5H2O (12.5 mg) and sodium ascorbate (30 mg) in water (10 mL) was added, and the reaction mixture was stirred at room temperature for 8 h. The reaction mixture was filtered and the organic layer was washed with water, then dried over anhydrous Na2SO4 and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography (PET/EtOAc = 2:1, v/v) to get the target compound 10a.
The compounds 10b–e, 11a–e, 12a–e, 13a–e were synthesized by the same operation procedure of compound 10a (5a–e, 6a–e with 9a; 8a, 8b with 4a–e).
7-(2-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)ethoxy-4-methyl-2H-chromen-2-one (10a): White solid; yield 31%; m.p. 83–85 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.87 (d, J = 7.6 Hz, 3H), 0.92 (d, J = 6.4 Hz, 3H), 1.19–1.28 (m, 3H), 1.44 (s, 3H), 1.45–1.50 (m, 1H), 1.55–1.59 (m, 1H), 1.70–1.75 (m, 1H), 1.80–1.90 (m, 1H), 1.99–2.04 (m, 1H), 2.33–2.37 (m, 1H), 2.40 (s, 3H), 2.60–2.68 (m, 1H), 4.43–4.46 (m, 2H), 4.76–4.71 (d, J = 12.4 Hz, 2H), 4.80–4.83 (m, 2H), 4.91 (d, J = 3.6 Hz, 1H), 4.95 (d, J = 12.4 Hz, 1H), 5.42 (s, 1H), 6.16 (s, 1H), 6.79 (d, J = 2.4 Hz, 1H), 6.80–6.83 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.50 (d, J = 8.8 Hz, 1H), 7.69 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 160.90, 160.65, 155.12, 152.29, 145.40, 125.85, 123.49, 114.39, 112.56, 112.07, 104.13, 101.77, 101.58, 87.98, 81.06, 66.80, 61.61, 52.51, 49.41, 44.35, 37.36, 36.40, 34.56, 30.78, 26.14, 24.64, 24.42, 20.31, 18.65, 12.97. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H37N3O8Na: 590.2473. Found: 590.2462.
7-(2-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)ethoxy-4-trifluoromethyl-2H-chromen-2-one (10b): White solid; yield 29%; m.p. 69–71 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.88 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.19–1.30 (m, 3H), 1.44 (s, 3H), 1.55–1.59 (m, 1H), 1.62–1.80 (m, 3H), 1.84–2.05 (m, 3H), 2.33–2.41 (m, 1H), 2.61–2.68 (m, 1H), 4.48 (t, J = 4.8 Hz, 2H), 4.69 (d, J = 12.4 Hz, 1H), 4.84 (m, 2H), 4.91 (d, J = 3.6 Hz, 1H), 4.95 (d, J = 12.4 Hz, 1H), 5.42 (s, 1H), 6.66 (s, 1H), 6.86 (d, J = 2.8 Hz, 1H), 6.89 (dd, J = 2.4 Hz, J = 9.2 Hz, 1H), 7.63 (d, J = 8.8 Hz, 1H), 7.69 (s, H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.42, 158.89, 156.07, 145.40, 130.83, 128.74, 126.57, 123.37, 113.10, 112.97, 112.93, 112.90, 112.86, 107.73, 104.06, 102.12, 101.52, 87.90, 80.97, 66.88, 65.47, 61.53, 52.40, 49.16, 44.24, 36.29, 34.46, 30.68, 26.05, 24.55, 24.33, 20.21, 12.88. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H34F3N3O8Na: 644.2190. Found: 644.2183.
