1. Introduction
The majority of new drugs exhibit poor aqueous solubility, which affects their low bioavailability after oral delivery. Many strategies have been described to increase the dissolution rate of drugs by reducing their particle size and salt formation, using surfactants, cyclodextrins, liposomes or nanoparticles [
1,
2,
3,
4]. A relatively new approach for poorly soluble drugs is lipid-based formulations, particularly self-emulsifying drug delivery systems (SEDDS) [
5,
6].
SEDDS are isotropic mixtures of oils and surfactants with or without co-surfactants, which act as lipid-based formulations after oral application in aqueous gastrointestinal fluid and upon gentle agitation can form an oil-in-water emulsion [
7,
8,
9,
10]. SEDDS technology was employed to increase solubility and consequently the bioavailability of many poorly water soluble drugs such as phyllanthin, celastrol, ketoprofen, indomethacin and hydrocortisone [
11,
12,
13,
14].
SEDDS as liquid formulations have several disadvantages such as low drug loading capacity, drug leakage, low stability, and few choices of dosage forms. To overcome these limitations, liquid SEDDS (L-SEDDS) can be transformed to solid dosage forms by using different methods (filling capsules with liquid or semi-solid SEDDS, adsorption to solid carrier, melt granulation, spray drying, melt extrusion or nanoparticle formation) [
15,
16]. Solid self-emulsifying drug delivery systems (S-SEDDS) combine the advantages of liquid lipid formulations with those of solid dosage forms such as higher stability and longer period of storage [
17,
18]. S-SEDDS could be formulated in the form of self-emulsifying capsules, pellets/tablets, micro/nano-particles, suppositories or dry emulsions. One of the methods used for the conversion of L-SEDDS to S-SEDDS is spray drying. This technique allows preparation of the self-emulsifying dry emulsion by removing water from an emulsion containing a water-soluble solid carrier [
19,
20,
21].
Atorvastatin (ATR) inhibits 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, an enzyme found in liver tissue that plays a key role in cholesterol production [
22]. ATR is commonly used in hyperlipidemia and cardiovascular events. It is insoluble in pH < 4 and very slightly soluble in water and in pH 7.4 phosphate buffer (<1 mg/mL). Low oral bioavailability of ATR (only 12% after a 40 mg oral dose) is associated with its poor solubility in water and high (above 80%) presystemic clearance [
23,
24,
25]. Therefore, using different lipid carriers of ATR has been widely reported as a promising drug delivery system [
26,
27,
28,
29,
30]. As to our best knowledge there are no papers concerning solid SEDDS for ATR, the aim of this work was to design and to obtain both liquid and solid SEDDS from the same, optimized composition for solubility enhancement of ATR. To screen and to optimize the composition of L-SEDDS, solubility tests, pseudoternary phase diagrams, self-emulsifying grading tests, determination of percentage transmittance, refractive index and turbidity, droplet size and zeta potential analysis were performed. The optimized L-SEDDS were finally converted to S-SEDDS using the spray drying technique and characterized.
3. Experimental Section
3.1. Chemicals
Atorvastatin (ATR) was obtained as a gift from Biofarm (Poznań, Poland). Captex 200P and Caprol PGE-860 were gift samples from Abitec Corporation (Janesville, WI, USA). Capryol 90, Caprol MPGO, Lauroglycol FCC, Labrafil M19944CS and Labrasol were obtained from Gattefosse (Nanterre, France). Orthophosphoric acid, oleic acid, Span 20, Kolliphor®RH40, Tween 20, Tween 60, Tween 80 and polyoxyethylene glycol 400 (PEG 400) were purchased from Sigma Aldrich (Saint Louis, MO, USA). Rapeseed oil and Castor oil were obtained from Fagron (Kraków, Poland). Soja oil and Miglyol 812 were purchased from Caesar & Loretz GmbH (Hilden, Germany). Almond oil was obtained from Chempol (Wrocław, Poland). Linseed oil was purchased from Amara (Kraków, Poland). 1,2-propylene glycol was obtained from POCH (Gliwice, Poland). Potassium dihydrogen phosphate was obtained from Chempur (Piekary Śląskie, Poland). Lactochem® powder (lactose) was a gift from DFE pharma (Goch, Germany). HPLC-grade water was prepared by a Milli-Q Reagent Water System (Millipore, Billerica, MA, USA). Acetonitrile and methanol (Merck, Darmstadt, Germany) used in the present study were of HPLC-grade. All other reagents were of analytical grade.
