Inhibitory Effect of Crocin(s) on Lens α-Crystallin Glycation and Aggregation, Results in the Decrease of the Risk of Diabetic Cataract
Abstract
:1. Introduction
2. Results
2.1. In Vitro Studies
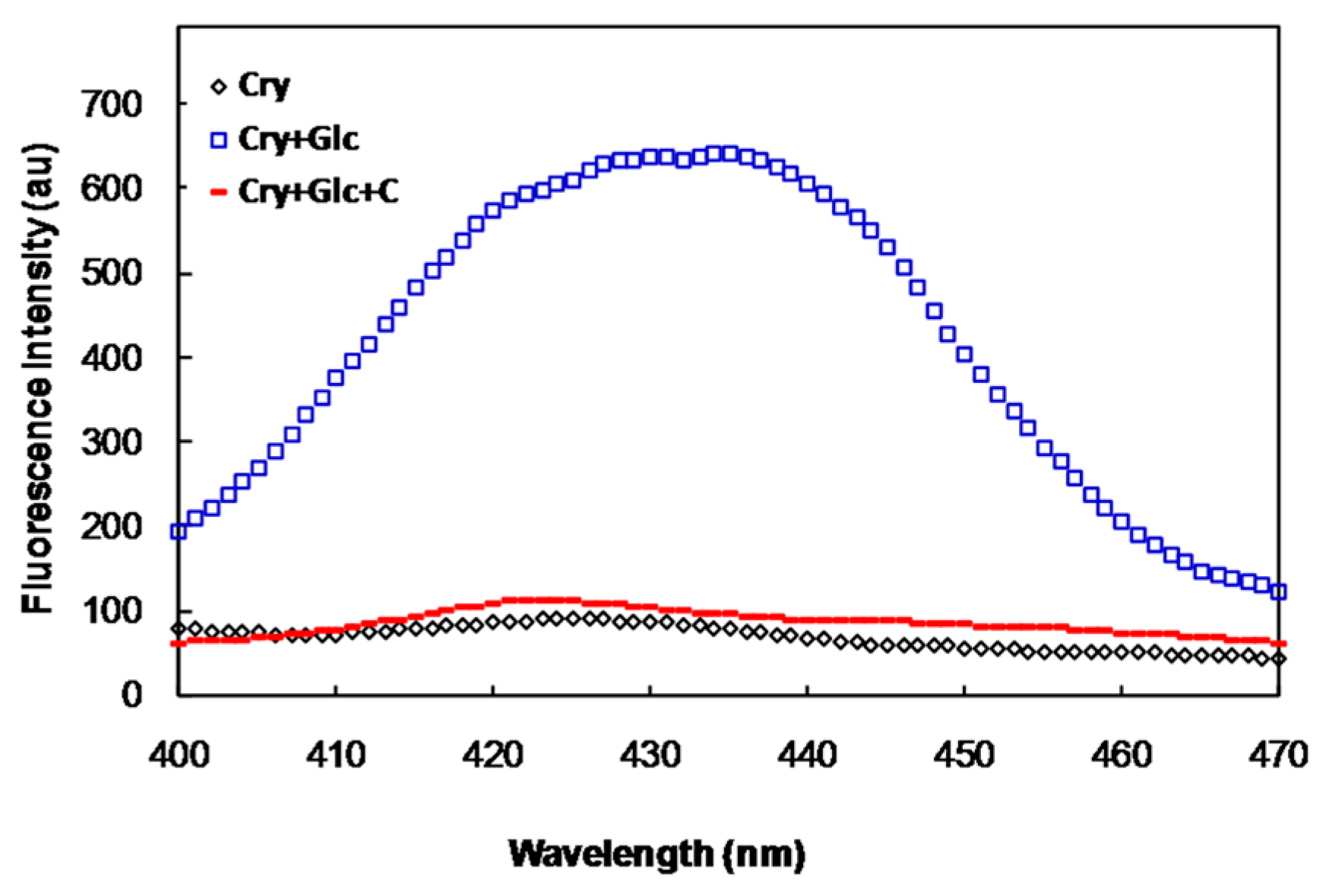
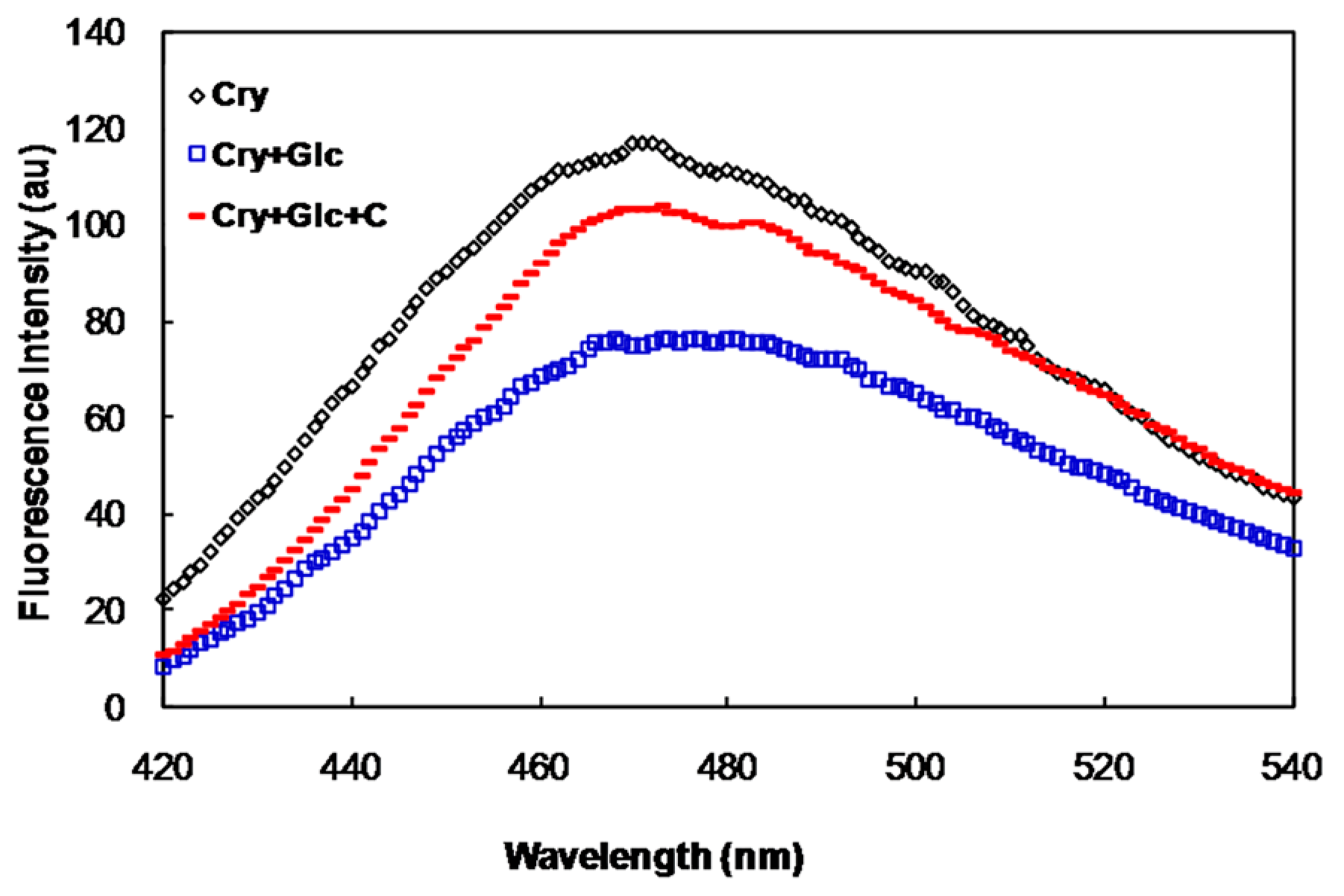
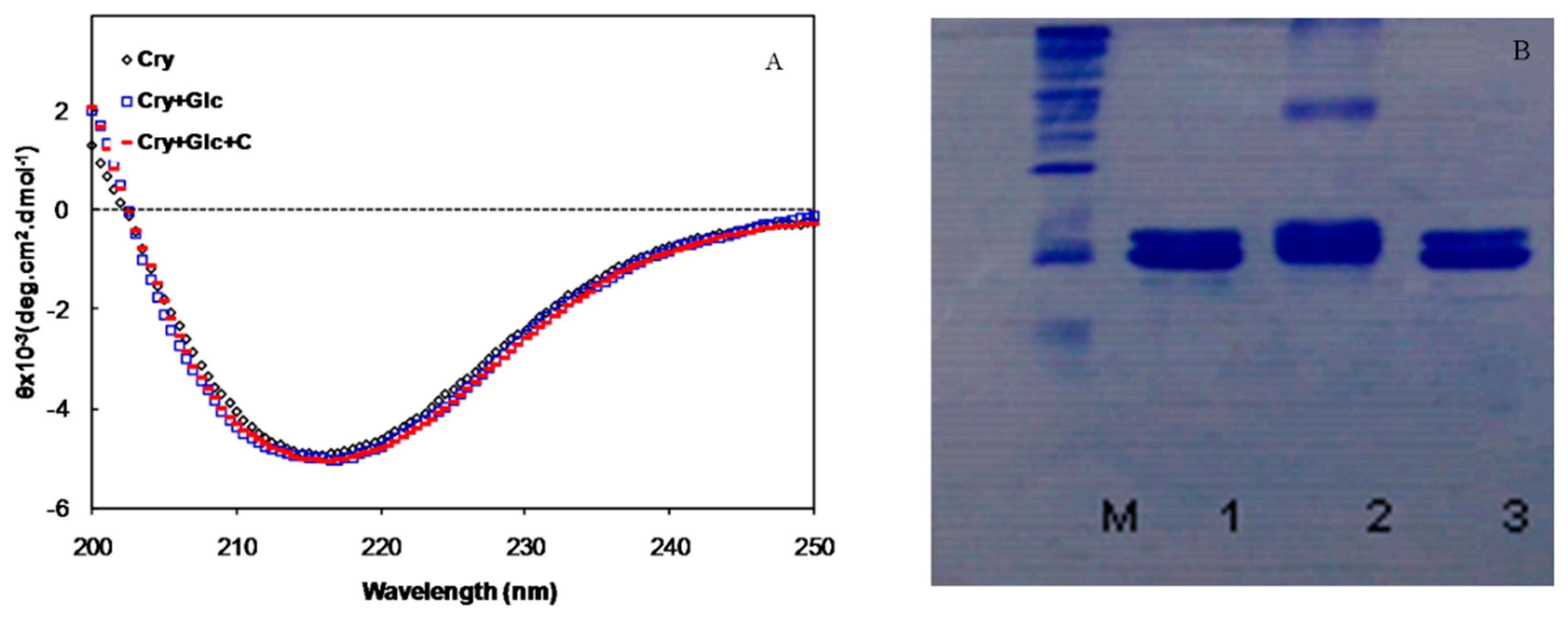
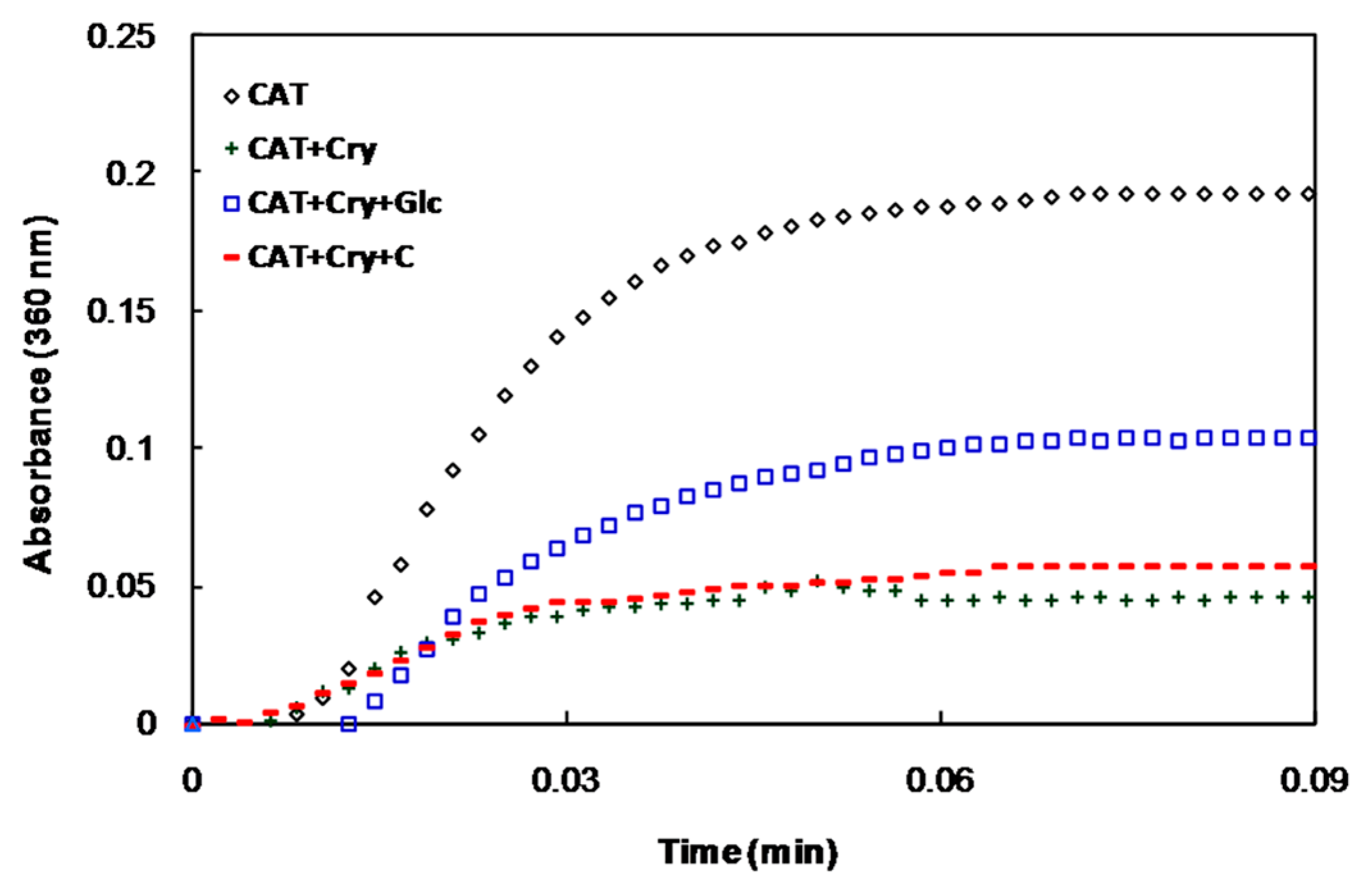
2.2. In Vivo Studies
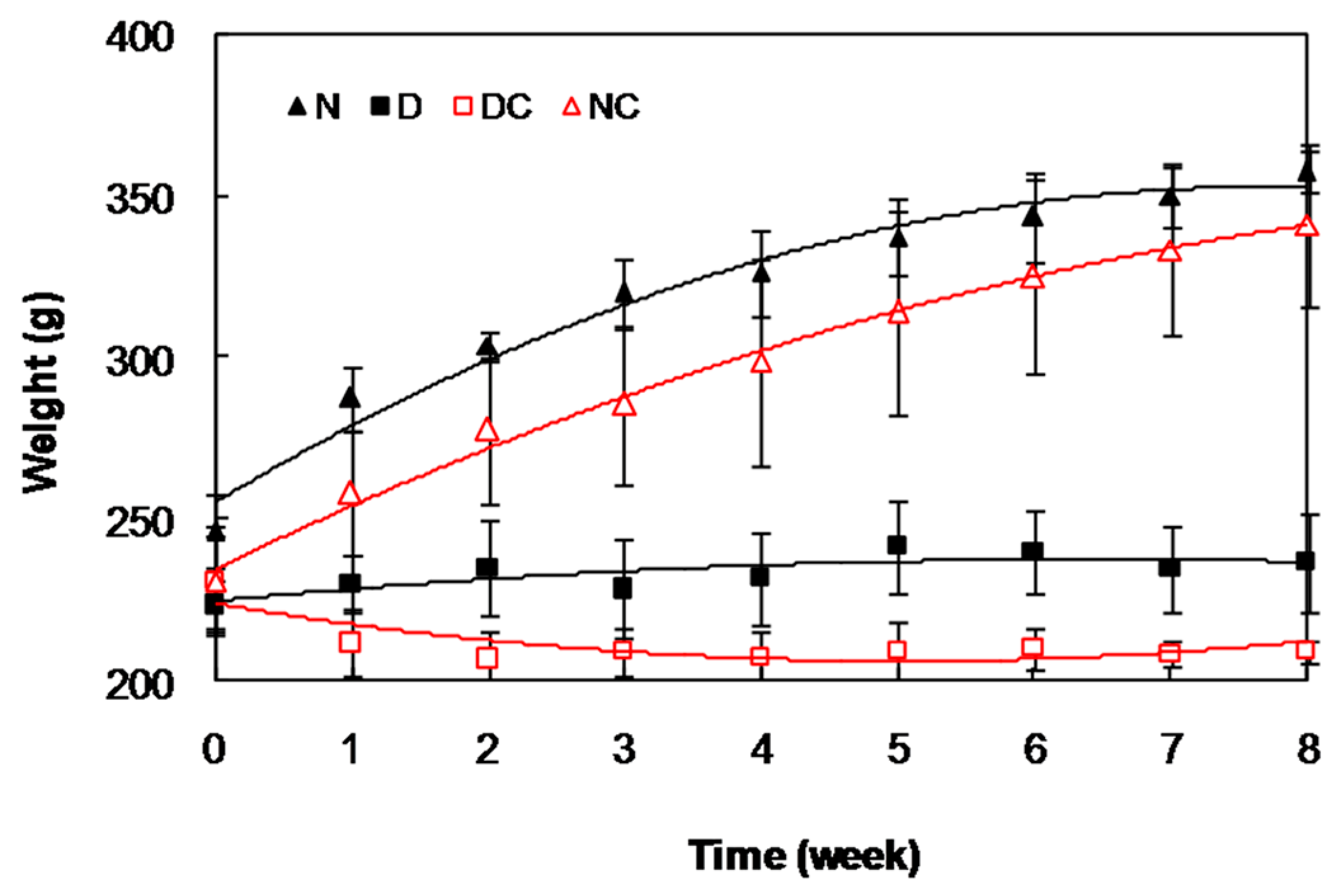
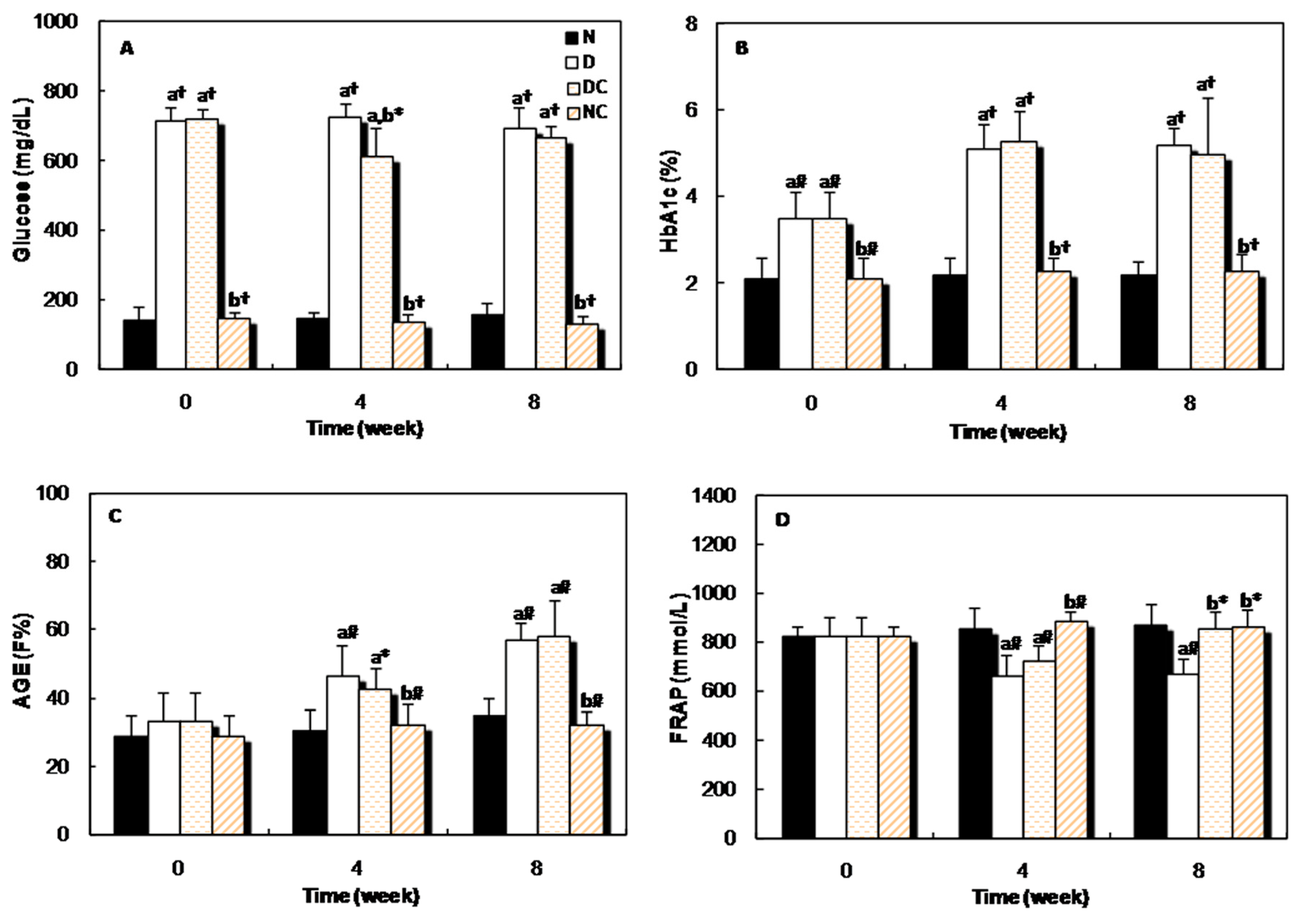
| Average of Cataract Score Time (week) | |||||
|---|---|---|---|---|---|
| Group | 0 | 2 | 4 | 6 | 8 |
| N | 0 | 0 | 0 | 0 | 0 |
| D | 0 | 0 | 0.3 | 2 | 2.6 |
| DC | 0 | 0 | 0.3 | 1.5 | 2.3 |
| NC | 0 | 0 | 0 | 0 | 0 |

| Group Name | GSH (mg/g Lens) | CAT Activity (μmol/g Lens pr) | SOD Activity (Units/min/mg Lens pr) |
|---|---|---|---|
| N | 214.5 ± 43.2 | 57.6 ± 2.4 | 6.59 ± 0.95 |
| D | 54.9 ± 12.7 a,† | 31.1 ± 5.2 a,* | 14.76 ± 5.46 a,* |
| DC | 144.9 ± 25.8 a,b,# | 53.3 ± 9.7 b,* | 13.81 ± 2.37 a,* |
| NC | 239.3 ± 23.7 b,† | 56.8 ± 11.2 b,* | 6.61 ± 0.58 b,# |
| Group Name | Lens AGE (FI%) | Glycated Proteins (μmol HMF/mg Lens prot.) | Total Protein (mg/Lens) | Soluble Protein (mg/Lens) |
|---|---|---|---|---|
| N | 18.0 ± 2.2 | 0.55 ± 0.01 | 17.40 ± 0.47 | 13.72 ± 1.23 |
| D | 70.1 ± 5.7 a,† | 1.22 ± 0.22 a,* | 11.89 ± 1.39 a,* | 5.45 ± 1.85 a,# |
| DC | 65.4 ± 7.7 a,† | 1.35 ± 0.42 a,# | 12.98 ± 2.60 a,* | 6.87 ± 1.11a,# |
| NC | 19.3 ± 4.6 b,† | 0.57 ± 0.17 b,* | 17.63 ± 1.15 b,# | 14.11 ± 1.32 b,# |
3. Discussion
4. Experimental Section
4.1. Materials
4.2. Methods
4.2.1. In Vitro Studies
4.2.2. In Vivo Studies
