An Efficient Agrobacterium-Mediated Transformation of Strawberry cv. Camarosa by a Dual Plasmid System
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of Different Concentrations of TDZ on Shoot Regeneration of Strawberry Leaves
2.2. Effects of Different Antibiotics on Shoot Regeneration from Strawberry Leaves
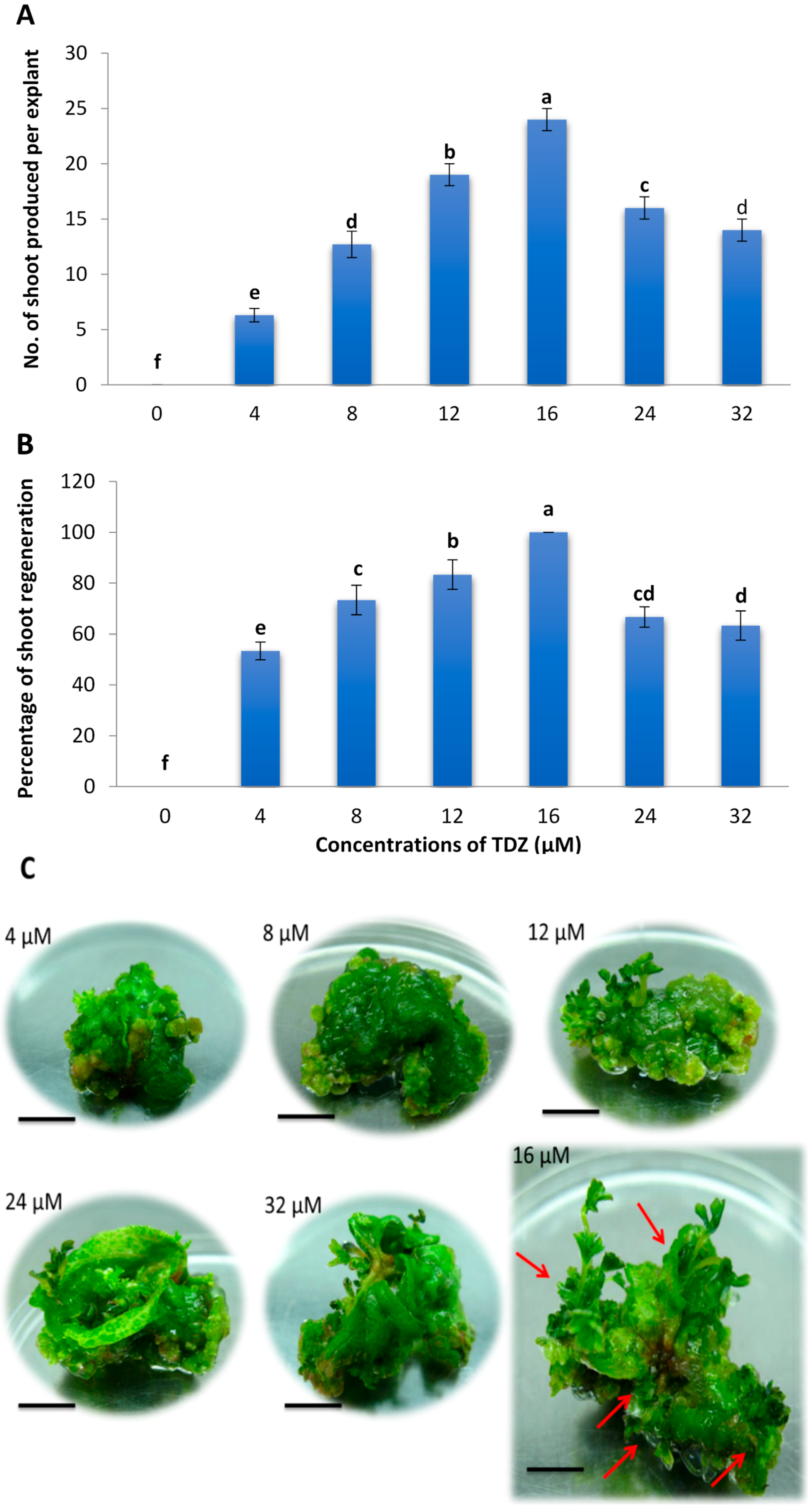
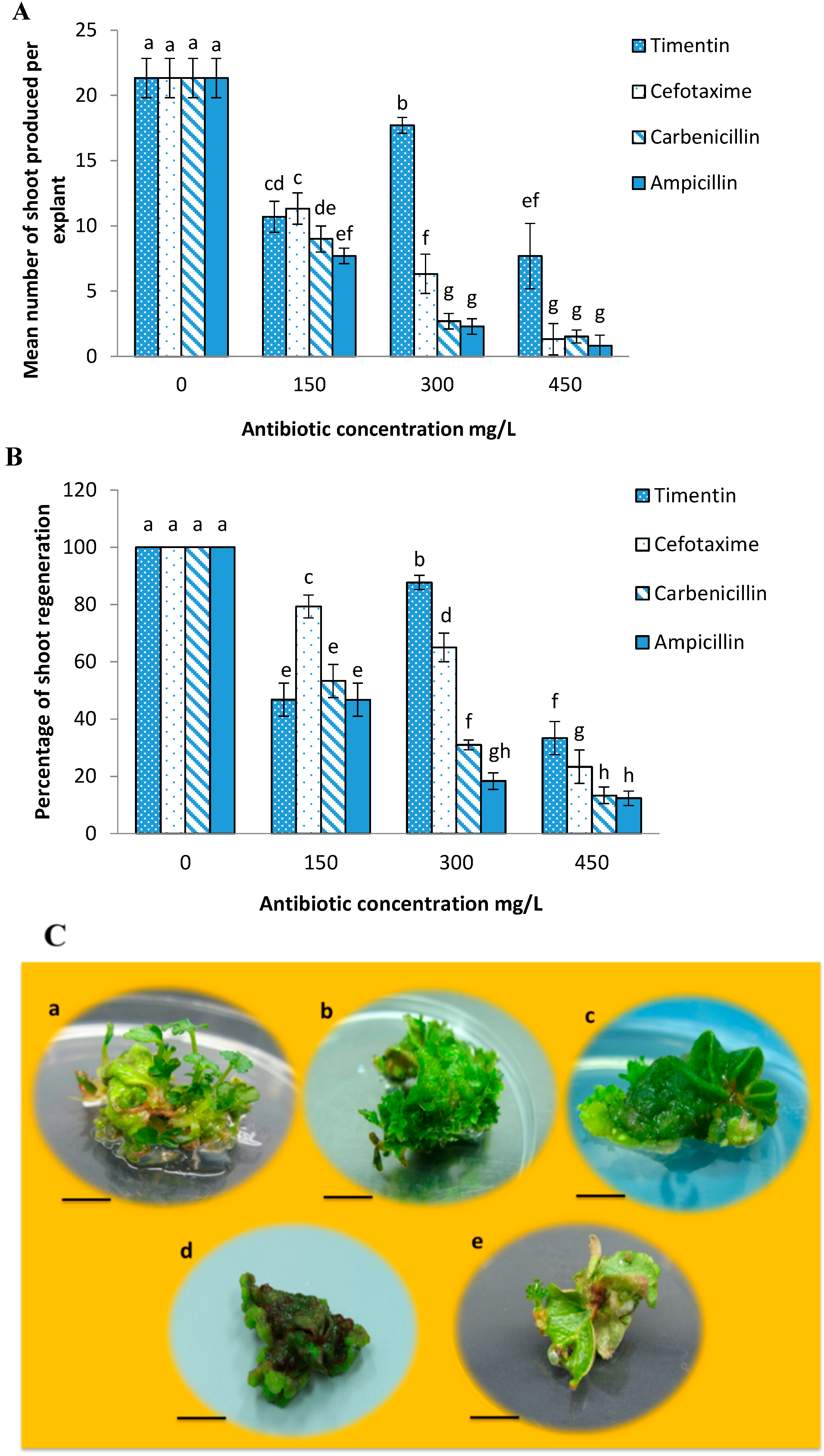
2.3. Minimum Inhibitory Concentration of Kanamycin on Leaf Explant
| Kanamycin (mg/L) | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
|---|---|---|---|---|---|---|---|---|---|
| Percentage of Regeneration (%) | 100 a | 73 b | 33 c | 13 d | 6 de | 0 e | 0 e | 0 e | 0 e |

2.4. Optimization of Different Factors Affecting Transformation of Strawberry
| Parameters measured | Preculture | ||
|---|---|---|---|
| No Preculture | TDZ 16 µM | BAP 9 µM + TDZ 4 µM | |
| OD600 = 0.6 Cefotaxime 150 mg/L | |||
| Mean No. | 6.7 b | 2.3 de | 6.3 b |
| Percentage % | 83 a | 23 ef | 67 cd |
| OD600 = 0.6 Timentin 300 mg/L | |||
| Mean No. | 7.3 b | 3.7 cd | 1.7 e |
| Percentage % | 60 bc | 57 bcd | 17 fg |
| OD600 = 1 Cefotaxime 150 mg/L | |||
| Mean No. | 4.7 c | 3.3 cd | 1 e |
| Percentage % | 43 d | 30 e | 7 g |
| OD600 = 1 Timentin 300 mg/L | |||
| Mean No. | 4.3 c | 9.7 a | 7.3 b |
| Percentage % | 50 cd | 67 b | 50 b |
2.5. Luciferase Reporter Gene Assay
| Preculture | OD600 = 0.6 | OD600 = 1 | ||
|---|---|---|---|---|
| Cefotaxime 150 mg/L | Timentin 300 mg/L | Cefotaxime 150 mg/L | Timentin 300 mg/L | |
| No preculture | 14 | 24 | 18 | 23 |
| TDZ 16 µM | 15 | 18 | 23 | 12 |
| BAP 9 µM + TDZ 4 µM | 17 | 19 | 12 | 25 |
2.6. Nested PCR
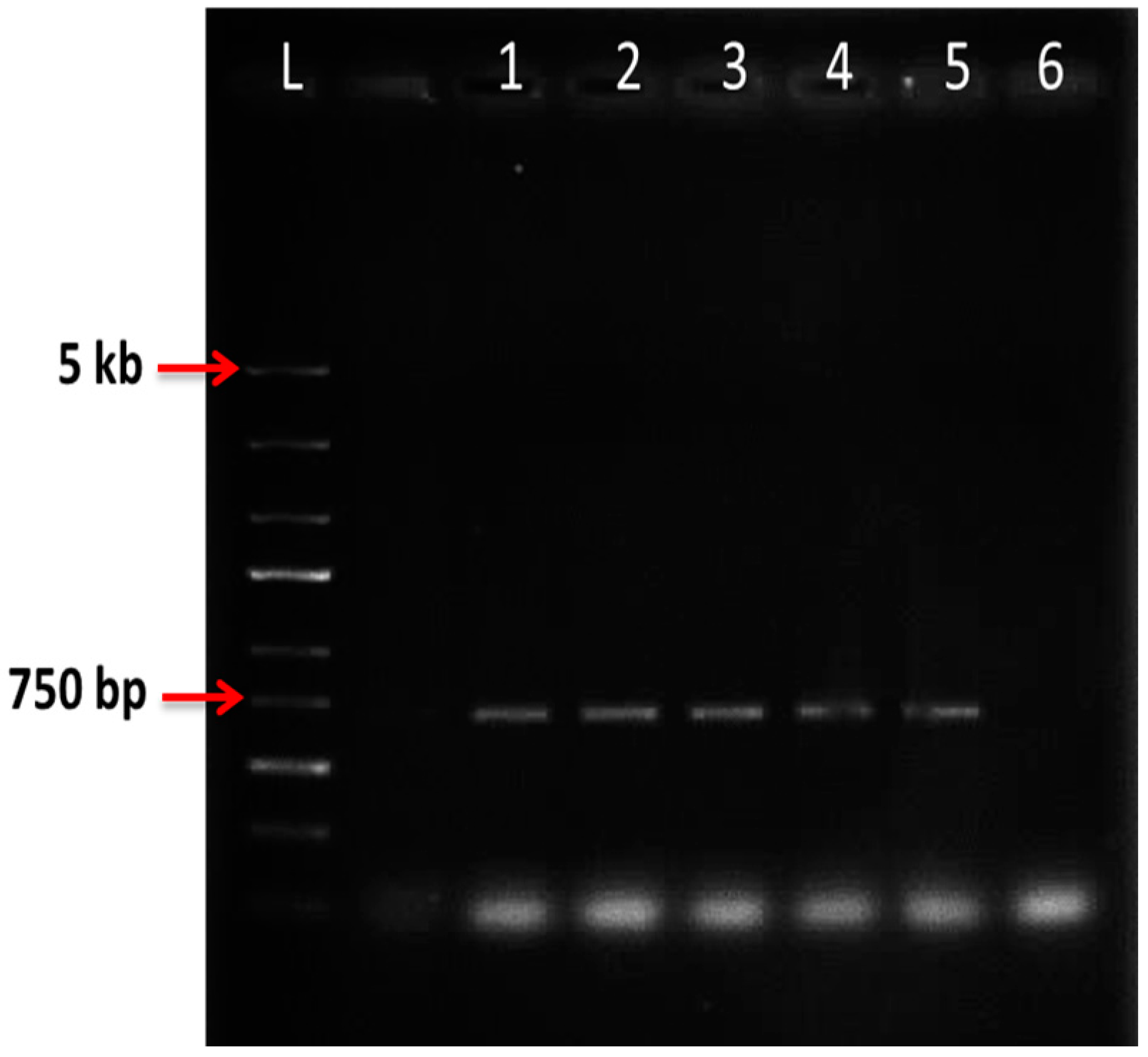
| Preculture | OD600 = 0.6 | OD600 = 1 | ||
|---|---|---|---|---|
| Cefotaxime 150 mg/L | Timentin 300 mg/L | Cefotaxime 150 mg/L | Timentin 300 mg/L | |
| No preculture | 100 | 60 | 40 | 53 |
| TDZ 16 µM | 33 | 53 | 20 | 67 |
| BAP 9 µM + TDZ 4 µM | 87 | 20 | 13 | 93 |
2.7. DNA Dot Blot Analysis
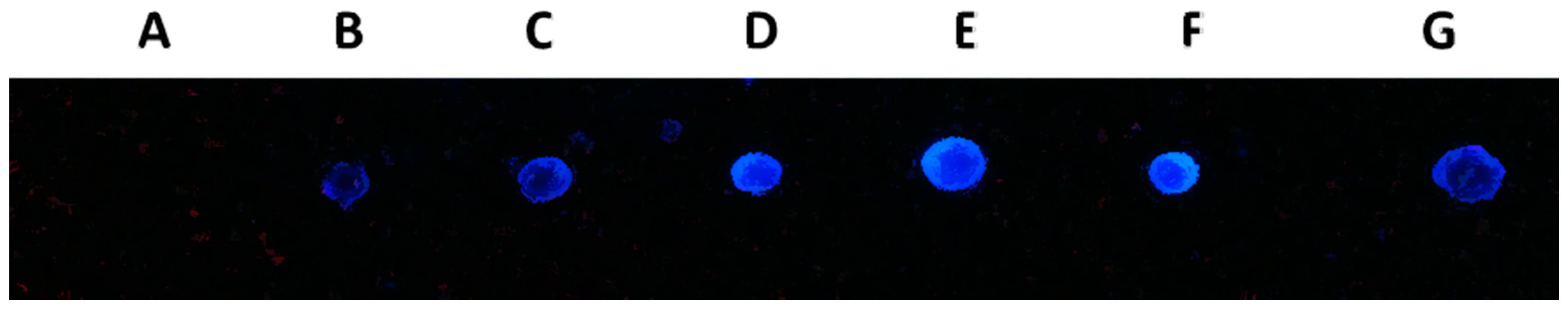
2.8. Discussion
3. Experimental Section
3.1. Binary Vector and Agrobacterium
3.1.1. Effects of Different Concentrations of TDZ on Shoot Regeneration of Strawberry Leaves
3.1.2. Effects of Different Antibiotics on Shoot Regeneration from Strawberry Leaves
