Multidimensional Transition Metal Complexes Based on 3-Amino-1H-1,2,4-triazole-5-carboxylic Acid: From Discrete Mononuclear Complexes to Layered Materials
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
- (i)
- the use NaOH or triethylamine (Et3N) to deprotonate H2atrc in reactions with transition metal salts (such as Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+ and Cd2+) led to poor crystalline powders;
- (ii)
- the use of temperatures above 60 °C in the reactions with the aforementioned metals also led to the formation of microcrystalline powders;
- (iii)
- with the use of less than ca. 6 mL of water in the systems mixed crystals of the new materials and the parent H2atrc ligand were easily produced;
- (iv)
- with 30 min of stirring, the clear mixed solutions become cloudy and no pure single crystals were produced.
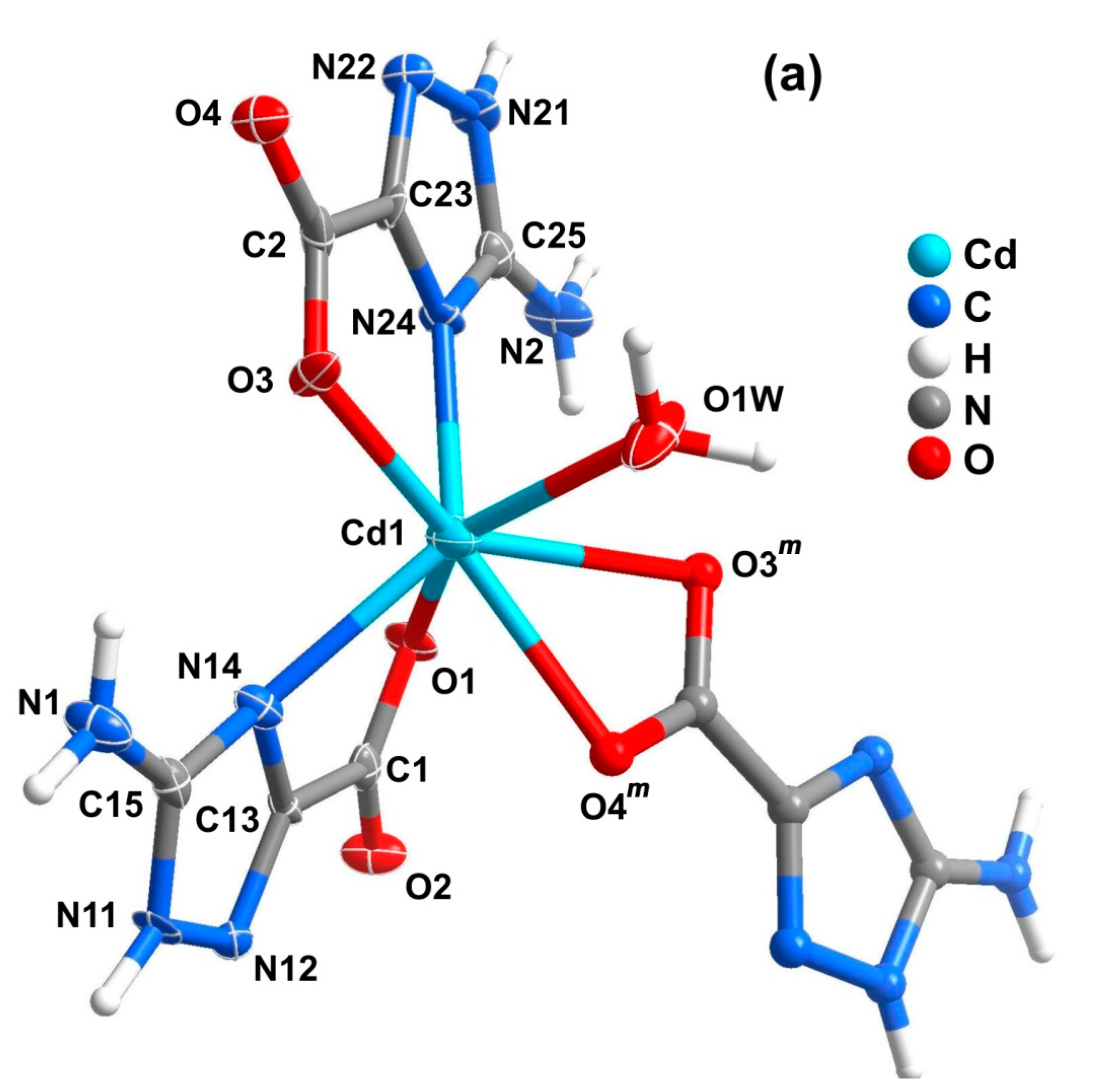
2.2. Mononuclear Complexes

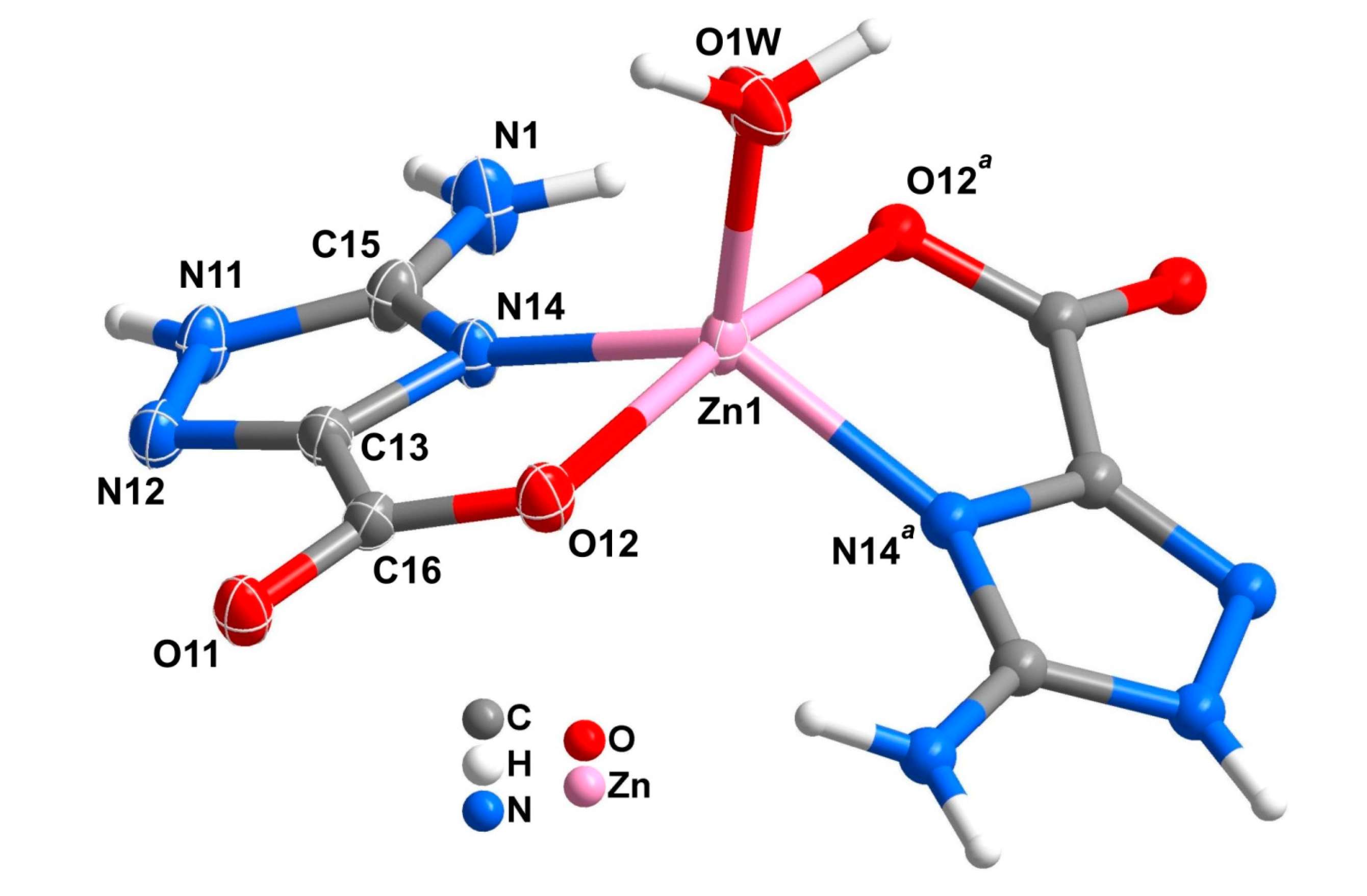
| 1 | 2 | Ref [34] | |||
|---|---|---|---|---|---|
| Zn1–O1W | 1.975(5) | Mn1–O1W | 2.206(2) | Cd1–O3 | 2.384(2) |
| Zn1–N14 | 1.981(3) | Mn1–N14 | 2.239(3) | Cd1–N1 | 2.255(3) |
| Zn1–O12 | 2.171(3) | Mn1–O1 | 2.186(2) | Cd1–O1 | 2.345(2) |
| N14–Zn1–O12 | 79.57(12) | O1–Mn1–N14 | 76.23(8) | O1–Cd1–N1 | 73.14(5) |
| O12–Zn1–O12 a | 168.94(17) | O1–Mn1–O1 b | 180.0 | ||
| O1W–Zn1–O12 | 95.53(9) | O1–Mn1–O1W | 87.38(8) | ||
| N14–Zn1–N14 a | 131.5(2) | N14–Mn1–N14 b | 180.0 | ||
| O1W–Zn1–N14 | 114.25(11) | O1W–Mn1–N14 | 90.44(8) | ||
| O1–Mn1–N14 b | 103.77(8) | ||||
| O1–Mn1–O1W b | 92.62(8) | ||||
| O1W–Mn1–N14 b | 89.56(9) | ||||
| O1W–Mn1–O1W b | 180.0 |
| D–H···A | D···A/Å | <DHA/° | |
|---|---|---|---|
| 1 | N11–H11A···O11 c | 2.749(4) | 171 |
| N1–H1A···O12 a | 3.191(5) | 141 | |
| N1–H1A···O1W d | 3.322(4) | 144 | |
| N1–H1A···N12 c | 2.964(5) | 167 | |
| O1W–H1WA···O11 e | 2.781(3) | 158 | |
| O1W–H1WA···O12 e | 3.031(3) | 131 | |
| 2 | N11–H11A···O2W f | 2.820(3) | 142 |
| N12–H12A···O2W g | 2.957(5) | 116 | |
| N1–H1A···O1W h | 2.957(4) | 173 | |
| N1–H1B···O2W f | 3.115(4) | 142 | |
| N1–H1B···O2W h | 3.141(4) | 128 | |
| O1W–H1WA···O1 i | 2.689(3) | 160 | |
| O1W–H1WB···O2 j | 2.639(3) | 162 | |
| O2W–H2WB···O2 j | 2.757(3) | 175 | |
| O2W–H2WA···N12 k | 2.959(3) | 152 |

| 3 | 4 | 5 | |||
|---|---|---|---|---|---|
| Fe1–O5 | 1.949(2) | Cd1–N24 | 2.239(5) | Mn1–N12 n | 2.192(2) |
| Fe1–O5 l | 1.979(2) | Cd1–N14 | 2.299(5) | Mn1–O1W | 2.194(2) |
| Fe1–O4 | 2.030(2) | Cd1–O3 | 2.344(5) | Mn1–O1 | 2.2179(19) |
| Fe1–O1 | 2.041(3) | Cd1–O4 m | 2.397(4) | Mn1–O2 o | 2.2531(19) |
| Fe1–N14 | 2.096(3) | Cd1–O1W | 2.407(5) | Mn1–N11 o | 2.255(2) |
| Fe1–N24 | 2.113(3) | Cd1–O2 | 2.434(4) | Mn1–N14 | 2.299(2) |
| Cd1–O3 m | 2.593(4) | ||||
| O5–Fe1–O5 l | 76.37(10) | N24–Cd1–N14 | 125.39(19) | N12 n–Mn1–O1W | 88.23(8) |
