Efficient Preparation of Streptochlorin from Marine Streptomyces sp. SYYLWHS-1-4 by Combination of Response Surface Methodology and High-Speed Counter-Current Chromatography
Abstract
:1. Introduction
2. Results
2.1. Optimization of Culture Conditions by RSM
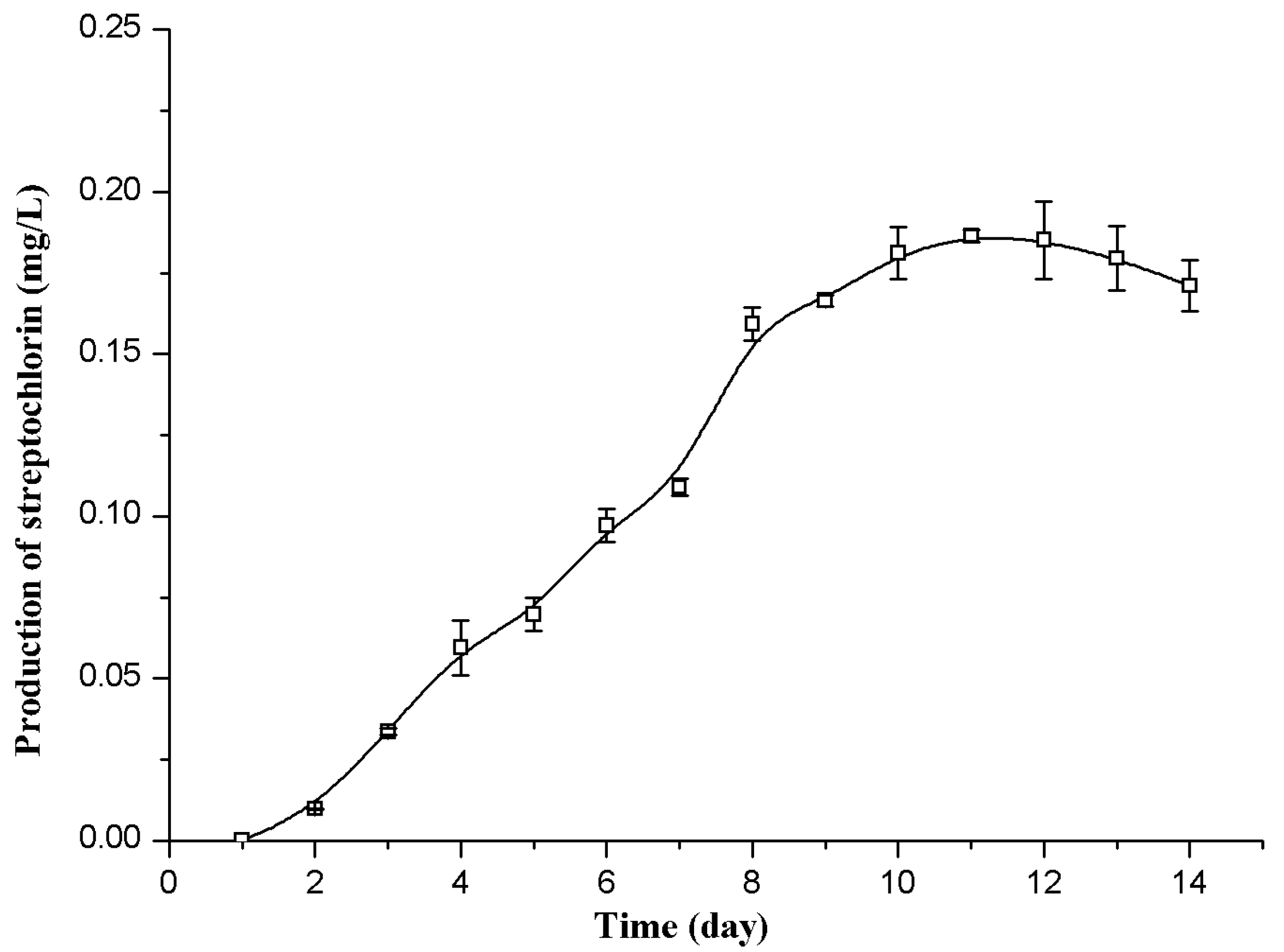
2.1.1. Determination of the Best Time in Flask Fermentation
2.1.2. Plackett-Burman Design and Identification of Significant Variables that Affect Streptochlorin Production
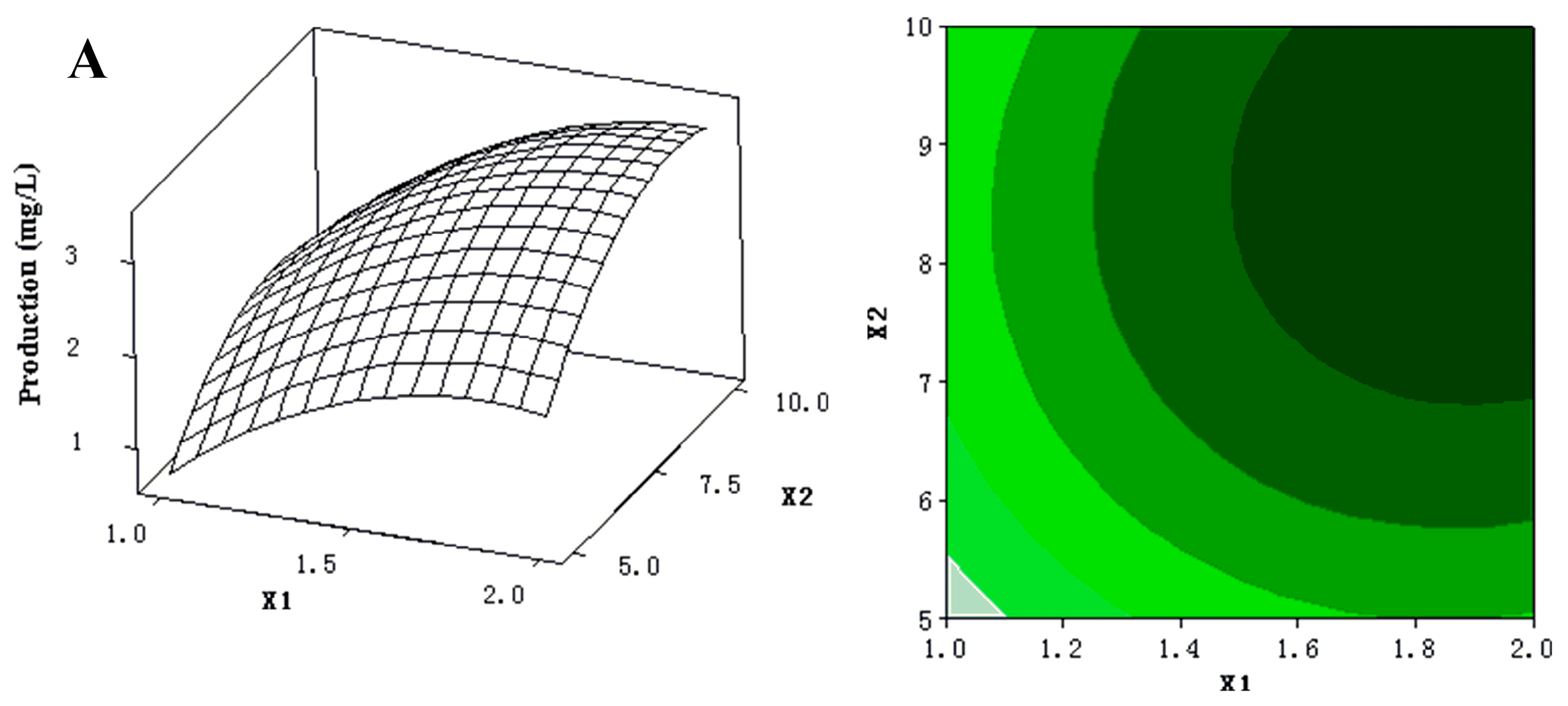
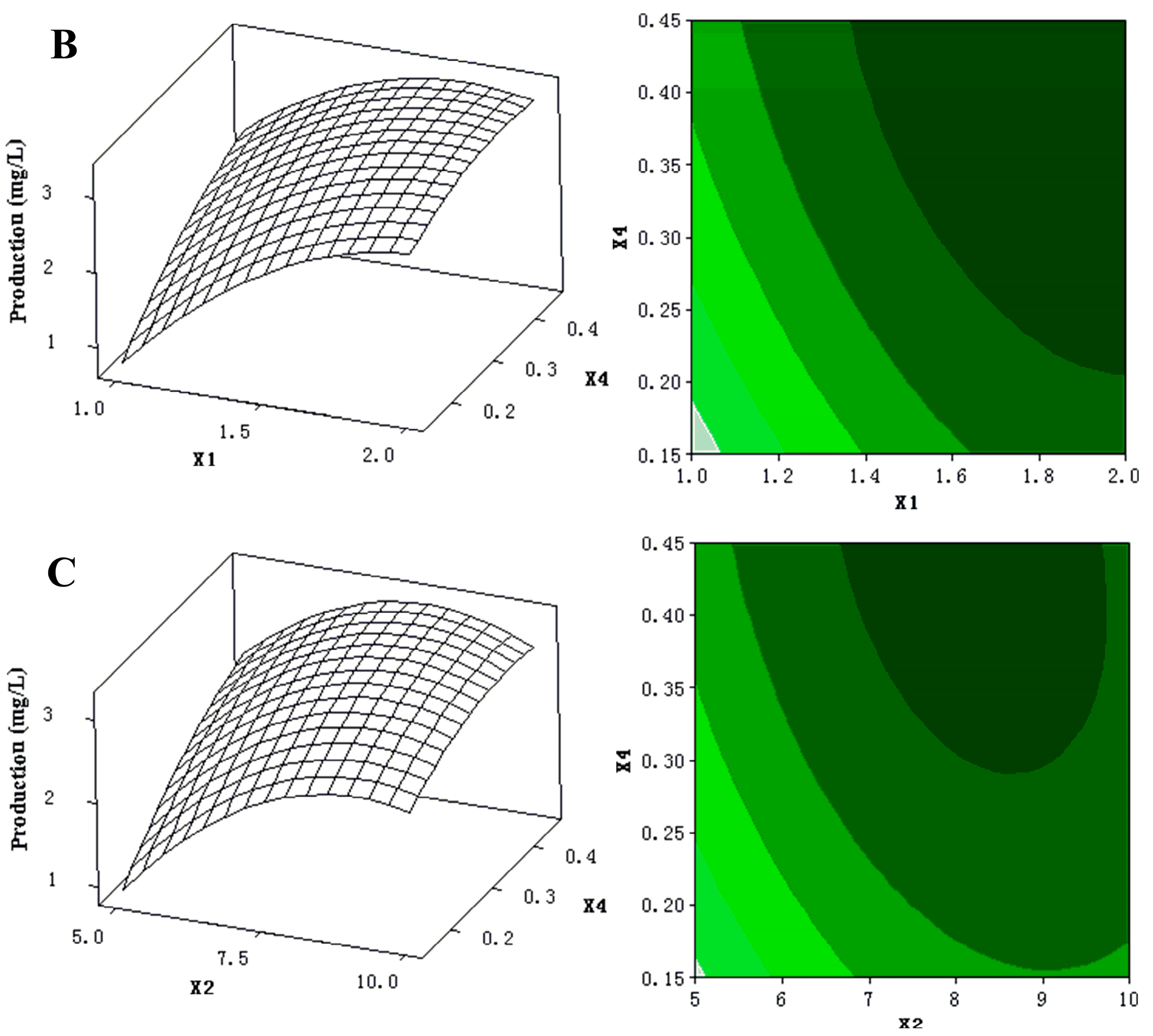
2.1.3. Optimization by Box-Behnken Design
2.1.4. Validation of the Optimized Condition
2.2. Preparation of Streptochlorin by HSCCC
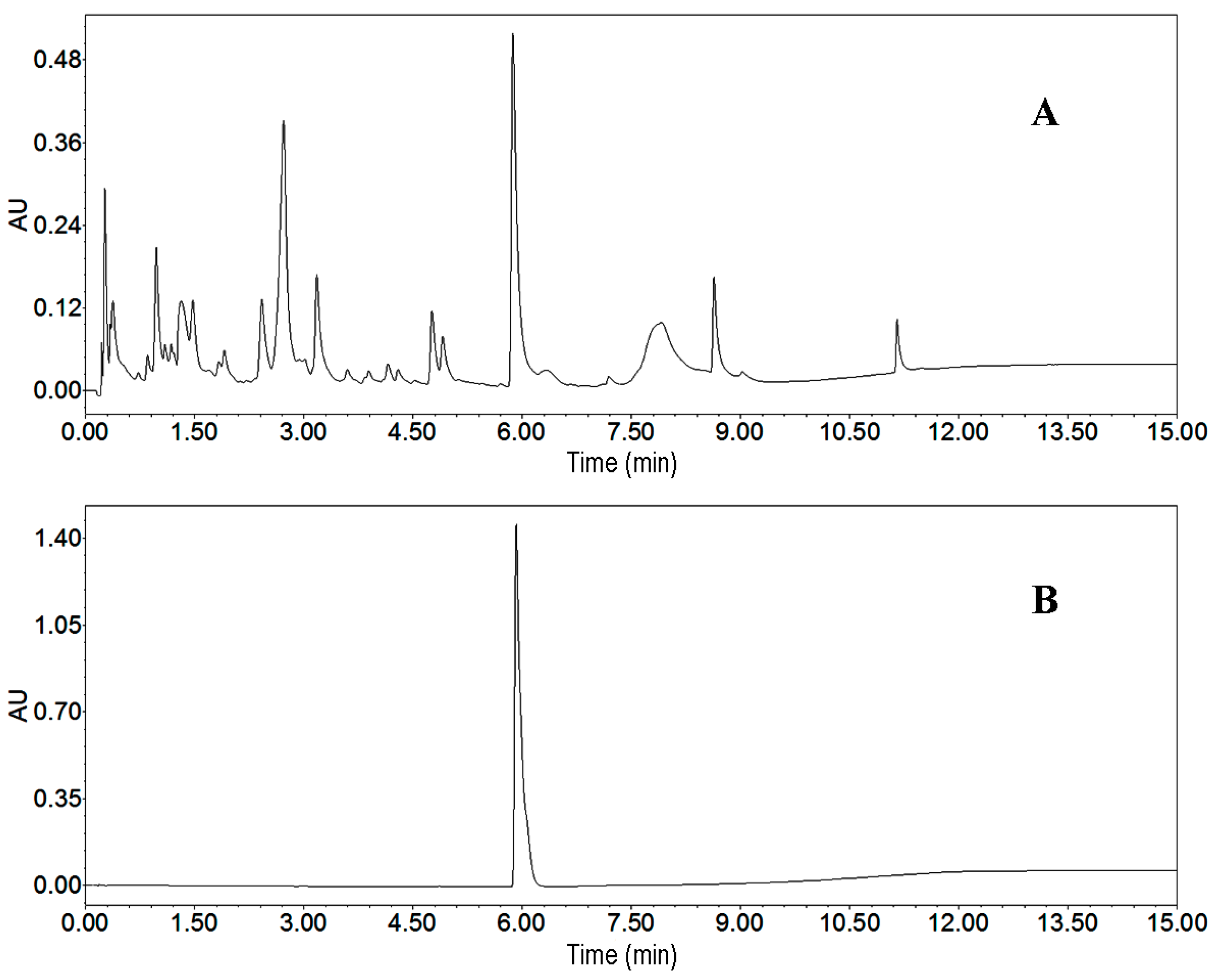
2.2.1. UPLC Analysis of the Crude Sample
2.2.2. Selection of Suitable Two-Phase Solvent System
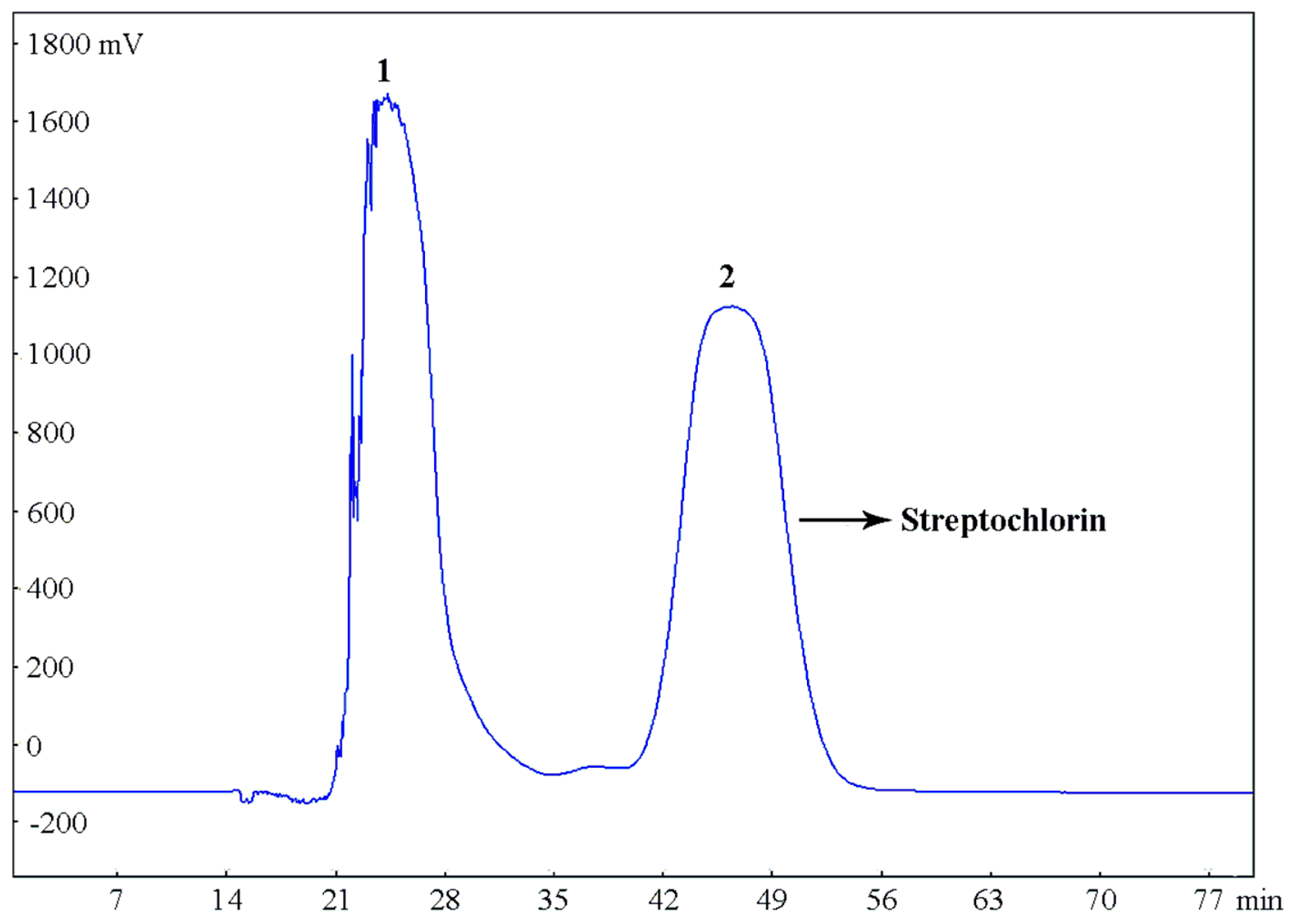
2.2.3. HSCCC Separation
2.2.4. UPLC Analysis and Identification of Peak Fraction
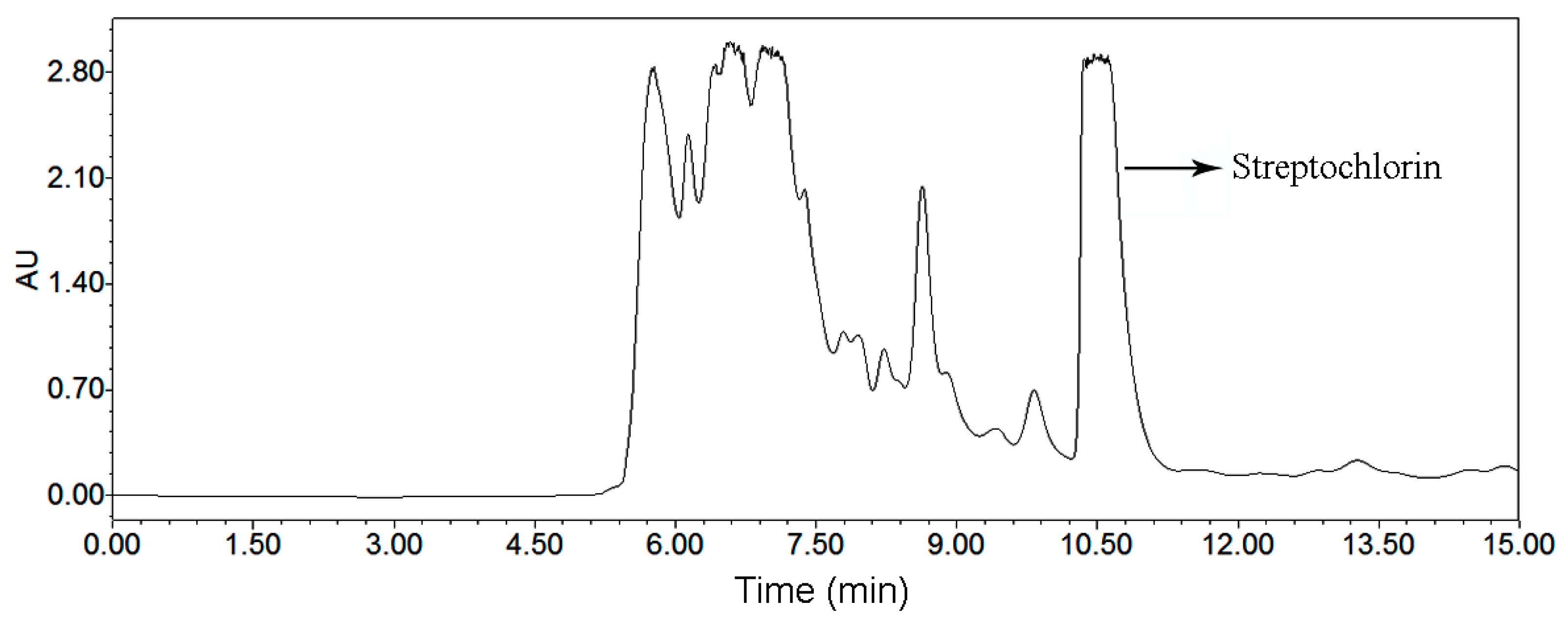
2.2.5. Comparison of Two Chromatographic Systems for Preparative Separation of Streptochlorin
3. Discussion
4. Materials and Methods
4.1. Microorganism and Fermentation Conditions
4.2. Experimental Design and RSM Optimization
4.3. Production of Streptochlorin from the Streptomyces Strain SYYLWHS-1-4 by a Jar Fermenter
4.4. UPLC Analysis, Preparative HPLC Separation and Structure Identification
4.5. Preparation of Crude Sample
4.6. Evaluation of Partition Coefficient Value
4.7. Preparation of Solvent System and Sample Solution for HSCCC Separation
