Cytoproliferative and Cytoprotective Effects of Striatisporolide A Isolated from Rhizomes of Athyrium multidentatum (Doell.) Ching on Human Umbilical Vein Endothelial Cells
Abstract
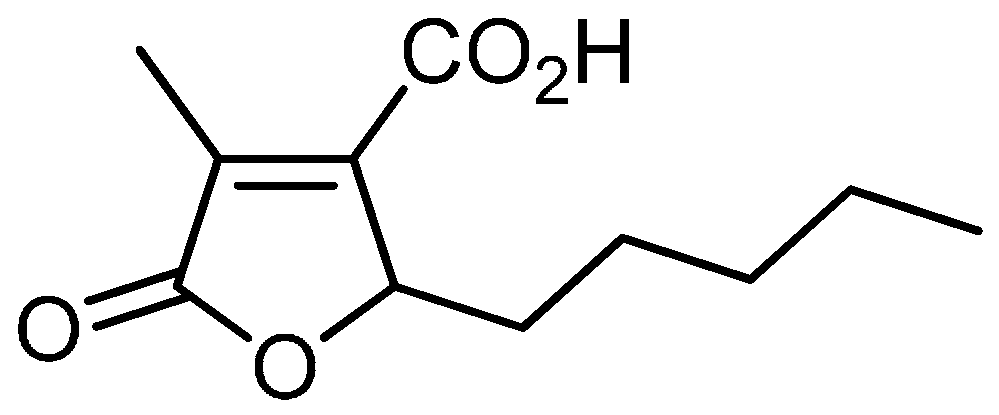
:1. Introduction
2. Results and Discussion
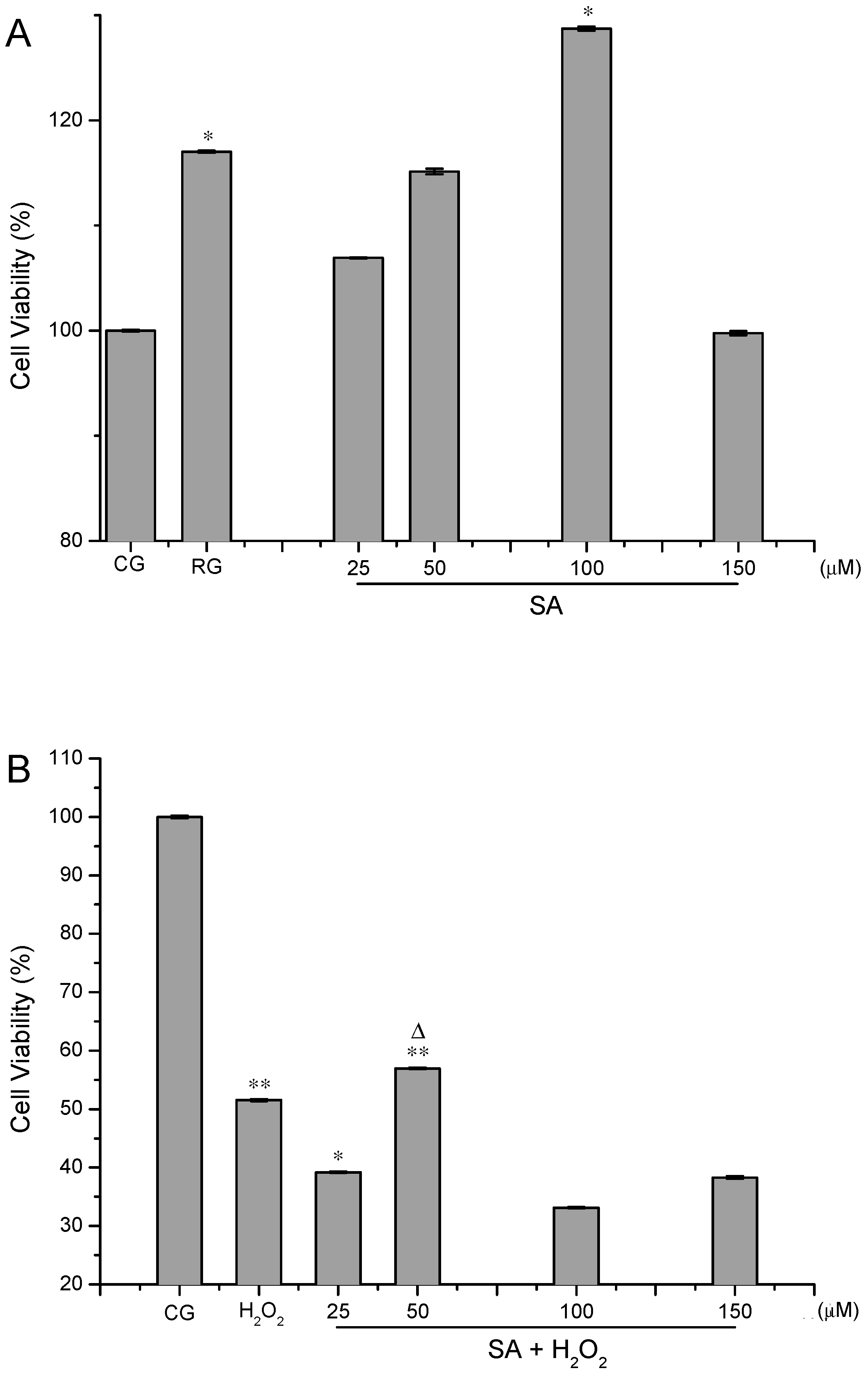
2.1. Analysis of Cell Viability
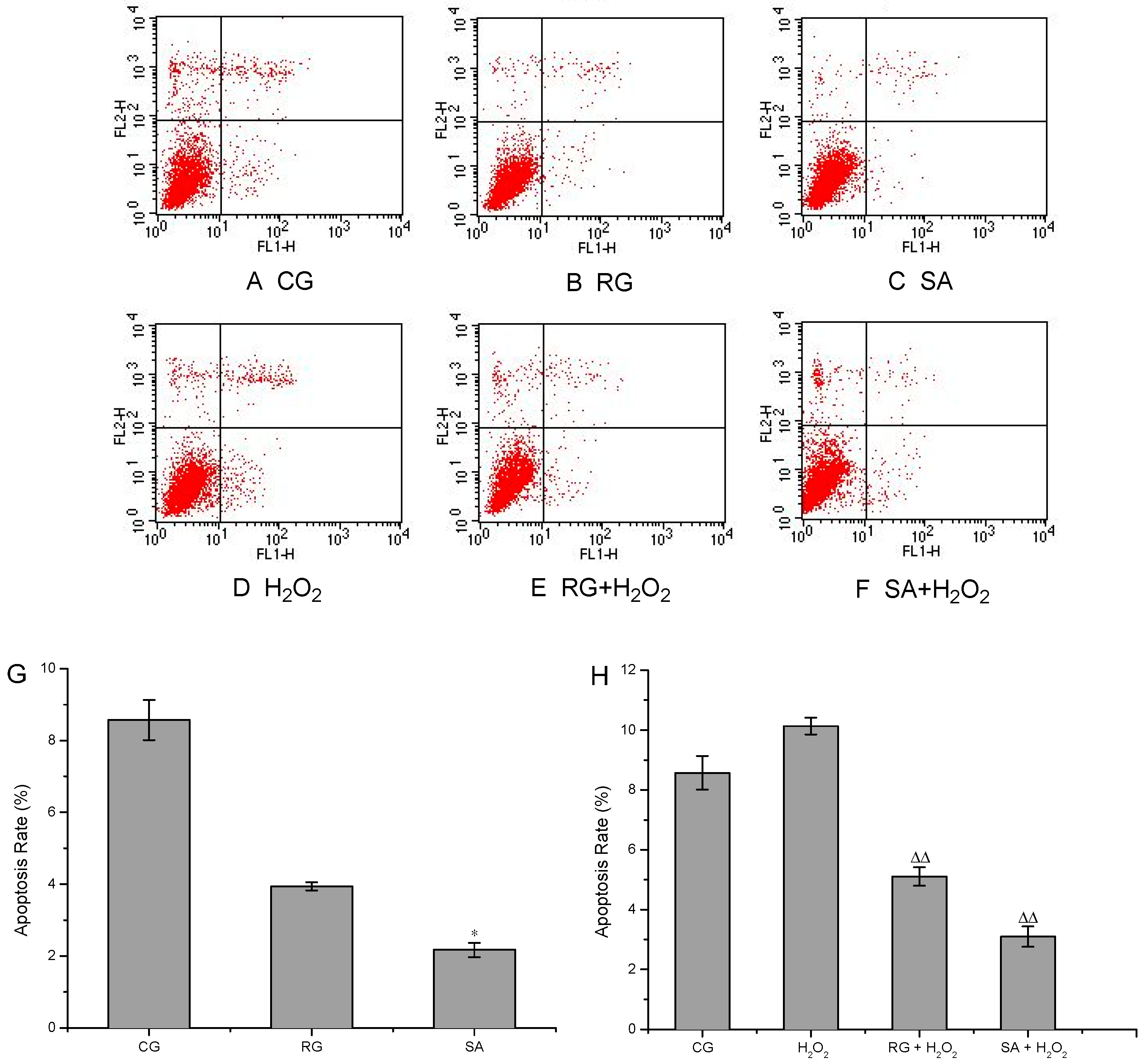
2.2. Cell Apoptosis Assays
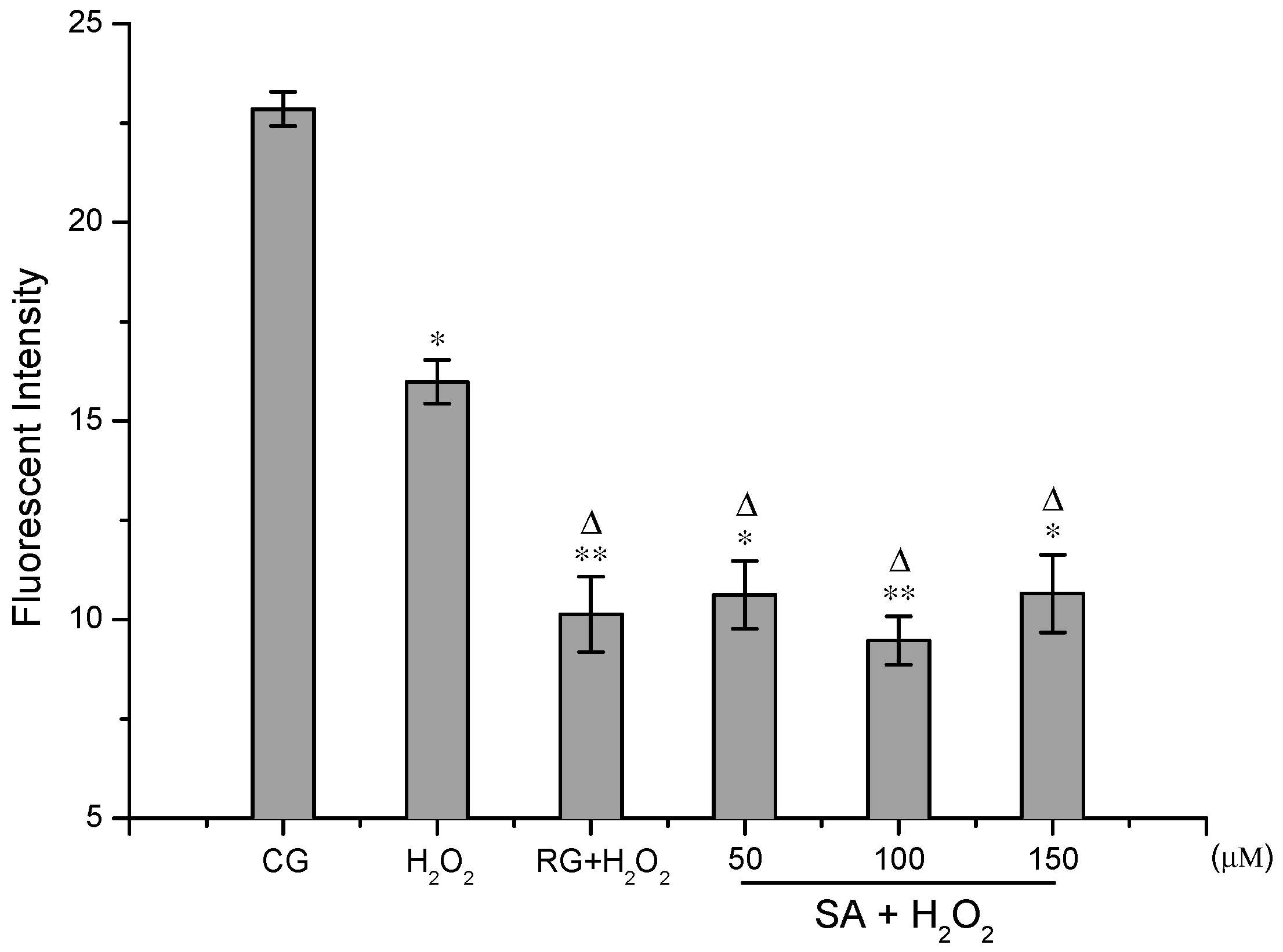
2.3. ROS Detection Assays
3. Materials and Methods
3.1. Materials
3.2. Preparation of Samples
3.3. Cell Culture
3.4. Analysis of Cell Viability
3.5. Cell Apoptosis Assays
3.6. ROS Detection Assays
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, D.H.; Zhu, T.J.; Liu, H.B.; Fang, Y.C.; Gu, Q.Q.; Zhu, W.M. Four Butenolides are Novel Cytotoxic Compounds Isolated from the Marine-Derived Bacterium, Streptoverticillium luteoverticillatum 11014. Arch. Pharm. Res. 2006, 29, 624–626. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.F.; Sun, T.T.; Ke, S.Y. Progress in the Studies on Synthesis and Biological Properties of Butenolide and Its Derivatives. Chin. J. Org. Chem. 2010, 30, 1113–1125. [Google Scholar]
- Prem, P.Y.; Ashish, A.; Hemant, K.B.; Ritu, R.K.; Sanjeev, K. New cassane butenolide hemiketal diterpenes from the marine creeper Caesalpinia bonduc and their antiproliferative activity. Tetrahedron Lett. 2007, 48, 7194–7198. [Google Scholar]
