Discovery of 5-(5,5-Dimethylbutenolide-3-ethylidene)-2-amino-imidazolinone Derivatives as Fungicidal Agents
Abstract
:1. Introduction
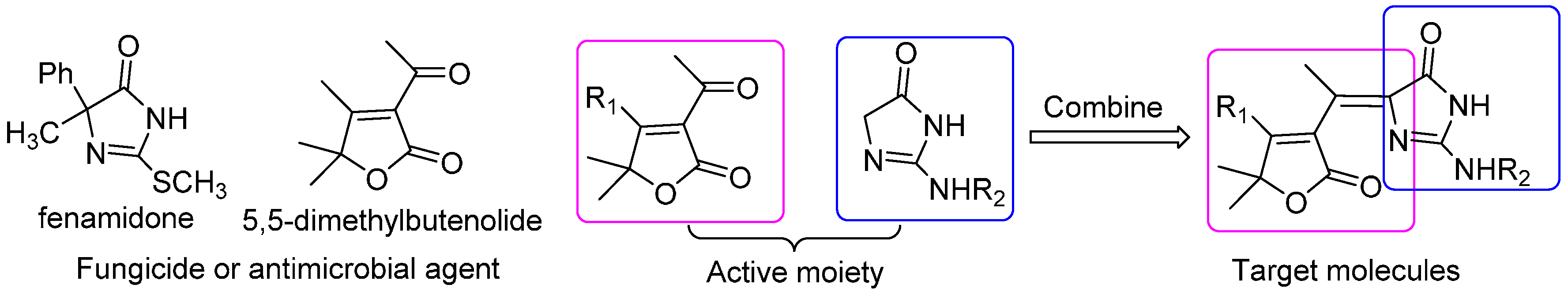
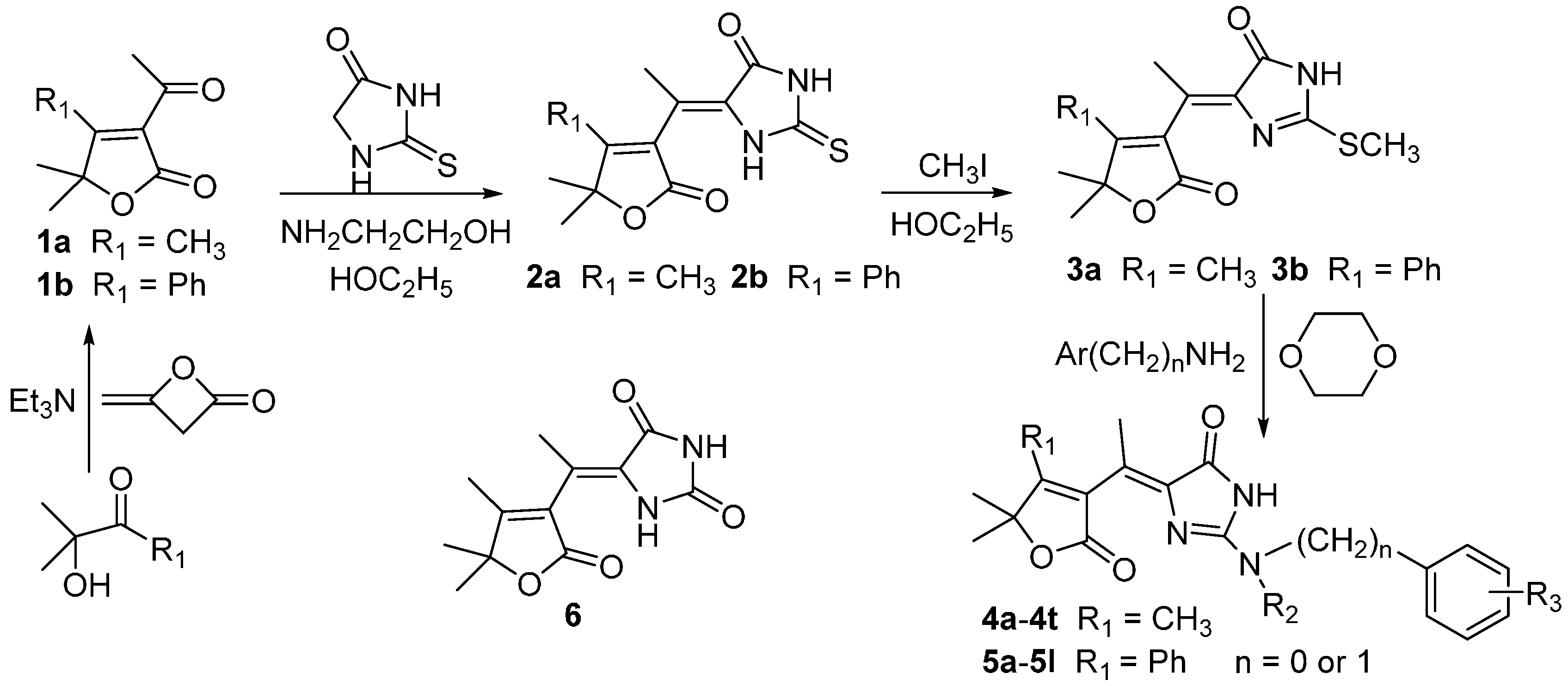
2. Results and Discussion
| Compd. | n R2 R3 | S. scleotiorum | R. solani | A. solani | B. cinerea | P. capsici | F. graminearum |
|---|---|---|---|---|---|---|---|
| 1a | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | |
| 1b | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | |
| 2a | 49.7 | 66.8 | 42.3 | 52.2 | 69.8 | 31.8 | |
| 2b | 3.3 | 7.7 | 1.5 | 36.2 | 59.3 | 3.9 | |
| 3a | 25.4 | 72.7 | 51.9 | 20.0 | 79.4 | 34.0 | |
| 3b | 0.0 | 18.6 | 0.0 | 16.8 | 34.2 | 10.6 | |
| 4a | 0 H H | 94.9 | 68.4 | 50.9 | 60.5 | 24.0 | 21.1 |
| 4b | 0 H 4-CH3 | 33.1 | 63.5 | 60.3 | 27.2 | 29.3 | 31.6 |
| 4c | 0 H 4-F | 19.1 | 59.6 | 63.2 | 31.8 | 16.7 | 38.2 |
| 4d | 0 H 4-OCH3 | 32.3 | 72.9 | 60.8 | 75.3 | 15.5 | 38.6 |
| 4e | 0 H 4-CF3 | 92.8 | 61.2 | 64.8 | 32.2 | 0.0 | 48.7 |
| 4f | 0 CH3 H | 31.7 | 57.6 | 53.9 | 16.5 | 36.1 | 0.0 |
| 4g | 0 H 3-CF3 | 65.0 | 72.9 | 55.7 | 69.3 | 47.7 | 38.0 |
| 4h | 0 H 2-CH3 | 16.5 | 56.4 | 37.0 | 36.2 | 15.7 | 0.0 |
| 4i | 0 H 2-F | 38.9 | 76.1 | 67.7 | 40.2 | 0.0 | 23.9 |
| 4j | 0 H 2-OCH3 | 22.2 | 75.7 | 22.0 | 21.4 | 33.9 | 31.5 |
| 4k | 0 H 2-Cl | 18.7 | 44.6 | 44.6 | 45.5 | 41.8 | 37.4 |
| 4l | 0 H 2,6-(CH3)2 | 30.8 | 65.3 | 45.9 | 29.1 | 2.1 | 39.2 |
| 4m | 1 H H | 52.2 | 72.0 | 53.9 | 48.4 | 23.0 | 22.9 |
| 4n | 1 H 4-OCH3 | 36.9 | 66.5 | 39.0 | 28.6 | 0.0 | 51.3 |
| 4o | 1 H 4-Cl | 16.3 | 68.2 | 48.1 | 40.3 | 5.3 | 30.7 |
| 4p | 1 H 4-F | 21.5 | 48.5 | 45.4 | 39.2 | 33.6 | 38.5 |
| 4q | 1 H 3-CF3 | 21.7 | 64.9 | 61.2 | 33.3 | 56.6 | 45.1 |
| 4r | 1 H 2-F | 15.4 | 59.5 | 41.2 | 31.6 | 27.6 | 23.5 |
| 4s | 1 H 2-Cl | 21.1 | 60.4 | 38.5 | 27.0 | 21.3 | 13.0 |
| 4t | 1 H 2-OCH3 | 22.2 | 72.5 | 42.1 | 41.5 | 0.0 | 10.1 |
| 5a | 0 H H | 67.8 | 7.6 | 0.0 | 21.3 | 27.7 | 26.4 |
| 5b | 0 H 4-CH3 | 29.4 | 10.9 | 11.6 | 25.1 | 13.3 | 22.0 |
| 5c | 0 H 4-F | 44.6 | 0.0 | 34.9 | 25.2 | 48.0 | 37.1 |
| 5d | 0 H 4-OCH3 | 21.4 | 6.9 | 0.0 | 15.2 | 20.0 | 16.7 |
| 5e | 0 H 4-CF3 | 58.1 | 11.4 | 31.4 | 38.6 | 50.4 | 24.1 |
