Structural Characterization and Antimicrobial Activities of 7H-Benzo[h]chromeno[2,3-d]pyrimidine and 14H-Benzo[h]chromeno[3,2-e][1,2,4]triazolo[1,5-c] pyrimidine Derivatives
Abstract
:1. Introduction
2. Results and Discussion
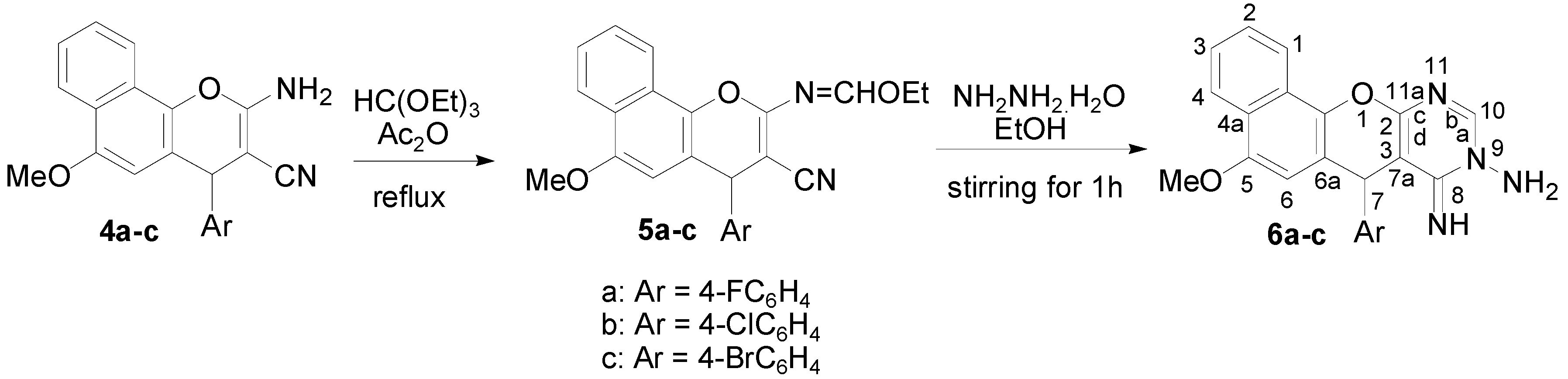
2.1. Chemistry
2.2. Antibacterial Evaluation
2.3. SAR Studies
3. Experimental Section
3.1. General Information
3.2. Synthesis
3.2.1. Starting Materials
3.2.2. General Procedure for the Synthesis of Compounds 7a–c
3.2.3. General Procedure for the Preparation of Compounds 7d–f
3.2.4. General Procedure for the Preparation of Compounds 7g–i
3.2.5. General Procedure for the Preparation of Compounds 7j–l
3.2.6. General Procedure for the Preparation of Compounds 7m–o
3.2.7. General Procedure for the Preparation of Compounds 8a–c
3.2.8. Synthesis of Compounds 12a–c
3.3. Antimicrobial Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saini, M.S.; Kumar, A.; Dwivedi, J.; Singh, R. A Review: Biological Significances of Heterocyclic Compounds. Int. J. Pharm. Sci. Res. 2013, 4, 66–77. [Google Scholar]
- Li, Q.-Z.; Nie, X.-Y.; Liang, J. Novel Coumarin and 4H-Chromene Derivatives Containing 4,5-Dihydropyrazole Moiety: Synthesis and Antibacterial Activity. Lett. Drug Des. Discov. 2011, 8, 558–561. [Google Scholar]
- Liu, X.-H.; Liu, J.-X.; Bai, L.-S.; Lan, G.-L.; Pan, C.-X. Novel dihydropyrazole Derivatives Linked with 4H-Chromene: Microwave-Promoted Synthesis and Antibacterial Activity. Lett. Drug. Des. Discov. 2010, 7, 487–490. [Google Scholar] [CrossRef]
- Foroumadi, A.; Emami, S.; Sorkhi, M.; Nakhjiri, M.; Nazarian, Z.; Heydar, S.; Ardestani, S.; Poorrajab, F.; Shafiee, A. Chromene-Based Synthetic Chalcones as Potent Antileishmanial Agents: Synthesis and Biological Activity. Chem. Biol. Drug Des. 2010, 75, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Narender, T.; Shweta Gupta, S. A convenient and biogenetic type synthesis of few naturally occurring chromeno dihydrochalcones and their in vitro antileishmanial activity. Bioorg. Med. Chem. Lett. 2009, 14, 3913–3916. [Google Scholar] [CrossRef] [PubMed]
- Sabry, N.M.; Mohamed, H.M.; Khattab, E.S.A.E.H.; Motlaq, S.S.; El-Agrody, A.M. Synthesis of 4H chromene, coumarin, 12H-chromeno[2,3-d]pyrimidine derivatives and some of their antimicrobial and cytotoxicity activities. Eur. J. Med. Chem. 2011, 46, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Rampa, A.; Bisi, A.; Belluti, F.; Gobbi, S.; Piazzi, L.; Valenti, P.; Zampiron, A.; Caputo, A.; Varani, K.; Borea, P.A.; Carrara, M. Homopterocarpanes as bridged triarylethylene analogues:Synthesis and antagonistic effects in human MCF-7 