3.2. Solubility Studies
The study was carried out by taking 1.0 mL of various solvents (oils, surfactants and co-surfactants) in a capped vial containing an excess amount (100 mg) of ATR. The mixtures were vortexed for 1 min to facilitate uniform dispersion, then were agitated with shaker for 30 min at 40 °C, and next for 48 h at room temperature. Afterwards, the samples were centrifuged at 3000 rpm for 15 min. The supernatant was collected and diluted with methanol. The concentration of dissolved ATR was determined by the HPLC method.
3.3. HPLC Analysis
The concentration of ATR was determined by the high-performance liquid chromatographic (HPLC) method. The system consists of Agilent Technologies 1260 Infinity with a UV detector (Agilent, Waldbronn, Germany). Isocratic separation was achieved on a Waters Spherisorb® ODS, 4.6 mm × 250 mm, 5 μm column (Waters Corporation, Milford, MA, USA). Mobile phase was acetonitrile/potassium dihydrogen phosphate buffer pH 3.0 (60:40; v/v), the flow rate was 1.0 mL/min and UV detection was performed at a wavelength of 247 nm. The column temperature was maintained at 25 °C. For the injection into the HPLC system, 20 μL of sample was used. The retention time of ATR was 5.03 min. Standard calibration curve was linear over the range of 5–100 μg/mL with the correlation coefficient (R2) 0.999.