| O5–Fe1–O4 | 165.65(10) | N24–Cd1–O3 | 73.44(17) | N12 n–Mn1–O1 | 100.41(8) |
| O5–Fe1–O1 | 92.88(10) | N24–Cd1–O4 m | 140.30(17) | N12 n–Mn1–O2 o | 168.57(8) |
| O5–Fe1–N14 | 97.93(10) | N24–Cd1–O1W | 88.15(19) | N12 n–Mn1–N11 o | 97.87(8) |
| O5–Fe1–N24 | 97.79(10) | N24–Cd1–O2 | 87.70(17) | N12 n–Mn1–N14 | 94.18(8) |
| O5 l–Fe1–O4 | 90.56(10) | N24–Cd1–O3 m | 87.73(17) | O1W–Mn1–O1 | 94.82(7) |
| O5 l–Fe1–O1 | 164.41(10) | N14–Cd1–O3 | 88.21(18) | O1W–Mn1–O2 o | 83.68(7) |
| O5 l–Fe1–N14 | 92.25(11) | N14–Cd1–O4 m | 85.21(17) | O1W–Mn1–N11 o | 98.93(8) |
| O5 l–Fe1–N24 | 95.89(11) | N14–Cd1–O1W | 137.85(19) | O1W–Mn1–N14 | 170.92(9) |
| O4–Fe1–O1 | 101.08(10) | N14–Cd1–O2 | 70.68(17) | O1–Mn1–O2 o | 88.34(7) |
| O4–Fe1–N14 | 88.36(10) | N14–Cd1–O3 m | 127.60(17) | O1–Mn1–N11 o | 157.40(8) |
| O4–Fe1–N24 | 77.40(10) | O3–Cd1–O4 m | 138.78(15) | O1–Mn1–N14 | 76.15(8) |
| O1–Fe1–N14 | 77.89(11) | O3–Cd1–O1W | 77.05(17) | O2 o–Mn1–N11 o | 75.53(8) |
| O1–Fe1–N24 | 96.75(11) | O3–Cd1–O2 | 135.96(16) | O2 o–Mn1–N14 | 95.06(8) |
| N14–Fe1–N24 | 163.63(11) | O3–Cd1–O3 m | 143.57(3) | N11 o–Mn1–N14 | 89.42(8) |
| O4 m–Cd1–O1W | 80.97(16) | ||||
| O4 m–Cd1–O2 | 79.08(15) | ||||
| O4 m–Cd1–O3 m | 52.60(14) | ||||
| O1W–Cd1–O2 | 143.05(16) | ||||
| O1W–Cd1–O3 m | 71.32(16) | ||||
| O2–Cd1–O3 m | 71.83(15) |
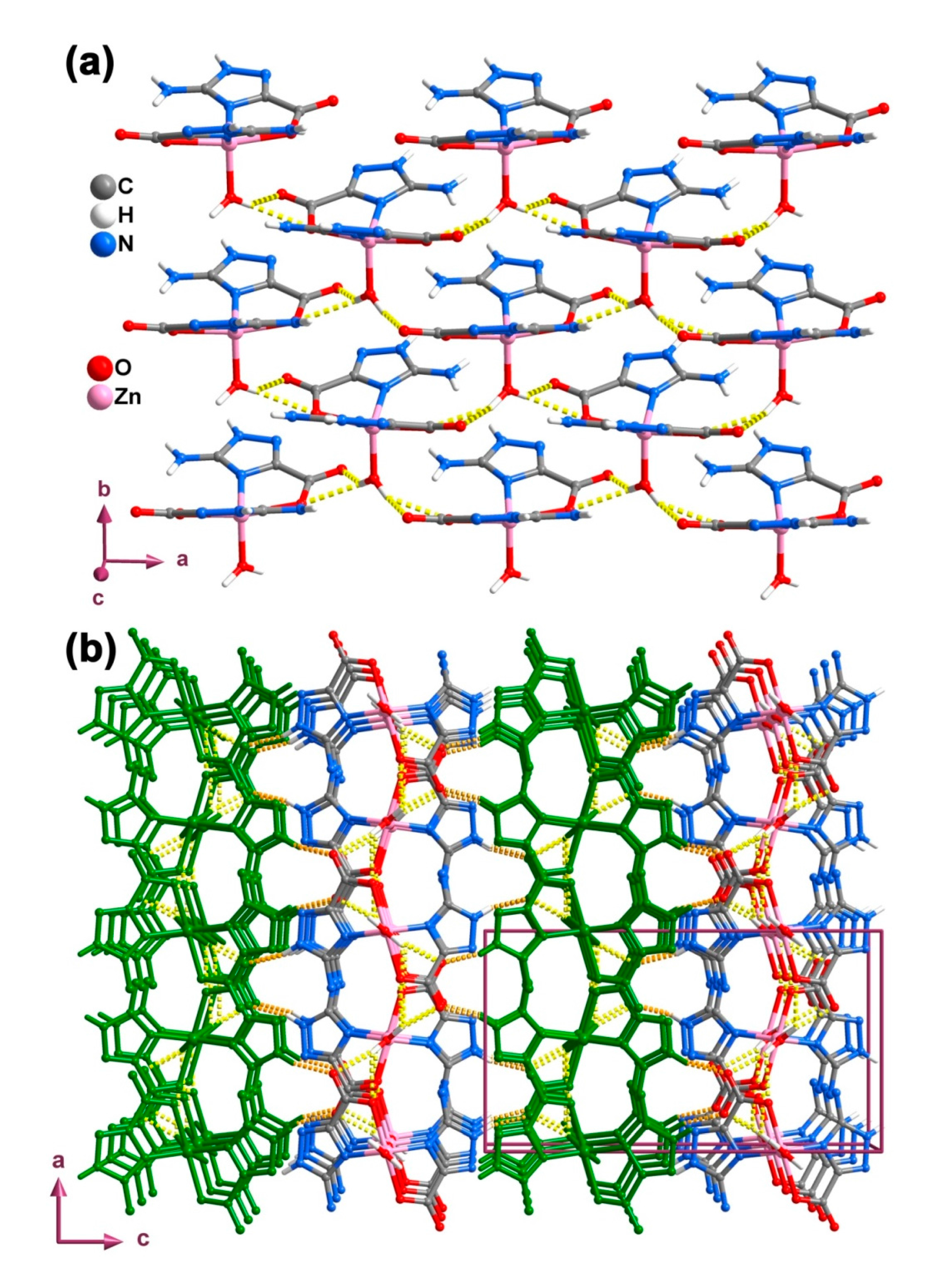
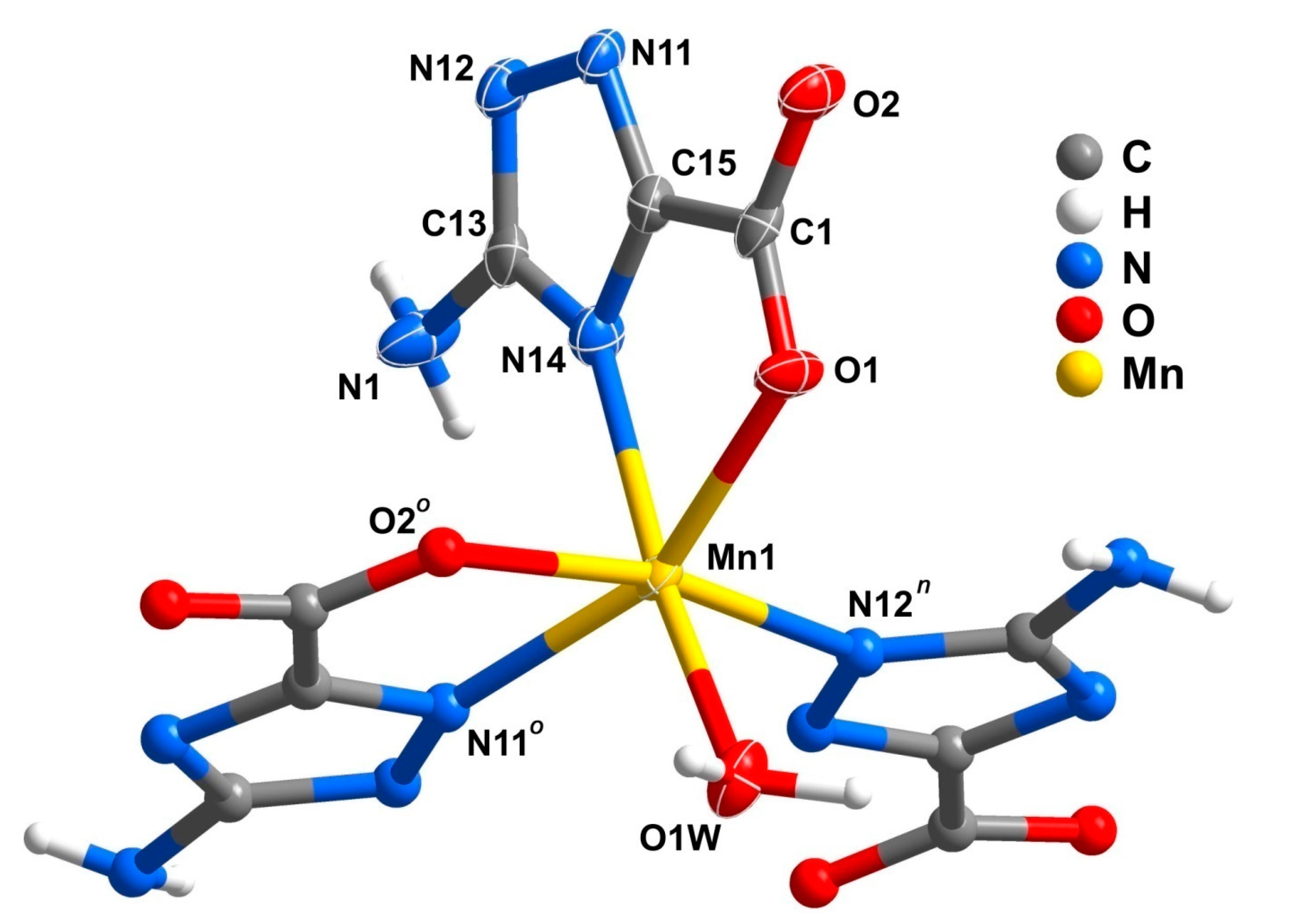
2.3. Binuclear Complex

| D–H···A | D···A/Å | <DHA/° | D–H···A | D···A/Å | <DHA/° |
|---|---|---|---|---|---|
| N11–H11A···O2W | 2.746(4) | 171 | O1W–H1WA···O3Wu | 2.847(4) | 174 |
| N1–H1A···O3W p | 2.905(4) | 151 | O1W–H1WB···N22t | 2.914(4) | 179 |
| N1–H1B···O2 q | 3.023(4) | 173 | O2W–H2WA···O1Wv | 2.678(4) | 154 |
| N21–H21A···O2W r | 2.903(4) | 135 | O2W–H2WB···O2 q | 2.842(4) | 145 |
| N21–H21A···O1 s | 2.866(4) | 128 | O3W–H3WA···O4 | 2.719(3) | 166 |
| N2–H2A···O3 t | 2.914(4) | 163 | O3W–3WB···N12 k | 2.830(4) | 175 |
| N2–H2B···O1W | 2.872(4) | 154 | O5–H5A···O3 t | 2.829(3) | 159 |

2.4. One-Dimensional Chain (1D)
| D–H···A | D···A/Å | <DHA/° | D–H···A | D···A/Å | <DHA/° | ||
|---|---|---|---|---|---|---|---|
| 4 | N11–H11A···O4 w | 2.769(7) | 169 | 5 | N1–H1A···O A | 2.966(3) | 156 |
| N1–H1A···O1W x | 3.200(8) | 162 | N1–H1B···O1W f | 3.224(3) | 151 | ||
| N1–H1B···N22 w | 3.211(8) | 153 | O1W–H1WA···O2 b | 2.748(3) | 161 | ||
| N21–H21A···O1 y | 2.684(6) | 164 | O1W–H1WB···O2W B | 2.714(3) | 152 | ||
| N2–H2A···O1W m | 3.064(8) | 155 | O2W–H2WB···N1 | 2.872(3) | 160 | ||
| N2–H2B···N12 y | 3.037(8) | 158 | O2W–H2WA···N14 C | 3.186(3) | 138 | ||
| O1W–H1WB···O1 z | 2.747(7) | 165 | O2W–H2WA···N12 D | 3.183(3) | 129 | ||
| O1W–H1WA···O2 x | 2.698(7) | 158 | O2W–H2WA···N11 D | 3.159 (3) | 121 |

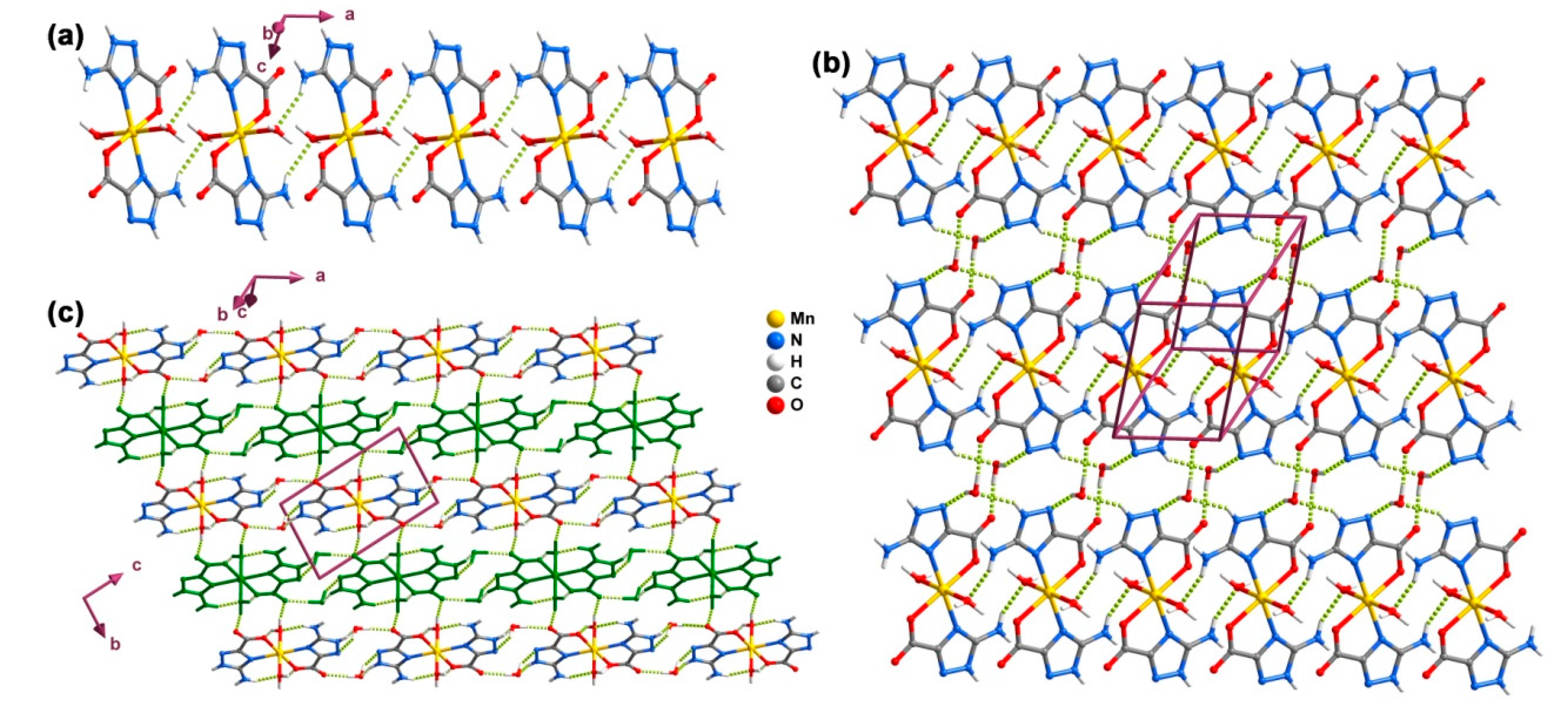
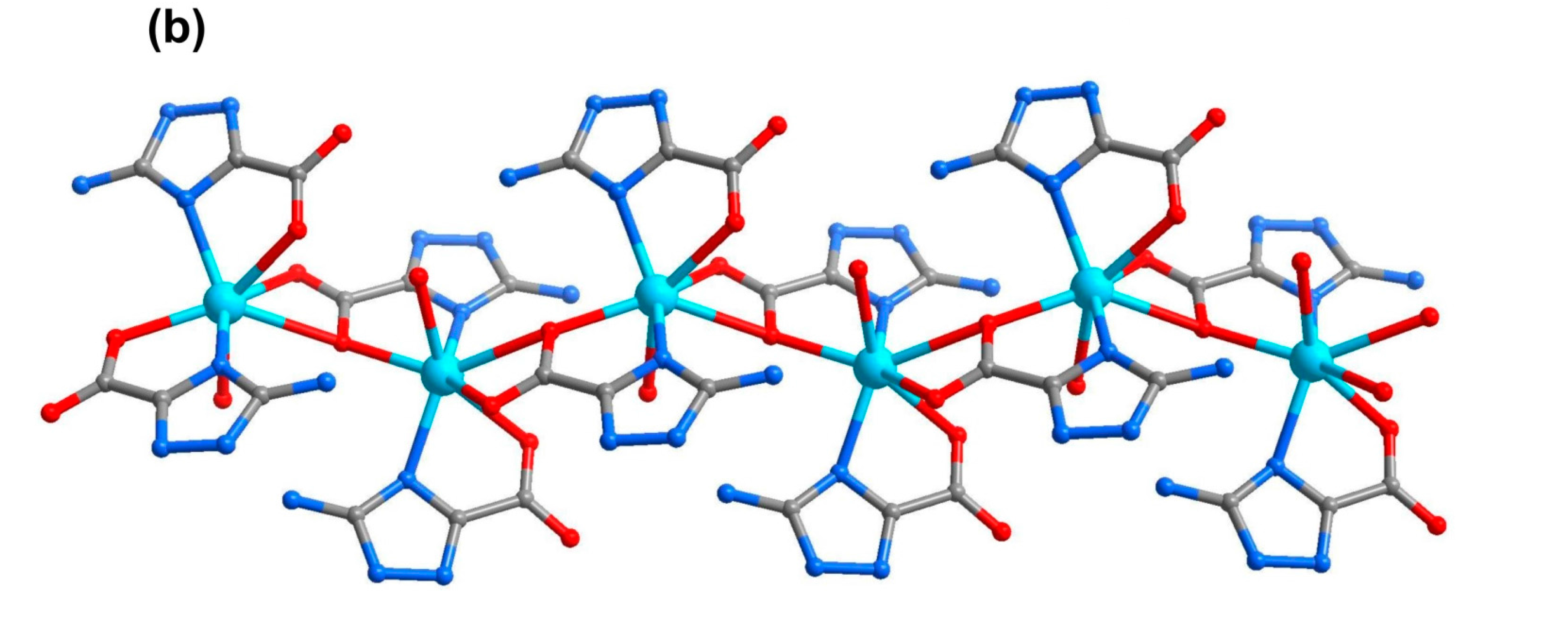
