Synthesis of Novel Triazoles, Tetrazine, Thiadiazoles and Their Biological Activities
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
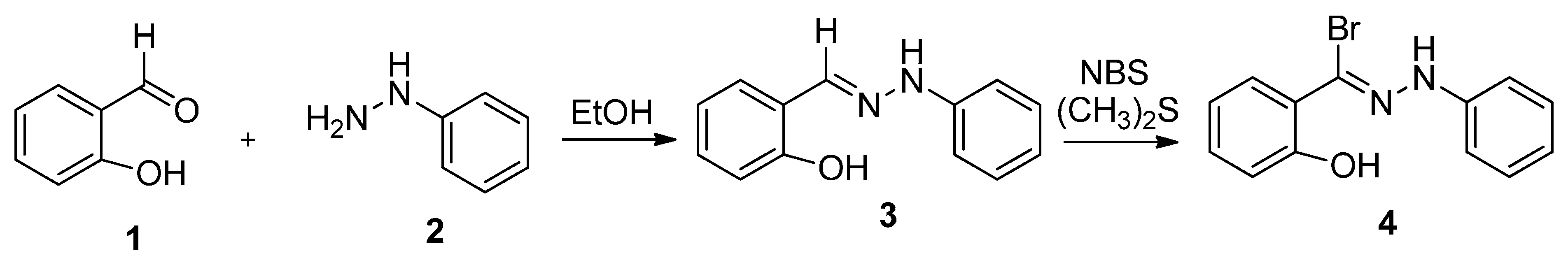
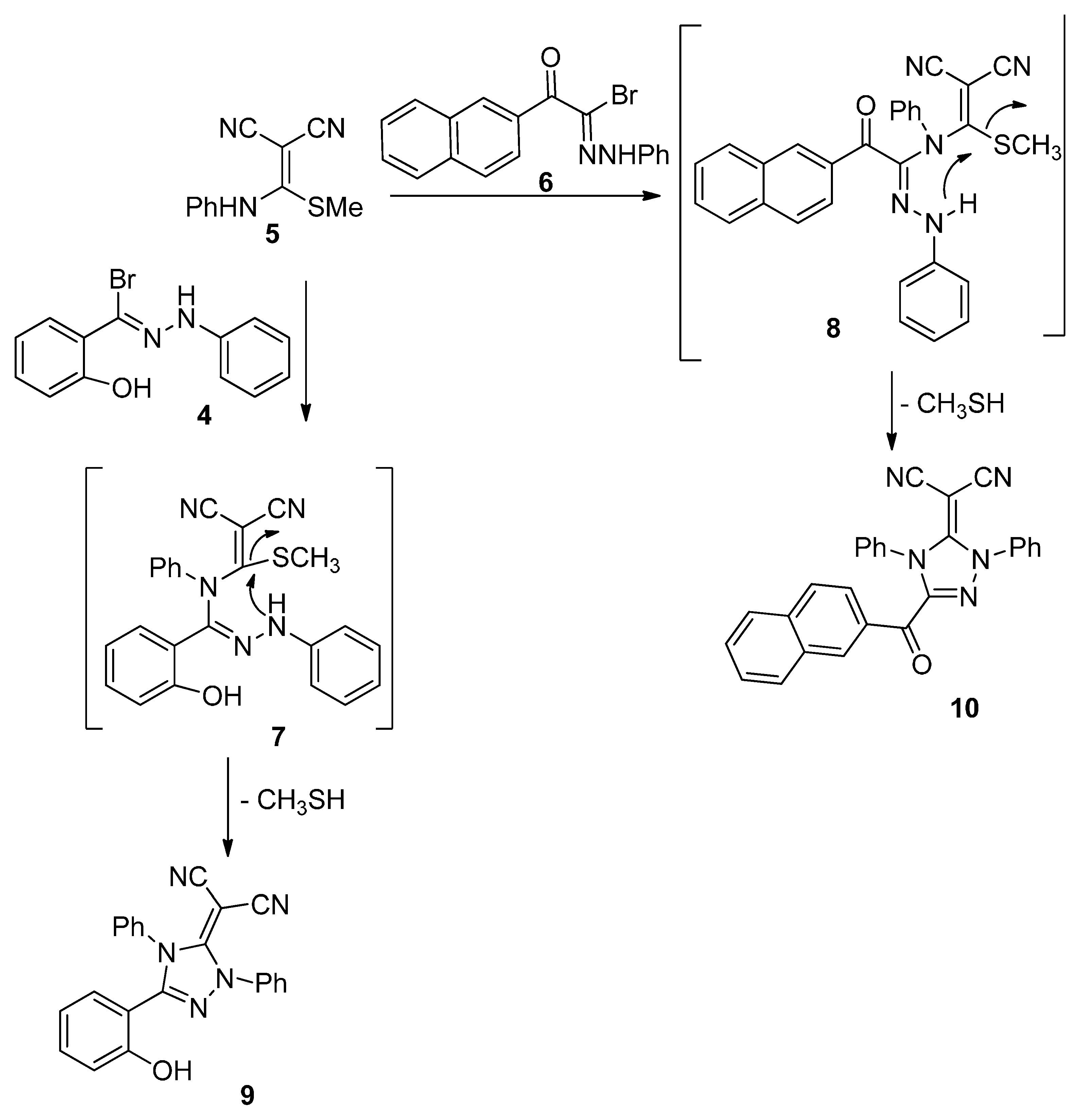
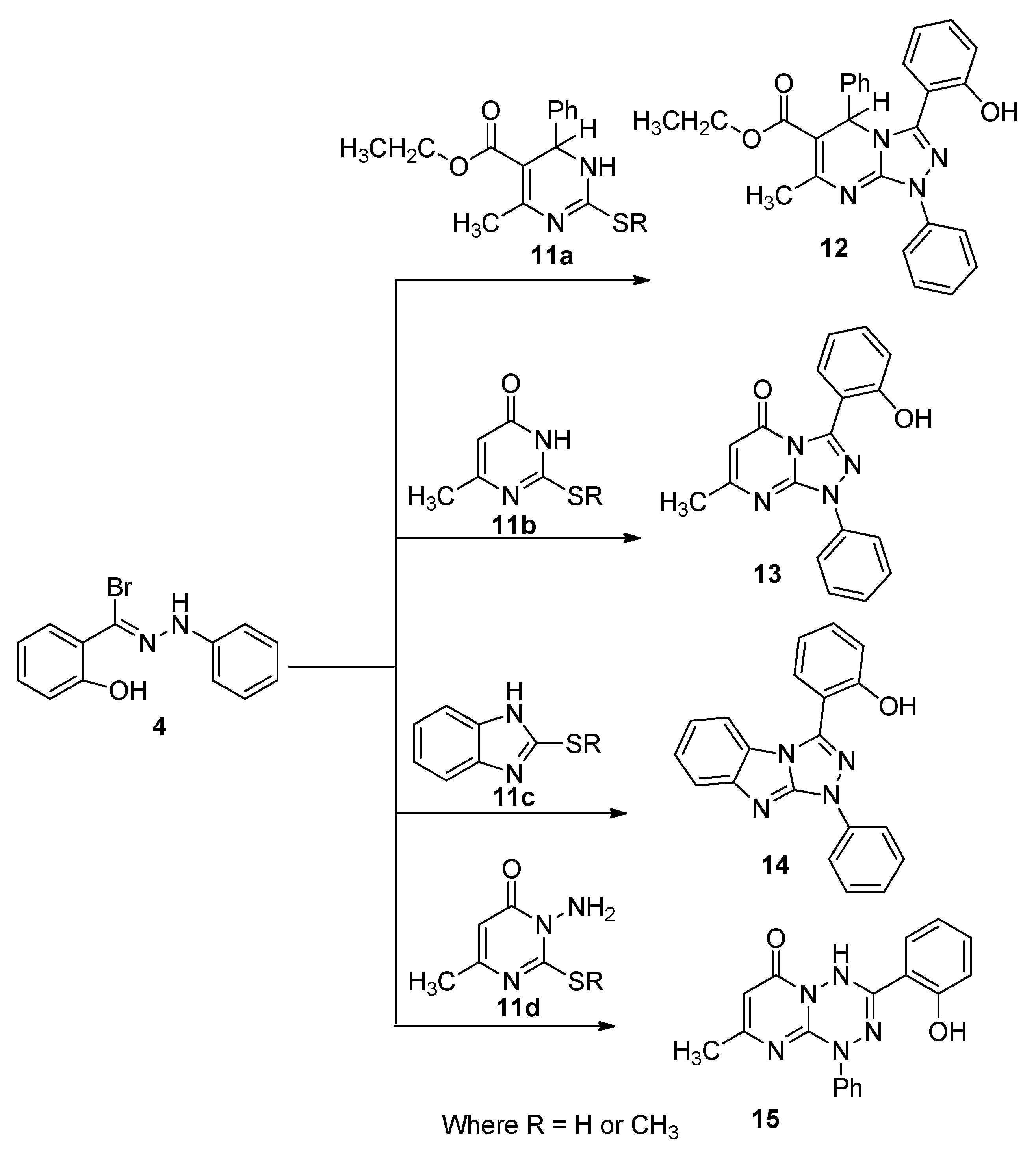

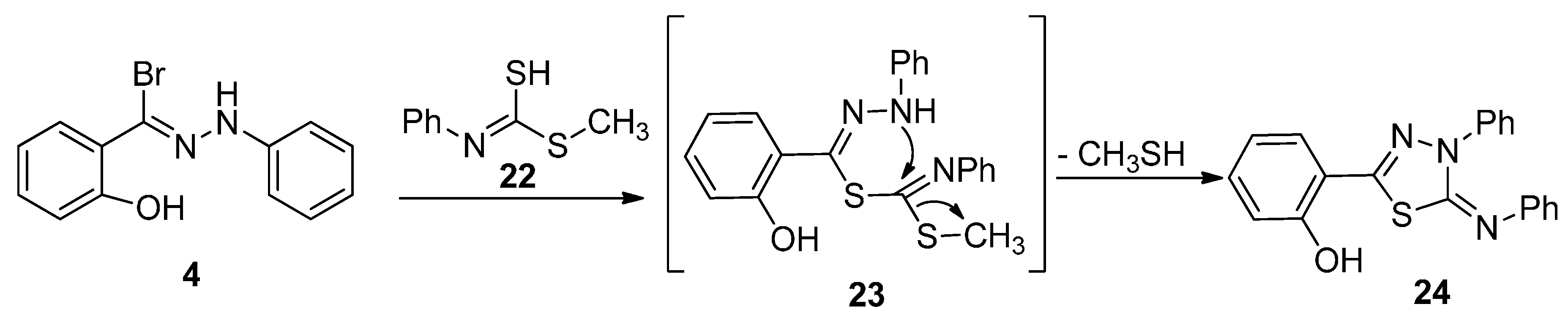
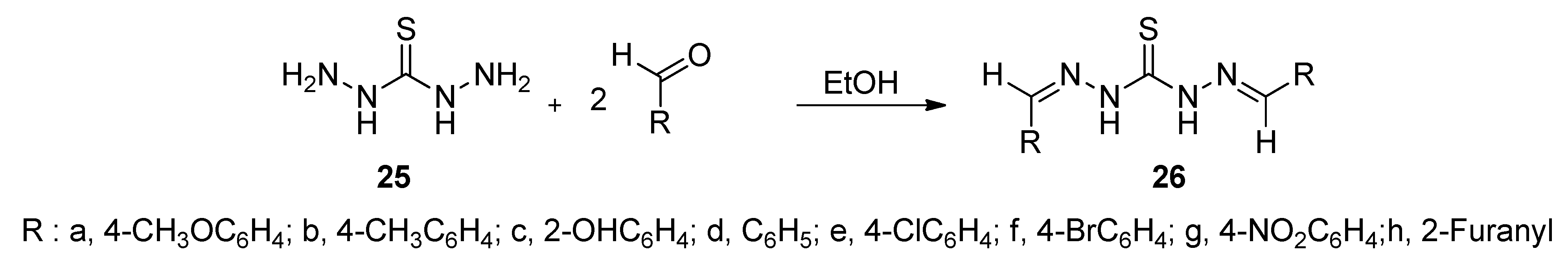
2.2. Biology
2.2.1. Antibacterial Activities
| Compound No. | Gram-Negative | Gram-Positive | ||
|---|---|---|---|---|
| E. coli | P. aeruginosa | S. aureus | B. megaterium | |
| 4 | 15 | 13 | 16 | 18 |
| 5 | 17 | 14 | 19 | 20 |
| 9 | 13 | 12 | 14 | 15 |
| 12 | 12 | 11 | 13 | 14 |
| 14 | 14 | 13 | 15 | 16 |
| 15 | 12 | 11 | 14 | 14 |
| 20 | 13 | 11 | 14 | 16 |
| 21 | 12 | 12 | 13 | 15 |
| 24 | 18 | 16 | 18 | 21 |
| 26a | 13 | 12 | 13 | 15 |
| 26b | 12 | 14 | 12 | 16 |
| 26c | 13 | 14 | 12 | 17 |
| 26d | 12 | 11 | 13 | 14 |
| 26e | 13 | 12 | 14 | 13 |
| 26f | 14 | 13 | 15 | 17 |
| 26g | 9 | 11 | 11 | 10 |
| 26h | 21 | 20 | 20 | 21 |
| Tetracycline | 20 | 18 | 22 | 24 |
2.2.2. Minimum Inhibitory Concentrations
| Compound No. | Gram-Negative | Gram-Positive | ||
|---|---|---|---|---|
| E. coli | P. aeruginosa | S. aureus | B. megaterium | |
| 4 | 25 | 30 | 30 | 35 |
| 5 | 15 | 15 | 20 | 20 |
| 9 | 35 | 40 | 35 | 45 |
| 12 | 40 | 45 | 50 | 50 |
| 14 | 20 | 30 | 35 | 35 |
| 15 | 45 | 40 | 45 | 50 |
| 20 | 40 | 45 | 50 | 40 |
| 21 | 35 | 40 | 50 | 45 |
| 24 | 15 | 20 | 20 | 20 |
| 26a | 30 | 35 | 35 | 35 |
| 26b | 30 | 30 | 35 | 35 |
| 26c | 25 | 35 | 40 | 35 |
| 26d | 40 | 45 | 45 | 55 |
| 26e | 45 | 45 | 55 | 50 |
| 26f | 35 | 40 | 40 | 35 |
| 26g | 55 | 50 | 70 | 50 |
| 26h | 15 | 20 | 20 | 20 |
| Tetracycline | 10 | 15 | 15 | 15 |
2.2.3. DNA Cleavage Assay

2.2.4. Superoxide Dismutase Mimetic Catalytic Activity
| Compound No. | Δ through 4 min | % Inhibition |
|---|---|---|
| Control | 0.548 | — |
| Horseradish peroxidase | 0.169 | 69.2 |
| 4 | 0.297 | 45.8 |
| 5 | 0.264 | 51.8 |
| 9 | 0.326 | 40.5 |
| 12 | 0.364 | 33.6 |
| 14 | 0.272 | 50.4 |
| 15 | 0.381 | 30.5 |
| 20 | 0.332 | 39.4 |
| 21 | 0.337 | 38.5 |
| 24 | 0.273 | 50.2 |
| 26a | 0.305 | 44.3 |
| 26b | 0.314 | 42.7 |