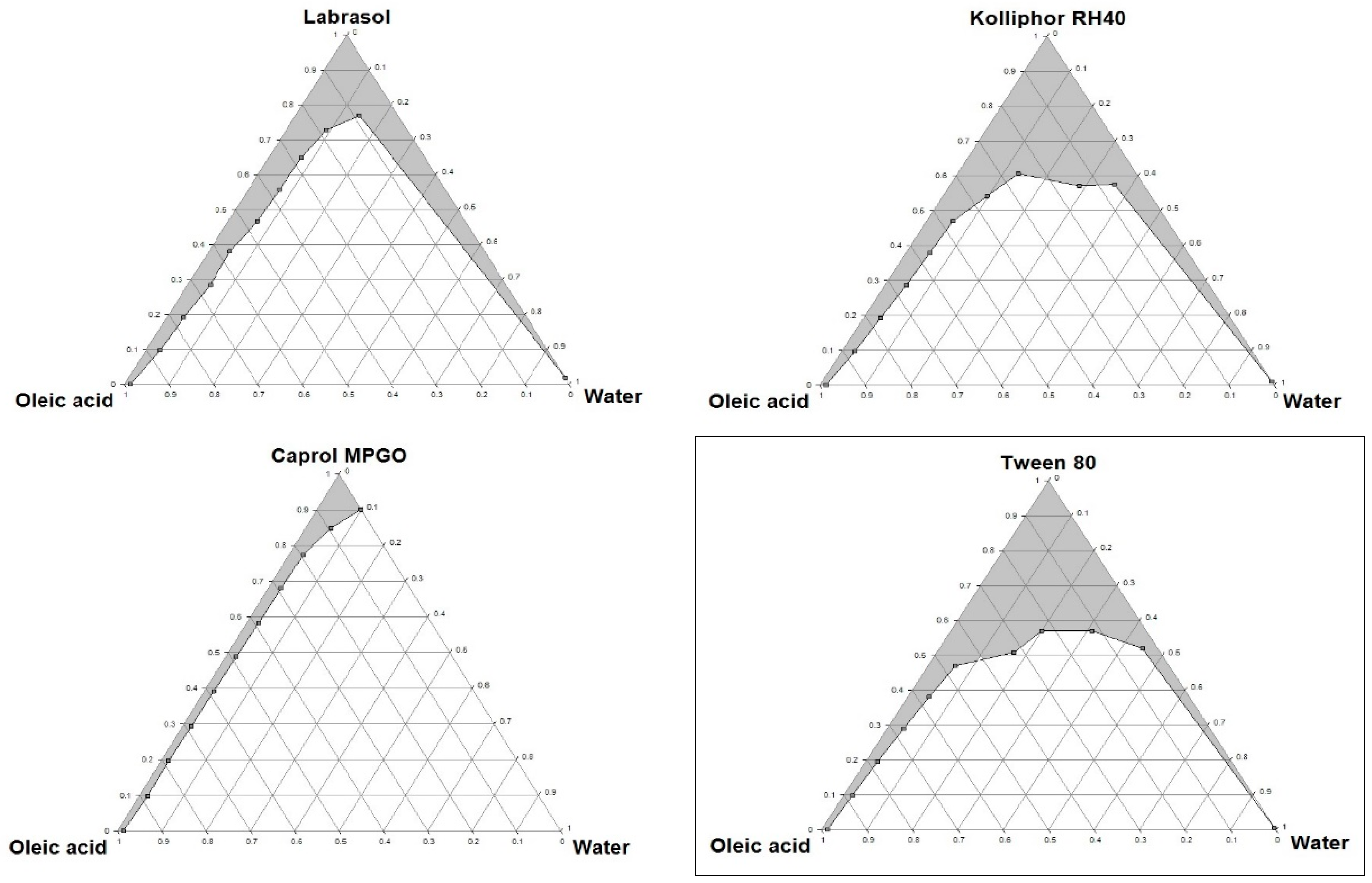
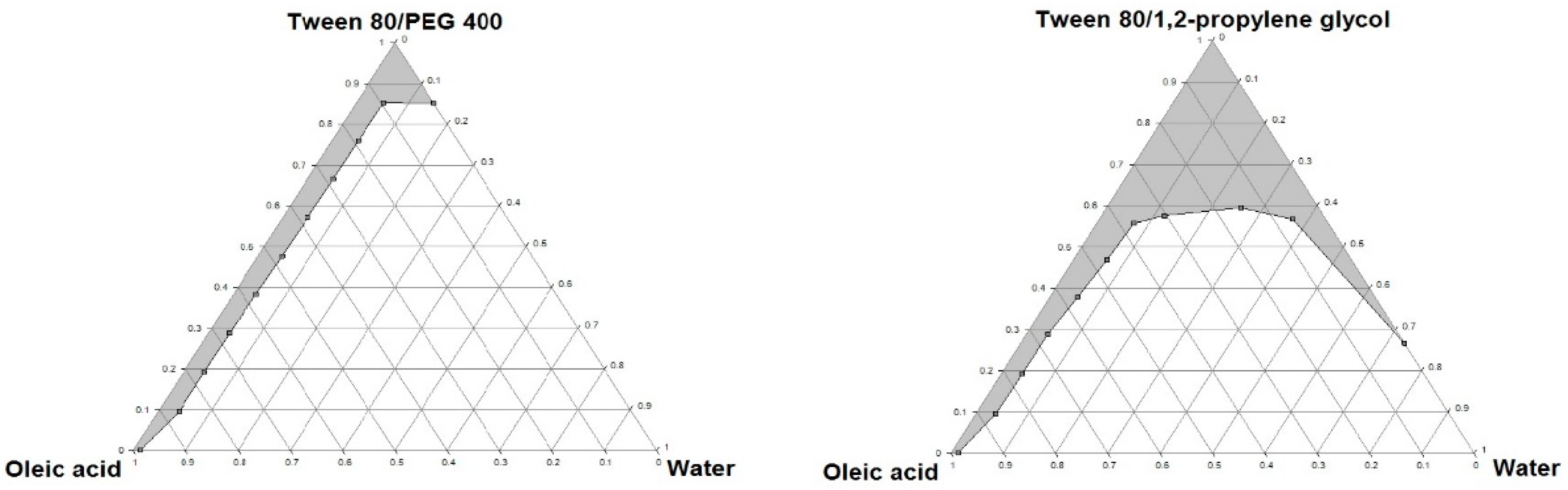
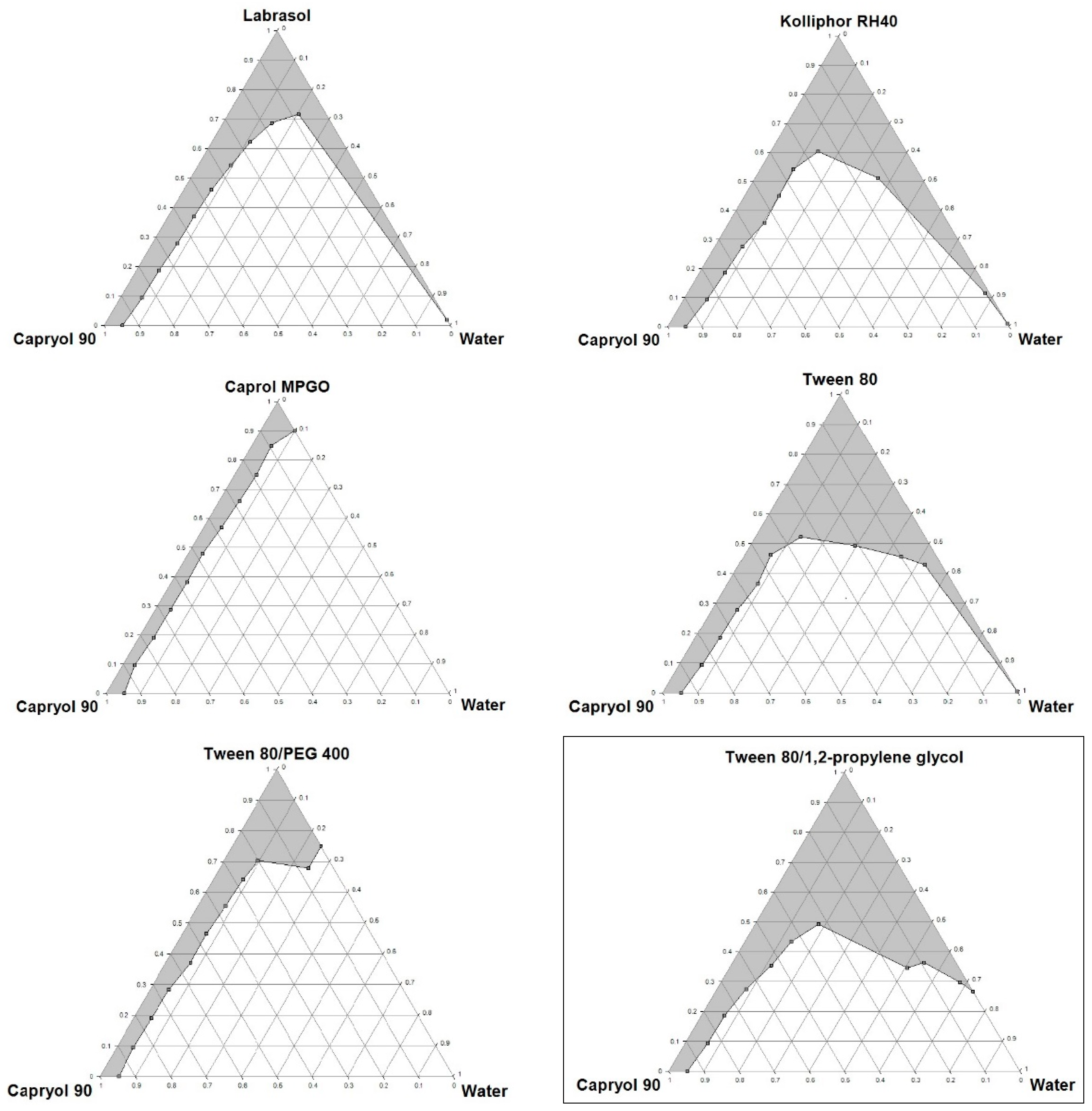
3.4. Construction of Ternary Phase Diagrams
Ternary phase diagrams were constructed for mixtures of oil, surfactant/co-surfactant and water at room temperature, using the water titration method. The mixtures of oil and surfactant/co-surfactant at certain weight ratios were diluted with water in a dropwise manner. For each phase diagram, oil and surfactant were mixed thoroughly in different weight ratios from 1:9 to 9:1 (1:9, 2:8, 3:7, 4:6, 5:5, 6:4, 7:3, 8:2, 9:1) in different glass vials. The homogenous mixture of oil and surfactant/co-surfactant was formed by vortexing for 5 min. Then each mixture was titrated with water and visually observed for phase clarity and flowability. The amount of water at which turbidity-to-transparency and transparency-to-turbidity transitions occurred was derived from the weight measurements. These values were then used to determine the boundaries of the microemulsion area corresponding to the values of oil and surfactant/co-surfactant. The phase diagrams were constructed using ProSim Ternary Diagram software (ProSim SA, Labege, France).
