Variation in Phenolics, Flavanoids, Antioxidant and Tyrosinase Inhibitory Activity of Peach Blossoms at Different Developmental Stages
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Phenolics and Flavanoids
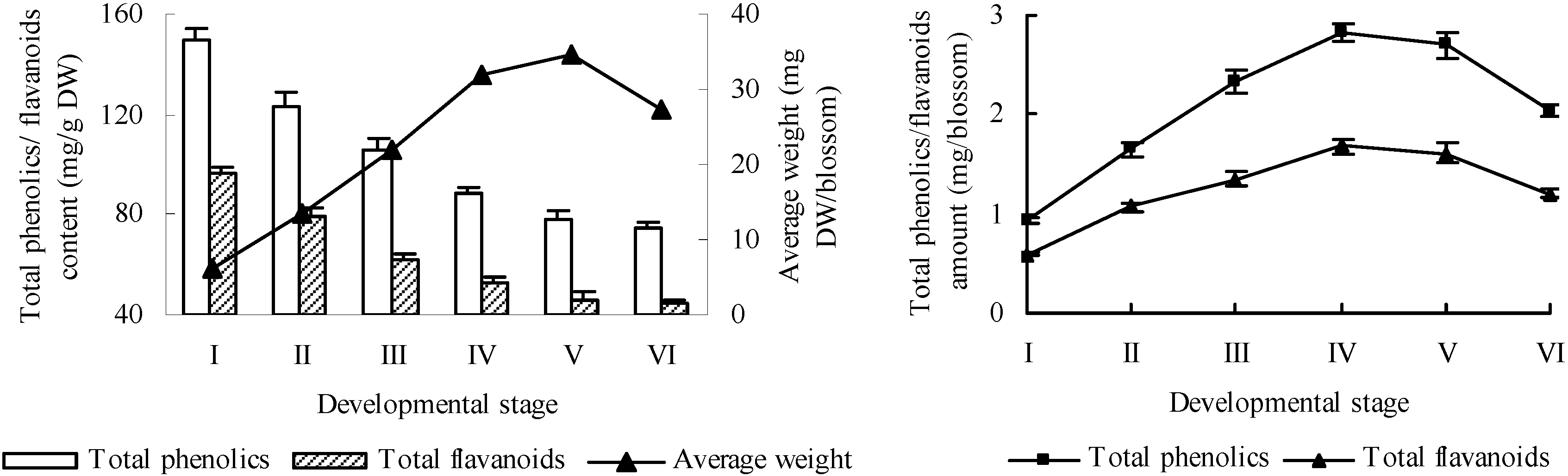
2.2. High Performance Liquid Chromatography (HPLC) Analysis of Selected Phenolic Compounds

| Developmental Stage | I | II | III | IV | V | VI |
|---|---|---|---|---|---|---|
| Chlorogenic acid | 72.87 ± 3.41 a | 59.58 ± 3.67 b | 44.21 ± 1.79 c | 37.32 ± 2.30 d | 32.53 ± 2.85 de | 30.67 ± 3.13 e |
| Quercetin-3-O-rhamnoside | 3.72 ± 0.30 ab | 4.00 ± 0.18 a | 4.00 ± 0.26 a | 3.76 ± 0.14 ab | 3.36 ± 0.26 bc | 3.07 ± 0.21 c |
| Kaempferol-3-O-galactoside | 9.96 ± 0.43 a | 10.08 ± 0.52 a | 8.45 ± 0.35 b | 7.50 ± 0.46 c | 6.52 ± 0.29 d | 7.05 ± 0.23 cd |
| Quercetin-3-O-galactoside | 0.80 ± 0.05 a | 0.89 ± 0.05 a | 0.86 ± 0.02 a | 0.83 ± 0.04 a | 0.81 ± 0.03 a | 0.84 ± 0.03 a |
| Kaempferol-4-O-glucoside | 5.57 ± 0.22 b | 5.86 ± 0.19 b | 5.62 ± 0.31 b | 5.61 ± 0.28 b | 5.99 ± 0.17 b | 6.96 ± 0.33 a |
| Cinnamic acid | 1.48 ± 0.10 a | 1.39 ± 0.06 a | 1.13 ± 0.03 b | 0.98 ± 0.03 b | 0.82 ± 0.05 c | 0.82 ± 0.02 c |
2.3. Antioxidant Activity
2.3.1. Scavenging Capacity towards Superoxide Anion Radicals

2.3.2. Scavenging Capacity towards Hydroxyl Radicals

2.3.3. Scavenging Capacity towards DPPH Radicals

2.4. Tyrosinase Inhibitory Activity

2.5. Correlation Analysis
| ACW | ACL | HR | DPPH | TI | |
|---|---|---|---|---|---|
| Total phenolics | 0.9795 ** | 0.9875 ** | 0.9593 ** | 0.9372 ** | 0.7242 |
| Total flavanoids | 0.9775 ** | 0.9845 ** | 0.9608 ** | 0.9302 ** | 0.7124 |
| Chlorogenic acid | 0.9856 ** | 0.9795 ** | 0.9496 ** | 0.9285 ** | 0.6995 |
| Quercetin-3-O-rhamnoside | 0.4525 | 0.6602 | 0.8037 * | 0.7063 | 0.8648 * |
| Kaempferol-3-O-galactoside | 0.8825 ** | 0.9510 ** | 0.9585 ** | 0.8369 * | 0.6570 |
| Quercetin-3-O-galactoside | −0.1236 | 0.1176 | 0.2104 | −0.0640 | 0.0501 |
| Kaempferol-4-O-glucoside | −0.4788 | −0.5769 | −0.7043 | −0.7862 * | −0.9560 ** |
| Cinnamic acid | 0.9333 ** | 0.9802 ** | 0.9851 ** | 0.9136 ** | 0.7379 |
| TI | 0.6282 | 0.7427 | 0.8368 * | 0.9001 ** | |
| DPPH | 0.9011 ** | 0.9355 ** | 0.9435 ** | ||
| HR | 0.8899 ** | 0.9652 ** | |||
| ACL | 0.9509 ** |
3. Experimental Section
3.1. Materials
3.2. Preparation of Peach Blossom Extracts
3.3. Determination of Total Phenolic Content
3.4. Determination of Total Flavonoid Content
3.5. High Performance Liquid Chromatography (HPLC) Analysis of Selected Phenolic Compounds
3.6. Photochem Assay
3.7. DPPH Assay
3.8. Hydroxyl Radical Assay
3.9. Tyrosinase Inhibition Assay
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kohen, R.; Nyska, A. Oxidation of biological systems: Oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification. Toxicol. Pathol. 2002, 30, 620–650. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell. Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Comporti, M. Three models of free radical-induced cell injury. Chem. Biol. Interact. 1989, 72, 1–56. [Google Scholar] [CrossRef]
- Liochev, S.I. Reactive oxygen species and the free radical theory of aging. Free Radical Biol. Med. 2013, 60, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Aruoma, O.I. Free radicals, oxidative stress, and antioxidants in human health and disease. J. Am. Oil Chem. Soc. 1998, 75, 199–212. [Google Scholar] [CrossRef]