3.1.3. Minimum Inhibitory Concentration of Kanamycin on Leaf Explants of Strawberry
3.1.4. Optimization of Factors Affecting Transformation of Strawberry
3.2. Culture Condition
3.2.1. Statistical Analysis
3.2.2. Luciferase Reporter Gene Assay
3.2.3. Nested PCR Analysis of Putatively Transformed Strawberry Plantlets
| Primer | Forward Primer Sequence | Reverse Primer Sequence | Temperature (°C) | Size of Amplified Fragment (bp) |
|---|---|---|---|---|
| First primer pairs | 5' TCTTCGCCAAAAGCACTCTGA 3' | 5' TGGTGATTTCAGCGTACCGAATTA 3' | 60 | 858 |
| Nested primer pairs | 5' CCTCTTTCGAAAGAAGTCGGGG 3' | 5' ACAATTTGGACTTTCCGCCCT 3' | 57.4 | 700 |
3.2.4. DNA Dot Blot Analysis of the Transgenic Strawberry Lines
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Biswas, M.; Dutt, M.; Roy, U.; Islam, R.; Hossain, M. Development and evaluation of in vitro somaclonal variation in strawberry for improved horticultural traits. Sci. Hortic. 2009, 122, 409–416. [Google Scholar] [CrossRef]
- Mezzetti, B. GMO strawberry: methods, risk and benefits. Genet Genom Rosaceae 2009, 6, 487–506. [Google Scholar]
- Qin, Y.; Teixeira da Silva, J.A.; Zhang, L.; Zhang, S. Transgenic strawberry: State of the art for improved traits. Biotechnol. Adv. 2008, 26, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhang, S. Factors influencing the efficiency of Agrobacterium-mediated transformation in strawberry cultivar Toyonaka. J. Nucl. Agric. Sci. 2007, 21, 461–465. [Google Scholar]
- Folta, K.M.; Dhingra, A. Invited review: Transformation of strawberry: The basis for translational genomics in Rosaceae. In Vitro Cell. Dev. Biol.-Plant 2006, 42, 482–490. [Google Scholar] [CrossRef]
- Barcelo, M.; El-Mansouri, I.; Mercado, J.A.; Quesada, M.A.; Pliego Alfaro, F. Regeneration and transformation via Agrobacterium tumefaciens of the strawberry cultivar Chandler. Plant Cell Tiss Org. Cul. 1998, 54, 29–36. [Google Scholar] [CrossRef]
- Gruchała, A.; Korbin, M.; Żurawicz, E. Conditions of transformation and regeneration of Induka’ and Elista’ strawberry plants. Plant Cell Tiss Org. Cul. 2004, 79, 153–160. [Google Scholar] [CrossRef]
- Qin, Y.; Teixeira da Silva, J.A.; Bi, J.; Zhang, S.; Hu, G. Response of in vitro strawberry to antibiotics. Plant Growth Regul. 2011, 65, 1–11. [Google Scholar] [CrossRef]