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Obrosova, I.G.; Chung, S.S.M.; Kador, P.F. Diabetic cataract: Mechanisms and management. Diabetes Metab. Res. Rev. 2010, 3, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Luthra, M.; Balasubramanian, D. Nonenzymatic glycation alters protein structure and stability: A study of two eye lens crystallins. J. Biol. Chem. 1993, 268, 18119–18127. [Google Scholar] [PubMed]
- Kyselova, Z.; Stefek, M.; Bauer, V. Pharmacological prevention of diabetic cataract. J. Diabetes Compl. 2004, 18, 129–140. [Google Scholar] [CrossRef]
- Horwitz, J. α-crystallin. Exp. Eye Res. 2003, 76, 145–153. [Google Scholar] [CrossRef]
- Takemoto, L.; Boyle, D. Determination of the in vivo deamidation rate of asparagine-101 from α-A crystallin using microdissected sections of the aging human lens. Exp. Eye Res. 1998, 67, 119–120. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, L.; Boyle, D. Deamidation of specific glutamine residues from α-A crystallin during aging of the human lens. Biochemistry 1998, 37, 13681–13685. [Google Scholar] [CrossRef] [PubMed]
- Thampi, P.; Zarina, S.; Abraham, E.C. α-Crystallin chaperone function in diabetic rat and human lenses. Mol. Cell. Biochem. 2002, 229, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.A.; Kumar, M.S.; Reddy, G.B. Effect of glycation on α-crystallin structure and chaperone-like function. Biochem. J. 2007, 408, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Pascolini, D.; Mariotti, S.P. Global estimates of visual impairment. Br. J. Ophthalmol. 2012, 96, 614–618. [Google Scholar]
- Agte, V.; Gite, S. Diabetic Cataract and Role of Antiglycating Phytochemicals. In Handbook of Nutrition, Diet and the Eye; Preedy, V.R., Ed.; Academic Press: London, UK, 2014; pp. 131–140. [Google Scholar]