- Wang, X.J.; Xu, H.W.; Guo, L.L.; Zheng, J.X.; Xu, B.; Guo, X.; Zheng, C.X.; Liu, H.M. Synthesis and in vitro antitumor activity of new butenolide-containing Dithiocarbamates. Bioorg. Med. Chem. Lett. 2011, 21, 3074–3077. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Nishigaya, K.; Uchikoshi, H.; Chen, I.S. Cochinolide, a new γ-alkylidene bicyclic butenolide with antiviral activity, and its β-glucopyranoside from homalium cochinchinensis. J. Nat. Prod. 1998, 61, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Igoli, J.O.; Gray, A.I.; Clements, C.J.; Kantheti, P.; Singla, R.K. Antitrypanosomal activity & docking studies of isolated constituents from the lichen Cetraria islandica: Possibly multifunctional scaffolds. Curr. Top. Med. Chem. 2014, 14, 1–8. [Google Scholar]
- Stewart, M.; Capon, O.J.; Lacey, E.; Tennant, S.; Gill, J.H. Calbistrin e and two other new metabolites from an Australian isolate of Penicillium striatisporum. J. Nat. Prod. 2005, 68, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.S.; Huang, Y.L.; Ma, L.Y.; Lu, C.J.; Liu, W.Z. Chemical constituents and their cytotoxic activities of the secondary metabolites of Penicillium janthinellum. Chin. Trad. Patent Med. 2016, 38, 830–834. [Google Scholar]
- Patel, R.M.; Argade, N.P. General approach to 2,4-dialkyl-3-carboxybuty-rolactones: An efficient synthesis of (±)-striatisporolide A and (±)-lichesterinic acid. Indian J. Chem. 2010, 49B, 1071–1075. [Google Scholar]
- Liu, D.M.; Sheng, J.W.; Wang, S.H.; Zhang, W.F.; Zhang, X.H.; Wang, D.Y. Chemical constituents from Athyrium multidentatum (Doell.) Ching rhizome and their reducing power. Chin. J. Exp.Tradit. Med. Form. 2016, 22, 199–202. [Google Scholar]