7-(2-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)ethoxy-3,4-dimethyl-2H-chromen-2-one (10c): White solid; yield 37%; m.p. 82–84 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.87 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.20–1.31 (m, 3H), 1.44 (s, 3H), 1.47–1.50 (m, 1H), 1.55–1.59 (m, 1H), 1.71–1.78 (m, 2H), 1.83–1.89 (m, 1H), 2.00–2.13 (m, 2H), 2.19 (s, 3H), 2.32–2.40 (m, 1H), 2.37 (s, 3H), 2.6–2.68 (m, 1H), 4.43 (t, J = 4.8 Hz, 2H), 4.68 (d, J = 12.8 Hz, 1H), 4.80–4.83 (m, 2H), 4.91 (d, J = 3.6 Hz, 1H), 4.95 (d, J = 12.8 Hz, 1H), 5.42 (s, 1H), 6.78 (s, 1H), 6.80 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.50 (d, J = 8.8 Hz, 1H), 7.70 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.13, 159.52, 153.41, 145.96, 145.41, 125.56, 123.49, 119.67, 115.01, 111.87, 104.14, 101.60, 101.43, 87.99, 81.09, 66.71, 61.65, 52.51, 49.45, 44.34, 37.37, 36.40, 34.56, 30.79, 26.18, 24.64, 24.43, 20.34, 15.18, 13.23, 13.00. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H39N3O8Na: 604.2629. Found: 604.2636.
3-Chloro-7-(2-(4-(10β-dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)ethoxy-4-methyl-2H-chromen-2-one (10d): White solid; yield 39%; m.p. 75–77 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.87 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.4 Hz, 3H), 1.21–1.28 (m, 3H), 1.44 (s, 3H), 1.47–1.50 (m, 1H), 1.55–1.59 (m, 1H), 1.71–1.75 (m, 2H), 1.84–1.89 (m, 1H), 2.00–2.05 (m, 2H), 2.32–2.40 (m, 1H), 2.54 (s, 3H), 2.61–2.65 (m, 1H), 4.46 (m, 2H), 4.68–4.70 (d, J = 12.4 Hz, 2H), 4.81–4.84 (m, 2H), 4.90–4.91 (d, J = 3.6 Hz, 1H), 4.93–4.96 (d, J = 12.4 Hz, 1H), 5.42 (s, 1H), 6.80 (d, J = 2.4 Hz, 1H), 6.87 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.53 (d, J = 8.8 Hz, 1H), 7.70 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 160.55, 157.08, 152.92, 147.70, 145.39, 126.18, 123.51, 118.42, 114.07, 112.76, 104.14, 101.67, 101.55, 87.99, 81.06, 66.88, 61.57, 52.50, 49.36, 44.34, 37.37, 36.39, 34.56, 30.77, 26.16, 24.65, 24.43, 20.33, 16.19, 12.98. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H36ClN3O8Na: 624.2083. Found: 624.2088.
Ethyl 2-oxo-7-(2-(4-(10β-dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)ethoxy-2H-chromene-3-carboxylate (10e): White solid; yield 23%; m.p. 153–155 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.87 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.4 Hz, 3H), 1.19–1.30 (m, 3H), 1.40 (t, J = 6.8 Hz, 3H), 1.44 (s, 3H), 1.46–1.60 (m, 3H), 1.70–1.78 (m, 2H), 1.83–1.90 (m, 1H), 2.00–2.06 (m, 1H), 2.33–2.41 (m, 1H), 2.61–2.68 (m, 1H), 4.40 (q, J = 7.2 Hz, 2H), 4.48 (t, J = 4.8 Hz, 2H), 4.69 (d, J = 12.8 Hz, 1H), 4.84 (m, 2H), 4.91 (d, J = 3.2 Hz, 1H), 4.95 (d, J = 12.4 Hz, 1H), 5.42 (s, 1H), 6.80 (d, J = 2.4 Hz, 1H), 6.86 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.69 (s, 1H), 8.50 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 163.21, 162.98, 157.26, 156.79, 148.62, 145.44, 130.98, 123.49, 114.93, 113.35, 112.33, 104.15, 101.58, 101.21, 87.99, 81.07, 67.01, 61.82, 61.58, 52.49, 49.25, 44.32, 37.38, 36.38, 34.55, 30.77, 26.17, 24.65, 24.43, 20.34, 14.27, 12.99. ESI-HRMS [M + Na]+: (m/z) Calcd. for C32H39N3O10Na: 648.2528. Found: 648.2525.