3.5. Preparation of Liquid SEDDS
L-SEDDS (
Table 2) were prepared by dissolving ATR in selected mixtures of oil and surfactant/co-surfactant. The amount of formulation should solubilize the whole drug dose (for ATR single dose is 10 mg). Hence, based on the solubility studies, 10 mg of ATR was dissolved in 1 g of formulations with oleic acid (OF1–OF10) and in 0.5 g of formulations with Capryol 90 (CF1–CF10). The mixtures were shaken and heated at 40 °C for a time necessary to dissolve the drug completely.
3.6. Characterization of Liquid SEDDS
3.6.1. Thermodynamic Stability and Phase Separation Study
SEDDS formulations were subjected to 3 freeze-thaw cycles, which included freezing at −18 °C for 24 h followed by thawing at 40 °C for 24 h. After centrifugation at 3000 rpm for 15 min, the formulations were observed for phase separation. Only stable formulations were selected for further experiments.
Phase separation study was assessed by exposing SEDDS formulations to 100 fold dilution with distilled water. Examined formulations were stored at 25 °C for 24 h and observed visually for phase separation and precipitation of drug.
3.6.2. Emulsification Study
The emulsification study was performed in a USP dissolution tester (DT 600 HH, Erweka, Heusenstamm, Germany). Each formulation (1 mL) was added to 100 mL distilled water maintained at 37 °C, with paddle rotating at 100 rpm for gentle agitation. The
in vitro performance of designed formulations was visually assessed by using the grading system as shown below [
7]:
- Grade A:
Rapidly forming (within 1 min) emulsion, with a clear or bluish appearance
- Grade B:
Rapidly forming (within 1 min), slightly less clear emulsion, with a bluish white appearance
- Grade C:
Fine milky emulsion that formed within 2 min
- Grade D:
Dull, greyish white emulsion having slightly oily appearance that is slow to emulsify (longer than 2 min)
- Grade E:
Formulation exhibiting either poor or minimal emulsification with large oil droplets on the surface
3.6.3. Determination of Percentage Transmittance, Refractive Index and Turbidity
The SEDDS formulations were diluted 100 times with water. The percentage of transmittance of the prepared emulsions was measured using UV spectrophotometer (Hitachi, Tokyo, Japan) keeping distilled water as blank at 630 nm. The refractive index was measured using Abbe’s refractometer (Atago, Tokyo, Japan). Turbidity of all formulations was studied using turbidimeter (Hach Lange, Düsseldorf, Germany), results were given as nephelometric turbidity unit (NTU).