- Tang, J.; Liang, S.; Zhang, J.; Gao, Z.; Zhang, S. pGreen-S: A clone vector bearing absence of enhanced green fluorescent protein for screening recombinants. Anal. Biochem. 2009, 388, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Alsheikh, M.; Suso, H.P.; Robson, M.; Battey, N.; Wetten, A. Appropriate choice of antibiotic and Agrobacterium strain improves transformation of antibiotic-sensitive Fragaria vesca and F. v. semperflorens. Plant Cell Rep. 2002, 20, 1173–1180. [Google Scholar] [CrossRef]
- Ieamkhang, S.; Chatchawankanphanich, O. Augmentin as an alternative antibiotic for growth suppression of Agrobacterium for tomato (Lycopersicon esculentum) transformation. Plant Cell Tiss Org. Cul. 2005, 82, 213–220. [Google Scholar] [CrossRef]
- Karami, O.; Esna-Ashari, M.; Karimi Kurdistani, G.; Aghavaisi, B. Agrobacterium-mediated genetic transformation of plants: The role of host. Biol. Plantarum. 2009, 53, 201–212. [Google Scholar] [CrossRef]
- Břiza, J.; Pavingerová, D.; Přikrylová, P.; Gazdova, J.; Vlasák, J.; Niedermeierová, H. Use of phosphomannose isomerase-based selection system for Agrobacterium-mediated transformation of tomato and potato. Biol. Plantarum. 2008, 52, 453–461. [Google Scholar] [CrossRef]
- Cho, M.A.; Moon, C.Y.; Liu, J.R.; Choi, P.S. Agrobacterium-mediated transformation in Citrullus lanatus. Biol. Plantarum. 2008, 52, 365–369. [Google Scholar] [CrossRef]
- Cheng, M.; Lowe, B.A.; Spencer, T.M.; Ye, X.; Armstrong, C.L. Factors influencing Agrobacterium-mediated transformation of monocotyledonous species. In Vitro Cell. Dev. Biol.-Plant 2004, 40, 31–45. [Google Scholar] [CrossRef]
- Olhoft, P.M.; Flagel, L.E.; Donovan, C.M.; Somers, D.A. Efficient soybean transformation using hygromycin B selection in the cotyledonary-node method. Planta 2003, 216, 723–735. [Google Scholar] [PubMed]
- Wu, H.; Sparks, C.; Amoah, B.; Jones, H. Factors influencing successful Agrobacterium-mediated genetic transformation of wheat. Plant Cell Rep. 2003, 21, 659–668. [Google Scholar] [PubMed]
- Zambre, M.; Terryn, N.; de Clercq, J.; de Buck, S.; Dillen, W.; van Montagu, M.; van der Straeten, D.; Angenon, G. Light strongly promotes gene transfer from Agrobacterium tumefaciens to plant cells. Planta 2003, 216, 580–586. [Google Scholar] [PubMed]
