Inorganic Mercury Sequestration by a Poly(ethylene imine) Dendrimer in Aqueous Solution
Abstract
:1. Introduction
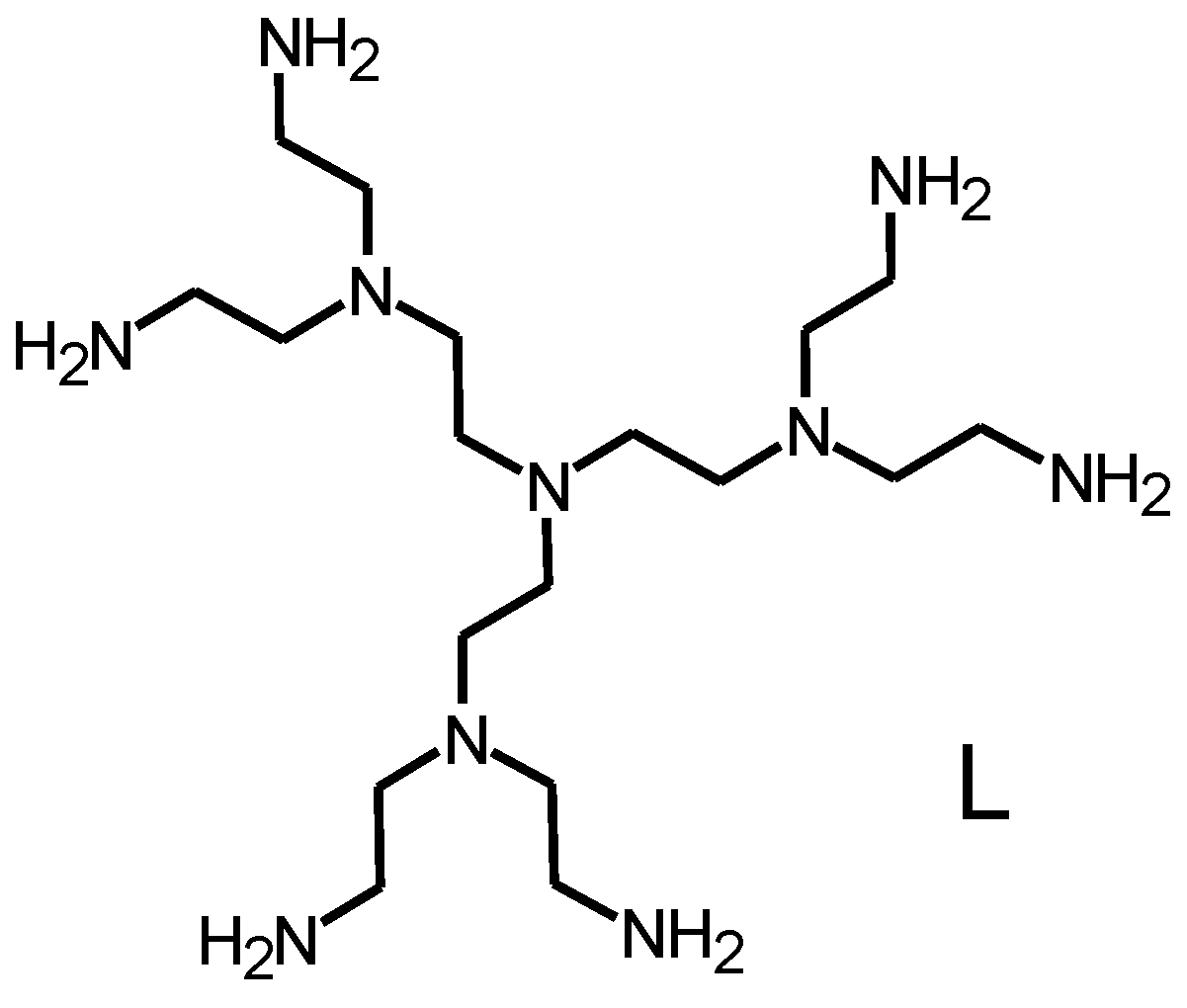
2. Results and Discussion
| Equilibria | logK |
|---|---|
| L + Hg2+ = HgL2+ | 28.17(5) |
| HgL2+ + H+ = HgLH3+ | 9.64(6) |
| HgLH3+ + H+ = HgLH24+ | 8.91(7) |
| HgLH24+ + H+ = HgLH35+ | 8.45(5) |
| HgLH35+ + H+ = HgLH46+ | 6.13(4) |
| HgLH46+ + H+ = HgLH57+ | 4.85(5) |
| L + 2Hg2+ = Hg2L4+ | 48.38(8) |
| Hg2L4+ + H+ = Hg2LH5+ | 9.41(9) |
| Hg2LH5+ + H+ = Hg2LH26+ | 6.63(6) |
| HgL2+ + Hg2+ = Hg2L4+ | 20.21(9) |
| 2L + 3Hg2+ = Hg3L26+ | 79.4(1) |