4.8. HSCCC Separation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| RSM | Response surface methodology |
| HSCCC | High-speed counter-current chromatography |
| PBD | Plackett-Burman design |
| BBD | Box-Bohnken design |
| UPLC | Ultra Performance Liquid Chromatography |
| HPLC | High Performance Liquid Chromatography |
| ESI-MS | Electro Spray Ionization-Mass Spectrometry |
| NMR | Nuclear Magnetic Resonance |
| HRESIMS | High Resolution Electrospray Ionization Mass Spectroscopy |
| TMS | Tetramethylsilane |
| DKPs | Diketopiperazines |
References
- Subramani, R.; Aalbersberg, W. Culturable rare Actinomycetes: Diversity, isolation and marine natural product discovery. Appl. Microbiol. Biotechnol. 2013, 97, 9291–9321. [Google Scholar] [CrossRef] [PubMed]
- Takano, H.; Nishiyama, T.; Amano, S.; Beppu, T.; Kobayashi, M.; Ueda, K. Streptomyces metabolites in divergent microbial interactions. J. Ind. Microbiol. Biotechnol. 2016, 43, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Amano, S.; Yoshida, J.; Takase, Y.; Miyadoh, S.; Sasaki, T.; Hatsu, M.; Takeuchi, Y.; Komada, Y. A new antibiotic SF2583A, 4-chloro-5-(3′-indolyl) oxazole, produced by Streptomyces. Meiji Seika Kenkyu Nenpo 1988, 27, 55–62. [Google Scholar]
- Kroiss, J.; Kaltenpoth, M.; Schneider, B.; Schwinger, M.G.; Hertweck, C.; Maddula, R.K.; Strohm, E.; Svatos, A. Symbiotic streptomycetes provide antibiotic combination prophylaxis for wasp offspring. Nat. Chem. Biol. 2010, 6, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chen, Q.; Xie, C.; Mulholland, N.; Turner, S.; Irwin, D.; Gu, Y.; Yang, G.; Clough, J. Synthesis