2.5. Coordination Layer (2D)
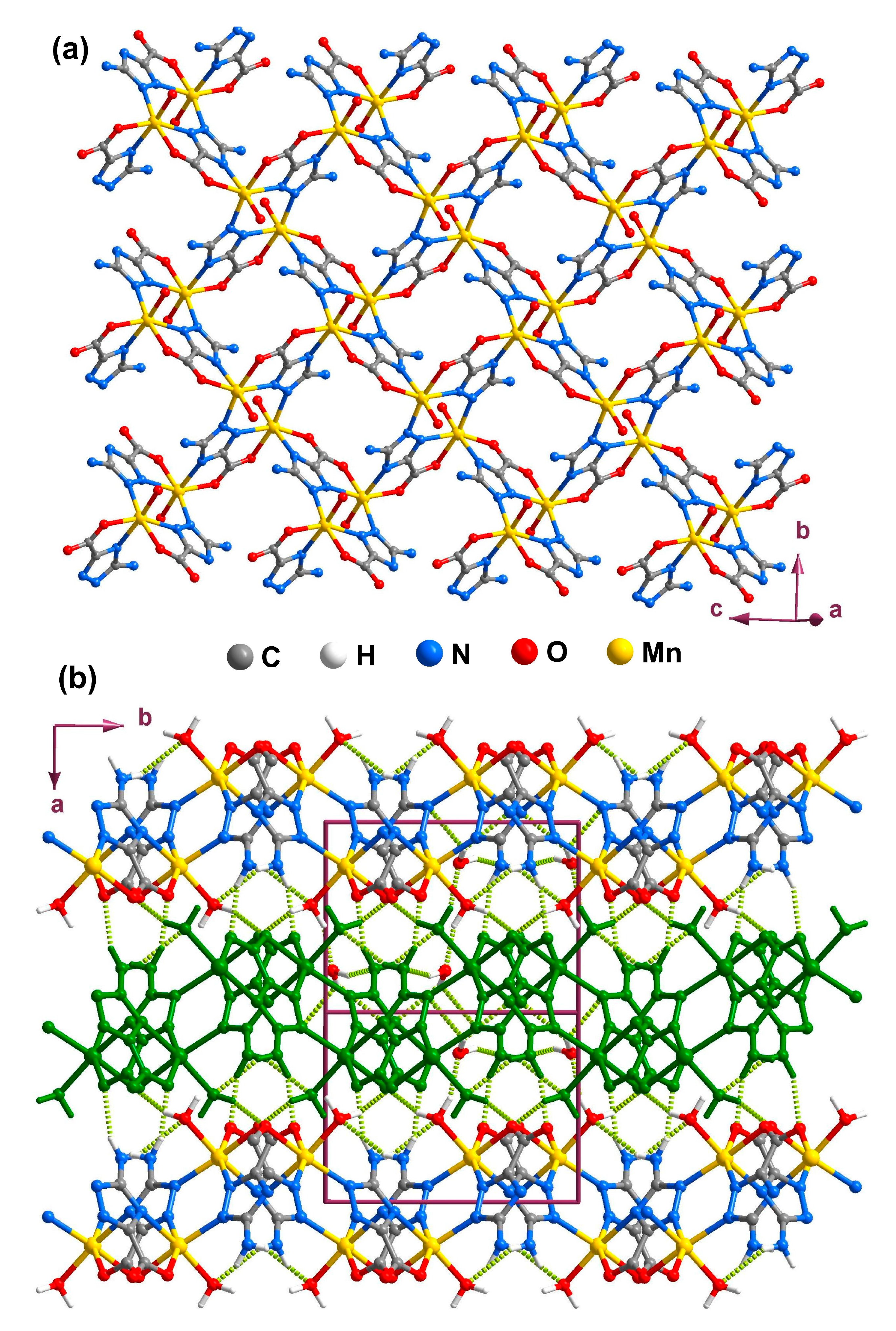
3. Experimental Section
3.1. Materials and Methods
3.2. Synthesis
3.3. Single Crystal X-ray Diffraction
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| Empirical formula | C6H8N8O5Zn | C6H14MnN8O8 | C12H26Fe2N16O16 | C6H8CdN8O5 | C3H6MnN4O4 |
| Color and Habit | Colorless Plate | Colorless block | Orange block | Colorless block | Colorless block |
| Crystal Size (mm3) | 0.42 × 0.21 × 0.07 | 0.08 × 0.04 × 0.03 | 0.07 × 0.02 × 0.02 | 0.07 × 0.03 × 0.04 | 0.12 × 0.11 × 0.05 |
| Crystal system | Orthorhombic | Triclinic | Orthorhombic | Monoclinic | Monoclinic |
| Space group | Pbcn | Pī | Pnna | P21/n | P21/c |
| a (Ǻ) | 9.537(3) | 5.2482(9) | 27.111(6) | 8.9893(13) | 8.1472(6) |
| b Ǻ | 6.865(2) | 6.5330(11) | 12.697(3) | 8.9785(13) | 9.9846(7) |
| c (Ǻ) | 17.120(5) | 10.6128(18) | 7.7352(17) | 13.9642(16) | 9.3679(6) |
| α (°) | 90 | 90.532(10) | 90 | 90 | 90 |
| β (°) | 90 | 102.774(11) | 90 | 108.117(5) | 113.994(4) |
| γ (°) | 90 | 109.139(10) | 90 | 90 | 90 |
| V (Ǻ3) | 1120.8(6) | 333.93(10) | 2662.6(11) | 1071.2(3) | 696.20(8) |
| Z | 4 | 2 | 4 | 4 | 4 |
| Dc (Mg m−3) | 2.000 | 1.896 | 1.901 | 2.385 | 2.071 |
| μ (mm−1) | 2.232 | 1.052 | 1.197 | 2.081 | 1.877 |
| F (000) | 680 | 195 | 1560 | 752 | 436 |
| θ (°) | 3.66 to 26.37 | 4.01 to 29.46 | 3.41 to 25.35 | 3.07 to 25.02 | 2.74 to 26.37 |
| Data completeness | |||||
| Reflections measured | 8019 | 5074 | 12255 | 13793 | 7382 |
| Independent reflections | 1146 (Rint = 0.0331) | 1055 (Rint = 0.0411) | 2418 (Rint = 0.0652) | 1878 (Rint = 0.0512) | 1418(Rint = 0.1497) |
| Final R1, wR2 [I > 2σ(I)] | 0.0393, 0.0867 | 0.0514, 0.0828 | 0.0436, 0.0807 | 0.0432, 0.1128 | 0.0388, 0.0896 |
| R1, wR2 (all data) | 0.0568, 0.0944 | 0.0603, 0.0857 | 0.087, 0.0925 | 0.0481, 0.1150 | 0.0474, 0.0935 |
| Δρmax/min (eǺ−3) | 0.389, −0.555 | 0.384, −0.487 | 0.364, −0.433 | 0.729, −0.974 | 0.555, −0.569 |
| Δρmax/(eǺ−3) | Distance/Ǻ | from | Δρmin/(eǺ−3) | Distance/Ǻ | from | |
|---|---|---|---|---|---|---|
| 1 | 0.389 | 0.85 | O1W | −0.555 | 0.70 | Zn1 |
| 2 | 0.384 | 1.17 | Mn1 | −0.487 | 0.61 | Mn1 |
| 3 | 0.364 | 0.52 | N24 | −0.433 | 0.64 | Fe1 |
| 4 | 0.729 | 0.63 | O3 | −0.974 | 1.29 | H1A |
| 5 | 0.555 | 0.90 | Mn1 | −0.569 | 0.96 | Mn1 |
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eddaoudi, M.; Li, H.L.; Yaghi, O.M. Highly porous and stable metal-organic frameworks: Structure design and sorption properties. J. Am. Chem. Soc. 2000, 122, 1391–1397. [Google Scholar] [CrossRef]
- James, S.L. Metal-organic frameworks. Chem. Soc. Rev. 2003, 32, 276–288. [Google Scholar] [CrossRef] [PubMed]