breast cancer cells. IL Farmco 2005, 60, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Magedov, I.V.; Manpadi, M.; Evdokimov, N.M.; Elias, E.M.; Rozhkova, E.; Ogasawara, M.A.; Bettale, J.D.; Przheval’skii, N.M.; Rogelj, S; Kornienko, A. Antiproliferative and apoptosis inducing properties of pyrano[3,2-c]pyridones accessible by a onestep multicomponent synthesis. Bioorg. Med Chem. Lett. 2007, 17, 3872–3876. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.M.; Devi, N.S.; Thokchom, D.S.; Sharma, G.J. Novel 3-alkanoyl/aroyl/heteroaroyl-2H-chromene-2-thiones: Synthesis and evaluation of their antioxidant activities. Eur. J. Med. Chem. 2010, 45, 2250–2257. [Google Scholar] [CrossRef] [PubMed]
- Vukovic, N.; Sukdolak, S.; Solujic, S.; Niciforovic, N. Substituted imino and amino derivatives of 4-hydroxycoumarins as novel antioxidant, antibacterial and antifungal agents: synthesis and in vitro assessments. Food Chem. 2010, 120, 1011–1018. [Google Scholar] [CrossRef]
- Tandon, V.K.; Vaish, M.; Jain, S.; Bhakuni, D.S.; Srimal, R.C. Synthesis, carbon-13 NMR and hypotensive action of 2,3-dihydro-2,2-dimethyl-4H-naphtho [1,2-b] pyran-4-one. Indian J. Pharm. Sci. 1991, 53, 22–23. [Google Scholar]
- Mahmoodi, M.; Aliabadi, A.; Emami, S.; Safavi, M.; Rajabalian, S.; Mohagheghi, M.A.; Khoshzaban, A.; Samzadeh-Kermani, A.; Lamei, N.; Shafiee, A. Synthesis and in vitro Cytotoxicity of Polyfunctionalized 4-(2-Arylthiazol-4-yl)-4H-Chromemes. Arch. Pharm. Chem. 2010, 343, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Endo, S.; Matsunaga, T.; Kuwata, K.; Zhao, H.-T.; El-Kabbani, O.; Kitade, Y.; Hara, A. Chromene-3-carboxamide derivatives discov ered from virtual screening as potent inhibitors of the tumor maker, AKR1B10. Bioorg. Med. Chem. 2010, 18, 2485–2490. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.-H.; Chuang, S.-K.; Hu, C.-C.; Chang, C.-F.; Huang, Y.-C.; Lin, C.-W.; Lee, Y.-J. The synthesis of morusin as a potent antitumor agent. Tetrahedron 2010, 66, 1335–1340. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; Fouda, A.M.; Khattab, E.S.A.E.H. Synthesis, antitumor activity of 2-amino-4H-benzo[h]chromene derivatives and Structure-activity relationships of the 3- and 4-positions. Med. Chem. Res. 2013, 22, 6105–6120. [Google Scholar] [CrossRef]
- Bruhlmann, C.; Ooms, F.; Carrupt, P.; Testa, B.; Catto, M.; Leonetti, F.; Altomare, C.; Cartti, A. Coumarins derivatives as dual inhibitors of acetylcholinesterase and monoamine oxidase. J. Med. Chem. 2001, 44, 3195–3198. [Google Scholar] [CrossRef] [PubMed]