7-(3-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)propoxy-4-methyl-2H-chromen-2-one (11a): White solid; yield 32%; m.p. 69–70 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.84 (d, J = 7.6 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.21–1.28 (m, 3H), 1.44 (s, 3H), 1.46–1.50 (m, 1H), 1.55–1.59 (m, 1H), 1.65–1.76 (m, 3H), 1.84–1.89 (m, 1H), 1.99–2.05 (m, 2H), 2.41 (s, 3H), 2.46 (m, 2H), 2.61–2.64 (m, 1H), 4.04 (t, J = 5.6 Hz, 2H), 4.60–4.63 (m, 2H), 4.67 (d, J = 12.8 Hz, 1H), 4.89 (d, J = 3.2 Hz, 1H), 4.93 (d, J = 12.4 Hz, 1H), 5.39 (s, 1H), 6.16 (s, 1H), 6.79 (d, J = 2.0 Hz, 1H), 6.84 (dd, J = 3.2 Hz, J = 8.8 Hz, 1H), 7.50 (d, J = 3.2 Hz, 1H), 7.51 (d, J = 5.2 Hz, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.35, 161.11, 155.21, 152.43, 145.23, 125.75, 122.83, 114.02, 112.29, 112.11, 104.13, 101.71, 101.66, 87.96, 81.08, 64.69, 61.72, 52.49, 46.87, 44.34, 37.38, 36.40, 34.55, 30.79, 29.70, 26.16, 24.67, 24.42, 20.32, 18.69, 12.96. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H39N3O8Na: 604.2629. Found: 604.2618.
7-(3-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)propoxy-4-trifluoromethyl-2H-chromen-2-one (11b): White solid; yield 19%; m.p. 67–69 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.85 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.19–1.29 (m, 3H), 1.44 (s, 3H), 1.46–1.50 (m, 2H), 1.55–1.59 (m, 1H), 1.66–1.76 (m, 2H), 1.84–1.89 (m, 1H), 1.99–2.05 (m, 1H), 2.32–2.40 (m, 1H), 2.48 (m, 2H), 2.59–2.67 (m, 1H), 4.07 (t, J = 6.0 Hz, 2H), 4.60–4.64 (m, 2H), 4.68 (d, J = 12.4 Hz, 1H), 4.90 (d, J = 3.2 Hz, 1H), 4.93 (d, J = 12.4 Hz, 1H), 5.39 (s, 1H), 6.64 (s, 1H), 6.85 (d, J = 2.8 Hz, 1H), 6.91 (dd, J = 2.4 Hz, J = 9.2 Hz, 1H), 7.52 (s, 1H), 7.64 (dd, J = 1.6 Hz, J = 8.8 Hz, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.23, 159.19, 156.25, 145.29, 126.57, 122.807, 120.19, 113.27, 112.66, 112.60, 107.44, 104.14, 102.10, 101.71, 87.96, 81.07, 64.95, 61.70, 52.48, 46.76, 44.33, 37.38, 36.39, 34.54, 30.78, 29.60, 26.15, 24.67, 24.41, 20.31, 12.95. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H36F3N3O8Na: 658.2347. Found: 658.2350.
7-(3-(4-(10β-Dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)propoxy-3,4-dimethyl-2H-chromen-2-one (11c): White solid; yield 35%; m.p. 83–85 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.85 (d, J = 5.2 Hz, 3H), 0.87 (d, J = 6.0 Hz, 3H), 0.90–0.99 (m, 1H), 1.15–1.27 (m, 2H), 1.30–1.31 (m, 1H), 1.55 (s, 3H), 1.58–1.72 (m, 3H), 1.74–1.79 (m, 1H), 1.82–1.89 (m, 1H), 2.19 (s, 3H), 2.38 (s, 3H), 2.40–2.48 (m, 3H), 3.57 (m, 1H), 4.00–4.03 (m, 2H), 4.59–4.65 (m, 3H), 4.87 (d, J = 4.4 Hz, 1H), 4.90 (d, J = 8.8 Hz, 1H), 5.26 (s, 1H), 6.77 (d, J = 2.4 Hz, 1H), 6.83 (dd, J = 2.8 Hz, J = 8.8 Hz, 1H), 7.49 (s, 1H), 7.51 (d, J = 9.2 Hz, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.30, 160.24, 153.48, 146.11, 145.32, 125.44, 122.75, 119.31, 114.61, 111.96, 107.99, 101.32, 99.46, 93.80, 84.08, 77.26, 69.56, 64.53, 61.81, 46.90, 42.41, 40.63, 34.82, 34.70, 30.32, 29.72, 24.98, 20.99, 18.82, 15.10, 13.19, 12.26. ESI-HRMS [M + Na]+: (m/z) Calcd. for C32H41N3O8Na: 618.2786. Found: 618.2784.