- Kaczorowski, T.; Justyniak, I.; Lipinska, T.; Lipkowski, J.; Lewinski, J. Metal Complexes of Cinchonine as Chiral Building Blocks: A Strategy for the Construction of Nanotubular Architectures and Helical Coordination Polymers. J. Am. Chem. Soc. 2009, 131, 5393–5395. [Google Scholar] [CrossRef] [PubMed]
- Maspoch, D.; Ruiz-Molina, D.; Veciana, J. Old materials with new tricks: Multifunctional open-framework materials. Chem. Soc. Rev. 2007, 36, 770–818. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.; Harms, K.; Dehnen, S. New Three-Dimensional Metal-Organic Framework with Heterometallic Fe-Ag Building Units: Synthesis, Crystal Structure, and Functional Studies. Inorg. Chem. 2011, 50, 2714–2716. [Google Scholar] [CrossRef] [PubMed]
- Robin, A.Y.; Fromm, K.M. Coordination polymer networks with O- and N-donors: What they are, why and how they are made. Coord. Chem. Rev. 2006, 250, 2127–2157. [Google Scholar] [CrossRef]
- Zhao, Y.G.; Wu, H.H.; Emge, T.J.; Gong, Q.H.; Nijem, N.; Chabal, Y.J.; Kong, L.Z.; Langreth, D.C.; Liu, H.; Zeng, H.P.; et al. Enhancing Gas Adsorption and Separation Capacity through Ligand Functionalization of Microporous Metal-Organic Framework Structures. Chem. Eur. J. 2011, 17, 5100–5108. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.M.; Zhang, J.; Wu, T.; Feng, P.Y.; Bu, X.H. Multiroute Synthesis of Porous Anionic Frameworks and Size-Tunable Extraframework Organic Cation-Controlled Gas Sorption Properties. J. Am. Chem. Soc. 2009, 131, 16027–16029. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, R.; Kitaura, R.; Kitagawa, S.; Kubota, Y.; Belosludov, R.V.; Kobayashi, T.C.; Sakamoto, H.; Chiba, T.; Takata, M.; Kawazoe, Y.; et al. Highly controlled acetylene accommodation in a metal-organic microporous material. Nature 2005, 436, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Moulton, B.; Zaworotko, M.J. From molecules to crystal engineering: Supramolecular isomerism and polymorphism in network solids. Chem. Rev. 2001, 101, 1629–1658. [Google Scholar] [CrossRef] [PubMed]
- Noro, S.; Kitaura, R.; Kondo, M.; Kitagawa, S.; Ishii, T.; Matsuzaka, H.; Yamashita, M. Framework engineering by anions and porous functionalities of Cu(II)/4,4′-bpy coordination polymers. J. Am. Chem. Soc. 2002, 124, 2568–2583. [Google Scholar] [CrossRef] [PubMed]
- Su, C.Y.; Cai, Y.P.; Chen, C.L.; Smith, M.D.; Kaim, W.; zur Loye, H.C. Ligand-directed molecular architectures: Self-assembly of two-dimensional rectangular metallacycles and three-dimensional trigonal or tetragonal prisms. J. Am. Chem. Soc. 2003, 125, 8595–8613. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Tominaga, M.; Hori, A.; Therrien, B. Coordination assemblies from a Pd(II)-cornered square complex. Acc. Chem. Res. 2005, 38, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Leininger, S.; Olenyuk, B.; Stang, P.J. Self-assembly of discrete cyclic nanostructures mediated by transition metals. Chem. Rev. 2000, 100, 853–907. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, L.; Ciani, G.; Proserpio, D.M. Polycatenation, polythreading and polyknotting in coordination network chemistry. Coord. Chem. Rev. 2003, 246, 247–289. [Google Scholar] [CrossRef]
