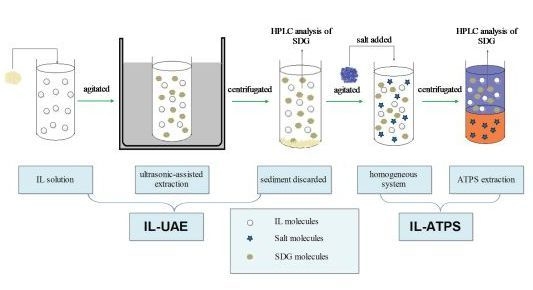
Ionic Liquid-Based Ultrasonic-Assisted Extraction of Secoisolariciresinol Diglucoside from Flaxseed (Linum usitatissimum L.) with Further Purification by an Aqueous Two-Phase System
Abstract
:1. Introduction
2. Results and Discussion
2.1. Ionic Liquid-Based Ultrasonic-Assisted Extraction (IL-UAE)
2.1.1. Selection of IL
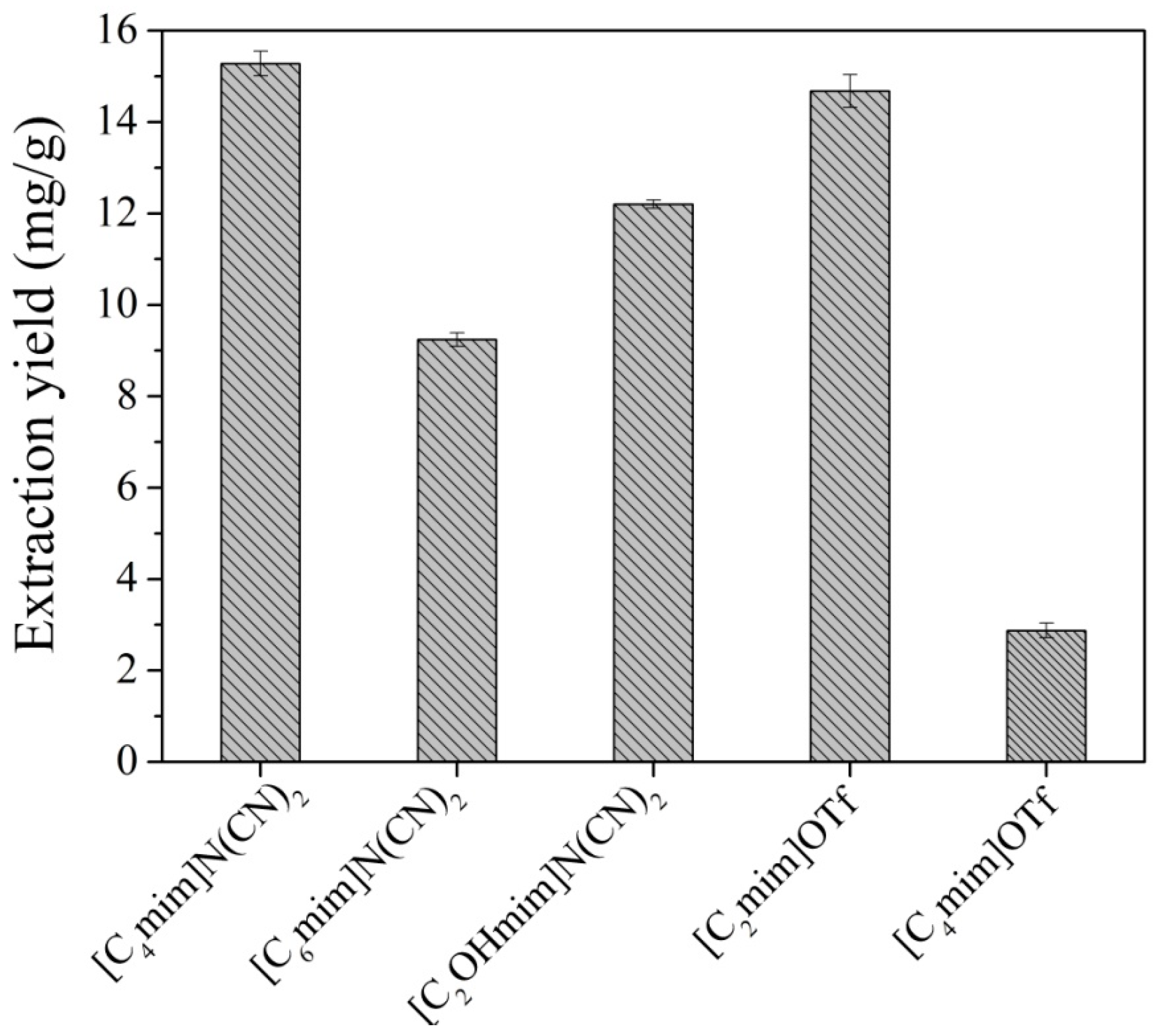
2.1.2. Single Factor Experiments
Effect of IL Concentration
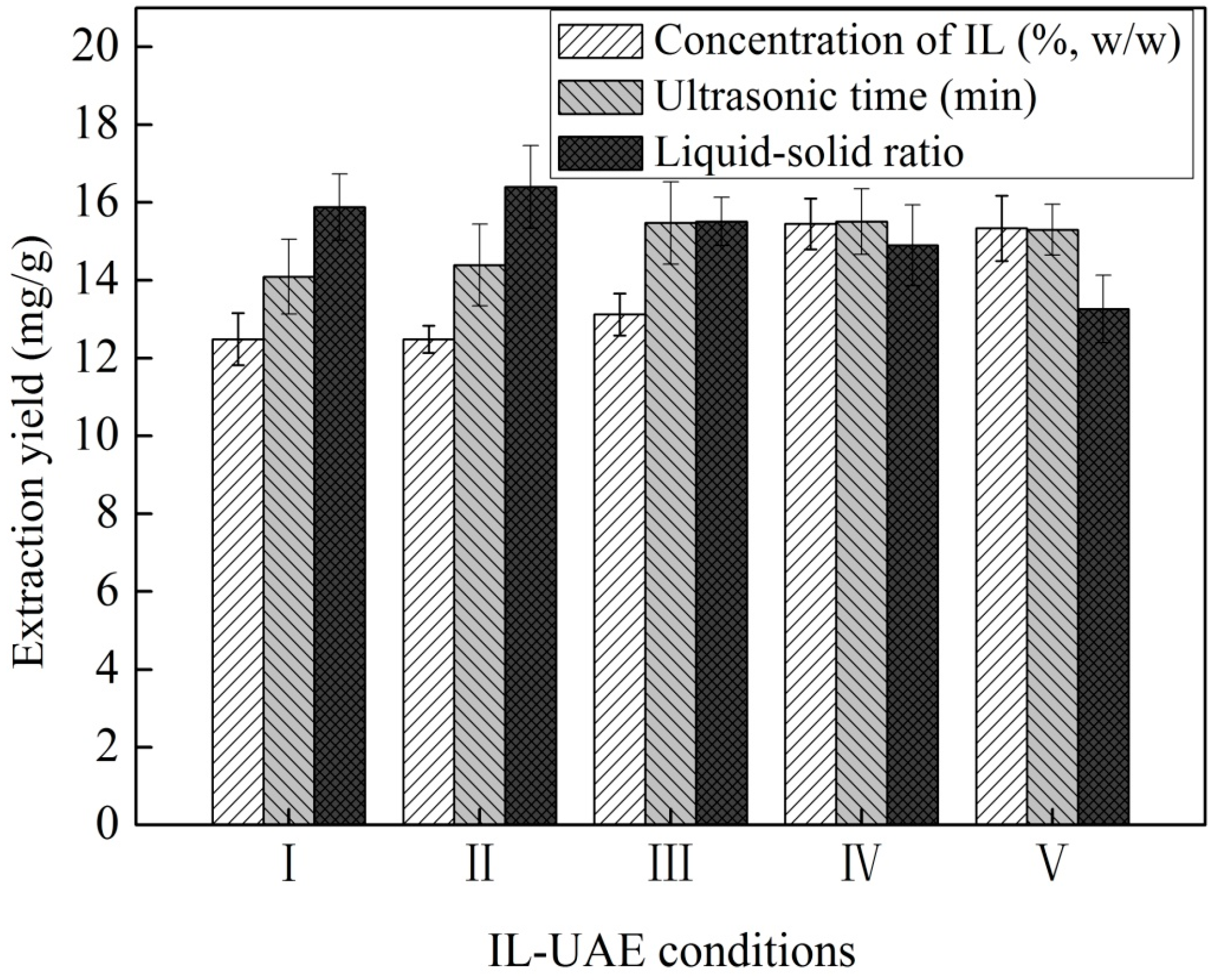
Effect of Liquid–Solid Ratio
Effect of Ultrasonic Irradiation Time
2.1.3. Optimization of IL-UAE Conditions by the Response Surface Method (RSM)
| Run | Factor A: IL Concentration (%, w/w) | Factor B: Liquid–Solid Ratio | Factor C:Ultrasonic Irradiation Time (min) | Response Average Extraction Yield (mg/g) |
|---|---|---|---|---|
| 1 | 60 | 10:1 | 50 | 12.5521 |
| 2 | 50 | 20:1 | 50 | 15.7939 |
| 3 | 50 | 20:1 | 50 | 15.9827 |
| 4 | 60 | 30:1 | 50 | 15.4370 |
| 5 | 50 | 10:1 | 40 | 13.7683 |
| 6 | 40 | 30:1 | 50 | 12.0817 |
| 7 | 50 | 30:1 | 60 | 15.3657 |
| 8 | 50 | 10:1 | 60 | 14.5096 |
| 9 | 50 | 30:1 | 40 | 15.5716 |
| 10 | 40 | 10:1 | 50 | 12.6094 |
| 11 | 60 | 20:1 | 60 | 15.5444 |
| 12 | 60 | 20:1 | 40 | 15.7341 |
| 13 | 50 | 20:1 | 50 | 15.8536 |
| 14 | 50 | 20:1 | 50 | 15.7837 |
| 15 | 40 | 20:1 | 40 | 14.3129 |
| 16 | 50 | 20:1 | 50 | 15.5716 |
| 17 | 40 | 20:1 | 60 | 14.9624 |
| Source | Degrees of Freedom | Sum of Squares | Mean Square | F-value | p-value Prob > F |
|---|---|---|---|---|---|
| Model | 9 | 26.15 | 2.91 | 65.53 | <0.0001 |
| A | 1 | 3.51 | 3.51 | 79.23 | <0.0001 |
| B | 1 | 3.15 | 3.15 | 70.95 | <0.0001 |
| C | 1 | 0.12 | 0.12 | 2.79 | 0.1386 |
| AB | 1 | 2.91 | 2.91 | 65.66 | <0.0001 |
| AC | 1 | 0.18 | 0.18 | 3.97 | 0.0865 |