and antifungal activity of novel streptochlorin analogues. Eur. J. Med. Chem. 2015, 92, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Shim, D.W.; Shin, H.J.; Han, J.W.; Shin, W.Y.; Sun, X.; Shim, E.J.; Kim, T.J.; Kang, T.B.; Lee, K.H. Anti-inflammatory effect of streptochlorin via TRIF-dependent signaling pathways in cellular and mouse models. Int. J. Mol. Sci. 2015, 16, 6902–6910. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Shin, H.J.; Kim, D.Y.; Shim, D.W.; Kim, T.J.; Ye, S.K.; Won, H.S.; Koppula, S.; Kang, T.B.; Lee, K.H. Streptochlorin suppresses allergic dermatitis and mast cell activation via regulation of Lyn/Fyn and Syk signaling pathways in cellular and mouse models. PLoS ONE 2013, 8, e74194. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.Y.; Shin, H.J.; Kim, G.Y.; Cheong, J.H.; Choi, I.W.; Kim, S.K.; Moon, S.K.; Kang, H.S.; Choi, Y.H. Streptochlorin isolated from Streptomyces sp. Induces apoptosis in human hepatocarcinoma cells through a reactive oxygen species-mediated mitochondrial pathway. J. Microbiol. Biotechnol. 2008, 18, 1862–1868. [Google Scholar] [PubMed]
- Shin, H.J.; Jeong, H.S.; Lee, H.S.; Park, S.K.; Kim, H.M.; Kwon, H.J. Isolation and structure determination of streptochlorin, an antiproliferative agent from a marine-derived Streptomyces sp. 04DH110. J. Microbiol. Biotechnol. 2007, 17, 1403–1406. [Google Scholar] [PubMed]