| 5f | 0 H 3-CF3 | 36.8 | 11.4 | 65.7 | 48.6 | 44.4 | 32.6 |
| 5g | 0 H 2-F | 81.4 | 12.9 | 14.4 | 26.3 | 45.1 | 59.9 |
| 5h | 0 H 2-Cl | 57.8 | 2.3 | 37.3 | 30.3 | 37.1 | 40.2 |
| 5i | 0 H 2-OCH3 | 31.2 | 1.5 | 0.0 | 6.8 | 8.7 | 20.0 |
| 5j | 1 H H | 18.3 | 9.6 | 5.2 | 0.0 | 2.4 | 28.3 |
| 5k | 1 H 4-Cl | 29.2 | 0.0 | 24.4 | 12.6 | 13.3 | 15.4 |
| 5l | 1 H 2-OCH3 | 30.0 | 6.9 | 3.0 | 8.5 | 0.0 | 27.7 |
| 6 | 41.7 | 46.1 | 70.7 | 40.9 | 4.2 | 31.6 | |
| Chlorothalonil | 100 | 99.9 | 100 | 100 | 99.1 | 95.8 | |
| Carbendazim | 81.0 | 100 | 100 | 4.2 | 34.7 | 100 | |
| Fenamidone | 97.8 | 96.4 | 93.5 | 57.1 | 79.8 | 71.9 |
| Compd. | Fungi | Equation | Coefficent γ2 | EC50 (µM) |
|---|---|---|---|---|
| 4a | S. scleotiorum | Y = 4.151X − 16.812 | 0.966 | 268.58 |
| 4e | S. scleotiorum | Y = 2.181X − 7.968 | 0.990 | 220.54 |
| 4g | S. scleotiorum | Y = 2.236X − 9.418 | 0.950 | 14.73 |
| 5g | S. scleotiorum | Y = 0.902X − 3.382 | 0.978 | 3.23 |
| Carbendazim | S. scleotiorum | Y = 1.143X − 4.821 | 0.981 | 4.12 |
| 2a | R. solani | Y = 2.993X − 12.458 | 0.949 | 120.78 |
| 3a | R. solani | Y = 2.136X − 8.651 | 0.972 | 38.25 |
| 4a | R. solani | Y = 3.052X − 12.407 | 0.956 | 218.48 |
| 4d | R. solani | Y = 2.152X − 8.824 | 0.953 | 24.18 |
| 4g | R. solani | Y = 2.170X − 8.084 | 0.971 | 149.00 |
| 4i | R. solani | Y = 1.797X − 6.847 | 0.963 | 48.73 |
| 4j | R. solani | Y = 8.084X − 36.222 | 0.972 | 4425.75 |
| 4l | R. solani | Y = 3.608X − 15.344 | 0.981 | 146.93 |
| 4m | R. solani | Y = 2.80X − 12.685 | 0.953 | 6.07 |
| 4n | R. solani | Y = 1.440X − 5.298 | 0.962 | 21.68 |
| 4o | R. solani | Y = 3.995X − 17.315 | 0.985 | 122.76 |
| 4t | R. solani | Y = 2.538X − 11.106 | 0.950 | 10.38 |
| 4e | A. solani | Y = 1.283X − 5.263 | 0.981 | 4.14 |
| 4i | A. solani | Y = 1.459X − 6.242 | 0.997 | 3.27 |
| 6 | A. solani | Y = 1.263X − 4.926 | 0.978 | 9.86 |
| Fenamidone | A. solani | Y = 0.802X − 3.709 | 0.973 | 0.90 |
| 4d | B. cinerea | Y = 2.127X − 8.363 | 0.957 | 52.49 |
| 4g | B. cinerea | Y = 1.514X − 5.267 | 0.986 | 51.02 |
| Fenamidone | B. cinerea | Y = 1.329X − 4.712 | 0.963 | 38.92 |

3. Experimental Section
3.1. General Information
3.2. Synthesis