| 26c | 0.308 | 43.8 |
| 26d | 0.382 | 30.3 |
| 26e | 0.358 | 34.7 |
| 26f | 0.294 | 46.4 |
| 26g | 0.386 | 29.6 |
| 26h | 0.256 | 53.3 |
2.2.5. DPPH Free Radical Scavenging Activity
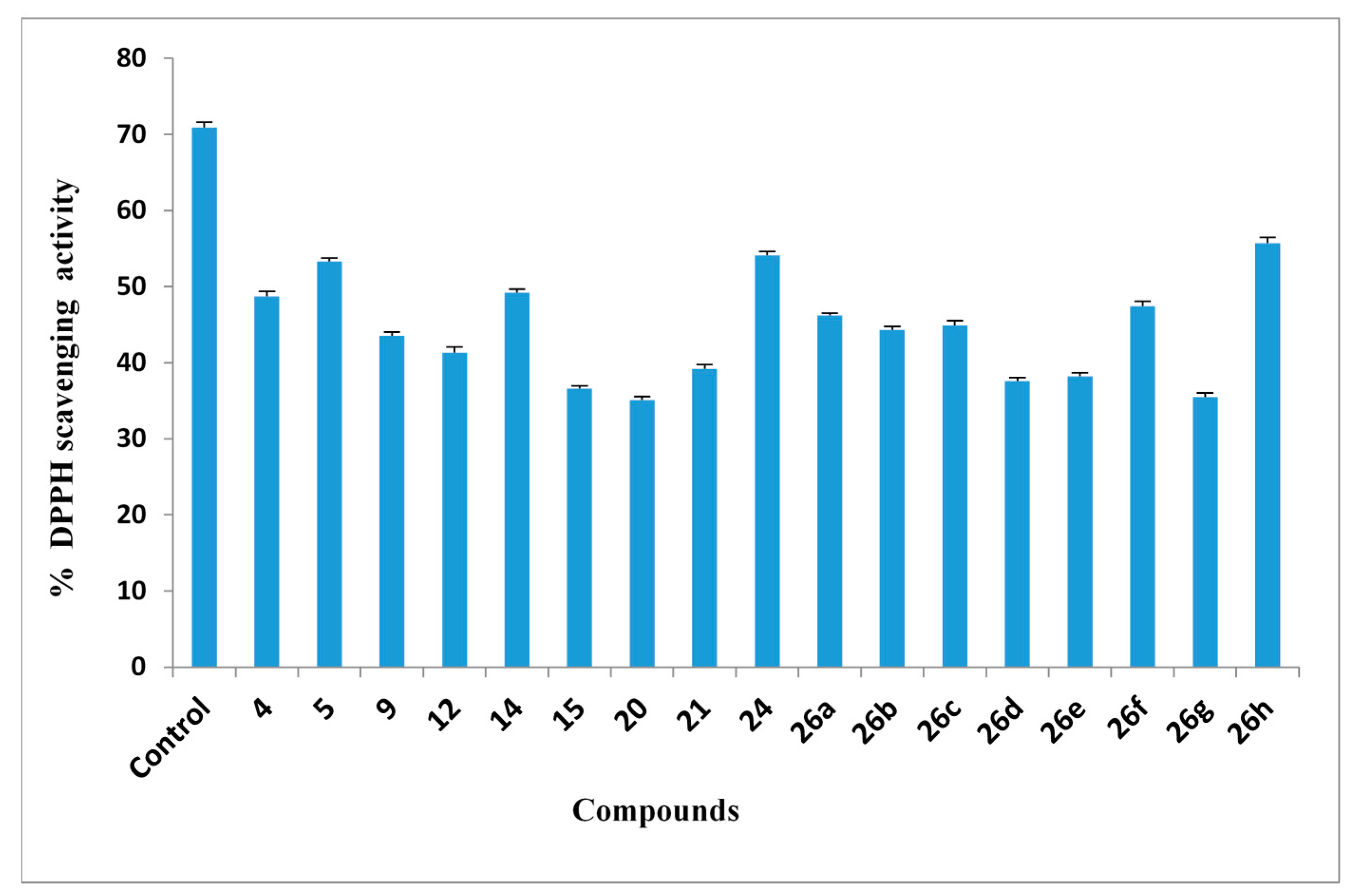
2.2.6. ABTS Radical Cation Decolorization Assay

2.2.7. Nitric Oxide (NO) Scavenging Assay

3. Experimental Section
3.1. General Information
3.1.1. Synthesis of 2-Hydroxy-N-phenylbenzohydrazonoyl Bromide (4)
3.1.2. Synthesis of 2-(5-(2-Hydroxyphenyl)-2,4-diphenyl-2H-1,2,4-triazol-3(4H)-ylidene)malononitrile (9) and 2-(3-(2-Naphthoyl-1,4-diphenyl-1H-1,2,4-triazol-5(4H)-ylidene)malononitrile (10)
3.1.3. General Procedure for the Preparation of Bis([1,2,4]triazolo[4,3-a]pyrimidine) Derivatives 12,13), Bis([1,2,4]triazolo[4,5-a]benzimidazole) Derivative 14 and 3-(2-Hydroxyphenyl)-8-methyl-1-phenyl-1H-pyrimido[1,2-b][1,2,4,5]tetrazine-6-(4H)-one (15)
3.1.4. Synthesis of 2-(4,5-Dihydro-5-imino-4-phenyl-1,3,4-thiadiazol-2-yl)phenol (20)
3.1.5. Synthesis of 1-(2-Hydroxyphenyl-3-phenyl-1,3,4-thiadiazol-4,(3H)-ylidene)hydrazine (21) and 4,5-Dihydro-4-phenyl-5-(phenylimino)-1,3,4-thiadiazol-2-yl)phenol (24)
3.2. Biology
3.2.1. Antibacterial Activities
3.2.2. Minimum Inhibitory Concentrations (MIC)
3.2.3. DNA Cleavage Assay
3.2.4. Superoxide Dismutase (SOD) Mimetic Catalytic Activity Assay
3.2.5. 2,2,-Diphenyl-1-Picrylhydrazyl (DPPH) Free Radical Scavenging Activity Assay
3.2.6. 2,2'-Azino-bis-3-ethylbenzthiazoline-6-sulphonic Acid (ABTS) Radical Cation Assay
3.2.7. Nitric Oxide (NO) Scavenging Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, X.H.; Tan, C.X.; Weng, J.Q. Phase transfer-catalyzed, one-pot synthesis of some novel N-pyrimidinyl-N'-nicotinylthiourea derivatives. Phosphorus Sulfur Silicon Relat. Elem. 2011, 186, 552–557. [Google Scholar] [CrossRef]
- Su, N.N.; Li, Y.; Yu, S.J.; Zhang, X.; Liu, X.H.; Zhao, W.G. Microwave-assisted synthesis of some novel 1,2,3-triazoles by click chemistry, and their biological activity. Res. Chem. Intermed. 2013, 39, 759–766. [Google Scholar] [CrossRef]