- Salas, M.; Park, S.; Srivatanakul, M.; Smith, R. Temperature influence on stable T-DNA integration in plant cells. Plant Cell Rep. 2001, 20, 701–705. [Google Scholar] [CrossRef]
- Hellens, R.P.; Edwards, E.A.; Leyland, N.R.; Bean, S.; Mullineaux, P.M. pGreen: A versatile and flexible binary Ti vector for Agrobacterium-mediated plant transformation. Plant Mol. Biol. 2000, 42, 819–832. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Vinocur, B.; Shoseyov, O.; Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci. 2004, 9, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Folta, K.M.; Dhingra, A.; Howard, L.; Stewart, P.J.; Chandler, C.K. Characterization of LF9, an octoploid strawberry genotype selected for rapid regeneration and transformation. Planta 2006, 224, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Debnath, S.C. Strawberry sepal: Another explant for thidiazuron-induced adventitious shoot regeneration. In Vitro Cell. Dev. Biol.-Plant 2005, 41, 671–676. [Google Scholar] [CrossRef]
- Qin, Y.; Shanglong, Z.; Asghar, S.; Lingxiao, Z.; Qiaoping, Q.; Kunsong, C.; Changjie, X. Regeneration mechanism of Toyonoka strawberry under different color plastic films. Plant Sci. 2005, 168, 1425–1431. [Google Scholar] [CrossRef]
- Landi, L.; Mezzetti, B. TDZ, auxin and genotype effects on leaf organogenesis in Fragaria. Plant Cell Rep. 2006, 25, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Husaini, A.M.; Abdin, M.Z. Development of transgenic strawberry (Fragaria x ananassa Duch.) plants tolerant to salt stress. Plant Sci. 2008, 174, 446–455. [Google Scholar] [CrossRef]
- Petri, C.; Lopez-Noguera, S.; Alburquerque, N.; Egea, J.; Burgos, L. An antibiotic-based selection strategy to regenerate transformed plants from apricot leaves with high efficiency. Plant Sci. 2008, 175, 777–783. [Google Scholar] [CrossRef]
- Grewal, D.; Gill, R.; Gosal, S.S. Influence of antibiotic cefotaxime on somatic embryogenesis and plant regeneration in indica rice. Biotechnol. J. 2006, 1, 1158–1162. [Google Scholar] [CrossRef] [PubMed]
- Aoshima, Y. Efficient embryogenesis in the callus of tea (Camellia sinensis) enhanced by osmotic stress or antibiotic treatment. Plant Biotechnol. 2005, 22, 277–280. [Google Scholar] [CrossRef]
- Teixeira da Silva, J.A.T. Filter paper type affects the morphogenic programs and buffers the phytotoxic effect of antibiotics in chrysanthemum and tobacco thin cell layers. HortScience 2003, 38, 1403–1407. [Google Scholar]