- Mousavi, S.Z.; Bathaie, S.Z. Historical uses of saffron: Identifying potential new avenues for modern research. Avicenna J. Phytomed. 2011, 1, 57–66. [Google Scholar]
- Bolhassani, A.; Khavari, A.; Bathaie, S.Z. Saffron and natural carotenoids: Biochemical activities and anti-tumor effects. Biochim. Biophys. Acta 2014, 1845, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Mousavi, S.Z. New applications and mechanisms of action of saffron and its important ingredients. Crit. Rev. Food Sci. Nutr. 2010, 50, 761–786. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, T.; Ohno, S.; Soeda, S.; Tanaka, H.; Shoyama, Y.; Shimeno, H. Crocin prevents the death of rat pheochromyctoma (PC-12) cells by its antioxidant effects stronger than those of α-tocopherol. Neurosci. Lett. 2004, 362, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Shirali, S.; Bathaie, S.Z.; Nakhjavani, M. Effect of crocin on the insulin resistance and lipid profile of streptozotocin-induced diabetic rats. Phytother. Res. PTR 2013, 27, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Fadai, F.; Mousavi, B.; Ashtari, Z.; Ali beigi, N.; Farhang, S.; Hashempour, S.; Shahhamzei, N.; Bathaie, S.Z. Saffron aqueous extract prevents metabolic syndrome in patients with schizophrenia on olanzapine treatment: A randomized triple blind placebo controlled study. Pharmacopsychiatry 2014, 47, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Karami, M.; Bathaie, S.Z.; Tiraihi, T.; Habibi-Rezaei, M.; Arabkheradmand, J.; Faghihzadeh, S. Crocin improved locomotor function and mechanical behavior in the rat model of contused spinal cord injury through decreasing calcitonin gene related peptide (CGRP). Phytomed. Int. J. Phytother. Phytopharmacol. 