3.6.4. Droplet Size and Zeta Potential Analysis
The particle size and zeta potential of obtained emulsions (after dilution 100 times with water) was determined by using Zetasizer Nano ZS90 (Malvern Instruments, Malvern, UK).
3.6.5. Drug Content in L-SEDDS
An amount of SEDDS equivalent to 10 mg of ATR was carefully weighted and placed in 100 mL volumetric flask containing methanol. After agitating for 24 h in a water bath (250 rpm), the extracted solution was analyzed for ATR content as described in
Subsection 3.3.
3.7. Preparation of Solid SEDDS
The S-SEDDS were prepared using the spray-drying technique using aBüchi Mini Spray Dryer B-290 apparatus (Büchi, Flawil, Switzerland). 10 g of lactose (Lactochem® powder) was dissolved in 100 mL distilled water and then the liquid SEDDS (10 g) was added. The solution was kept at 50 °C for 15 min to obtain an emulsion. The emulsion was spray dried under the conditions set during preliminary experiments: inlet temperature 60 °C; outlet temperature 40 °C; aspiration 100%, spray flow 550 L/h; feeding rate of the emulsion 4 mL/min.
3.8. Characterization of Solid SEDDS
3.8.1. Drug Content in S-SEDDS
ATR loading in S-SEDDS was determined by dissolving accurately weighted amount of dried emulsion (50 mg) in 10 mL of methanol and agitating for 24 h at 250 rpm in a water bath. After filtration using a 0.45 μm cellulose acetate membrane filter (Chromafil
®, Düren, Germany), the concentration of ATR was determined as described in
Subsection 3.3. ATR encapsulation efficiency (EE %) in S-SEDDS was calculated by the formula: EE = Q
a/Q
t × 100, where: EE—percentage of encapsulation efficiency, Q
a—actual drug content, Q
t—theoretical drug content [
38].
3.8.2. Morphological Analysis
The morphological features of solid atorvastatin-SEDDS were assessed by scanning electron microscope (SEM) (Hitachi S4200, Tokyo, Japan). Before imaging samples were sputter-coated with gold.
3.8.3. Droplet Size Determination
Droplet size of emulsions obtained from solid SEDDS was determined by using Zetasizer Nano ZS90 as described in