- Teixeira da Silva, J.A.T.; Fukai, S. The impact of carbenicillin, cefotaxime and vancomycin on chrysanthemum and tobacco TCL morphogenesis and Agrobacterium growth. J. Appl. Hortic. 2001, 3, 3–12. [Google Scholar]
- Finstad, K.; Martin, R.R. Transformation of strawberry for virus resistance. Acta Hortic. 1995, 385, 86–90. [Google Scholar]
- Husaini, A.M. Pre-and post-agroinfection strategies for efficient leaf disk transformation and regeneration of transgenic strawberry plants. Plant Cell Rep. 2010, 29, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Eapen, S.; George, L. Influence of phytohormones, carbohydrates, amino acids, growth supplements and antibiotics on somatic embryogenesis and plant differentiation in finger millet. Plant Cell Tiss Org. Cul. 1990, 22, 87–93. [Google Scholar] [CrossRef]
- Borrelli, G.; di Fonzo, N.; Lupotto, E. Effect of cefotaxime on callus culture and plant regeneration in durum wheat. J. Plant Physiol. 1992, 140, 372–374. [Google Scholar] [CrossRef]
- Plus, J.; George, L.; Eapen, S.; Rao, P. Enhanced plant regeneration in pearl millet (Pennisetum americanum) by ethylene inhibitors and cefotaxime. Plant Cell Tiss Org. Cul. 1993, 32, 91–96. [Google Scholar] [CrossRef]
- Rao, A.; Sree, K.P.; Kishor, P.B.K. Enhanced plant regeneration in grain and sweet sorghum by asparagine, proline and cefotaxime. Plant Cell Rep. 1995, 15, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Channappagoudar, S.B. Studies on In Vitro Regeneration and Genetic Transformation in Chilli (Capsisum annuum L.). Ph.D. Thesis, University of Agricultural Sciences, Dharwad, India, 2007. [Google Scholar]
- Kaur, A.; Gill, M.; Ruma, D.; Gosal, S. Enhanced in vitro shoot multiplication and elongation in sugarcane using cefotaxime. Sugar Technol. 2008, 10, 60–64. [Google Scholar] [CrossRef]
- Costa, M.; Nogueira, F.; Figueira, M.; Otoni, W.; Brommonschenkel, S.; Cecon, P. Influence of the antibiotic timentin on plant regeneration of tomato (Lycopersicon esculentum Mill.) cultivars. Plant Cell Rep. 2000, 19, 327–332. [Google Scholar] [CrossRef]
- Holford, P.; Newbury, H. The effects of antibiotics and their breakdown products on the in vitro growth of Antirrhinum majus. Plant Cell Rep. 1992, 11, 93–96. [Google Scholar] [PubMed]