- Gordon, M.H. Dietary antioxidants in disease prevention. Nat. Prod. Rep. 1996, 13, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Koo, N.; Min, D.B. Reactive oxygen species, aging, and antioxidative nutraceuticals. Compr. Rev. Food Sci. Food Saf. 2004, 3, 21–33. [Google Scholar] [CrossRef]
- Gordon, M.H. Significance of dietary antioxidants for health. Int. J. Mol. Sci. 2012, 13, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Melnyk, J.P.; Tsao, R.; Marcone, M.F. How natural dietary antioxidants in fruits, vegetables and legumes promote vascular health. Food Res. Int. 2011, 44, 14–22. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.D.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Ferrer, A.; Rodrıguez-Lopez, J.N.; Garcia-Canovas, F.; Garcia-Carmona, F. Tyrosinase: A comprehensive review of its mechanism. Biochim. Biophys. Acta 1995, 1247, 1–11. [Google Scholar] [CrossRef]
- Hearing, V.J.; Tsukamoto, K. Enzymatic control of pigmentation in mammals. FASEB J. 1991, 5, 2902–2909. [Google Scholar] [PubMed]
- Costin, G.E.; Hearing, V.J. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. 2007, 21, 4976–4994. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M. Food browning and its prevention: An overview. J. Agric. Food Chem. 1996, 44, 631–653. [Google Scholar] [CrossRef]
- Martinez, M.V.; Whitaker, J.R. The biochemistry and control of enzymatic browning. Trends Food Sci. Technol. 1995, 6, 195–200. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Tundis, R.; Menichini, F. Natural and synthetic tyrosinase inhibitors as antibrowning agents. Compr. Rev. Food Sci. Food Saf. 2012, 11, 378–398. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Uyama, H. Tyrosinase inhibitors from natural and synthetic sources: Structure, inhibition mechanism and perspective for the future. Cell. Mol. Life Sci. 2005, 62, 1707–1723. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.-S.; Bae, H. Naturally occurring tyrosinase inhibitors: Mechanism and applications in skin health, cosmetics and agriculture industries. Phytother. Res. 2007, 21, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.-S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Khan, M.T.H. Flavonoid derivatives as potent tyrosinase inhibitors—A survey of recent findings between 2008 and 2013. Curr. Top. Med. Chem. 2014, 14, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.T.; Kim, B.J.; Kim, J.H.; Heo, M.Y.; Kim, H.P. Biological screening of 100 plant extracts for cosmetic use (I): Inhibitory activities of tyrosinase and DOPA auto-oxidation. Int. J. Cosmet. Sci. 1997, 19, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Dung, N.T.; Bajpai, V.K.; Rahman, A.; Yoon, J.I.; Kang, S.C. Phenolic contents, antioxidant and tyrosinase inhibitory activities of Lonicera japonica Thumb. J. Food Biochem. 2011, 35, 148–160. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Lim, Y.Y.; Wong, L.F.; Lianto, F.S.; Wong, S.K.; Lim, K.K.; Joe, C.E.; Lim, T.Y. Antioxidant and tyrosinase inhibition properties of leaves and rhizomes of ginger species. Food Chem. 2008, 109, 477–483. [Google Scholar] [CrossRef]