- Kesten, S.R.; Heffner, T.G.; Johnson, S.J.; Pugsley, T.A.; Wright, J.L.; Wise, D.L. Design, Synthesis, and Evaluation of Chromen-2- ones as Potent and Selective Human Dopamine D4 Antagonists. J. Med. Chem. 1999, 42, 3718–3725. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-S.; Khil, L.-Y.; Chae, S.-H.; Kim, D.; Lee, B.-H.; Hwang, G.-S.; Moon, C.-H.; Chang, T.-S.; Moon, C.-K. Effects of DK-002, a synthesized(6aS,cis)-9,10-Dimethoxy-7,11b-dihydro-indeno[2,1-c]chromene-3,6a-diol, on platelet activity. Life Sci. 2006, 78, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Coudert, P.; Coyquelet, J.M.; Bastide, J.; Marion, Y.; Fialip, J. Synthesis and anti-allergic properties of N-arylnitrones with furo-pyran structure. Ann. Pharm. Fr. 1988, 46, 91–96. [Google Scholar] [PubMed]
- El-Sayed, A.T.; Ibrahim, M.A. Synthesis and Antimicrobial Activity of Chromone- linked-2-Pyridone Fused with 1,2,4-Triazoles, 1,2,4-Triazines and 1,2,4-Triazepines Ring Systems. J. Braz. Chem. 2010, 21, 1006–1007. [Google Scholar]
- Narasimhan, B.; Kumar, P.; Sharma, D. Biological activities of hydrazide derivatives in the new millennium. Acta Pharm. Sci. 2010, 52, 169–180. [Google Scholar]
- Keri, R.S.; Hosamani, K.M.; Shingalapur, R.V.; Hugar, M.H. Analgesic, anti-pyretic and DNA cleavage studies of novel pyrimidine derivatives of coumarin moiety. Eur. J. Med. Chem. 2010, 45, 2597–2605. [Google Scholar] [CrossRef] [PubMed]
- Sashidhara, K.V.; Kumar, M.; Modukuri, R.K.; Srivastava, A.; Puri, A. Discovery and synthesis of novel substituted benzocoumarins as orally active lipid modulating agents. Bioorg. Med Chem. Lett. 2011, 21, 6709–6713. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Nasuhara, A.; Michiloshi, K.; Kato, T.; Kikugawa, K. DNA strand-breaking activity and mutagenicity of 2,3-dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one (DDMP), a Maillard reaction product of glucose and glycine. Mutat. Res. 1997, 395, 47–56. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; El-Hakium, M.H.; Abd El-Latif, M.S.; Fekry, A.H.; Sayed, S.M.; El-Gareab, K.A. Synthesis of Pyrano[2,3-d]pyrimidine and Pyrano[3,2-e][1,2,4]triazolo[2,3-c]pyri- midine Derivatives with Promising Antimicrobial Activities. Acta Pharm. 2000, 50, 111–120. [Google Scholar]
- Bedair, A.H.; Emam, H.A.; El-Hady, N.A.; Ahmed, K.A.R.; El-Agrody, A.M. Synthesis and Antimicrobial Activities of Novel Naphtho[2,1-b]pyran, Pyrano[3,2-d]pyrimidine and Pyrano-[3,2-e][1,2,4]triazolo-[2,3-c]pyrimidine Derivatives. Il Farmaco 2001, 56, 965–973. [Google Scholar] [CrossRef]