- Obembe, O.; Adesoye, A.; Machuka, J. Evaluation of the effectiveness of selected antibiotics in the suppression of Agrobacterium from Cowpea (Vigna unguiculata L. Walp.) embryo explants and as potential selective agents in Agrobacterium-mediated transformation. J. Trop. Microbiol. Biotechnol. 2005, 1, 27–33. [Google Scholar]
- Murthy, B.; Singh, R.; Saxena, P.K. Induction of high-frequency somatic embryogenesis in geranium (Pelargonium x hortorum Bailey cv. Ringo Rose) cotyledonary cultures. Plant Cell Rep. 1996, 15, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Bermúdez, S.; Redondo-Nevado, J.; Muñoz-Blanco, J.; Caballero, J.L.; López-Aranda, J.M.; Valpuesta, V.; Pliego-Alfaro, F.; Quesada, M.A.; Mercado, J.A. Manipulation of strawberry fruit softening by antisense expression of a pectate lyase gene. Plant Physiol. 2002, 128, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Dhir, S.; Hinchee, M.A.W.; Layton, J.G.; Oakes, J.V. Methods for Strawberry Transformation Using Agrobacterium Tumefaciens. U.S. Patent 6274791 B1, 14 Agust 2001. [Google Scholar]
- Cordeno de Mesa, C.; Jiménez-Bermúdez, S.; Pliego-Alfaro, F.; Quesada, M.A.; Mercado, J.A. Agrobacterium cells as microprojectile coating: a novel approach to enhance stable transformation rates in strawberry. Funct Plant Biol. 2000, 27, 1093–1100. [Google Scholar] [CrossRef]
- Mathews, H.; Dewey, V.; Wagoner, W.; Bestwick, R.K. Molecular and cellular evidence of chimaeric tissues in primary transgenics and elimination of chimaerism through improved selection protocols. Transgenic Res. 1998, 7, 123–129. [Google Scholar] [CrossRef]
- El-Mansouri, I.; Mercado, J.A.; Valpuesta, V.; López-Aranda, J.M.; Pliego-Alfaro, F.; Quesada, M.A. Shoot regeneration and Agrobacterium-mediated transformation of Fragaria vesca L. Plant Cell Rep. 1996, 15, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Wadl, P.A. Improved Regeneration and Agrobacterium-Mediated Transformation of Wild Strawberry (Fragaria vesca L.). Master’s Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2006. [Google Scholar]