- Chang, L.W.; Juang, L.J.; Wang, B.S.; Wang, M.Y.; Tai, H.M.; Hung, W.J.; Chen, Y.J.; Huang, M.H. Antioxidant and antityrosinase activity of mulberry (Morus alba L.) twigs and root bark. Food Chem. Toxicol. 2011, 49, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Watanabe, Y.; Kasai, K.; Yamakoshi, J.; Koga, T. Inhibitory effect of an ellagic acid-rich pomegranate extract on tyrosinase activity and ultraviolet-induced pigmentation. Biosci. Biotechnol. Biochem. 2005, 69, 2368–2373. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.-H.; Yuk, H.-G.; Lee, J.-H.; Kim, J.-W.; Kim, R.; Lee, S.-C. Antioxidant, tyrosinase inhibitory, and acetylcholinesterase inhibitory activities of green tea (Camellia sinensis L.) seed and its pericarp. Food Sci. Biotechnol. 2012, 21, 761–768. [Google Scholar] [CrossRef]
- Park, J.W.; Yuk, H.G.; Lee, S.C. Antioxidant and tyrosinase inhibitory activities of different parts of oriental cherry (Prunus serrulata var. spontanea). Food Sci. Biotechnol. 2012, 21, 339–343. [Google Scholar] [CrossRef]
- Maisuthisakul, P.; Gordon, M.H. Antioxidant and tyrosinase inhibitory activity of mango seed kernel by product. Food Chem. 2009, 117, 332–341. [Google Scholar] [CrossRef]
- Reig, G.; Iglesias, I.; Gatius, F.; Alegre, S. Antioxidant capacity, quality, and anthocyanin and nutrient contents of several peach cultivars [Prunus persica (L.) Batsch] grown in spain. J. Agric. Food Chem. 2013, 61, 6344–6357. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.I.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Kader, A.A. Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. J. Agric. Food Chem. 2002, 50, 4976–4982. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, M.-H. Antioxidant activity of peach blossom extracts. J. Korean Soc. Appl. Biol. Chem. 2011, 54, 46–53. [Google Scholar] [CrossRef]
- Liu, J.C.; Zhang, C.L.; Lv, Z.Z.; Liu, H.; Jiao, Z.G. Extraction of total phenolics and flavanoids from peach blossoms and their antioxidant activity. J. Food Saf. Quality 2013, 4, 1750–1756. [Google Scholar]
- Liu, J.C.; Zhang, Q.L.; Jiao, Z.G.; Zhang, C.L.; Lv, Z.Z.; Liu, H.; Wang, S.X. Inhibitory effect of peach flower extract on tyrosinase and its kinetics analysis. J. Fruit Sci. 2014, 31, 836–841. [Google Scholar]
- Schmitzer, V.; Mikulic-Petkovsek, M.; Stampar, F. Sepal phenolic profile during Helleborus niger flower development. J. Plant Physiol. 2013, 170, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Xu, F.; Sun, X.; Bao, J.; Beta, T. Phenolic acids, anthocyanins, and antioxidant capacity in rice (Oryza sativa L.) grains at four stages of development after flowering. Food Chem. 2014, 143, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyüre, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (IUPAC technical report). Pure Appl. Chem. 2013, 85, 957–998. [Google Scholar] [CrossRef]
- Niki, E. Assessment of antioxidant capacity in vitro and in vivo. Free Radical Biol. Med. 2010, 49, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.F.; Li, S.Y.; Zheng, Y.M.; Dong, L. The substitute substrate for soilless culture of potted ornamental peach. Hunan Agric. Sci. 2012, 7, 119–121, 125. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method. Enzymol. 1999, 299, 152–178. [Google Scholar]