- Pitt, M.A.; Johnson, D.W. Main group supramolecular chemistry. Chem. Soc. Rev. 2007, 36, 1441–1453. [Google Scholar] [CrossRef] [PubMed]
- Andraud, C.; Maury, O. Lanthanide Complexes for Nonlinear Optics: From Fundamental Aspects to Applications. Eur. J. Inorg. Chem. 2009, 2009, 4357–4371. [Google Scholar] [CrossRef]
- Bauer, C.A.; Timofeeva, T.V.; Settersten, T.B.; Patterson, B.D.; Liu, V.H.; Simmons, B.A.; Allendorf, M.D. Influence of connectivity and porosity on ligand-based luminescence in zinc metal-organic frameworks. J. Am. Chem. Soc. 2007, 129, 7136–7144. [Google Scholar] [CrossRef] [PubMed]
- Deplano, P.; Pilia, L.; Espa, D.; Mercuri, M.L.; Serpe, A. Square-planar d8 metal mixed-ligand dithiolene complexes as second order nonlinear optical chromophores: Structure/property relationship. Coord. Chem. Rev. 2010, 254, 1434–1447. [Google Scholar] [CrossRef]
- Kuang, G.C.; Ji, Y.; Jia, X.R.; Chen, E.Q.; Gao, M.; Yeh, J.M.; Wei, Y. Supramolecular Self-Assembly of Dimeric Dendrons with Different Aliphatic Spacers. Chem. Mater. 2009, 21, 456–462. [Google Scholar] [CrossRef]
- Zhang, J.J.; Wojtas, L.; Larsen, R.W.; Eddaoudi, M.; Zaworotko, M.J. Temperature and Concentration Control over Interpenetration in a Metal-Organic Material. J. Am. Chem. Soc. 2009, 131, 17040–17041. [Google Scholar] [CrossRef] [PubMed]
- Aromi, G.; Barrios, L.A.; Roubeau, O.; Gamez, P. Triazoles and tetrazoles: Prime ligands to generate remarkable coordination materials. Coord. Chem. Rev. 2011, 255, 485–546. [Google Scholar] [CrossRef]
- Dias, H.V.R.; Singh, S.; Campana, C.F. Toluene-sandwiched trinuclear copper(I) and silver(I) triazolates and phosphine adducts of dinuclear copper(I) and silver(I) triazolates. Inorg. Chem. 2008, 47, 3943–3945. [Google Scholar] [CrossRef] [PubMed]
- Fillat, M.F.; Gimeno, M.C.; Laguna, A.; Latorre, E.; Ortego, L.; Villacampa, M.D. Synthesis, Structure and Bactericide Activity of (Aminophosphane)gold(I) Thiolate Complexes. Eur. J. Inorg. Chem. 2011, 1487–1495. [Google Scholar] [CrossRef]
- Kitchen, J.A.; Brooker, S. Spin crossover in iron(II) complexes of 3,5-di(2-pyridyl)-1,2,4-triazoles and 3,5-di(2-pyridyl)-1,2,4-triazolates. Coord. Chem. Rev. 2008, 252, 2072–2092. [Google Scholar] [CrossRef]
- Kitchen, J.A.; White, N.G.; Jameson, G.N.L.; Tallon, J.L.; Brooker, S. Effect of Counteranion X on the Spin Crossover Properties of a Family of Diiron(II) Triazole Complexes Fe2II(PMAT)2X4. Inorg. Chem. 2011, 50, 4586–4597. [Google Scholar] [CrossRef] [PubMed]