- Liu, X.H.; Pan, L.; Tan, C.X.; Weng, J.Q.; Wang, B.L.; Li, Z.M. Synthesis, crystal structure, bioactivity and DFT calculation of new oxime ester derivatives containing cyclopropane moiety. Pestic. Biochem. Physiol. 2011, 101, 143–147. [Google Scholar] [CrossRef]
- Liu, X.H.; Pan, L.; Ma, Y.; Weng, J.Q.; Tan, C.X.; Li, Y.H.; Shi, Y.X.; Li, B.J.; Li, Z.M.; Zhang, Y.G. Design, synthesis, biological activities, and 3D-QSAR of new N,N'-diacylhydrazines containing 2-(2,4-dichlorophenoxy)propane moiety. Chem. Biol. Drug Des. 2011, 78, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Q.; Tan, C.X.; Weng, J.Q.; Liu, X.H. Synthesis, structure and DFT calculation of chlorimuron-ethyl. Asian J. Chem. 2012, 24, 2808–2810. [Google Scholar]
- Liu, X.H.; Weng, J.Q.; Tan, C.X. Synthesis, crystal structure, and fungicidal activity of 5-(4-cyclopropyl-5-((3-fluorobenzyl)thio)-4H-1,2,4-triazol-3-yl)-4-methyl-1,2,3-thiadiazole. J. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Weng, J.Q.; Wang, L.; Liu, X.H. Synthesis, crystal structure and herbicidal activity of a 1,2,4-triazol-5(4H)-one derivative. J. Chem. Soc. Pak. 2012, 34, 1248–1252. [Google Scholar]
- Liu, X.H.; Zhao, W.G.; Wang, B.L.; Li, Z.M. Synthesis, bioactivity and DFT structure-activity relationship study of novel 1,2,3-thiadiazole derivatives. Res. Chem. Intermed. 2012, 38, 1999–2008. [Google Scholar] [CrossRef]
- Metwally, M.A.; Gouda, M.A.; Harmal, A.N.; Khalil, A.M. Synthesis, antitumor, cytotoxic and antioxidant evaluation of some new pyrazolotriazines attached to antipyrine moiety. Eur. J. Med. Chem. 2012, 56, 254–262. [Google Scholar] [CrossRef]
- Sun, N.B.; Tong, J.Y.; Wu, H.K. Synthesis and fungicidal activity of 1,3,4-oxadiazole derivatives containing pyrazole moiety. Chin. J. Org. Chem. 2013, 33, 101–105. [Google Scholar] [CrossRef]
- Tan, C.X.; Shi, Y.X.; Weng, J.Q.; Liu, X.H.; Li, B.J.; Zhao, W.G. Synthesis and antifungal activity of 1,2,4-triazole derivatives containing cyclopropane moiety. Lett. Drug Des. Discov. 2012, 9, 431–435. [Google Scholar] [CrossRef]
- El-Serwy, W.S.; Mohamed, N.A.; Abbas, E.M.; Abdel-Rahman, R.F. Synthesis and anti-inflammatory properties of novel 1,2,4-triazole derivatives. Res. Chem. Intermed. 2013, 39, 2543–2554. [Google Scholar] [CrossRef]