- Yang, L.; Cao, Y.L.; Jiang, J.G.; Lin, Q.S.; Chen, J.; Zhu, L. Response surface optimization of ultrasound-assisted flavonoids extraction from the flower of Citrus aurantium L. var. amara. Engl. J. Sep. Sci. 2010, 33, 1349–1355. [Google Scholar] [PubMed]
- Towler, M.J.; Weathers, P.J. Variations in key artemisinic and other metabolites throughout plant development in Artemisia annua L. for potential therapeutic use. Ind. Crop. Prod. 2015, 67, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.P.; Aradhya, S.M. Chemical changes and antioxidant activity in pomegranate arils during fruit development. Food Chem. 2005, 93, 319–324. [Google Scholar] [CrossRef]
- Shi, G.A.; Guo, X.F.; Fan, B.Y.; Bao, M.Z. Changes of antioxidative activity during florescence and flower senescence of peonies petal. Acta Hort. Sin. 2009, 36, 1685–1690. [Google Scholar]
- Wang, S.X.; Liu, J.C.; Jiao, Z.G.; Zhang, S.N.; Yang, L. Changes of polyphenols during fruit development in apples. J. Fruit Sci. 2003, 20, 427–431. [Google Scholar]
- Wei, Z.-F.; Jin, S.; Luo, M.; Pan, Y.-Z.; Li, T.-T.; Qi, X.-L.; Efferth, T.; Fu, Y.-J.; Zu, Y.-G. Variation in contents of main active components and antioxidant activity in leaves of different pigeon pea cultivars during growth. J. Agric. Food Chem. 2013, 61, 10002–10009. [Google Scholar] [CrossRef] [PubMed]
- Pegg, R.B.; Amarowicz, R.; Naczk, M.; Shahidi, F. PHOTOCHEM® for determination of antioxidant capacity of plant extracts. ACS Sym. Ser. 2007, 956, 140–158. [Google Scholar]
- Besco, E.; Braccioli, E.; Vertuan, S.; Ziosi, P.; Brazzo, F.; Bruni, R.; Sacchetti, G.; Manfredini, S. The use of photochemiluminescence for the measurement of the integral antioxidant capacity of baobab products. Food Chem. 2007, 102, 1352–1356. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, X.; Zhang, Y.; Wang, L.; Zhang, R. In vitro antioxidant properties of different parts of pomegranate flowers. Food Bioprod. Process. 2011, 89, 234–240. [Google Scholar] [CrossRef]
- Jo, Y.-H.; Seo, G.-U.; Yuk, H.-G.; Lee, S.-C. Antioxidant and tyrosinase inhibitory activities of methanol extracts from Magnolia denudata and Magnolia denudata var. purpurascens flowers. Food Res. Int. 2012, 47, 197–200. [Google Scholar] [CrossRef]
- Vallisuta, O.; Nukoolkarn, V.; Mitrevej, A.; Sarisuta, N.; Leelapornpisid, P.; Phrutivorapongkul, A.; Sinchaipanid, N. In vitro studies on the cytotoxicity, and elastase and tyrosinase inhibitory activities of marigold (Tagetes erecta L.) flower extracts. Exp. Ther. Med. 2014, 7, 246–250. [Google Scholar] [PubMed]
- Nithitanakool, S.; Pithayanukul, P.; Bavovada, R.; Saparpakorn, P. Molecular docking studies and anti-tyrosinase activity of thai mango seed kernel extract. Molecules 2009, 14, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhao, X.; Sun, C.; Li, X.; Chen, K. Phenolic composition from different loquat (Eriobotrya japonica Lindl.) cultivars grown in China and their antioxidant properties. Molecules 2015, 20, 542–555. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.M.; Guo, X.; Fu, X.; Zhou, L.; Chen, Y.; Zhu, Y.; Yan, H.; Liu, R.H. Comparative assessment of phenolic content and in vitro antioxidant capacity in the pulp and peel of mango cultivars. Int. J. Mol. Sci. 2015, 16, 13507–13527. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Cao, J.; Jiang, W. Evaluation and comparison of vitamin C, phenolic compounds, antioxidant properties and metal chelating activity of pulp and peel from selected peach cultivars. LWT-Food Sci. Technol. 2015, 63, 1042–1048. [Google Scholar] [CrossRef]