| BC | 1 | 0.22 | 0.22 | 5.06 | 0.0595 |
| A2 | 1 | 5.53 | 5.53 | 124.76 | <0.0001 |
| B2 | 1 | 9.23 | 9.23 | 208.24 | <0.0001 |
| C2 | 1 | 1.00 | 1.00 | 22.57 | 0.0021 |
| Residual | 7 | 0.31 | 0.044 | ||
| Lack of fit | 3 | 0.22 | 0.074 | 3.33 | 0.1376 |
| Pure Error | 4 | 0.089 | 0.022 | ||
| Cor total | 16 | 26.46 |


2.1.4. Comparison with Other Extraction Methods
| Extraction Methods | HRE | MAE | UAE | ||
|---|---|---|---|---|---|
| Time (min) | 20 | 40 | 60 | 40 | 40 |
| Extraction yield (mg/g) | 2.6953 | 9.6581 | 12.6324 | 16.1263 | 16.0854 |
2.2. Ionic Liquid-Based Aqueous Two-Phase System (IL-ATPS)
2.2.1. Effect of Salt
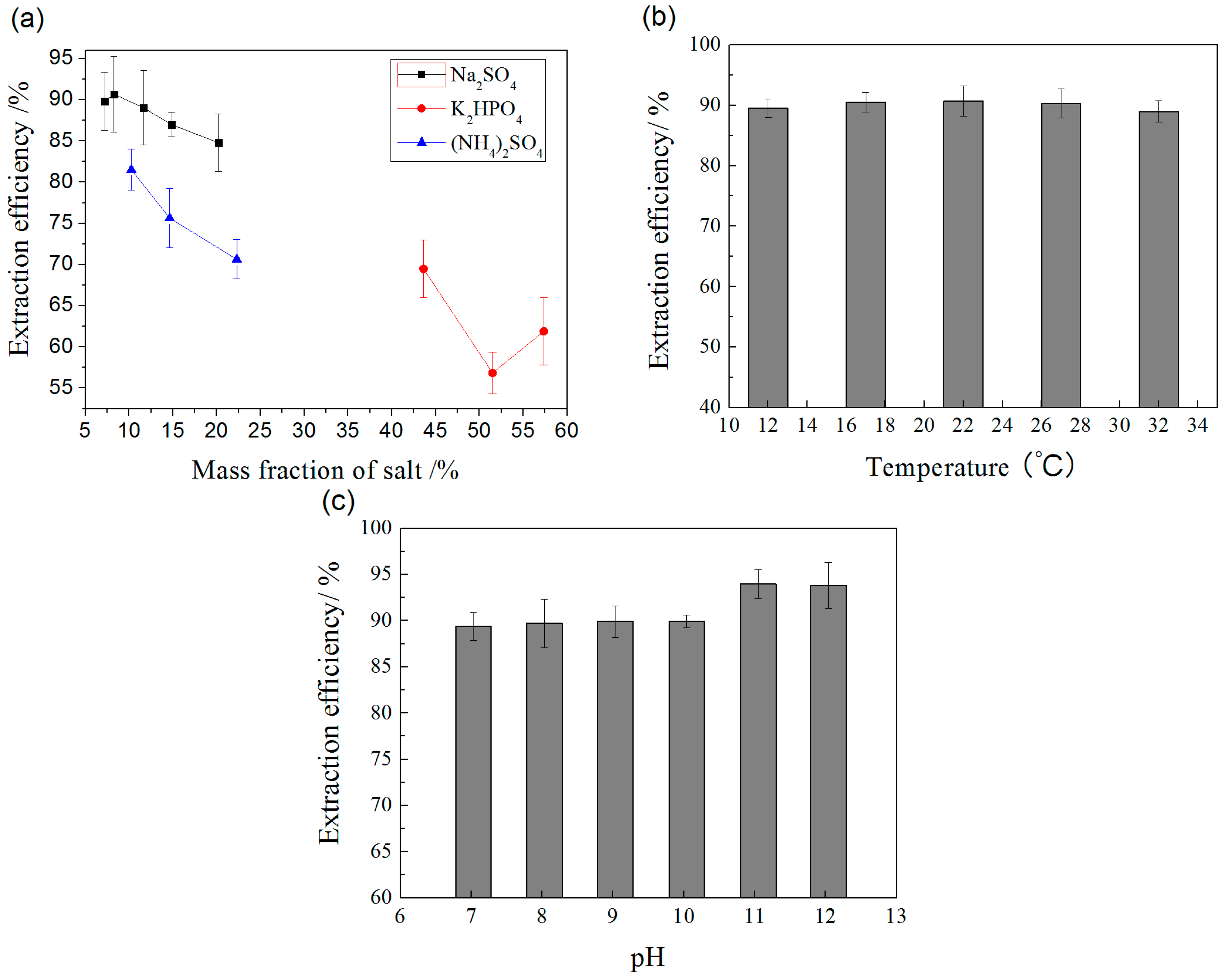
2.2.2. Effect of Temperature
2.2.3. Effect of pH

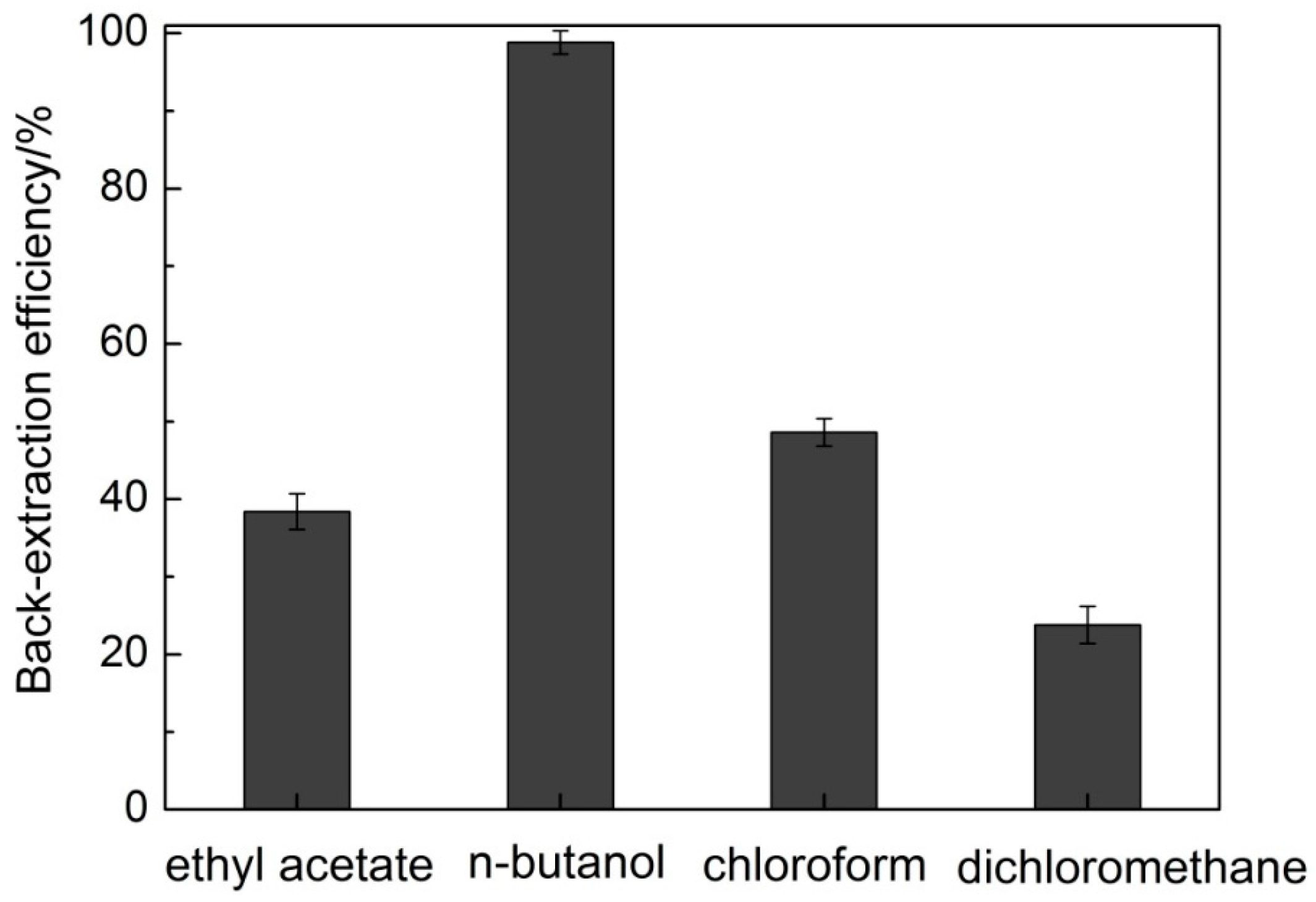
2.2.4. SDG Isolation and IL Recycling
3. Experimental Section
3.1. Materials and Reagents
3.2. Ionic Liquid-Based Ultrasound-Assisted Extraction
3.3. Ionic Liquids-Based Aqueous Two-Phase System
3.4. HPLC Conditions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peterson, J.; Dwyer, J.; Adlercreutz, H.; Scalbert, A.; Jacques, P.; McCullough, M.L. Dietary lignans: Physiology and potential for cardiovascular disease risk reduction. Nutr. Rev. 2010, 68, 571–603. [Google Scholar] [CrossRef] [PubMed]
- Nemes, S.M.; Orsat, V.; Raghavan, G.V. Calibration of artificial neural network and partial least squares regression models for the prediction of secoisolariciresinol diglucoside contents in microwave-assisted extracts of various flaxseed (Linum usitatissimum L.) samples. Food Chem. 2012, 133, 1588–1595. [Google Scholar] [CrossRef]
- Hano, C.; Renouard, S.; Molinie, R.; Corbin, C.; Barakzoy, E.; Doussot, J.; Lamblin, F.; Laine, E. Flaxseed (Linum usitatissimum L.) extract as well as (+)-secoisolariciresinol diglucoside and its mammalian derivatives are potent inhibitors of α-amylase activity. Bioorg. Med. Chem. Lett. 2013, 23, 3007–3012. [Google Scholar] [CrossRef] [PubMed]