3-Chloro-7-(3-(4-(10β-dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)propoxy-4-methyl-2H-chromen-2-one (11d): White solid; yield 34%; m.p. 150–152 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.85 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.19–1.28 (m, 3H), 1.44 (s, 3H), 1.45–1.49 (m, 1H), 1.55–1.59 (m, 1H), 1.65–1.81 (m, 3H), 1.83–1.90 (m, 1H), 1.99–2.05 (m, 1H), 2.32–2.40 (m, 1H), 2.47 (m, 2H), 2.56 (s, 3H), 2.59–2.66 (m, 1H), 4.05 (m, 2H), 4.61 (m, 2H), 4.67 (d, J = 12.8 Hz, 1H), 4.89 (d, J = 3.2 Hz, 1H), 4.92 (d, J = 12.8 Hz, 1H), 5.39 (s, 1H), 6.80 (d, J = 2.4 Hz, 1H), 6.88 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (s, 1H), 7.54 (d, J = 8.8 Hz, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.26, 157.26, 153.01, 147.84, 145.22, 126.09, 122.84, 118.10, 113.68, 112.81, 104.13, 101.67, 101.56, 87.96, 81.07, 64.84, 61.67, 52.48, 46.86, 44.33, 37.38, 36.39, 34.55, 30.78, 29.68, 26.16, 24.67, 24.42, 20.33, 16.21, 12.97. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H38ClN3O8Na: 638.2240. Found: 638.2243.
Ethyl 2-oxo-7-(3-(4-(10β-dihydroartemisininoxy)methyl)-1H-1,2,3-triazol-1-yl)propoxy-2H-chromene-3-carboxylate (11e): White solid; yield 29%; m.p. 124–126 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.85 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 6.0 Hz, 3H), 1.20–1.32 (m, 3H), 1.41 (t, J = 7.2 Hz, 3H), 1.44 (s, 3H), 1.46–1.50 (m, 1H), 1.55–1.59 (m, 1H), 1.64–1.81 (m, 3H), 1.83–1.90 (m, 1H), 1.99–2.05 (m, 1H), 2.32–2.40 (m, 1H), 2.48 (m, 2H), 2.59–2.67 (m, 1H), 4.08 (t, J = 6.0 Hz, 2H), 4.41 (q, J = 7.2 Hz, 2H), 4.61 (td, J = 2.0 Hz, J = 6.8 Hz, 2H), 4.68 (d, J = 12.4 Hz, 1H), 4.90 (d, J = 3.2 Hz, 1H), 4.93 (d, J = 12.8 Hz, 1H), 5.39 (s, 1H), 6.79 (d, J = 2.0 Hz, 1H), 6.87 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 6.4 Hz, 1H), 7.53 (d, J = 2.4 Hz, 1H), 8.50 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 163.78, 163.31, 157.37, 156.96, 148.80, 145.26, 130.91, 122.83, 114.48, 113.45, 111.97, 104.13, 101.69, 101.12, 87.95, 81.07, 65.11, 61.76, 61.68, 52.47, 46.82, 44.32, 37.37, 36.381, 34.54, 30.78, 29.62, 26.16, 24.66, 24.41, 20.32, 14.29, 12.97. ESI-HRMS [M + Na]+: (m/z) Calcd. for C33H41N3O10Na: 662.2684. Found: 662.2679.