- Hou, Y.L.; Liu, L.; Ding, X.; Zhao, D.Q.; Hou, W.R. Structure elucidation, proliferation effect on macrophage and its mechanism of a new heteropolysaccharide from Lactarius deliciosus Gray. Carbohyd. Polym. 2016, 152, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Young, Y.J.; Dolly, S.; Deepti, S.; Mi, L.E.; Soonmo, C.; Soo, H.S.; Seon, J.P. Terminalia bellirica extracts loaded on stimuli responsive HEMA-DEA hydrogel for enhanced growth and proliferation of mesenchymal stem cells. J. Biomater. Tissue Eng. 2014, 4, 37–45. [Google Scholar] [CrossRef]
- Udalamaththa, V.L.; Jayasinghe, C.D.; Udagama, P.V. Potential role of herbal remedies in stem cell therapy: Proliferation and differentiation of human mesenchymal stromal cells. Stem Cell Res. Ther. 2016, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.H.; Wang, B.L.; Huang, P.; Wang, H.Y.; Xu, K.L.; Wang, X.F.; Xu, L.H.; Guo, Z.L. Microcystin-LR promotes cell proliferation in the mice liver by activating Akt and p38/ERK/JNK cascades. Chemosphere 2016, 163, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Kitada, M.; Koya, D. Renal Protective Effects of Resveratrol. Oxid. Med. Cell. Longev. 2013, 2013, 568093. [Google Scholar] [CrossRef] [PubMed]
- Masella, R.; Santangelo, C.; D’Archivio, M.; LiVolti, G.; Giovannini, C.; Galvano, F. Protocatechuic acid and human disease prevention: Biological activities and molecular mechanisms. Curr. Med. Chem. 2012, 19, 2901–2917. [Google Scholar] [CrossRef] [PubMed]
- Alberts, B.; Johnson, A.; Lewis, J.L.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell; Garland Science: New York, NY, USA; Milton Park, UK, 2002; p. 1119. [Google Scholar]