- Xiong, L.N.; Yang, J.J.; Jiang, Y.R.; Lu, B.Y.; Hu, Y.Z.; Zhou, F.; Mao, S.Q.; Shen, C.X. Phenolic compounds and antioxidant capacities of 10 common edible flowers from China. J. Food Sci. 2014, 79, C517–C525. [Google Scholar] [CrossRef] [PubMed]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Lin, H.M.; Chen, X.; Xie, J.; Wang, P. Mechanochemical-assisted extraction and antioxidant activities of kaempferol glycosides from Camellia oleifera Abel. meal. J. Agric. Food Chem. 2011, 59, 3986–3993. [Google Scholar] [CrossRef] [PubMed]
- Tseye-Oidov, O.; Mkami, I.; Watanabe, J.; Tsushida, T.; Demberel, B.; Kimura, T.; Ide, T. Antioxidant capacities and total quercetin content of several species of polygonaceae in Mongolia. Food Sci. Technol. Res. 2010, 16, 169–174. [Google Scholar] [CrossRef]
- Sevgi, K.; Tepe, B.; Sarikurkcu, C. Antioxidant and DNA damage protection potentials of selected phenolic acids. Food Chem. Toxicol. 2015, 77, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Oskoueian, E.; Abdullah, N.; Hendra, R.; Karimi, E. Bioactive compounds, antioxidant, xanthine oxidase inhibitory, tyrosinase inhibitory and anti-inflammatory activities of selected agro-industrial by-products. Int. J. Mol. Sci. 2011, 12, 8610–8625. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Ng, C.C.; Lin, H.T.; Shyu, Y.T. Free radical-scavenging and tyrosinase-inhibiting activities of extracts from sorghum distillery residue. J. Biosci. Bioeng. 2011, 111, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Yamashita, D.; Takeda, Y.; Yonemori, S. Screening for tyrosinase inhibitors among extracts of seashore plants and identification of potent inhibitors from Garcinia subelliptica. Biosci. Biotechnol. Biochem. 2005, 69, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Smirnoff, N.; Cumbes, Q.J. Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 1989, 28, 1057–1060. [Google Scholar] [CrossRef]
- Hsu, C.-K.; Chang, C.-T.; Lu, H.-Y.; Chung, Y.-C. Inhibitory effects of the water extracts of Lavendula sp. on mushroom tyrosinase activity. Food Chem. 2007, 105, 1099–1105. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the peach blossom are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.-C.; Jiao, Z.-G.; Yang, W.-B.; Zhang, C.-L.; Liu, H.; Lv, Z.-Z. Variation in Phenolics, Flavanoids, Antioxidant and Tyrosinase Inhibitory Activity of Peach Blossoms at Different Developmental Stages. Molecules 2015, 20, 20460-20472. https://doi.org/10.3390/molecules201119709
Liu J-C, Jiao Z-G, Yang W-B, Zhang C-L, Liu H, Lv Z-Z. Variation in Phenolics, Flavanoids, Antioxidant and Tyrosinase Inhibitory Activity of Peach Blossoms at Different Developmental Stages. Molecules. 2015; 20(11):20460-20472. https://doi.org/10.3390/molecules201119709
Chicago/Turabian StyleLiu, Jie-Chao, Zhong-Gao Jiao, Wen-Bo Yang, Chun-Ling Zhang, Hui Liu, and Zhen-Zhen Lv. 2015. "Variation in Phenolics, Flavanoids, Antioxidant and Tyrosinase Inhibitory Activity of Peach Blossoms at Different Developmental Stages" Molecules 20, no. 11: 20460-20472. https://doi.org/10.3390/molecules201119709