- Schaart, J.G.; Krens, F.A.; Pelgrom, K.T.B.; Mendes, O.; Rouwendal, G.J.A. Effective production of marker-free transgenic strawberry plants using inducible site-specific recombination and a bifunctional selectable marker gene. Plant Biotechnol. J. 2004, 2, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, A.H.; El-Shafy, Y.H.; El-Shihy, O.M.; Madkour, M. Highly efficient regeneration system via somatic embryogenesis from immature embryos of Egyptian wheat cultivars (Triticum aestivum L.) using different growth regulators. World J. Agr. Sci. 2006, 22, 82–289. [Google Scholar]
- Marcotrigiano, M.; McGlew, S.P.; Hackett, G.; Chawla, B. Shoot regeneration from tissue-cultured leaves of the American cranberry (Vaccinium macrocarpon). Plant Cell Tiss Org. Cul. 1996, 44, 195–199. [Google Scholar] [CrossRef]
- Huetteman, C.A.; Preece, J.E. Thidiazuron: A potent cytokinin for woody plant tissue culture. Plant Cell Tiss. Org. Cul. 1993, 33, 105–119. [Google Scholar] [CrossRef]
- Vellicce, G.R.; Ricci, J.C.D.; Hernández, L.; Castagnaro, A.P. Enhanced resistance to Botrytis cinerea mediated by the transgenic expression of the chitinase gene ch5B in strawberry. Transgenic Res. 2006, 15, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Nasri, S.; Bahramnejad, B. Regeneration and Agrobacterium-mediated transformation of three economically important strawberry cultivars Kurdistan, Camarosa and Paros. Prog. Biol. Sci. 2013, 2, 60–66. [Google Scholar]
- Morgan, A.; Baker, C.M.; Chu, J.S.F.; Lee, K.; Crandall, B.A.; Jose, L. Production of herbicide tolerant strawberry through genetic engineering. Acta Hortic. 2002, 567, 113–115. [Google Scholar]
- Chalavi, V.; Tabaeizadeh, Z.; Thibodeau, P. Enhanced resistance to Verticillium dahliae in transgenic strawberry plants expressing a Lycopersicon chilense chitinase gene. J. Am. Soc. Hortic. Sci. 2003, 128, 747–753. [Google Scholar]
- Liu, F.; Guo, Y.; Gu, D.; Xiao, G.; Chen, Z.; Chen, S. Salt tolerance of transgenic plants with BADH cDNA. Acta Genet Sin. 1997, 24, 54–58. [Google Scholar]
- Kost, B.; Schnorf, M.; Potrykus, I.; Neuhaus, G. Non‐destructive detection of firefly luciferase (LUC) activity in single plant cells using a cooled, slow‐scan CCD camera and an optimized assay. Plant J. 1995, 8, 155–166. [Google Scholar] [CrossRef]
- Liu, C.W.; Chen, J.J.W.; Kang, C.C.; Wu, C.H.; Yiu, J.C. Transgenic lettuce (Lactuca sativa L.) expressing H1N1 influenza surface antigen (neuraminidase). Sci. Hortic. 2012, 139, 8–13. [Google Scholar] [CrossRef]