2013, 21, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Hoshyar, R.; Bathaie, S.Z.; Sadeghizadeh, M. Crocin triggers the apoptosis through increasing the Bax/Bcl-2 ratio and caspase activation in human gastric adenocarcinoma, AGS, cells. DNA Cell. Biol. 2013, 32, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Miri, H.; Mohagheghi, M.A.; Mokhtari-Dizaji, M.; Shahbazfar, A.A.; Hasanzadeh, H. Saffron Aqueous Extract Inhibits the Chemically-induced Gastric Cancer Progression in the Wistar Albino Rat. Iran. J. Basic Med. Sci. 2013, 16, 27–38. [Google Scholar] [PubMed]
- Bathaie, S.Z.; Hoshyar, R.; Miri, H.; Sadeghizadeh, M. Anticancer effects of crocetin in both human adenocarcinoma gastric cancer cells and rat model of gastric cancer. Biochem. Cell Biol. 2013, 91, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Hoshyar, R.; Bathaie, S.Z.; Kyani, A.; Mousavi, M.F. Is there any interaction between telomeric DNA structures, G-quadruplex and I-motif, with saffron active metabolites? Nucleosides Nucleotides Nucleic Acids 2012, 31, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Yan, H.; Li, M.Y.; Harding, J.J. Effect of a combination of carnosine and aspirin eye drops on streptozotocin-induced diabetic cataract in rats. Mol. Vis. 2009, 15, 2129–2138. [Google Scholar] [PubMed]
- Yan, H.; GUO, Y.; Zhang, J.; Ding, Z.; Ha, W.; Harding, J.J. Effect of carnosine, aminoguanidine, and aspirin drops on the prevention of cataracts in diabetic rats. Mol. Vis. 2008, 14, 2282–2291. [Google Scholar] [PubMed]
- Zhang, S.; Chai, F.Y.; Yan, H.; Guo, Y.; Harding, J.J. Effects of N-acetylcysteine and glutathione ethyl ester drops on streptozotocin-induced diabetic cataract in rats. Mol. Vis. 2008, 14, 862–870. [Google Scholar] [PubMed]