- Li, B.Y.; Jin, D.; Ma, B.H.; Liu, D.; Li, G.H.; Shi, Z.; Feng, S.H. Two Coordination Polymers with Rare Topologies Based on Copper(II) and Ligands Generated by in Situ Reactions. Eur. J. Inorg. Chem. 2011, 2011, 35–38. [Google Scholar] [CrossRef]
- Riederer, S.K.U.; Bechlars, B.; Herrmann, W.A.; Kühn, F.E. Synthesis and Structural Characterization of New Chiral (Biscarbene)platinum(II) Complexes. Eur. J. Inorg. Chem. 2011, 2011, 249–254. [Google Scholar] [CrossRef]
- Tekarli, S.M.; Cundari, T.R.; Omary, M.A. Rational design of macrometallocyclic trinuclear complexes with superior π-acidity and π-basicity. J. Am. Chem. Soc. 2008, 130, 1669–1675. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.T.; Lu, Y.Z.; Chen, W.; Chen, S.W. One-Pot Synthesis, Photoluminescence, and Electrocatalytic Properties of Subnanometer-Sized Copper Clusters. J. Am. Chem. Soc. 2011, 133, 2060–2063. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Maruta, G.; Takeda, S. Reversible solid-state structural conversion between a three-dimensional network and a one-dimensional chain of Cu(II) triazole coordination polymers in acidic/basic-suspensions or vapors. Chem. Commun. 2011, 47, 653–655. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.A.; Liu, B.; Tomé, J.P.C.; Cunha-Silva, L.; Paz, F.A.A. 5-Amino-1H-1,2,4-triazol-4-ium-3-carboxylate hemihydrate. Acta Cryst. E 2011, 67, O2073–U2868. [Google Scholar] [CrossRef] [PubMed]
- Allen, F.H. The Cambridge Structural Database: A quarter of a million crystal structures and rising. Acta Cryst. B 2002, 58, 380–388. [Google Scholar] [CrossRef]
- Wang, J.; Li, W.Z.; Wang, J.G.; Xiao, H.P. Crystal structure of diaquabis(3-amino-1,2,4-triazole-5-carboxylato)-cadmium(II), Cd(H2O)2(C3H3N4O2)2. Z. Krist. New Cryst. Struct. 2011, 226, 163–164. [Google Scholar] [CrossRef]
- Siddiqui, K.A.; Mehrotra, G.K.; Narvi, S.S.; Butcher, R.J. Molecular self-assembly of cadmium-triazolate complexes via hydrogen bonding: Synthesis, structures and photoluminescent properties. Inorg. Chem. Commun. 2011, 14, 814–817. [Google Scholar] [CrossRef]
- Chen, Y.C.; Xu, J.J.; Wang, K.B.; Wang, Y. Syntheses and characterization of two new alkaline earth metal-organic topological frameworks with 3-amino-1H-1,2,4-triazole-5-carboxylate. Chin. J. Struct. Chem. 2011, 30, 799–804. [Google Scholar]
- Tseng, T.W.; Luo, T.T.; Lu, K.H. Impeller-like dodecameric water clusters in metal-organic nanotubes. CrystEngComm 2014, 16, 5516–5519. [Google Scholar] [CrossRef]
- Shi, F.N.; Cunha-Silva, L.; Hardie, M.J.; Trindade, T.; Paz, F.A.A.; Rocha, J. Heterodimetallic germanium(IV) complex structures with transition metals. Inorg. Chem. 2007, 46, 6502–6515. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.N.; Cunha-Silva, L.; Paz, F.A.A.; Hardie, M.J.; Klinowski, J.; Rocha, J.; Trindade, T. A novel germanium(IV) oxalate complex: Ge(OH)2(C2O4)22−. Inorg. Chem. Commun. 2008, 11, 283–287. [Google Scholar] [CrossRef]