- Kocyigit-Kaymakcioglu, B.; Celen, A.O.; Tabanca, N.; Ali, A.; Khan, S.I.; Khan, I.A.; Wedge, D.E. Synthesis and biological activity of substituted urea and thiourea derivatives containing 1,2,4-triazole moieties. Molecules 2013, 18, 3562–3576. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Tan, C.X.; Weng, J.Q. Synthessis, dimeric crystal structure and fungicidal activity of 1-(4-methylphenyl)-2-(5-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-phenyl-4H-1,2,4-triazol-3-ylthio)ethanone. Phosphorus Sulfur Silicon Relat. Elem. 2011, 186, 558–564. [Google Scholar] [CrossRef]
- Tong, J.Y.; Wu, H.K.; Sun, N.B.; Liu, X.H. Synthesis, crystal structure and biological activity of a new 1,2,4-triazole derivative. Chin. J. Struct. Chem. 2013, 32, 607–611. [Google Scholar]
- Ke, W.; Sun, N.B.; Wu, H.K. Microwave assistant synthesis, crystal structure and biological activity of a 1,2,4-triazole compound. J. Chem. Soc. Pak. 2013, 35, 1239–1244. [Google Scholar]
- Al-Omar, M.A.; Al-Abdullah, E.S.; Shehata, I.A.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activities of novel 5-(1-adamantyl)-4-arylideneamino-3-mercapto-1,2,4-triazoles and related derivatives. Molecules 2010, 15, 2526–2550. [Google Scholar] [CrossRef] [PubMed]
- Franklim, T.N.; Freire-de-Lima, L.; Diniz, J.D.S.; Previato, J.O.; Castro, R.N.; Mendonca-Previato, L.; de Lima, M.E.F. Design, synthesis and trypanocidal evaluation of novel 1,2,4-triazoles-3-thiones derived from natural piperine. Molecules 2013, 18, 6366–6382. [Google Scholar] [CrossRef] [PubMed]
- Benci, K.; Mandic, L.; Suhina, T.; Sedic, M.; Klobucar, M.; Pavelic, S.K.; Pavelic, K.; Wittine, K.; Mintas, M. Novel coumarin derivatives containing 1,2,4-triazole, 4,5-dicyanoimidazole and purine moieties: Synthesis and evaluation of their cytostatic activity. Molecules 2012, 17, 11010–11025. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Yoshizawa, Y.; Oh, K. Synthesis of 2RS,4RS-1-[2-phenyl-4-[2-(2-trifluromethoxyphenoxy)-ethyl]-1,3-dioxolan-2-yl-methyl]-1H-1,2,4-triazole derivatives as potent inhibitors of brassinosteroid biosynthesis. Molecules 2012, 17, 4460–4473. [Google Scholar] [CrossRef] [PubMed]