- Gavrish, E.; Bollmann, A.; Epstein, S.; Lewis, K. A trap for in situ cultivation of filamentous actinobacteria. J. Microbiol. Meth. 2008, 72, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; He, S.; Yan, X. Efficient preparation of pseudoalteromone A from marine Pseudoalteromonas rubra QD1-2 by combination of response surface methodology and high-speed counter-current chromatography: A comparison with high-performance liquid chromatography. Appl. Microbiol. Biotechnol. 2014, 98, 4369–4377. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Wang, H.; Yan, X.; Zhu, P.; Chen, J.; Yang, R. Preparative isolation and purification of macrolactin antibiotics from marine bacterium Bacillus amyloliquefaciens using high-speed counter-current chromatography in stepwise elution mode. J. Chromatogr. A 2013, 1272, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Takahashi, H.; Esumi, Y.; Arie, T.; Morita, T.; Koshino, H.; Uzawa, J.; Uramoto, M.; Yamaguchi, I. Haematocin, a new antifungal diketopiperazine produced by Nectriahaematococca Berk. et Br. (880701a-1) causing nectria blight disease on ornamental plants. J. Antibio (Tokyo) 2007, 53, 45–49. [Google Scholar] [CrossRef]
- Ito, Y. Golden rules and pitfalls in selecting optimum conditions for high-speed counter-current chromatography. J. Chromatogr. A 2005, 1065, 145–168. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.B.; Xin, X.L.; Yang, Y.; Lee, S.S.; Aisa, H.A. Highly conjugated norditerpenoid and pyrroloquinoline alkaloids with potent PTP1B inhibitory activity from Nigella glandulifera. J. Nat. Prod. 2014, 77, 807–812. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Lu, Y.; Jiang, L.; Wu, B.; Zhang, F.; Pan, Y. Preparative isolation and purification of antioxidative stilbene oligomers from Vitis chunganeniss using high-speed counter-current chromatography in stepwise elution mode. J. Sep. Sci. 2009, 32, 2339–2345. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, S.N.; McAlpine, J.B.; Klein, L.L.; Friesen, J.B.; Lankin, D.C.; Pauli, G.F. Quantification of a botanical negative marker without an identical standard: Ginkgotoxin in Ginkgo biloba. J. Nat. Prod. 2014, 77, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Dong, H.; Liu, Y.; Yang, B.; Wang, X.; Huang, L. Application of high-speed counter-current chromatography for preparative separation of cyclic peptides from Vaccaria segetalis. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 811–814. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Su, Z.; Yang, Y.; Ba, H.; Aisa, H.A. Isolation of three sesquiterpene lactones from the roots of Cichorium glandulosum Boiss. et Huet. by high-speed counter-current chromatography. J. Chromatogr. A 2007, 1176, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Luo, Q.; Ding, L.; Fang, F.; Yuan, Y.; Chen, J.; Zhang, J.; Jin, H.; He, S. Preparative isolation and purification of lignans from Justicia procumbens using high-speed counter-current chromatography in stepwise elution mode. Molecules 2015, 20, 7048–7058. [Google Scholar] [CrossRef] [PubMed]
- Katz, L.; Baltz, R.H. Natural product discovery: Past, present, and future. J. Ind. Microbiol. Biotechnol. 2016, 43, 155–176. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, E.K.; Hoepfner, D.; Krastel, P. Natural products as probes in pharmaceutical research. J. Ind. Microbiol. Biotechnol. 