- Khafagy, M.M.; Abd El-Wahab, A.H.F.; Eid, F.A.; El-Agrody, A.M. Synthesis of Halogen Derivatives of Benzo[h]cheromene and Benzo[a]anthracene with Promising Antimicrobial Activities. Il Farmaco 2002, 57, 715–722. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; Sabry, N.M.; Motlaq, S.S. Synthesis of some new 2-substituted 12H-chromeno[3,2-e][1,2,4]triazolo[1,5-c]pyrimidine, 3-ethoxycarbonyl-12H-chromeno[3,2-e]-[3,2-e][1,2,4]triazolo[1,5-c]pyrimidine-2-one, ethyl 2-formylamino/acetylamino-4H-chromene-3-carboxylate and some of their ntimicrobial activities. J. Chem. Res. 2011, 35, 77–83. [Google Scholar]
- Halawa, A.H.; Fouda, A.M.; Al-Dies, A.M.; El-Agrody, A.M. Synthesis, Biological Evaluation and Molecular Docking Studies of 4Hbenzo[h]chromenes, 7H-benzo[h]chromeno-[2,3-d]pyrimidines as Antitumor Agents. Lett. Drug. Des. Discov. 2016, 13, 77–88. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; Halawa, A.H.; Fouda, A.M.; Al-Dies, A.M. The anti-proliferative activity of novel 4H-benzo[h]chromenes, 7H-benzo[h]chromeno[2,3-d]pyrimidines and the structure-activity relationships of the 2-,3-positions and fused rings at the 2,3-positions. J. Saudi Chem. Soc. 2016. [Google Scholar] [CrossRef]
- El-Agrody, A.M.; Fouda, A.M.; Al-Dies, A.M. Studies on the synthesis, in vitro antitumor activity of 4H-benzochromene, 7H-benzo[h]chromeno[2,3-d]pyrimidine derivatives and Structure activity relationships of the 2-,3- and 2,3-positions. Med. Chem. Res. 2014, 23, 3187–3199. [Google Scholar] [CrossRef]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution. Clin. Microbiol. Infect. 2000, 6, 509–515. [Google Scholar]
- National Committee for Clinical Laboratory Standards. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 5th ed.; National Committee for Clinical Laboratory Standards (NCCLS): Wayne, PA, USA, 2000; Approved Standard M7-A5. [Google Scholar]
- Al-Dies, A.M.; Amr, A.-G.E.; El-Agrody, A.M.; Chia, T.S.; Fun, H.-K. 2-Amino-4-(4-fluorophenyl)-6-methoxy-4H-benzo[h]chromene-3-carbonitrile. Acta Cryst. 2012, E68, 1934–1935. [Google Scholar] [CrossRef] [PubMed]
- Al-Sehemi, A.G.; Irfan, A.; El-Agrody, A.M. Synthesis, characterization and DFT study of 4H-benzo[h]chromene derivatives. J. Mol. Struct. 2012, 1018, 171–175. [Google Scholar] [CrossRef]
- Al-Dies, A.M.; El-Agrody, A.M.; Al-Omar, M.A.; Amr, A.-G.E.; Ng, S.W.; Tiekink, E.R.T. 2-Amino-4-(4-bromophenyl)-6-methoxy-4H-benzo[h]chromene-3-carbonitrile. Acta Cryst. 2013, E69, 0480–0481. [Google Scholar] [CrossRef] [PubMed]
- El-Agrody, A.M.; Al-Dies, A.M.; Fouda, A.M. Microwave assisted synthesis of 2-amino-6-methoxy-4H-benzo[h]chromene derivatives. Eur. J. Chem. 2014, 5, 133–137. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds (7a–o), (8a–c) and (12a–c) are available from the authors.