- Ram, V.J.; Upadhyay, D.N. Synthesis of pyrimidines and azolopyrimidines as biodynamic agents. Indian J. Chem. 1999, 38B, 173–177. [Google Scholar]
- Ram, V.J.; Singha, U.K. Chemotherapeutic agents. XI. Synthesis of pyrimidines and azolopyrimidines as leishmanicides. Eur. J. Med. Chem. 1990, 25, 533–538. [Google Scholar] [CrossRef]
- Nakamura, H.; Hosoi, Y.H.; Fukawa, J. High-contract Photographic Material for Printing Platemaking and Its Processing. JP 03010245, 17 January 1991. [Google Scholar]
- Clark, J.; Shohhet, M.S.; Korakas, D.; Varvounis, G. Synthesis of thieno[2,3-d]pyrimidines from 4,6-dichloropyrimidin-5-carbaldehydes. J. Heterocycl. Chem. 1993, 30, 1065–1072. [Google Scholar] [CrossRef]
- Kogowwra, I.Y.; Yimatsusita, N.N.; Pfkador, J.K. Synthesis of substituted 2,4-dioxo thieno pyrimidin-1-acetic acid and their evalution as aldose reductase inhibitors. Eur. J. Med. Chem. 1993, 28, 769–781. [Google Scholar] [CrossRef]
- Mawad, E.B.; Yousif, M.Y.; Metwaly, M.A. Synthesis of certain heteroaryl-fused pyrimidines and pyridines and selena- and thia-diazoles with naphthyl substituent as potential antifungal agents. Pharmazie 1989, 44, 820–822. [Google Scholar] [PubMed]
- Khalil, Z.H.; Abdel Hafez, A.A.; Abdo, A.A. New pyrimidine derivatives: Synthesis and application of thiazolo [3,2-a]triazolo[4,3-a]pyrimidine as bactericides, fungicides and bioregulators. Phosphorus Sulfur Silicon Relat. Elem. 1989, 45, 81–93. [Google Scholar] [CrossRef]
- Bleehen, N.M.; Newlands, E.S.; Lee, S.M.; Thatcher, N.; Selby, P.; Calvert, A.H.; Rustin, G.J.S.; Brampton, M.; Stevens, M.F.G. Cancer research campaign phase II trial of temozolomide in metastatic melanoma. J. Clin. Oncol. 1995, 13, 910–913. [Google Scholar] [PubMed]
- Liu, X.H.; Weng, J.Q.; Tan, C.X. Microwave synthesis and biological activity of hydrazine derivatives containing 1,2,3-thiadiazole. Asian J. Chem. 2011, 23, 4064–4066. [Google Scholar]
- Tan, C.X.; Weng, J.Q.; Liu, Z.X.; Liu, X.H.; Zhao, W.G. Synthesis, crystal structure, and fungicidal activity of a novel 1,2,3-thiadiazole compound. Phosphorus Sulfur Silicon Relat. Elem. 2012, 187, 990–996. [Google Scholar] [CrossRef]