2016, 43, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Jiang, Y.; Zhang, H. Marine natural products as sources of novel scaffolds: Achievement and concern. Drug Discov. Today 2010, 15, 884–886. [Google Scholar] [CrossRef] [PubMed]
- Montaser, R.; Luesch, H. Marine natural products: A new wave of drugs? Future Med. Chem. 2011, 3, 1475–1489. [Google Scholar] [CrossRef] [PubMed]
- Friesen, J.B.; McAlpine, J.B.; Chen, S.N.; Pauli, G.F. Countercurrent separation of natural products: An update. J. Nat. Prod. 2015, 78, 1765–1796. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.; Zhang, Y.; Ding, L.; He, S.; Wu, B.; Dong, J.; Zhu, P.; Chen, J.; Zhang, J.; Yan, X. Preparative separation of sulfur-containing diketopiperazines from marine fungus Cladosporium sp. using high-speed counter-current chromatography in stepwise elution mode. Mar. Drugs 2015, 13, 354–365. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Wang, H.; Wu, B.; Zhou, H.; Zhu, P.; Yang, R.; Yan, X. Response surface methodology optimization of fermentation conditions for rapid and efficient accumulation of macrolactin A by marine Bacillus amyloliquefaciens ESB-2. Molecules 2013, 18, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Plackett, R.L.; Burman, J.P. The design of optimum multifactorial experiments. Biometrika 1946, 33, 305–325. [Google Scholar] [CrossRef]
- Sample Availability: Streptochlorin is available from the authors.
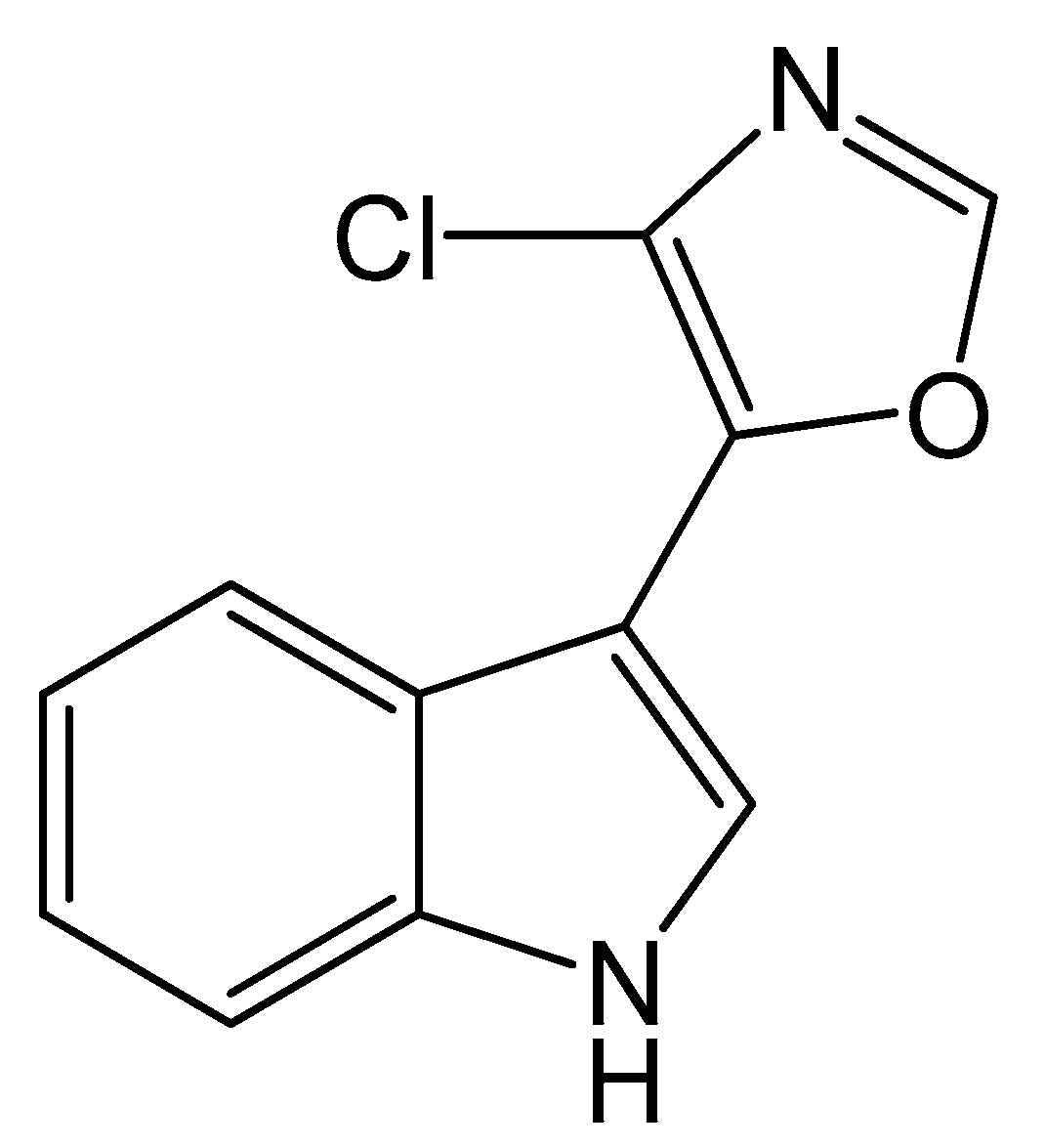
| Variable | Code | Low Level | High Level | Coefficient | t-Value | p-Value |