- Cunha-Silva, L.; Shi, F.N.; Paz, F.A.A.; Hardie, M.J.; Klinowski, J.; Trindade, T.; Rocha, J. Supramolecular salts containing the anionic Ge(C2O4)32− complex and heteroaromatic amines. Inorg. Chim. Acta 2009, 362, 263–270. [Google Scholar] [CrossRef]
- Neves, P.; Gago, S.; Balula, S.S.; Lopes, A.D.; Valente, A.A.; Cunha-Silva, L.; Almeida Paz, F.A.; Pillinger, M.; Rocha, J.; Silva, C.M.; et al. Synthesis and Catalytic Properties of Molybdenum(VI) Complexes with Tris(3,5-dimethyl-1-pyrazolyl)methane. Inorg. Chem. 2011, 50, 3490–3500. [Google Scholar] [CrossRef] [PubMed]
- Ananias, D.; Kostova, M.; Paz, F.A.A.; Ferreira, A.; Carlos, L.D.; Klinowski, J.; Rocha, J. Photoluminescent layered lanthanide silicates. J. Am. Chem. Soc. 2004, 126, 10410–10417. [Google Scholar] [CrossRef] [PubMed]
- Klinowski, J.; Paz, F.A.A.; Silva, P.; Rocha, J. Microwave-Assisted Synthesis of Metal-Organic Frameworks. Dalton Trans. 2011, 40, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.; Carlos, L.D.; Paz, F.A.A.; Ananias, D. Luminescent multifunctional lanthanides-based metal-organic frameworks. Chem. Soc. Rev. 2011, 40, 926–940. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.; Paz, F.A.A.; Shi, F.N.; Ananias, D.; Silva, N.J.O.; Carlos, L.D.; Trindade, T. Mixed-Metal d-f Phosphonate Frameworks—Photoluminescence and Magnetic Properties. Eur. J. Inorg. Chem. 2011, 2011, 2035–2044. [Google Scholar] [CrossRef]
- Shi, F.N.; Cunha-Silva, L.; Ferreira, R.A.S.; Mafra, L.; Trindade, T.; Carlos, L.D.; Paz, F.A.A.; Rocha, J. Interconvertable modular framework and layered lanthanide(III)-etidronic acid coordination polymers. J. Am. Chem. Soc. 2008, 130, 150–167. [Google Scholar] [CrossRef] [PubMed]
- Soares-Santos, P.C.R.; Cunha-Silva, L.; Sousa, F.L.; Sousa, J.L.C.; Gates, P.J.; Klinowski, J.; Trindade, T.; Rocha, J.; Cavaleiro, A.M.V.; Paz, F.A.A.; et al. A new supramolecular organic-inorganic adduct: {[Eu(CH3OH)(H2O)8]2 [Eu(H2O)8][PW12O40]3}•8(C14H20O5)•2(C28H40O10)•6(CH3OH)•6(H2O). J. Mol. Struct. 2011, 989, 80–85. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; Vanrijn, J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen sulfur donor ligands-The crystal and molecular-structure of aqua 1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane copper(II) perchlorate. Dalton Trans. 1984, 7, 1349–1356. [Google Scholar] [CrossRef]
- Spek, A. Structure validation in chemical crystallography. Acta Cryst. D 2009, 65, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Grell, J.; Bernstein, J.; Tinhofer, G. Graph-set analysis of hydrogen-bond patterns: Some mathematical concept. Acta Cryst. B 1999, 55, 1030–1043. [Google Scholar] [CrossRef]