- An, L.J.; Guan, S.; Shi, G.F.; Bao, Y.M.; Duan, Y.L.; Jiang, B. Protocatechuic acid from Alpinia oxyphylla against MPP+-induced neurotoxicity in PC12 cells. Food Chem. Toxicol. 2006, 44, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.N.; An, C.N.; Xu, M.; Guo, D.A.; Li, M.; Pu, X.P. Protocatechuic acid inhibits rat pheochromocytoma cell damage induced by a dopaminergic neurotoxin. Biol. Pharm. Bull. 2009, 32, 1866–1869. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Pan, L.Q.; Zhang, L.; Miao, J.J.; Wang, J. Immune responses, ROS generation and the haemocyte damage of scallop Chlamys farreri exposed to Aroclor 1254. Fish Shellfish Immunol. 2009, 26, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Fiers, W.; Beyaert, R.; Declercq, W.; Vandenabeele, P. More than one way to die: apoptosis and necrosis and reactive oxygen damage. Oncogene 1999, 18, 7719–7730. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H. Berberine Hydrochloride Protects C2C12 Myoblast cells against oxidative stress-induced damage via induction of nrf-2-mediated ho-1 expression. Drug Dev. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Luo, Q.; Cao, Y.; Goulette, T.; Liu, X.; Xiao, H. Mechanism of different stereoisomeric astaxanthin in resistance to oxidative stress in caenorhabditis elegans. J. Food Sci. 2016. [Google Scholar] [CrossRef]
- Brandt, A.P.; Gozzi, G.J.; Pires Ado, R.A.; Martinez, G.R.; dos Santos Canuto, A.V.; Echevarria, A.; di Pietro, A.; Cadena, S.M. Impairment of oxidative phosphorylation increases the toxicity of SYD-1 on hepatocarcinoma cells (HepG2). Chem.-Biol. Interact. 