| Hg3L26+ + H+ = Hg3HL27+ | 10.4(2) |
| Hg3L2H7+ + H+ = Hg3L2H28+ | 9.0(2) |
| Hg2L4+ + HgL2+ = Hg3L26+ | 2.9(2) |
| L + 3Hg2+ = Hg3L6+ | 66.74(5) |
| Hg3L6+ + OH− = Hg3LOH5+ | 4.83(8) |
| Hg2L4+ + Hg2+ = Hg3L6+ | 18.36(8) |
| H7L7+ + HgCl42− = [(H7L)HgCl4]5+ | 2.7(1) |
| H8L8+ + HgCl42− = [(H8L)HgCl4]6+ | 2.8(1) |
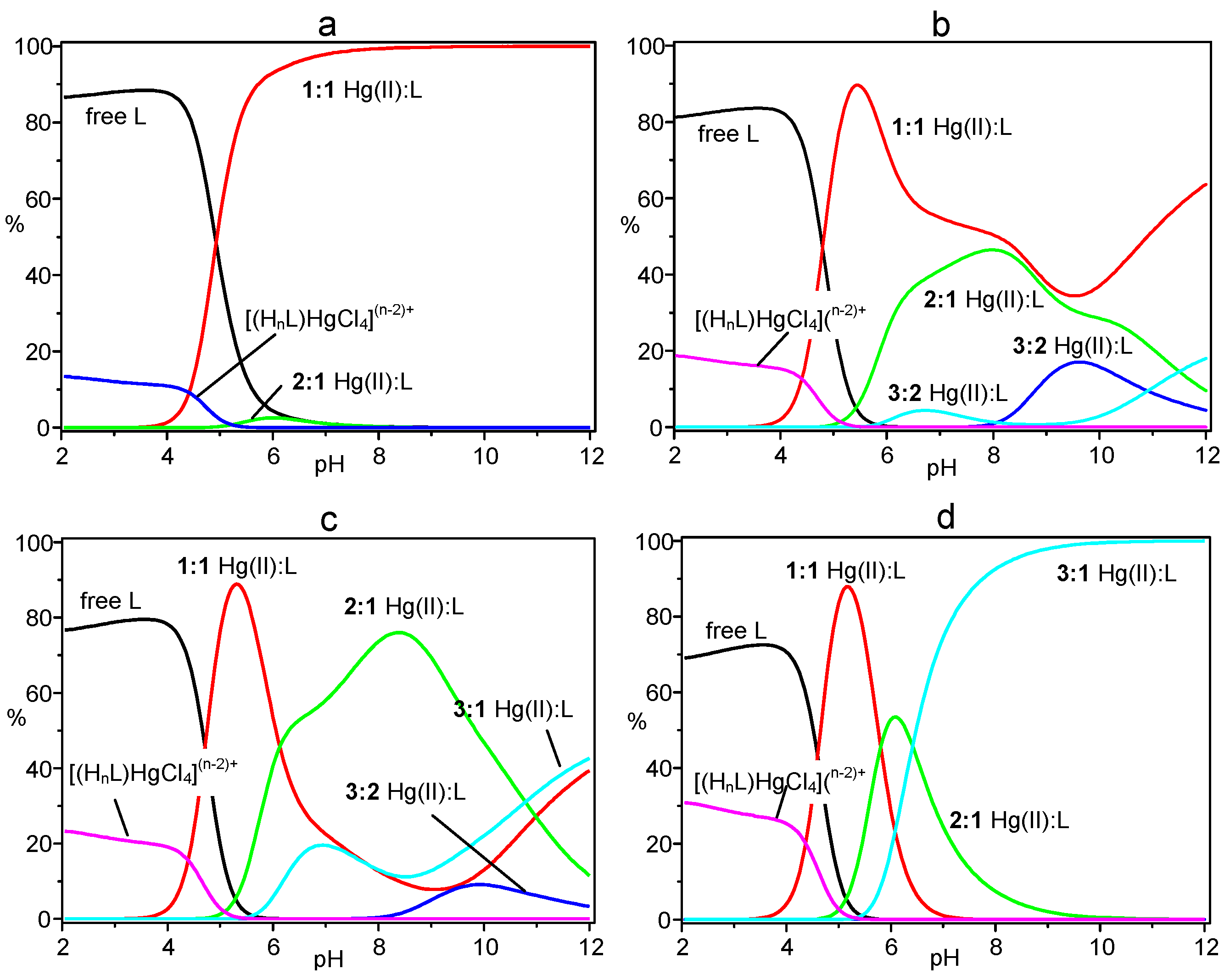
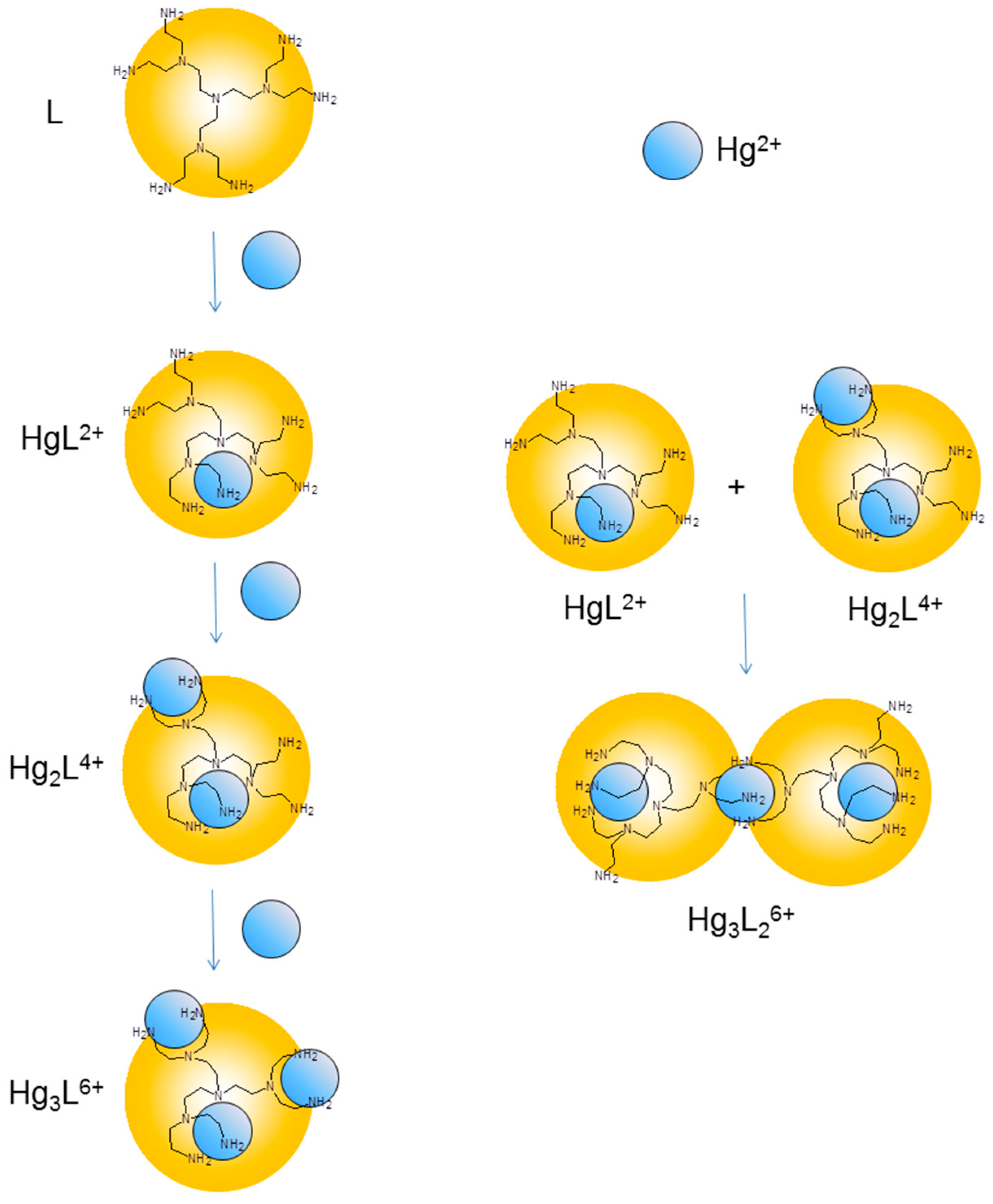
3. Experimental Section
3.1. General
3.2. Potentiometric Measurements
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Merian, E.; Anke, M.; Ihnat, M.; Stoeppler, M. (Eds.) Elements