- Bahmani, F.; Bathaie, S.Z.; Aldavood, S.J.; Ghahghaei, A. Glycine therapy inhibits the progression of cataract in streptozotocin-induced diabetic rats. Mol. Vis. 2012, 18, 439–448. [Google Scholar] [PubMed]
- Sai Varsha, M.K.; Raman, T.; Manikandan, R. Inhibition of diabetic-cataract by vitamin K1 involves modulation of hyperglycemia-induced alterations to lens calcium homeostasis. Exp. Eye Res. 2014, 128, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Varma, S.D.; Hegde, K.R.; Kovtun, S. Inhibition of selenite-induced cataract by caffeine. Acta Ophthalmol. 2010, 88, e245–e249. [Google Scholar] [CrossRef] [PubMed]
- Ghahghaei, A.; Bathaie, S.Z.; Kheirkhah, H.; Bahraminejad, E. The protective effect of crocin on the amyloid fibril formation of Aβ42 peptide in vitro. Cell. Mol. Biol. Lett. 2013, 18, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Farajzade, A.; Hoshyar, R. A review of the chemistry and uses of crocin and crocetin, the carotenoid natural dyes in saffron, with particular emphasis on applications as colorants including their use as biological stains. Biotech. Histochem.: Off. Publ. Biol. Stain Comm. 2014, 89, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Shams, A.; Moghadas Zadeh Kermani, F. Crocin Bleaching Assay Using Purified Di-gentiobiosyl Crocin (-crocin) from Iranian Saffron. Iran. J. Basic Med. Sci. 2011, 14, 399–406. [Google Scholar] [PubMed]
- Ordoudi, S.A.; Tsimidou, M.Z. Crocin bleaching assay step by step: Observations and suggestions for an alternative validated protocol. J. Agric. Food Chem. 2006, 54, 1663–1671. [Google Scholar] [CrossRef] [PubMed]
- Ordoudi, S.A.; Tsimidou, M.Z. Measuring antioxidant and prooxidant capacity using the Crocin Bleaching Assay (CBA). Methods Mol. Biol. 2015, 1208, 329–344. [Google Scholar] [PubMed]