- Fuentealba, C.; Figuerola, F.; Estévez, A.M.; González-Muñoz, A.; Muñoz, O. Optimization of secoisolariciresinol diglucoside extraction from flaxseed (Linum usitatissimum L.) and isolation by a simple HPLC-UV method. CyTA-J. Food 2015, 13, 273–281. [Google Scholar] [CrossRef]
- Rizwan, S.; Naqshbandi, A.; Farooqui, Z.; Khan, A.A.; Khan, F. Protective effect of dietary flaxseed oil on arsenic-induced nephrotoxicity and oxidative damage in rat kidney. Food Chem. Toxicol. 2014, 68, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Goyal, A.; Sharma, V.; Upadhyay, N.; Gill, S.; Sihag, M. Flax and flaxseed oil: An ancient medicine & modern functional food. J. Food Sci. Technol. 2014, 51, 1633–1653. [Google Scholar] [PubMed]
- Oomah, B.D. Flaxseed as a functional food source. J. Sci. Food Agric. 2001, 81, 889–894. [Google Scholar] [CrossRef]
- Moree, S.S.; Kavishankar, G.B.; Rajesha, J. Antidiabetic effect of secoisolariciresinol diglucoside in streptozotocin-induced diabetic rats. Phytomedicine 2013, 20, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yuan, Y.V.; Kitts, D.D. Antioxidant activities of the flaxseed lignan secoisolariciresinol diglucoside, its aglycone secoisolariciresinol and the mammalian lignans enterodiol and enterolactone in vitro. Food Chem. Toxicol. 2007, 45, 2219–2227. [Google Scholar] [CrossRef] [PubMed]