2016, 256, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Guan, S.; Bao, Y.M.; Jiang, B.; An, L.J. Protective effect of protocatechuic acid from Alpinia oxyphylla on hydrogen peroxide-induced oxidative PC12 cell death. Eur. J. Pharmacol. 2006, 538, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.H.; Hsieh, T.J.; Tsai, M.L.; Chen, C.H.; Lin, H.T.; Wu, S.J. Neuroprotective effects of Hu-Yi-Neng, a diet supplement, on SH-SY5Y human neuroblastoma cells. J. Nutr. Health Aging 2014, 18, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Striatisporolide A sample is available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, D.-M.; Sheng, J.-W.; Wang, S.-H.; Zhang, W.-F.; Zhang, W.; Zhang, D.-J. Cytoproliferative and Cytoprotective Effects of Striatisporolide A Isolated from Rhizomes of Athyrium multidentatum (Doell.) Ching on Human Umbilical Vein Endothelial Cells. Molecules 2016, 21, 1280. https://doi.org/10.3390/molecules21101280
Liu D-M, Sheng J-W, Wang S-H, Zhang W-F, Zhang W, Zhang D-J. Cytoproliferative and Cytoprotective Effects of Striatisporolide A Isolated from Rhizomes of Athyrium multidentatum (Doell.) Ching on Human Umbilical Vein Endothelial Cells. Molecules. 2016; 21(10):1280. https://doi.org/10.3390/molecules21101280
Chicago/Turabian StyleLiu, Dong-Mei, Ji-Wen Sheng, Si-Hong Wang, Wei-Fen Zhang, Wei Zhang, and Dai-Juan Zhang. 2016. "Cytoproliferative and Cytoprotective Effects of Striatisporolide A Isolated from Rhizomes of Athyrium multidentatum (Doell.) Ching on Human Umbilical Vein Endothelial Cells" Molecules 21, no. 10: 1280. https://doi.org/10.3390/molecules21101280