- Rajaei, Z.; Hadjzadeh, M.A.; Nemati, H.; Hosseini, M.; Ahmadi, M.; Shafiee, S. Antihyperglycemic and antioxidant activity of crocin in streptozotocin-induced diabetic rats. J. Med. Food 2013, 16, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Sheng, L.; Qian, Z.; Zheng, S.; Xi, L. Mechanism of hypolipidemic effect of crocin in rats: Crocin inhibits pancreatic lipase. Eur. J. Pharmacol. 2006, 543, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Makri, O.E.; Ferlemi, A.V.; Lamari, F.N.; Georgakopoulos, C.D. Saffron administration prevents selenite-induced cataractogenesis. Mol. Vis. 2013, 19, 1188–1197. [Google Scholar] [PubMed]
- Asai, A.; Nakano, T.; Takahashi, M.; Nagao, A. Orally administered crocetin and crocin are absorbed into blood plasma as crocetin and its glucuronide conjugates in mice. J. Agric. Food Chem. 2005, 53, 7302–7306. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, B.; Bathaie, S.Z.; Fadai, F.; Ashtari, Z.; Ali Beigi, N.; Farhang, S.; Hashempour, S.; Shahhamzei, N.; Heidarzadeh, H. Safety evaluation of saffron stigma (Crocus sativus L.) aqueous extract and crocin in patients with schizophrenia. Avicenna J. Phytomed. 2015, 5, 413–419. [Google Scholar] [PubMed]
- Mohamadpour, A.H.; Ayati, Z.; Parizadeh, M.R.; Rajbai, O.; Hosseinzadeh, H. Safety Evaluation of Crocin (a constituent of saffron) Tablets in Healthy Volunteers. Iran. J. Basic Med. Sci. 2013, 16, 39–46. [Google Scholar] [PubMed]