3.2.1. Synthesis of 3-Acetyl-4-methyl(phenyl)-5,5-dimethylbutenolide (1a and 1b)
3.2.2. Synthesis of 4-Methyl(phenyl)-5,5-dimethyl-3-ethylidenebutenolide-5-thiohydantoin (2a and 2b)
3.2.3. Synthesis of 4-Methyl(phenyl)-5,5-dimethyl-3-ethylidenebutenolide-5-(2-methylthioimidazo-4-one) (3a and 3b)
3.2.4. General Procedure for the Synthesis of Compounds 4 and 5
3.3. Bioassay of Fungicidal Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.; Xie, H.; Pan, Y.R.; Ding, M.W. Facile synthesis of 4-arylidene-1H-imidazol-5(4H)-ones by an Ugi-Aza-Wittig sequences. Synthesis 2014, 46, 336–342. [Google Scholar] [CrossRef]
- Revanasiddappa, B.C.; Kalsi, J.; Jisha, M.S.; Jose, N.; Varghese, S.S. Synthesis and biological evaluation of some novel imidazolinone derivatives. Indian J. Heterocycl. Chem. 2013, 22, 225–228. [Google Scholar]
- Miqdad, O.A.; Abunada, N.M.; Hassaneen, H.M.; Abu Samaha, A.M. Synthesis and biological activity evaluation of some new heterocyclic spirocompounds with imidazolinone and pyrazoline moieties. Int. J. Chem. 2011, 3, 20–31. [Google Scholar] [CrossRef]
- Patel, J.B.; Desai, V.A. Discovery of new imidazolinone derivatives as potential microbial agents. Int. J. Drug Des. Discov. 2011, 2, 393–402. [Google Scholar]
- Ivanna, S.; Dmytro, A.; Ewa, S.; Katarzyna, K.K.; Borys, Z.; Olexandr, V.; Andrezj, G.; Roman, L. Synthesis of 5-arylidene-2-amino-4-azolones and evaluation of their anticancer activity. Bioorg. Med. Chem. 2010, 18, 5090–5102. [Google Scholar]
- Huang, X.B.; Wu, D.Z.; Ding, J.C.; Zhang, X.C.; Liu, M.C.; Wu, H.Y.; Su, W.K. Synthesis and biological activities of new chiral imidazolone derivatives. Phosphorus Sulfur Silicon 2010, 185, 117–128. [Google Scholar] [CrossRef]
- Huang, X.B.; Liu, J.Z.; Yang, F.L.; Ding, M.W. Synthesis and properties of novel imidazolone derivatives containing a sulfur atom. Phosphorus Sulfur Silicon 2007, 182, 939–950. [Google Scholar] [CrossRef]
- Paola, V.; Athina, G.; Kitka, A.; Matteo, I.; Franca, Z. Synthesis and antimicrobial activity of novel 2-thiozolyimino-5-arylidene-2-amino-4-thiazolidinones. Bioorg. Med. Chem. 2006, 14, 3859–3964. [Google Scholar]