and Their Compounds in the Environment; Wiley-VCH: Weinheim, Germany, 2008; pp. 931–1005.
- Normile, D. In Minamata, Mercury Still Divides. Science 2013, 341, 1446–1447. [Google Scholar] [CrossRef] [PubMed]
- Hosohara, K.; Uezuma, H.K.; Kawasaki, K.; Tsuruta, T. Studies on the total amount of mercury in sea waters. Nippon Kagaku Zasshi 1961, 81, 1479–1480. [Google Scholar] [CrossRef]
- Kumagai, M.; Nishimura, H. Mercury distribution in seawater in Minamata Bay and the origin of particulate mercury. J. Oceanogr. Soc. Jpn. 1978, 34, 50–56. [Google Scholar] [CrossRef]
- Harada, M. Minamata disease: Methylmercury poisoning in Japan caused by environmental pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Gianguzza, A.; Pellizzetti, E.; Sammartano, S. (Eds.) Chemistry of Marine Water and Sediments; Springer: Berlin, Germany, 2002.
- Gianguzza, A.; Pellizzetti, E.; Sammartano, S. (Eds.) Chemical Processes in Marine Environments; Springer: Berlin, Germany, 2000.
- Global Mercury Assessment 2013, United Nations Environment Programme, Geneva, Switzerland. Available online: http://www.unep.org/publications/contents/pub_details_search.asp?ID=6282 (accessed on 23 February 2015).