- Dong, W.L.; Liu, Z.X.; Liu, X.H.; Li, Z.M.; Zhao, W.G. Synthesis and antiviral activity of new acrylamide derivatives containing 1,2,3-thiadiazole as inhibitors of hepatitis B virus replication. Eur. J. Med. Chem. 2010, 45, 1919–1926. [Google Scholar] [CrossRef] [PubMed]
- Rollas, S.; Gulerman, N.; Erdeniz, H. Synthesis and antimicrobial activity of some new hydrazones of 4-fluorobenzoic acid hydrazide and 3-acetyl-2,5-disubstituted-1,3,4-oxadiazolines. Farmaco 2002, 57, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Kucukguzel, S.G.; Rollas, S.; Kucukguzel, I.; Kiraz, M. Synthesis and antimycobacterial activity of some coupling products from 4-aminobenzoic acid hydrazones. Eur. J. Med. Chem. 1999, 34, 1093–1100. [Google Scholar] [CrossRef]
- Kucukguzel, S.G.; Oruc, E.E.; Rollas, S.; Sahin, F.; Ozbek, A. Synthesis, characterization and biological activity of novel 4-thiazolidinones, 1,3,4-oxadiazoles and some related compounds. Eur. J. Med. Chem. 2002, 37, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Raja, G.; Jayabalakrishnan, C. Organoruthenium(II) thiosemicarbazone complexes: Synthesis, spectral characterization, DNA binding and DNA cleavage studies. Cent. Eur. J. Chem. 2013, 11, 1010–1018. [Google Scholar] [CrossRef]
- Sayed, A.R. Synthesis of novel bisthiadiazoles, bistriazoles and polypyrazole derivatives based on hydrazonoyl halides. Tetrahedron 2013, 69, 5293–5298. [Google Scholar] [CrossRef]
- Sayed, A.R. Synthesis of 1,3,4-thiadiazines, bis-1,3,4-thiadiazoles, [1,2,4]triazino[3,4-b][1,3,4]thiadiazine, thiazolines from carbonothioic dihydrazide. Tetrahedron 2012, 68, 2784–2784. [Google Scholar] [CrossRef]
- Shawali, A.S.; Sayed, A.R.; Zayed, M.M.A. Convenient method for synthesis of bis-2,2'-(1,3,4-thiadiazole) and bis-3,3'-(1,2,4-triazole) derivatives. J. Sulfur Chem. 2011, 32, 311–314. [Google Scholar] [CrossRef]
- Sayed, A.R.; Wiggins, J.S. Alternating copolymer of pyridine and 1,4-diphenyl-1,2,4,5-tetrazine from bis-1,3-dipolar cycloaddition polymerization. J. Appl. Poly. Sci. 2010, 120, 623–630. [Google Scholar] [CrossRef]