- Ayella, A.; Lim, S.; Jiang, Y.; Iwamoto, T.; Lin, D.; Tomich, J.; Wang, W. Cytostatic inhibition of cancer cell growth by lignan secoisolariciresinol diglucoside. Nutr. Res. 2010, 30, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Saggar, J.K.; Corey, P.; Thompson, L.U. Flaxseed and pure secoisolariciresinol diglucoside, but not flaxseed hull, reduce human breast tumor growth (MCF-7) in athymic mice. J. Nutr. 2009, 139, 2061–2066. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Hypocholesterolemic and antiatherosclerotic effect of flax lignan complex isolated from flaxseed. Atherosclerosis 2005, 179, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Rogers, R.D.; Seddon, K.R. Ionic liquids—Solvents of the future? Science 2003, 302, 792–793. [Google Scholar] [CrossRef] [PubMed]
- Rogers, R.D. Materials science—Reflections on ionic liquids. Nature 2007, 447, 917–918. [Google Scholar] [CrossRef] [PubMed]
- Neves, C.M.; Ventura, S.P.; Freire, M.G.; Marrucho, I.M.; Coutinho, J.A. Evaluation of cation influence on the formation and extraction capability of ionic-liquid-based aqueous biphasic systems. J. Phys. Chem. B 2009, 113, 5194–5199. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Baker, G.A.; Zhao, H. Ether- and alcohol-functionalized task-specific ionic liquids: Attractive properties and applications. Chem. Soc. Rev. 2012, 41, 4030–4066. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Z.; Li, Y.; Chi, R. Optimization of ionic liquid-based microwave-assisted extraction of isoflavones from Radix puerariae by response surface methodology. Sep. Purif. Technol. 2014, 135, 285–285. [Google Scholar] [CrossRef]
- Ge, L.; Xia, F.; Song, Y.; Yang, K.; Qin, Z.; Li, L. Solubility of luteolin in several imidazole-based ionic liquids and extraction from peanut shells using selected ionic liquid as solvent. Sep. Purif. Technol. 2014, 135, 223–228. [Google Scholar] [CrossRef]