- López-Garzón, R.; Godino-Salido, M.L.; Gutiérrez-Valero, M.D.; Arranz-Mascarós, P.; Melguizo, M.; García, C.; Domingo-García, M.; López-Garzón, F.J. Supramolecular assembling of molecular ion-ligands on graphite-based solid materials directed to specific binding of metal ions. Inorg. Chim. Acta 2014, 417, 208–221. [Google Scholar] [CrossRef]
- Peñas-Sanjuán, A.; López-Garzón, R.; López-Garzón, J.; Pérez-Mendoza, M.; Melguizo, M. Preparation of a poly-alkylamine surface-functionalized carbon with excellent performance as a Pd(II) scavenger. Carbon 2012, 50, 2350–2352. [Google Scholar] [CrossRef]
- García-Martín, J.; López-Garzón, R.; Godino-Salido, M.L.; Cuests-Martos, R.; Gutiérrez-Valero, M.D.; Arranz-Mascarós, P.; Stoeckli-Evans, H. Adsorption of Zn2+ and Cd2+ from aqueous solution onto a carbon sorbent containing a pyrimidine–polyamine conjugate as ion receptor. Eur. J. Inorg. Chem. 2005, 2005, 3093–3103. [Google Scholar] [CrossRef]
- Arranz, P.; Bianchi, A.; Cuesta, R.; Giorgi, C.; Godino, M.L.; Gutiérrez, M.D.; López, R.; Santiago, A. Binding and removal of sulphate, phosphate, arsenate, tetrachloromercurate, and chromate in aqueous solution by means of an activated carbon functionalized with a pyrimidine-based anion receptor (HL). Crystal structures of [H3L(HgCl4)]∙H2O and [H3L(HgBr4)]∙H2O showing anion-π interactions. Inorg. Chem. 2010, 49, 9321–9332. [Google Scholar]
- Bazzicalupi, C.; Bianchi, A.; Giorgi, C.; Gratteri, P.; Mariani, P.; Valtancoli, B. Metal ion binding by a G-2 poly(ethylene imine) dendrimer. Ion-directed self-assembling of hierearchical mono- and two-dimensional nanostructured materials. Inorg. Chem. 2013, 52, 2125–2137. [Google Scholar]
- Bazzicalupi, C.; Bianchi, A.; Giorgi, C.; Gratteri, P.; Mariani, P.; Valtancoli, B. Anion and ion-pair binding by a G-2 poly(ethylene imine) dendrimer. Dalton Trans. 2013, 42, 12130–12138. [Google Scholar] [CrossRef] [PubMed]
- Bazzicalupi, C.; Bianchi, A.; Giorgi, C.; Valtancoli, B. Zn(II) enhances nucleotide binding and dephosphorylation in the presence of a 2 poly(ethylene imine) dendrimer. Inorg. Chim. Acta 2014, 417, 163–170. [Google Scholar] [CrossRef]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the HYPERQUAD suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar]
- Bianchi, A.; García-España, E. The Use of calculated species distribution diagrams in the analysis of thermodynamic selectivity. J. Chem. Educ. 1999, 76, 1727–1732. [Google Scholar] [CrossRef]
- Bazzicalupi, C.; Bianchi, A.; Giorgi, C.; Clares, M.P.; García-España, E. Addressing selectivity criteria in binding equilibria. Coord. Chem. Rev. 2012, 256, 13–27. [Google Scholar] [CrossRef]
- Smith, R.M.; Martell, A.E. NIST Stability Constants Database, Version 4.0; National Institute of Standards and Technology: Washington, DC, USA, 1997. [Google Scholar]
- Crea, F.; de Stefano, C.; Foti, C.; Milea, D.; Sammartano, S. Chelating agents for the sequestration of mercury(II) and monomethyl mercury(II). Curr. Med. Chem. 2014, 21, 3819–3836. [Google Scholar] [CrossRef] [PubMed]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
- Gran, G. Determination of the equivalence point in potentiometric titration, Part II. Analyst (Lond.) 1952, 77, 661–671. [Google Scholar] [CrossRef]
- Sample Availability: Samples of L∙10HCl are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, E.S.; Savastano, M.; Bianchi, A. Inorganic Mercury Sequestration by a Poly(ethylene imine) Dendrimer in Aqueous Solution. Molecules 2015, 20, 3783-3790. https://doi.org/10.3390/molecules20033783
Serrano ES, Savastano M, Bianchi A. Inorganic Mercury Sequestration by a Poly(ethylene imine) Dendrimer in Aqueous Solution. Molecules. 2015; 20(3):3783-3790. https://doi.org/10.3390/molecules20033783
Chicago/Turabian StyleSerrano, Elena Salvador, Matteo Savastano, and Antonio Bianchi. 2015. "Inorganic Mercury Sequestration by a Poly(ethylene imine) Dendrimer in Aqueous Solution" Molecules 20, no. 3: 3783-3790. https://doi.org/10.3390/molecules20033783
APA StyleSerrano, E. S., Savastano, M., & Bianchi, A. (2015). Inorganic Mercury Sequestration by a Poly(ethylene imine) Dendrimer in Aqueous Solution. Molecules, 20(3), 3783-3790. https://doi.org/10.3390/molecules20033783