7-(1-(2-(10β-Dihydroartemisininoxy)ethyl)-1H-1,2,3-triazol-4-yl)methoxy-4-methyl-2H-chromen-2-one (12a): White solid; yield 32%; m.p. 79–81 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.78 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 5.6 Hz, 3H), 1.16–1.30 (m, 3H), 1.36–1.39 (m, 1H), 1.42 (s, 3H), 1.45–1.52 (m, 2H), 1.56–1.60 (m, 1H), 1.64–1.74 (m, 1H), 1.84–1.88 (m, 1H), 1.99–2.05 (m, 1H), 2.31–2.39 (m, 1H), 2.41 (s, 3H), 2.57–2.64 (m, 1H), 3.79–3.84 (m, 1H), 4.27–4.32 (m, 1H), 4.51–4.57 (m, 1H), 4.63–4.69 (m, 1H), 4.76 (d, J = 3.2 Hz, 1H), 5.15 (s, 1H), 5.27 (s, 2H), 6.16 (s, 1H), 6.92 (d, J = 2.0 Hz, 1H), 6.95 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.72 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.13, 155.15, 152.42, 143.10, 125.73, 123.49, 114.09, 112.33, 112.31, 104.21, 102.18, 102.13, 87.87, 80.80, 77.24, 66.40, 62.33, 52.38, 50.50, 44.04, 37.34, 36.31, 34.45, 30.59, 26.10, 24.61, 24.34, 20.36, 18.69, 12.81. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H37N3O8Na: 590.2473. Found: 590.2471.
7-(1-(2-(10β-Dihydroartemisininoxy)ethyl)-1H-1,2,3-triazol-4-yl)methoxy-4-trifluoromethyl-2H-chromen-2-one (12b): White solid; yield 24%; m.p. 77–79 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.78 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 5.6 Hz, 3H), 1.16–1.31 (m, 3H), 1.42 (s, 3H), 1.48–1.76 (m, 4H), 1.84–1.88 (m, 1H), 2.00–2.05 (m, 1H), 2.31–2.34 (m, 1H), 2.59–2.62 (m, 1H), 3.80–3.85 (m, 1H), 4.25–4.32 (m, 1H), 4.51–4.70 (m, 2H), 4.76 (d, J = 2.7 Hz, 1H), 5.16 (s, 1H), 5.29 (s, 1H), 6.64 (s, 1H), 7.01–7.03 (m, 2H), 7.64 (d, J = 9.2 Hz, 1H), 7.73 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.928, 159.095, 156.088, 142.555, 126.430, 124.988, 123.535, 113.398, 112.573, 107.385, 104.107, 102.503, 102.073, 87.770, 80.677, 77.191, 76.979, 76.767, 66.282, 62.356, 52.277, 50.429, 43.935, 37.235, 36.202, 34.358, 30.480, 25.974, 24.517, 24.229, 20.241, 19.083, 12.691. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H34F3N3O8Na: 644.2190. Found: 644.2187.
7-(1-(2-(10β-Dihydroartemisininoxy)ethyl)-1H-1,2,3-triazol-4-yl)methoxy-3,4-dimethyl-2H-chromen-2-one (12c): White solid; yield 36%; m.p. 84–86 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.78 (d, J = 7.2 Hz, 3H), 0.91 (d, J = 4.4 Hz, 3H), 0.97–1.29 (m, 3H), 1.42 (s, 3H), 1.45–1.55 (m, 2H), 1.55–2.05 (m, 5H), 2.18 (s, 3H), 2.30–2.34 (m, 1H), 2.37 (s, 3H), 2.57–2.61 (m, 1H), 3.79–3.84 (m, 1H), 4.27–4.32 (m, 1H), 4.51–4.57 (m, 1H), 4.63–4.69 (m, 1H), 4.76 (d, J = 3.2 Hz, 1H), 5.15 (m, 1H), 5.25 (m, 2H), 6.90–6.95 (m, 2H), 7.52 (dd, J = 3.6 Hz, J = 8.8 Hz, 1H), 7.72 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.26, 160.01, 153.39, 146.12, 143.21, 125.44, 123.52, 119.29, 114.66, 112.10, 104.16, 102.13, 101.76, 87.84, 80.78, 66.38, 62.25, 52.37, 50.47, 44.03, 37.29, 36.30, 34.43, 30.58, 26.07, 24.58, 24.31, 20.34, 15.08, 13.17, 12.80. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H39N3O8Na: 604.2629. Found: 604.2645.