- Ribeiro, B.D.; Coelho, M.A.Z.; Rebelo, L.P.N.; Marrucho, I.M. Ionic liquids as additives for extraction of saponins and polyphenols from mate (Ilex paraguariensis) and tea (Camellia sinensis). Ind. Eng. Chem. Res. 2013, 52, 12146–12153. [Google Scholar] [CrossRef]
- Ribeiro, B.D.; Coelho, M.A.Z.; Marrucho, I.M. Extraction of saponins from sisal (Agave sisalana) and juá (Ziziphus joazeiro) with cholinium-based ionic liquids and deep eutectic solvents. Eur. Food Res. Technol. 2013, 237, 965–975. [Google Scholar] [CrossRef]
- Tang, B.; Lee, Y.J.; Lee, Y.R.; Row, K.H. Examination of 1-methylimidazole series ionic liquids in the extraction of flavonoids from Chamaecyparis obtuse leaves using a response surface methodology. J. Chromatogr. B 2013, 933, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Wang, Y.; Gao, S.; Zhang, R.; Ren, R.; Li, N.; Zhang, H. Determination of the active constituents in Arnebia euchroma (Royle) Johnst. by ionic liquid-based ultrasonic-assisted extraction high-performance liquid chromatography. J. Chromatogr. B 2011, 879, 1833–1838. [Google Scholar] [CrossRef] [PubMed]
- Chatel, G.; Macfarlane, D.R. Ionic liquids and ultrasound in combination: Synergies and challenges. Chem. Soc. Rev. 2014, 43, 8132–8149. [Google Scholar] [CrossRef] [PubMed]
- Harde, S.M.; Lonkar, S.L.; Degani, M.S.; Singhal, R.S. Ionic liquid based ultrasonic-assisted extraction of forskolin from Coleus forskohlii roots. Ind. Crop. Prod. 2014, 61, 258–264. [Google Scholar] [CrossRef]
- Yang, L.; Li, L.L.; Liu, T.T.; Zu, Y.G.; Yang, F.J.; Zhao, C.J.; Zhang, L.; Chen, X.Q.; Zhang, Z.H. Development of sample preparation method for isoliquiritigenin, liquiritin, and glycyrrhizic acid analysis in licorice by ionic liquids-ultrasound based extraction and high-performance liquid chromatography detection. Food Chem. 2013, 138, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.C.; Li, Q.Y.; Liu, Y.H.; Chen, B.B. Ionic liquid-aqueous solution ultrasonic-assisted extraction of three kinds of alkaloids from Phellodendron amurense Rupr and optimize conditions use response surface. Ultrason. Sonochem. 