- Modaghegh, M.H.; Shahabian, M.; Esmaeili, H.A.; Rajbai, O.; Hosseinzadeh, H. Safety evaluation of saffron (Crocus sativus) tablets in healthy volunteers. Phytomed.: Int. J. Phytother. Phytopharmacol. 2008, 15, 1032–1037. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Olmo, D.C.; Riese, H.H.; Escribano, J.; Ontanon, J.; Fernandez, J.A.; Atienzar, M. Effects of long-term treatment of colon adenocarcinoma with crocin, a carotenoid from saffron (Crocus sativus L.): An experimental study in the rat. Nutr. Cancer 1999, 35, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Suryanarayana, P.; Saraswat, M.; Mrudula, T.; Krishna, P.; Krishnaswamy, K.; Reddy, G.B. Curcumin and turmeric delay streptozotocin-induced diabetic cataract in rats. Invest. Ophthalmol. Vis. Sci. 2005, 46, 2092–2099. [Google Scholar] [CrossRef] [PubMed]
- Saraswat, M.; Suryanarayana, P.; Reddy, P.Y.; Patil, M.A.; Balakrishna, N.; Reddy, G.B. Antiglycating potential of Zingiber officinalis and delay of diabetic cataract in rats. Mol. Vis. 2010, 16, 1525–1537. [Google Scholar] [PubMed]
- Kumar, P.A.; Reddy, P.Y.; Srinivas, P.N.B.S.; Reddy, G.B. Delay of diabetic cataract in rats by the antiglycating potential of cumin through modulation of α-crystallin chaperone activity. J. Nutr. Biochem. 2009, 20, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Bolhasani, A.; Bathaie, S.Z.; Yavari, I.; Moosavi-Movahedi, A.A.; Ghaffari, M. Separation and purification of some components of Iranian saffron. Asian J. Chem. 2005, 17, 725–729. [Google Scholar]
- Ghahghaei, A.; Rekas, A.; Carver, J.A.; Augusteyn, R.C. Structure/function studies of dogfish α-crystallin, comparison with bovine α-crystallin. Mol. Vis. 2009, 15, 2411–2420. [Google Scholar] [PubMed]