3-Chloro-7-(1-(2-(10β-dihydroartemisininoxy)ethyl)-1H-1,2,3-triazol-4-yl)methoxy-4-methyl-2H-chromen-2-one (12d): White solid; yield 33%; m.p. 76–78 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.78 (d, J = 7.2 Hz, 3H), 0.92 (d, J = 5.6 Hz, 3H), 1.16–1.32 (m, 3H), 1.32–1.42 (m, 2H), 1.42 (s, 3H), 1.44–1.69 (m, 5H), 1.84–1.88 (m, 1H), 1.99–2.15 (m, 2H), 2.31–2.39 (m, 1H), 2.58–2.62 (m, 1H), 3.79–3.85 (m, 1H), 4.27–4.32 (m, 1H), 4.51–4.58 (m, 1H), 4.63–4.69 (m, 1H), 4.76 (d, J = 3.2 Hz, 1H), 5.15 (s, 1H), 5.27 (s, 2H), 6.94 (d, J = 2.4 Hz, 1H), 7.00 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.55 (d, J = 8.8 Hz, 1H), 7.73 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.04, 157.29, 152.97, 147.83, 142.90, 126.07, 123.56, 118.17, 113.78, 113.02, 104.21, 102.18, 102.06, 87.87, 80.79, 66.40, 62.38, 52.38, 50.51, 44.03, 37.33, 36.31, 34.46, 30.59, 26.09, 24.61, 24.34, 20.36, 16.20, 12.82. ESI-HRMS [M + Na]+: (m/z) Calcd. for C30H36ClN3O8Na: 624.2083. Found: 624.2111.
Ethyl 2-oxo-7-(1-(2-(10β-dihydroartemisininoxy)ethyl)-1H-1,2,3-triazol-4-yl)methoxy-2H-chromene-3-carboxylate (12e): White solid; yield 26%; m.p. 71–73 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.78 (d, J = 7.6 Hz, 3H), 0.92 (d, J = 5.6 Hz, 3H), 1.16–1.28 (m, 3H), 1.39–1.42 (m, 8H), 1.47–1.52 (m, 1H), 1.55–1.60 (m, 1H), 1.65–1.69 (m, 1H), 1.84–1.89 (m, 1H), 1.99–2.05 (m, 1H), 2.31–2.39 (m, 1H), 2.57–2.64 (m, 1H), 3.80–3.85 (m, 1H), 4.27–4.32 (m, 1H), 4.40 (q, J = 3.2 Hz, 2H), 4.52–4.58 (m, 1H), 4.64–4.70 (m, 1H), 4.76 (d, J = 3.6 Hz, 1H), 5.15 (s, 1H), 5.29 (s, 2H), 6.94 (d, J = 2.0 Hz, 1H), 6.98 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.74 (s, 1H), 8.51 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 163.55, 163.36, 157.34, 156.98, 148.79, 142.60, 130.88, 123.64, 114.63, 113.63, 112.08, 104.23, 102.19, 101.62, 87.88, 80.78, 66.40, 62.52, 61.79, 52.37, 50.54, 44.02, 37.35, 36.31, 34.46, 30.58, 26.10, 24.62, 24.34, 20.36, 14.29, 12.82. ESI-HRMS [M + Na]+: (m/z) Calcd. for C32H39N3O10Na: 648.2528. Found: 648.2530.