2015, 24, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Gutowski, K.E.; Broker, G.A.; Willauer, H.D.; Huddleston, J.G.; Swatloski, R.P.; Holbrey, J.D.; Rogers, R.D. Controlling the aqueous miscibility of ionic liquids: Aqueous biphasic systems of water-miscible ionic liquids and water-structuring salts for recycle, metathesis, and separations. J. Am. Chem. Soc. 2003, 125, 6632–6633. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wang, Y.; Liu, Y.; Li, Y.; Lu, Y.; Yan, Y.; Ni, L. Ionic liquid-salt aqueous two-phase extraction based on salting-out coupled with high-performance liquid chromatography for the determination of sulfonamides in water and food. Anal. Bioanal. Chem. 2013, 405, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Passos, H.; Trindade, M.P.; Vaz, T.S.; da Costa, L.P.; Freire, M.G.; Coutinho, J.A.P. The impact of self-aggregation on the extraction of biomolecules in ionic-liquid-based aqueous two-phase systems. Sep. Purif. Technol. 2013, 108, 174–180. [Google Scholar] [CrossRef]
- Sha, O.; Zhu, X.S.; Feng, Y.L.; Ma, W.X. Aqueous two-phase based on ionic liquid liquid-liquid microextraction for simultaneous determination of five synthetic food colourants in different food samples by high-performance liquid chromatography. Food Chem. 2015, 174, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.J.; Li, F.F.; Xu, X.L.; Xing, J.M. Simultaneous extraction and purification of aloe polysaccharides and proteins using ionic liquid based aqueous two-phase system coupled with dialysis membrane. Desalination 2012, 286, 389–393. [Google Scholar] [CrossRef]