Subsubsection 3.6.4.
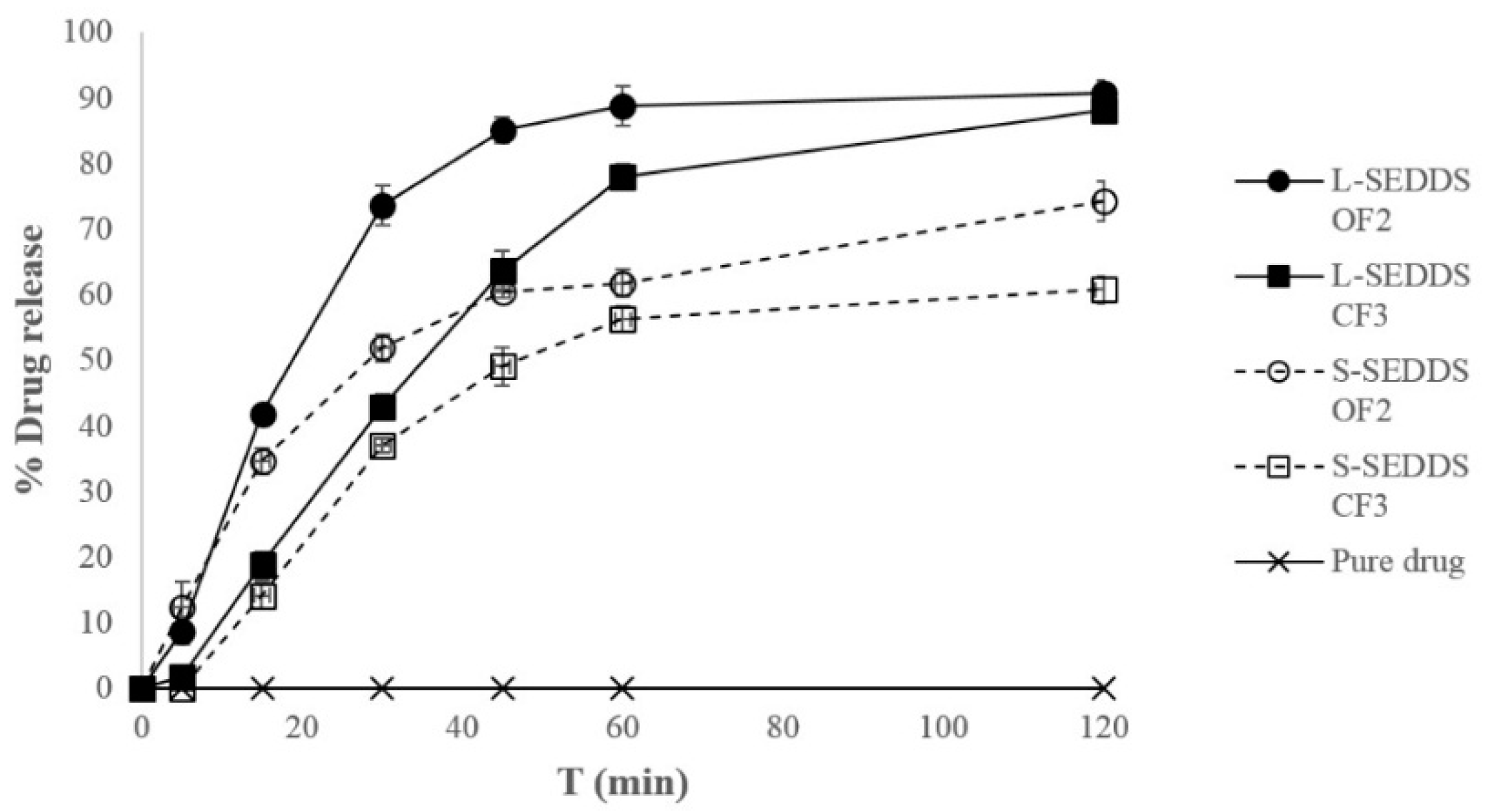
3.9. In Vitro Dissolution Study
For the
in vitro dissolution study USP apparatus type I (Erweka Dissolution tester type DT 600HH, Heusenstamm, Germany) was used. Selected liquid and solid SEDDS formulations equivalent to 10 mg of ATR and only 10 mg of ATR powder were filled in hard gelatin capsules (size 00) and were undertaken for dissolution study. 300 mL of water maintained at 37 °C and stirred at 100 rpm was used as dissolution medium. Dissolution samples (5 mL) were withdrawn at predetermined time intervals and replaced with an equivalent amount of fresh water. Samples were filtered through a 0.45 μm nylon membrane filter (Chromafil
®) and concentrations of ATR were determined by HPLC method as described in
Subsection 3.3.
3.10. Statistical Analysis
Results are expressed as the mean and standard deviation. The data were statistically analyzed using the Mann-Whitney test (Statistica 10.0 software, StatSoft, Tulsa, OK, USA). The level of significance was accepted with p < 0.05.
4. Conclusions
Liquid SEDDS for ATR with oleic acid or Capryol 90 as oily phase, Tween 80 as surfactant and 1,2-propylene glycol as co-surfactant were developed. Based on the thermodynamic stability test, phase separation, emulsification, percentage transmittance, refractive index, turbidity, droplet size and zeta potential studies, two optimal compositions of L-SEDDS were selected—OF2 and CF3. The optimized liquid atorvastatin-SEDDS were finally successfully converted, using the spray-drying technique, to S-SEDDS. Despite the fact that liquid formulation CF3 was characterized by lower turbidity, higher percentage transmittance and better self-emulsifying properties, based on the in vitro dissolution study, it can be concluded that better solubilization properties were exhibited by solid formulation OF2. Obtained S-SEDDS can be formed into solid dosage forms, as multicompartment tablets or capsules, but further studies are needed.