7-(1-(3-(10β-Dihydroartemisininoxy)propyl)-1H-1,2,3-triazol-4-yl)methoxy-4-methyl-2H-chromen-2-one (13a): White solid; yield 39%; m.p. 75–77 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.93 (d, J = 7.2 Hz, 3H), 0.96 (d, J = 6.4 Hz, 3H), 1.22–1.33 (m, 3H), 1.43 (s, 3H), 1.46–1.55 (m, 2H), 1.63–1.69 (m, 1H), 1.76–1.78 (m, 1H), 1.86–1.93 (m, 1H), 2.02–2.13 (m, 2H), 2.17–2.25 (m, 2H), 2.33–2.38 (m, 1H), 2.41 (s, 3H), 2.62–2.69 (m, 1H), 3.36–3.42 (m, 1H), 3.87–3.93 (m, 1H), 4.44–4.52 (m, 2H),4.77 (d, J = 3.6 Hz, 1H), 5.27 (s, 2H), 5.40 (s, 1H), 6.15 (s, 1H), 6.93 (d, J = 2.0 Hz, 1H), 6.96 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.66 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.18, 161.13, 155.12, 152.50, 143.05, 125.74, 123.05, 114.07, 112.39, 112.29, 104.19, 102.19, 102.13, 87.94, 80.97, 64.65, 62.311, 52.50, 47.65, 44.27, 37.46, 36.37, 34.56, 30.81, 30.44, 26.15, 24.66, 24.55, 20.36, 18.70, 13.09. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H39N3O8Na: 604.2629. Found: 604.2674.
7-(1-(3-(10β-Dihydroartemisininoxy)propyl)-1H-1,2,3-triazol-4-yl)methoxy-4-trifluoromethyl-2H-chromen-2-one (13b): White solid; yield 25%; m.p. 69–71 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.94 (d, J = 7.2 Hz, 3H), 0.97 (d, J = 6.0 Hz, 3H), 1.22–1.39 (m, 3H), 1.43 (s, 3H), 1.46–1.55 (m, 2H), 1.64–1.78 (m, 3H), 1.87–1.92 (m, 1H), 2.02–2.06 (m, 1H), 2.16–2.28 (m, 2H), 2.34–2.41 (m, 1H), 2.63–2.70 (m, 1H), 3.37–3.43 (m, 1H), 3.87–3.93 (m, 1H), 4.45–4.54 (m, 2H), 4.78 (d, J = 3.2 Hz, 1H), 5.29 (s, 2H), 5.40 (s, 1H), 6.64 (s, 1H), 7.02–7.03 (m, 2H), 7.64–7.67 (m, 2H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.03, 159.26, 156.18, 142.64, 141.33, 126.52, 123.13, 113.55, 112.67, 112.62, 107.50, 104.21, 102.61, 102.21, 87.95, 80.96, 64.66, 62.45, 52.50, 47.68, 44.27, 37.47, 36.37, 34.56, 30.81, 30.44, 26.14, 24.66, 24.55, 20.36, 13.09. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H36F3N3O8Na: 658.2347. Found: 658.2345.
7-(1-(3-(10β-Dihydroartemisininoxy)propyl)-1H-1,2,3-triazol-4-yl)methoxy-3,4-dimethyl-2H-chromen-2-one (13c): White solid; yield 37%; m.p. 68–70 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.93 (d, J = 7.6 Hz, 3H), 0.96 (d, J = 6.4 Hz, 3H), 1.22–1.40 (m, 3H), 1.43 (s, 3H), 1.46–1.52 (m, 2H), 1.54–1.78 (m, 3H), 1.86–1.95 (m, 1H), 2.01–2.17 (m, 2H), 2.19 (s, 3H), 2.22–2.35 (m, 2H), 2.38 (s, 3H), 2.60–2.69 (m, 1H), 3.36–3.41 (m, 1H), 3.87–3.93 (m, 1H), 4.42–4.53 (m, 2H), 4.77 (d, J = 3.6 Hz, 1H), 5.26 (s, 2H), 5.40 (s, 1H), 6.91 (d, J = 2.4 Hz, 1H), 6.94 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.65 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 162.33, 160.02, 153.40, 146.16, 143.22, 125.44, 122.99, 119.31, 114.68, 112.18, 104.18, 102.18, 101.82, 87.93, 80.97, 64.65, 62.28, 52.51, 47.62, 44.27, 37.45, 36.37, 34.56, 30.82, 30.44, 26.15, 24.66, 24.55, 20.36, 15.10, 13.19, 13.09. ESI-HRMS [M + Na]+: (m/z) Calcd. for C32H41N3O8Na: 618.2786. Found: 618.2785.