- Claudio, A.F.; Ferreira, A.M.; Freire, C.S.; Silvestre, A.J.; Freire, M.G.; Coutinho, J.A. Optimization of the gallic acid extraction using ionic-liquid-based aqueous two-phase systems. Sep. Purif. Technol. 2012, 97, 142–149. [Google Scholar] [CrossRef]
- Abdolrahimi, S.; Nasernejad, B.; Pazuki, G. Influence of process variables on extraction of cefalexin in a novel biocompatible ionic liquid based-aqueous two phase system. Phys. Chem. Chem. Phys. 2015, 17, 655–669. [Google Scholar] [CrossRef]
- Jiang, B.; Feng, Z.; Liu, C.; Xu, Y.; Li, D.; Ji, G. Extraction and purification of wheat-esterase using aqueous two-phase systems of ionic liquid and salt. J. Food Sci. Technol. 2015, 52, 2878–2885. [Google Scholar] [CrossRef] [PubMed]
- Jacquemin, J.; Husson, P.; Padua, A.A.; Majer, V. Density and viscosity of several pure and water-saturated ionic liquids. Green Chem. 2006, 8, 172–180. [Google Scholar] [CrossRef]
- Bogdanov, M.G.; Svinyarov, I. Ionic liquid-supported solid–liquid extraction of bioactive alkaloids. II. Kinetics, modeling and mechanism of glaucine extraction from Glaucium flavum Cr. (Papaveraceae). Sep. Purif. Technol. 2013, 103, 279–288. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, Z.; Wang, J.; Yang, S.; Li, B.; Xu, N. Aqueous ionic liquid based ultrasonic assisted extraction of four acetophenones from the Chinese medicinal plant Cynanchum bungei Decne. Ultrason. Sonochem. 2013, 20, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, M.G. Ionic liquids as alternative solvents for extraction of natural products. In Alternative Solvents for Natural Products Extraction; Chemat, F., Abert-Vian, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 127–166. [Google Scholar]