|---|---|---|---|---|---|---|
| Intercept | 1.0930 | 17.44 | 0.000 | |||
| Yeast extract (g/L) | X1 | 2 | 3 | −0.3460 | −5.52 | 0.000 |
| soluble starch (g/L) | X2 | 10 | 15 | −0.1520 | −2.42 | 0.036 |
| MgSO4 (g/L) | X3 | 0.5 | 0.75 | −0.0950 | −1.52 | 0.161 |
| K2HPO4 (g/L) | X4 | 0.5 | 0.75 | −0.1980 | −3.16 | 0.010 |
| CaCl2 (g/L) | X5 | 2 | 3 | 0.0370 | 0.59 | 0.568 |
| Initial pH value | X6 | 6 | 8 | 0.0100 | 0.16 | 0.876 |
| Medium volume (%) | X7 | 40 | 60 | −0.0330 | −0.53 | 0.610 |
| Temperature (°C) | X8 | 25 | 30 | −0.0840 | −1.34 | 0.210 |
| Marine salt (g/L) | X9 | 20 | 30 | 0.1130 | 1.80 | 0.102 |
| Variables | Code | Level | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Yeast extract (g/L) | X1 | 1 | 1.5 | 2 |
| Soluble starch (g/L) | X2 | 5 | 7.5 | 10 |
| K2HPO4 (g/L) | X4 | 0.15 | 0.3 | 0.45 |
| Source | SS | df | MS | F Value | p-Value |
|---|---|---|---|---|---|
| Model | 11.2888 | 9 | 1.25431 | 49.54 | <0.001 |
| Residual | 0.1266 | 5 | 0.02532 | ||
| Lack of Fit | 0.1162 | 3 | 0.03874 | 7.48 | 0.120 |
| Pure Error | 0.0104 | 2 | 0.00518 | ||
| Cor Total | 11.4154 | 14 |
| Variables | Coefficient | p-Value |
|---|---|---|
| Intercept | 2.9040 | <0.001 |
| X1 | 0.7703 | <0.001 |
| X2 | 0.5307 | <0.001 |
| X4 | 0.4665 | <0.001 |
| X12 | −0.4688 | 0.002 |
| X22 | −0.5895 | 0.001 |
| X42 | −0.1994 | 0.006 |
| X1 × X2 | 0.1315 | 0.500 |
| X1 × X4 | −0.2668 | 0.162 |
| X2 × X4 | −0.2163 | 0.153 |
| Petroleum Ether–Ethyl Acetate–Methanol–Water | KD |
|---|---|
| 5:5:5:5 | 6.47 |
| 8:2:5:5 | 3.47 |
| 9:1:5:5 | 1.33 |
| 9:0.8:5:5 | 1.05 |