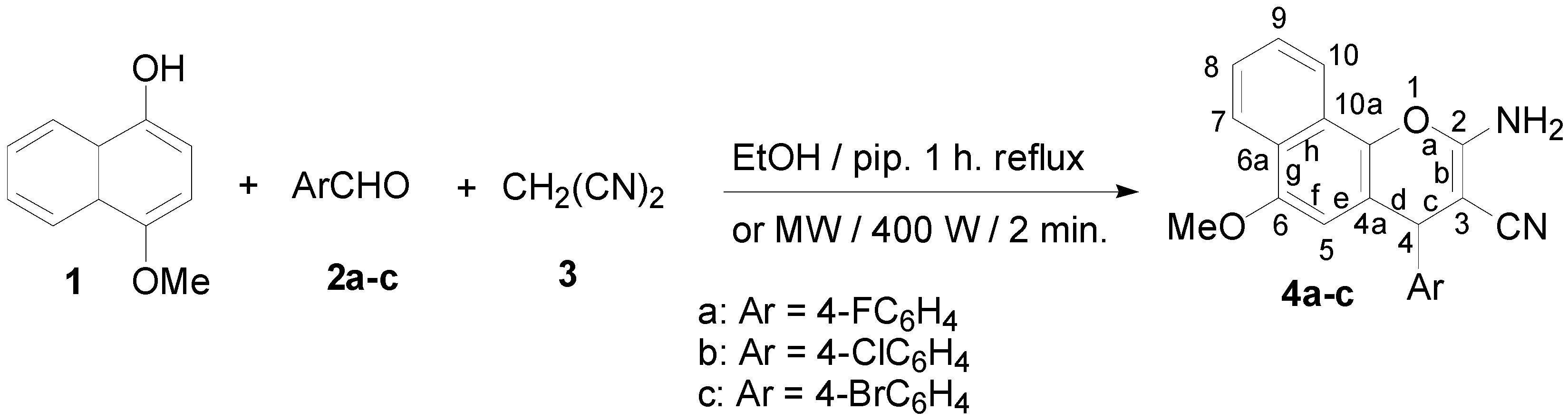


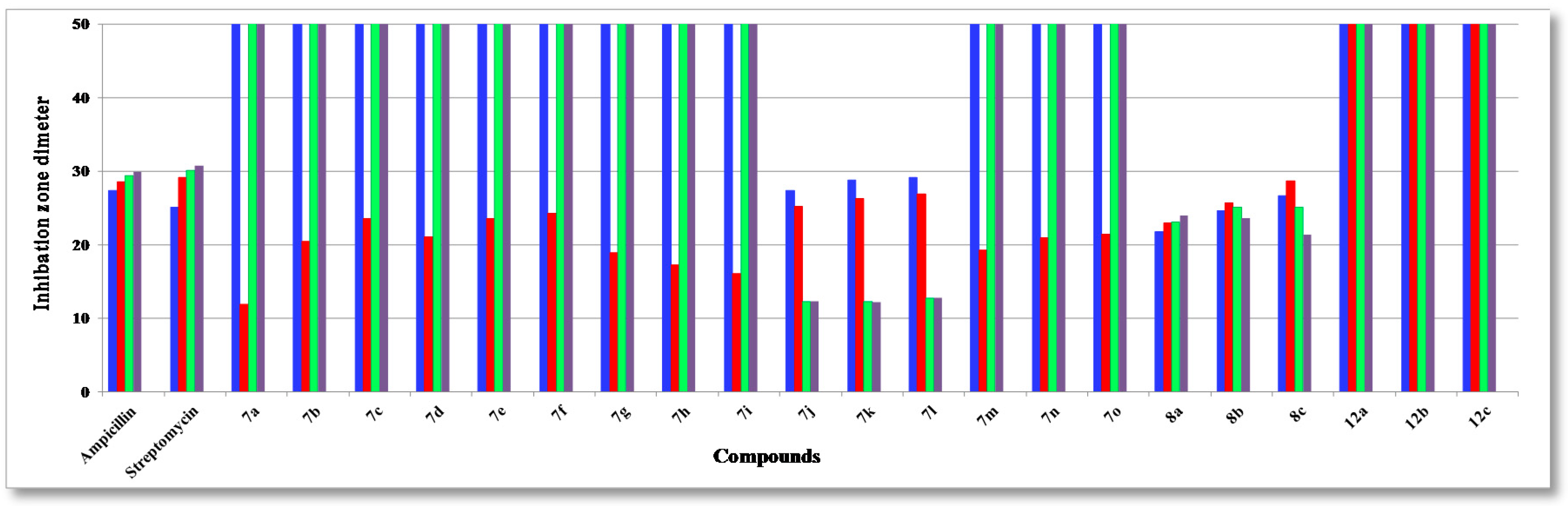
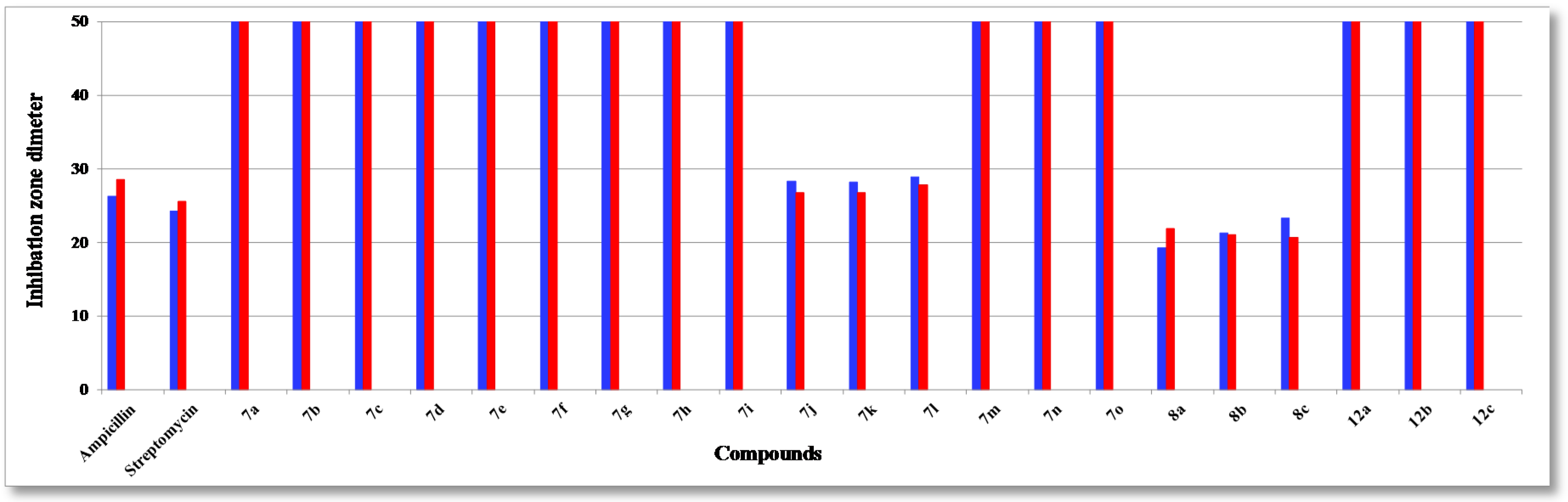

| Compound | R | Ar | S. aureus (+ve) | S. Epider. (+ve) | B. Subtilis (+ve) | B. pumilus (+ve) | P. Aerug. (−ve) | E. coli (−ve) | A. fumig. | C. albic. | S. cerev. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 7a | H | 4-FC6H5 | NA | 11.9 ± 0.3 | NA | NA | NA | NA | NA | NA | NA |
| 7b | H | 4-ClC6H5 | NA | 20.5 ± 0.2 | NA | NA | NA | NA | NA | NA | NA |
| 7c | H | 4-BrC6H5 | NA | 23.5 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7d | CH3 | 4-FC6H5 | NA | 21.1 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7e | CH3 | 4-ClC6H5 | NA | 23.5 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7f | CH3 | 4-BrC6H5 | NA | 24.3 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7g | CO2Et | 4-FC6H5 | NA | 18.9 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7h | CO2Et | 4-ClC6H5 | NA | 17.2 ± 0.2 | NA | NA | NA | NA | NA | NA | NA |
| 7i | CO2Et | 4-BrC6H5 | NA | 16.1 ± 0.3 | NA | NA | NA | NA | NA | NA | NA |
| 7j | CH2CN | 4-FC6H5 | 27.3 ± 0.5 | 25.2 ± 0.1 | 12.3 ± 0.3 | 12.3 ± 0.3 | 28.3 ± 0.07 | 26.7 ± 0.1 | 12.3 ± 0.3 | 19.3 ± 0.1 | 23.3 ± 0.1 |
| 7k | CH2CN | 4-ClC6H5 | 28.8 ± 0.1 | 26.3 ± 0.3 | 12.2 ± 0.4 | 12.2 ± 0.4 | 28.2 ± 0.03 | 26.8 ± 0.3 | 12.2 ± 0.1 | 21.2 ± 0.2 | 20.2 ± 0.1 |