- Yang, L.; Ge, H.; Wang, W.; Zu, Y.; Yang, F.; Zhao, C.; Zhang, L.; Zhang, Y. Development of sample preparation method for eleutheroside b and e analysis in Acanthopanax senticosus by ionic liquids-ultrasound based extraction and high-performance liquid chromatography detection. Food Chem. 2013, 141, 2426–2433. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, M.G.; Keremedchieva, R.; Svinyarov, I. Ionic liquid-supported solid-liquid extraction of bioactive alkaloids. III. Ionic liquid regeneration and glaucine recovery from ionic liquid-aqueous crude extract of Glaucium flavum Cr. (Papaveraceae). Sep. Purif. Technol. 2015. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds flaxseed powder are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, Z.-J.; Wang, C.-Y.; Yang, Z.-Z.; Yi, Y.-J.; Wang, H.-Y.; Zhou, W.-L.; Li, F.-F. Ionic Liquid-Based Ultrasonic-Assisted Extraction of Secoisolariciresinol Diglucoside from Flaxseed (Linum usitatissimum L.) with Further Purification by an Aqueous Two-Phase System. Molecules 2015, 20, 17929-17943. https://doi.org/10.3390/molecules201017929
Tan Z-J, Wang C-Y, Yang Z-Z, Yi Y-J, Wang H-Y, Zhou W-L, Li F-F. Ionic Liquid-Based Ultrasonic-Assisted Extraction of Secoisolariciresinol Diglucoside from Flaxseed (Linum usitatissimum L.) with Further Purification by an Aqueous Two-Phase System. Molecules. 2015; 20(10):17929-17943. https://doi.org/10.3390/molecules201017929
Chicago/Turabian StyleTan, Zhi-Jian, Chao-Yun Wang, Zi-Zhen Yang, Yong-Jian Yi, Hong-Ying Wang, Wan-Lai Zhou, and Fen-Fang Li. 2015. "Ionic Liquid-Based Ultrasonic-Assisted Extraction of Secoisolariciresinol Diglucoside from Flaxseed (Linum usitatissimum L.) with Further Purification by an Aqueous Two-Phase System" Molecules 20, no. 10: 17929-17943. https://doi.org/10.3390/molecules201017929