- Travert, N.; Al Mourabit, A. A likely biogenetic gateway linking 2-aminoimidazolinone metabolites of sponges to proline: Spontaneous oxidative conversion of the pyrrole-proline guanidine pseudo-peptide to dispacamide A. J. Am. Chem. Soc. 2004, 126, 10252–10253. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.P.; Han, J.T.; Xu, Z.H.; Dong, H.B.; Wang, M.A. Synthesis and fungicidal activity of 5-cyclohexylidene-2-aminoimidazolin-4-one derivatives. Chin. J. Org. Chem. 2012, 32, 1993–1998. [Google Scholar] [CrossRef]
- Lei, J.P.; Dong, H.B.; Tang, B.; Wang, M.A. Synthesis and fungicidal activity of 5-(2-ethylcyclohexylidene)-2-aminoimidazolin-4-one derivatives. Chin. J. Pestic. Sci. 2012, 14, 669–672. [Google Scholar]
- Wang, W.H.; Kim, H.Y.; Nam, S.; Rho, B.J.; Kang, H. Antibacterial butenolides from the Korean tunicate Pseudodistoma antinboja. J. Nat. Prod. 2012, 75, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
- Centko, R.M.; Ramón-García, S.; Taylor, T.; Patrick, B.O.; Thompson, C.J.; Miao, V.P.; Andersen, R.J. Ramariolides A–D, antimycobacterial butenolides isolated from the mushroom Ramaria cystidiophora. J. Nat. Prod. 2012, 75, 2178–2182. [Google Scholar] [CrossRef] [PubMed]
- Schulz, D.; Ohlendorf, B.; Zinecker, H.; Schmaljohann, R.; Imhoff, J.F. Eutypoids B–E produced by a Penicillium sp. strain from the North Sea. J. Nat. Prod. 2011, 74, 99–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinale, F.; Marra, R.; Scala, F.; Ghisalberti, E.L.; Lorito, M.; Sivasithamparam, K. Major secondary metabolites produced by two commercial Trichoderma strains active against different phytopathogens. Lett. Appl. Microbiol. 2006, 43, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Wangteeraprasert, R.; Lipipun, V.; Gunaratnam, M.; Neidle, S.; Gibbons, S.; Likhitwitayawuid, K. Bioactive compounds from Carissa spinarum. Phytother. Res. 2012, 26, 1496–1499. [Google Scholar] [CrossRef] [PubMed]
- Leite, L.; Jansone, D.; Veveris, M.; Cirule, H.; Popelis, J.; Melikyan, G.; Avetisyan, A.; Lukevics, E. Vasodilating and antiarrhythmic activity of heteryl lactones. Eur. J. Med. Chem. 1999, 34, 859–865. [Google Scholar] [CrossRef]
- Melikyan, G.S.; Hovhannisyan, A.A.; Hayotsyan, S.S. Synthesis of some heterocyclic compounds from enamines of 3-acetylfuran-2(5H)-ones. Synth. Commun. 2012, 42, 2267–2276. [Google Scholar] [CrossRef]