3-Chloro-7-(1-(3-(10β-Dihydroartemisininoxy)propyl)-1H-1,2,3-triazol-4-yl)methoxy-4-methyl-2H-chromen-2-one (13d): White solid; yield 38%; m.p. 71–73 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.93 (d, J = 7.6 Hz, 3H), 0.97 (d, J = 6.4 Hz, 3H), 1.22–1.29 (m, 2H), 1.32–1.38 (m, 1H), 1.43 (s, 3H), 1.46–1.51 (m, 2H), 1.58–1.82 (m, 3H), 1.87–1.92 (m, 1H), 2.02–2.07 (m, 1H), 2.17–2.25 (m, 2H), 2.33–2.41 (m, 1H), 2.56 (s, 3H), 2.62–2.70 (m, 1H), 3.36–3.42 (m, 1H), 3.87–3.92 (m, 1H), 4.41–4.56 (m, 2H), 4.77 (d, J = 3.2 Hz, 1H), 5.27 (s, 2H), 5.40 (s, 1H), 6.95 (d, J = 2.0 Hz, 1H), 7.01 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.55 (d, J = 8.8 Hz, 1H), 7.66 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 161.04, 157.34, 152.98, 147.86, 142.90, 126.07, 123.06, 118.17, 113.80, 113.08, 104.21, 102.21, 102.09, 87.95, 80.97, 64.65, 62.40, 52.51, 47.65, 44.28, 37.47, 36.38, 34.57, 30.82, 30.45, 26.16, 24.67, 24.56, 20.37, 16.21, 13.10. ESI-HRMS [M + Na]+: (m/z) Calcd. for C31H38ClN3O8Na: 638.2240. Found: 638.2240.
Ethyl 2-oxo-7-(1-(3-(10β-dihydroartemisininoxy)propyl)-1H-1,2,3-triazol-4-yl)methoxy-2H-chromene-3-carboxylate (13e): White solid; yield 30%; m.p. 79–81 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 0.93 (d, J = 7.6 Hz, 3H), 0.96 (d, J = 5.6 Hz, 3H), 1.22–1.32 (m, 2H), 1.39–1.43 (m, 6H), 1.47–1.56 (m, 2H), 1.61–1.73 (m, 2H), 1.76–1.78 (m, 1H), 1.86–1.93 (m, 1H), 1.97–2.07 (m, 2H), 2.18–2.26 (m, 2H), 2.33–2.41 (m, 1H), 2.62–2.68 (m, 1H), 3.37–3.43 (m, 1H), 3.87–3.92 (m, 1H), 4.40 (q, 2H), 4.45–4.55 (m, 2H), 4.78 (d, J = 3.2 Hz, 1H), 5.29 (s, 2H), 5.40 (s, 1H), 6.94 (d, J = 2.0 Hz, 1H), 6.99 (dd, J = 2.4 Hz, J = 8.8 Hz, 1H), 7.53 (d, J = 8.8 Hz, 1H), 7.68 (s, 1H), 8.50 (s, 1H). 13C-NMR (100 MHz, CDCl3) δ (ppm): 163.59, 163.40, 157.32, 157.03, 148.84, 146.18, 130.84, 114.59, 113.84, 112.13, 104.19, 102.13, 101.78, 87.97, 81.01, 80.63, 64.64, 62.26, 61.77, 52.54, 48.49, 44.34, 37.47, 36.39, 34.63, 30.85, 30.14, 29.71, 26.17, 24.68, 20.38, 14.29, 13.16. ESI-HRMS [M + Na]+: (m/z) Calcd. for C33H41N3O10Na: 662.2684. Found: 662.2680.