- Sayed, A.R. Synthesis of novel thiadiazoles and bis-thiadiazoles from carbonothioic dihydrazide. Tetrahedron Lett. 2010, 51, 4490–4493. [Google Scholar]
- Diganta, K.D.; Priyanka, G.; Smita, S. Salicylaldehyde phenylhydrazone: A new highly selective; fluorescent lead (II) probe. J. Fluoresc. 2013, 23, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Stephen, H.W.; Wilson, F.J. Some thiazole derivatives I. J. Chem. Soc. 1926, 129, 2531–2538. [Google Scholar] [CrossRef]
- Buu-Hoi, N.P.; Loc, T.B.; Xuong, N.D. The chemistry of thiocarbohydrazide. Bull. Soc. Chim. Fr. 1955, 694–697. [Google Scholar]
- El-Sayed, L.; El-Toukhy, A.; El Kady, N.; Iskander, M.F. Coordination compounds of hydrazine derivatives with transition metals. Part 21. Reaction of 1,5-thiocarbodihydrazones with nickel(II) and copper(II) salts. Trans. Metal Chem. 1980, 5, 293–297. [Google Scholar] [CrossRef]
- Filatov, M.A.; Baluschev, S.; Ilieva, I.Z.; Enkelmann, V.; Miteva, T.; Landfester, K.; Aleshchenkov, S.E.; Cheprakov, A.V. Tetraaryltetraanthra [2,3]porphyrins: Synthesis, structure, and optical properties. J. Org. Chem. 2012, 77, 11119–11131. [Google Scholar] [CrossRef] [PubMed]
- Nasr, T.; Bondock, S.; Eid, S. Design, synthesis, antimicrobial evaluation and molecular docking studies of some new thiophene, pyrazole and pyridone derivatives bearing sulfisoxazole moiety. Eur. J. Med. Chem. 2014, 84, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Finnerty, J.; Mitschke, U.; Wentrup, C. Linear ketenimines. Variable structures of C,C-dicyanoketenimines and C,C-bis-sulfonylketenimines. J. Org. Chem. 2002, 67, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Hassaneen, H.M.; Shawali, A.S.; Elwan, N.M.; Abounada, N.M. Reaction of 1-(2-naphthoyl) methyl-2-dimethylsulfonium bromide with N-nitroso-N-arylacetamides and reactions of the products with some nucleophiles. Sulfur Lett. 1992, 13, 273–285. [Google Scholar]
- Kappe, C.O.; Roschger, P. Synthesis and reactions of “Biginelli-compounds”. Part I. J. Heterocyc. Chem. 1989, 26, 55–64. [Google Scholar] [CrossRef]
- Okabe, T.; Taniguchi, E.; Maekawa, K. Ring cleaving reaction of thiadiazolo- and triazolopyrimidine derivatives. Bull. Chem. Soc. Jpn. 1974, 47, 2813–2817. [Google Scholar] [CrossRef]