- Goto, F.; Yoshihara, T.; Saiki, H. Iron accumulation and enhanced growth in transgenic lettuce plants expressing the iron-binding protein ferritin. Theor. Appl. Genet 2000, 100, 658–664. [Google Scholar] [CrossRef]
- Firsov, A.; Dolgov, S. Agrobacterial transformation and transfer of the antifreeze protein gene of winter flounder to the strawberry. Acta Hortic. 1999, 484, 581–586. [Google Scholar]
- Haymes, K.; Davis, T. Agrobacterium-mediated transformation of “Alpine” Fragaria vesca, and transmission of transgenes to R1 progeny. Plant Cell Rep. 1998, 17, 279–283. [Google Scholar] [CrossRef]
- Oosumi, T.; Gruszewski, H.A.; Blischak, L.A.; Baxter, A.J.; Wadl, P.A.; Shuman, J.L.; Veilleux, R.E.; Shulaev, V. High-efficiency transformation of the diploid strawberry (Fragaria vesca) for functional genomics. Planta 2006, 223, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Brown, T. Dot and slot blotting of DNA. In Current Protocols in Molecular Biology; John Wiley & Sons: Hoboken, NJ, USA, 2001. [Google Scholar] [CrossRef]
- An, G.; Ebert, P.R.; Mitra, A.; Ha, S.B. Binary vectors. In Plant Molecular Biology Manual; Gelvin, S.B., Schilperoort, R.A., Eds.; Kluwer Academic Publishers: Dordrecht, the Netherlands, 1988; Volume A3/7, pp. 29–47. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plantarum. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the Agrobacterium harboring the pGreen is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haddadi, F.; Abd Aziz, M.; Abdullah, S.N.A.; Tan, S.G.; Kamaladini, H. An Efficient Agrobacterium-Mediated Transformation of Strawberry cv. Camarosa by a Dual Plasmid System. Molecules 2015, 20, 3647-3666. https://doi.org/10.3390/molecules20033647
Haddadi F, Abd Aziz M, Abdullah SNA, Tan SG, Kamaladini H. An Efficient Agrobacterium-Mediated Transformation of Strawberry cv. Camarosa by a Dual Plasmid System. Molecules. 2015; 20(3):3647-3666. https://doi.org/10.3390/molecules20033647
Chicago/Turabian StyleHaddadi, Fatemeh, Maheran Abd Aziz, Siti Nor Akmar Abdullah, Soon Guan Tan, and Hossein Kamaladini. 2015. "An Efficient Agrobacterium-Mediated Transformation of Strawberry cv. Camarosa by a Dual Plasmid System" Molecules 20, no. 3: 3647-3666. https://doi.org/10.3390/molecules20033647