- Kiec-Kononowicz, K.; Szymanska, E. Antimycobacterial activity of 5-arylidene derivatives of hydantoin. Farmaco 2002, 57, 909–916. [Google Scholar] [CrossRef]
- Lacey, R.N. Derivatives of acetoacetic acid. Part IV. A new route to α-acetyltetronic acids. J. Chem. Soc. 1954, 832–839. [Google Scholar] [CrossRef]
- Han, J.T.; Dong, H.B.; Xu, Z.H.; Wang, J.M.; Wang, M.A. Synthesis and Activity of Novel Acylthiourea with Hydantoin. Int. J. Mol. Sci. 2013, 14, 19526–19539. [Google Scholar] [CrossRef] [PubMed]
- Han, J.T.; Dong, H.B.; Xu, Z.H.; Lei, J.P.; Wang, M.A. Facile synthesis of 5-arylidene thiohydantoin by sequential sulfonylation/desulfination reaction. Int. J. Mol. Sci. 2013, 14, 12484–12495. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Han, J.T.; Tang, B.; Wang, M.A. Synthesis and Fungicidal Activity of d-Ribose and d-Xylose with Hydantion. Chin. J. Org. Chem. 2014, 34, 1442–1446. [Google Scholar] [CrossRef]
- Chen, N.C. The Bioassay Technologies for Pesticides; Beijing Agricultural University Press: Beijing, China, 1991; pp. 161–162. [Google Scholar]
- Chen, W.Y. The Research & Development of New Pesticide—Methods & Progress; Chemical Industry Press: Beijing, China, 2007; pp. 132–134. [Google Scholar]
- Sample Availability: Samples of the compounds 4a–4t, 5a–5l and 6 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, B.; Yang, M.; Zhao, Y.; Kong, L.; Wang, W.; Wang, M. Discovery of 5-(5,5-Dimethylbutenolide-3-ethylidene)-2-amino-imidazolinone Derivatives as Fungicidal Agents. Molecules 2015, 20, 13740-13752. https://doi.org/10.3390/molecules200813740
Tang B, Yang M, Zhao Y, Kong L, Wang W, Wang M. Discovery of 5-(5,5-Dimethylbutenolide-3-ethylidene)-2-amino-imidazolinone Derivatives as Fungicidal Agents. Molecules. 2015; 20(8):13740-13752. https://doi.org/10.3390/molecules200813740
Chicago/Turabian StyleTang, Bo, Mingyan Yang, Yu Zhao, Lingqing Kong, Weiwei Wang, and Mingan Wang. 2015. "Discovery of 5-(5,5-Dimethylbutenolide-3-ethylidene)-2-amino-imidazolinone Derivatives as Fungicidal Agents" Molecules 20, no. 8: 13740-13752. https://doi.org/10.3390/molecules200813740