- Jafarnejad, A.; Bathaie, S.Z.; Nakhjavani, M.; Hassan, M.Z. Investigation of the mechanisms involved in the high-dose and long-term acetyl salicylic acid therapy of type I diabetic rats. JPET 2008, 324, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Jafarnejad, A.; Bathaie, S.Z.; Nakhjavani, M.; Hassan, A.; Banasadegh, S. The improvement effect of l-Lys as a chemical chaperone on STZ-induced diabetic rats, protein structure and function. Diabetes Metab. Res. Rev. 2008, 24, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Kalousova, M.; Skrha, J.; Zima, T. Advanced glycation end-products and advanced oxidation protein products in patients with diabetes mellitus. Physiol. Res. 2002, 51, 597–604. [Google Scholar] [PubMed]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Suryanarayana, P.; Krishnaswamy, K.; Reddy, G.B. Effect of curcumin on galactose-induced cataractogenesis in rats. Mol. Vis. 2003, 9, 223–230. [Google Scholar] [PubMed]
- Sample Availability: Samples of crocin(s) and α-crystallin are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahmani, F.; Bathaie, S.Z.; Aldavood, S.J.; Ghahghaei, A. Inhibitory Effect of Crocin(s) on Lens α-Crystallin Glycation and Aggregation, Results in the Decrease of the Risk of Diabetic Cataract. Molecules 2016, 21, 143. https://doi.org/10.3390/molecules21020143
Bahmani F, Bathaie SZ, Aldavood SJ, Ghahghaei A. Inhibitory Effect of Crocin(s) on Lens α-Crystallin Glycation and Aggregation, Results in the Decrease of the Risk of Diabetic Cataract. Molecules. 2016; 21(2):143. https://doi.org/10.3390/molecules21020143
Chicago/Turabian StyleBahmani, Fereshteh, Seyedeh Zahra Bathaie, Seyed Javid Aldavood, and Arezou Ghahghaei. 2016. "Inhibitory Effect of Crocin(s) on Lens α-Crystallin Glycation and Aggregation, Results in the Decrease of the Risk of Diabetic Cataract" Molecules 21, no. 2: 143. https://doi.org/10.3390/molecules21020143