| 7l | CH2CN | 4-BrC6H5 | 29.1 ± 0.2 | 26.9 ± 0.3 | 12.7 ± 0.4 | 12.8 ± 0.4 | 28.9 ± 0.03 | 27.8 ± 0.3 | 12.1 ± 0.2 | 21.1 ± 0.2 | 18.2 ± 0.1 |
| 7m | Ph | 4-FC6H5 | NA | 19.3 ± 0.5 | NA | NA | NA | NA | NA | NA | NA |
| 7n | Ph | 4-ClC6H5 | NA | 20.9 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 7o | Ph | 4-BrC6H5 | NA | 21.4 ± 0.1 | NA | NA | NA | NA | NA | NA | NA |
| 8a | - | 4-FC6H5 | 21.8 ± 0.1 | 22.9 ± 0.1 | 23.1 ± 0.2 | 24.0 ± 0.3 | 19.3 ± 0.1 | 21.9 ± 0.1 | 23.8 ± 0.1 | 22.9 ± 0.1 | 16.8 ± 0.4 |
| 8b | - | 4-ClC6H5 | 24.6 ± 0.2 | 25.7 ± 0.2 | 25.1 ± 0.4 | 23.6 ± 0.1 | 21.3 ± 0.2 | 21.0 ± 0.4 | 23.4 ± 0.1 | 22.4 ± 0.1 | 18.2 ± 0.3 |
| 8c | - | 4-BrC6H5 | 26.6 ± 0.1 | 28.5 ± 0.3 | 25.1 ± 0.4 | 21.3 ± 0.1 | 23.3 ± 0.1 | 20.7 ± 0.2 | 22.1 ± 0.1 | 21.7 ± 0.2 | 19.5 ± 0.1 |
| 12a | - | 4-FC6H5 | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| 12b | - | 4-ClC6H5 | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| 12c | - | 4-BrC6H5 | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Ampicillin | - | - | 27.4 ± 0.1 | 28.6 ± 0.5 | 29.4 ± 0.7 | 29.9 ± 0.3 | 26.3 ± 0.3 | 28.5 ± 0.1 | - | - | - |
| Streptomycin | - | - | 25.1 ± 0.2 | 29.1 ± 0.3 | 30.1 ± 0.9 | 30.8 ± 0.4 | 24.3 ± 0.8 | 25.6 ± 0.4 | - | - | - |
| Mycostatine | - | - | - | - | - | - | - | - | 29.5 ± 0.1 | 27.1 ± 0.1 | 24.1 ± 0.3 |