- Shimizu, M.; Shimazaki, T.; Kon, Y.; Konakahara, T. S-Methylation of N-containing heterocyclic thiols with conjugated acids of methoxy groups. Heterocycles 2010, 81, 413–420. [Google Scholar] [CrossRef]
- Jackman, D.E.; Westphal, D.B. A new method for the synthesis of heterocyclic S-alkyl thiolactams. Synthesis 1987, 12, 1134–1136. [Google Scholar] [CrossRef]
- Hu, W.X.; Sun, N.; Yang, Z.Y. A Study on synthesis and anticancer activity of thiosemicarbazones. Chin. J. Med. Chem. 2001, 11, 129–134. [Google Scholar]
- Peak, C.S.; Youn, I.; Lee, Y.S. A facile, modified synthesis of aryl isothiocyanates from methyl N-aryldithiocarbamates. Synthesis 1982, 11, 969–970. [Google Scholar] [CrossRef]
- Bauer, W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. J. Clinic. Pathol. 1966, 45, 493–496. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, Approved Standard, 7th ed.; CLSI: Wayne, PA, USA, 2006. [Google Scholar]
- Youssef, M.M.; Al-Omair, M.A.; Ismail, M.A. Synthesis, DNA affinity, and antimicrobial activity of 4-substituted phenyl-2,2'-bichalcophenes and aza-analogues. Med. Chem. Res. 2012, 21, 4074–4082. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Sreejayan, N.; Rao, M.N.A. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. J. Pharm. Pharmacol. 1997, 49, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 3,4,9,10, 12–15, 20 and 24 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Omair, M.A.; Sayed, A.R.; Youssef, M.M. Synthesis of Novel Triazoles, Tetrazine, Thiadiazoles and Their Biological Activities. Molecules 2015, 20, 2591-2610. https://doi.org/10.3390/molecules20022591
Al-Omair MA, Sayed AR, Youssef MM. Synthesis of Novel Triazoles, Tetrazine, Thiadiazoles and Their Biological Activities. Molecules. 2015; 20(2):2591-2610. https://doi.org/10.3390/molecules20022591
Chicago/Turabian StyleAl-Omair, Mohammed A., Abdelwahed R. Sayed, and Magdy M. Youssef. 2015. "Synthesis of Novel Triazoles, Tetrazine, Thiadiazoles and Their Biological Activities" Molecules 20, no. 2: 2591-2610. https://doi.org/10.3390/molecules20022591