| 10:0:5:5 | 0.12 |
| HSCCC | Preparative HPLC | |
|---|---|---|
| Stationary phase | Upper phase petroleum ether–ethyl acetate–methanol–water (9:0.8:5:5, v/v/v/v) | YMC C18 column 250 mm × 20 mm ID 5 μm |
| Mobile phase | Lower phase | Methanol–water (65:35, v/v) |
| Sample capacity per run (mg) | 300 | 40 |
| Run time (min) | 55 | 20 |
| Organic solvent consumption (L/mg) | 0.048 | 0.15 |
| Productivity (mg/min) | 0.3 | 0.06 |
| Purity of isolated compound | 97% | 98% |
| Sample recovery | 91% | 48% |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; He, S.; Ding, L.; Yuan, Y.; Zhu, P.; Epstein, S.; Fan, J.; Wu, X.; Yan, X. Efficient Preparation of Streptochlorin from Marine Streptomyces sp. SYYLWHS-1-4 by Combination of Response Surface Methodology and High-Speed Counter-Current Chromatography. Molecules 2016, 21, 693. https://doi.org/10.3390/molecules21060693
Li L, He S, Ding L, Yuan Y, Zhu P, Epstein S, Fan J, Wu X, Yan X. Efficient Preparation of Streptochlorin from Marine Streptomyces sp. SYYLWHS-1-4 by Combination of Response Surface Methodology and High-Speed Counter-Current Chromatography. Molecules. 2016; 21(6):693. https://doi.org/10.3390/molecules21060693
Chicago/Turabian StyleLi, Lin, Shan He, Lijian Ding, Ye Yuan, Peng Zhu, Slava Epstein, Jianzhong Fan, Xiaokai Wu, and Xiaojun Yan. 2016. "Efficient Preparation of Streptochlorin from Marine Streptomyces sp. SYYLWHS-1-4 by Combination of Response Surface Methodology and High-Speed Counter-Current Chromatography" Molecules 21, no. 6: 693. https://doi.org/10.3390/molecules21060693