| Clotrimazole | - | - | - | - | - | - | - | - | 26.1 ± 0.1 | 23.1 ± 0.3 | 18.3 ± 0.1 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okasha, R.M.; Albalawi, F.F.; Afifi, T.H.; Fouda, A.M.; Al-Dies, A.-A.M.; El-Agrody, A.M. Structural Characterization and Antimicrobial Activities of 7H-Benzo[h]chromeno[2,3-d]pyrimidine and 14H-Benzo[h]chromeno[3,2-e][1,2,4]triazolo[1,5-c] pyrimidine Derivatives. Molecules 2016, 21, 1450. https://doi.org/10.3390/molecules21111450
Okasha RM, Albalawi FF, Afifi TH, Fouda AM, Al-Dies A-AM, El-Agrody AM. Structural Characterization and Antimicrobial Activities of 7H-Benzo[h]chromeno[2,3-d]pyrimidine and 14H-Benzo[h]chromeno[3,2-e][1,2,4]triazolo[1,5-c] pyrimidine Derivatives. Molecules. 2016; 21(11):1450. https://doi.org/10.3390/molecules21111450
Chicago/Turabian StyleOkasha, Rawda M., Fawzia F. Albalawi, Tarek H. Afifi, Ahmed M. Fouda, Al-Anood M. Al-Dies, and Ahmed M. El-Agrody. 2016. "Structural Characterization and Antimicrobial Activities of 7H-Benzo[h]chromeno[2,3-d]pyrimidine and 14H-Benzo[h]chromeno[3,2-e][1,2,4]triazolo[1,5-c] pyrimidine Derivatives" Molecules 21, no. 11: 1450. https://doi.org/10.3390/molecules21111450
APA StyleOkasha, R. M., Albalawi, F. F., Afifi, T. H., Fouda, A. M., Al-Dies, A.-A. M., & El-Agrody, A. M. (2016). Structural Characterization and Antimicrobial Activities of 7H-Benzo[h]chromeno[2,3-d]pyrimidine and 14H-Benzo[h]chromeno[3,2-e][1,2,4]triazolo[1,5-c] pyrimidine Derivatives. Molecules, 21(11), 1450. https://doi.org/10.3390/molecules21111450