- Ding, B.; Yi, L.; Wang, Y.; Cheng, P.; Liao, D.Z.; Yan, S.P.; Jiang, Z.H.; Song, H.B.; Wang, H.G. Synthesis of a series of 4-pyridyl-1,2,4-triazole-containing cadmium(II) luminescent complexes. Dalton Trans. 2006, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Habib, H.A.; Hoffmann, A.; Hoeppe, H.A.; Janiak, C. Crystal structures and solid-state CPMAS 13C NMR correlations in luminescent zinc(II) and cadmium(II) mixed-ligand coordination polymers constructed from 1,2-bis(1,2,4-triazol-4-yl)ethane and benzenedicarboxylate. Dalton Trans. 2009, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Li, B.L.; Zhu, X.; Zhou, J.H.; Peng, Y.F.; Zhang, Y. Syntheses and structures of five cadmium(II) coordination polymers from 1,2-bis(1,2,4-triazol-l-yl)ethane. Polyhedron 2004, 23, 3133–3141. [Google Scholar] [CrossRef]
- Kottke, T.; Stalke, D. Crystal handling at low temperatures. J. Appl. Crystallogr. 1993, 26, 615–619. [Google Scholar] [CrossRef]
- APEX2. Data Collection Software Version 2.1-RC13, Bruker AXS: Delft, The Netherlands, 2006.
- Cryopad. Remote Monitoring and Control, Version 1.451, Oxford Cryosystems: Oxford, UK, 2006.
- SAINT+. Data Integration Engine, version 7.23a, Bruker AXS: Madison, WI, USA; 1997–2005.
- Sheldrick, G.M. SADABS v.2.01, Bruker/Siemens Area Detector Absorption Correction Program 1998; Bruker AXS: Madison, WI, USA.
- Sheldrick, G.M. SHELXS-97, Program for Crystal Structure Solution; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. SHELXS-97, Program for Crystal Structure Refinement; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Fernandes, J.A.; Tomé, J.P.C.; Paz, F.A.A.; Cunha-Silva, L. Multidimensional Transition Metal Complexes Based on 3-Amino-1H-1,2,4-triazole-5-carboxylic Acid: From Discrete Mononuclear Complexes to Layered Materials. Molecules 2015, 20, 12341-12363. https://doi.org/10.3390/molecules200712341
Liu B, Fernandes JA, Tomé JPC, Paz FAA, Cunha-Silva L. Multidimensional Transition Metal Complexes Based on 3-Amino-1H-1,2,4-triazole-5-carboxylic Acid: From Discrete Mononuclear Complexes to Layered Materials. Molecules. 2015; 20(7):12341-12363. https://doi.org/10.3390/molecules200712341
Chicago/Turabian StyleLiu, Bing, José A. Fernandes, João P. C. Tomé, Filipe A. Almeida Paz, and Luís Cunha-Silva. 2015. "Multidimensional Transition Metal Complexes Based on 3-Amino-1H-1,2,4-triazole-5-carboxylic Acid: From Discrete Mononuclear Complexes to Layered Materials" Molecules 20, no. 7: 12341-12363. https://doi.org/10.3390/molecules200712341
APA StyleLiu, B., Fernandes, J. A., Tomé, J. P. C., Paz, F. A. A., & Cunha-Silva, L. (2015). Multidimensional Transition Metal Complexes Based on 3-Amino-1H-1,2,4-triazole-5-carboxylic Acid: From Discrete Mononuclear Complexes to Layered Materials. Molecules, 20(7), 12341-12363. https://doi.org/10.3390/molecules200712341









