2.1. Chemical Characterization
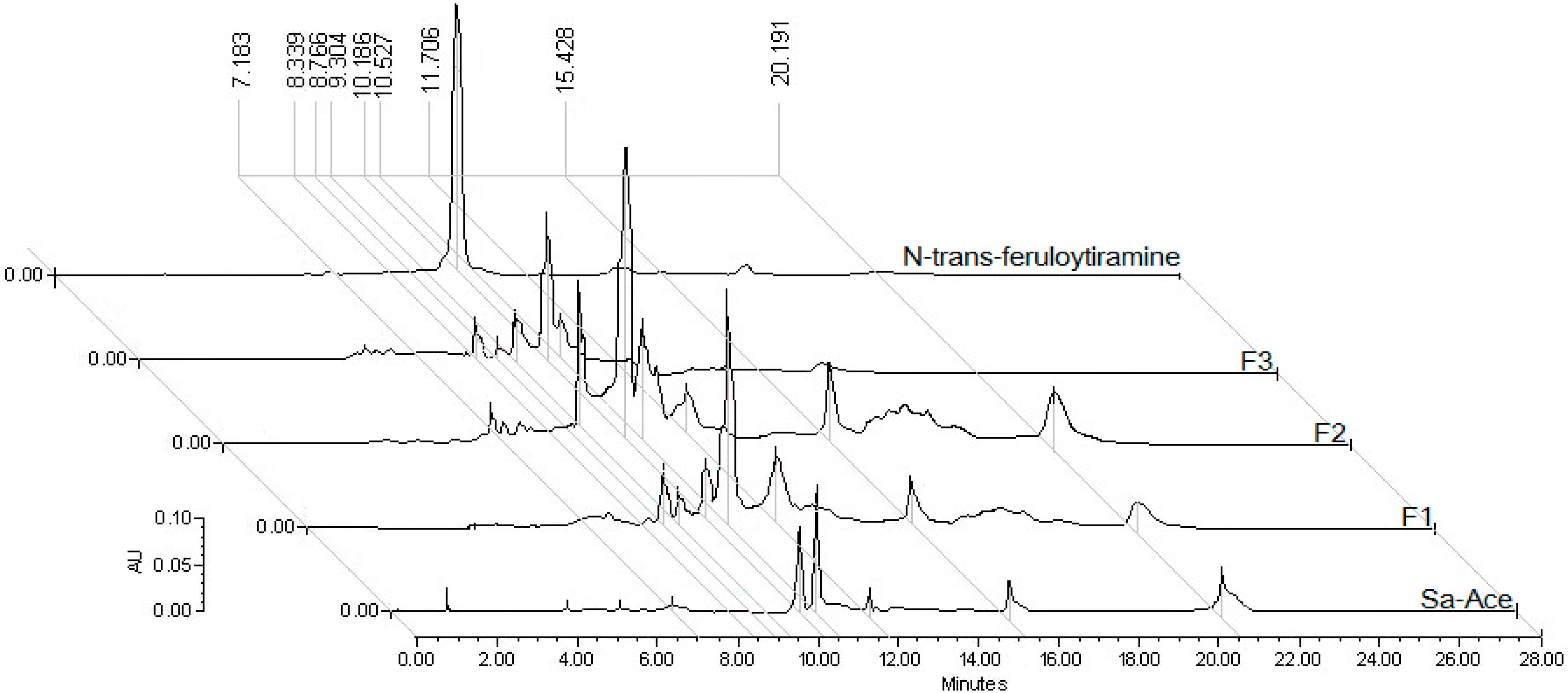
Chromatographic fractionation of the F2, allowed us to obtain compound
1 as a yellow precipitate, soluble in methanol, with a melting point of 74–75 °C. In the UV-vis spectrum, the compound showed the following λ
max: 220, 293, and 318 nm. The
1H-NMR spectrum showed four systems: an aromatic ABX system [δ 7.10 (1H, d,
J = 2 Hz), 6.79 (1H, d,
J = 8.2 Hz) and 7.0 (1H, dd,
J = 2, 8.2 Hz)] assigned a H-2, H-5 and H-6 respectively; another aromatic A
2B
2 system [δ 7.05 (2H, d,
J = 6.8 Hz) and 6.72 (2H, d,
J = 7.2 Hz)] assigned H-2', H-6' and H-3', H-5' respectively; a
trans double bond AB system [δ 7.43 (1H, d,
J = 15.6 Hz) and 6.40 (1H, d,
J = 15.6 Hz)] assigned a H-7 and H-8 respectively, and a AB system [δ 2.75 (2H, dd,
J = 7.1, 7.5 Hz) and 3.46 (2H, dd,
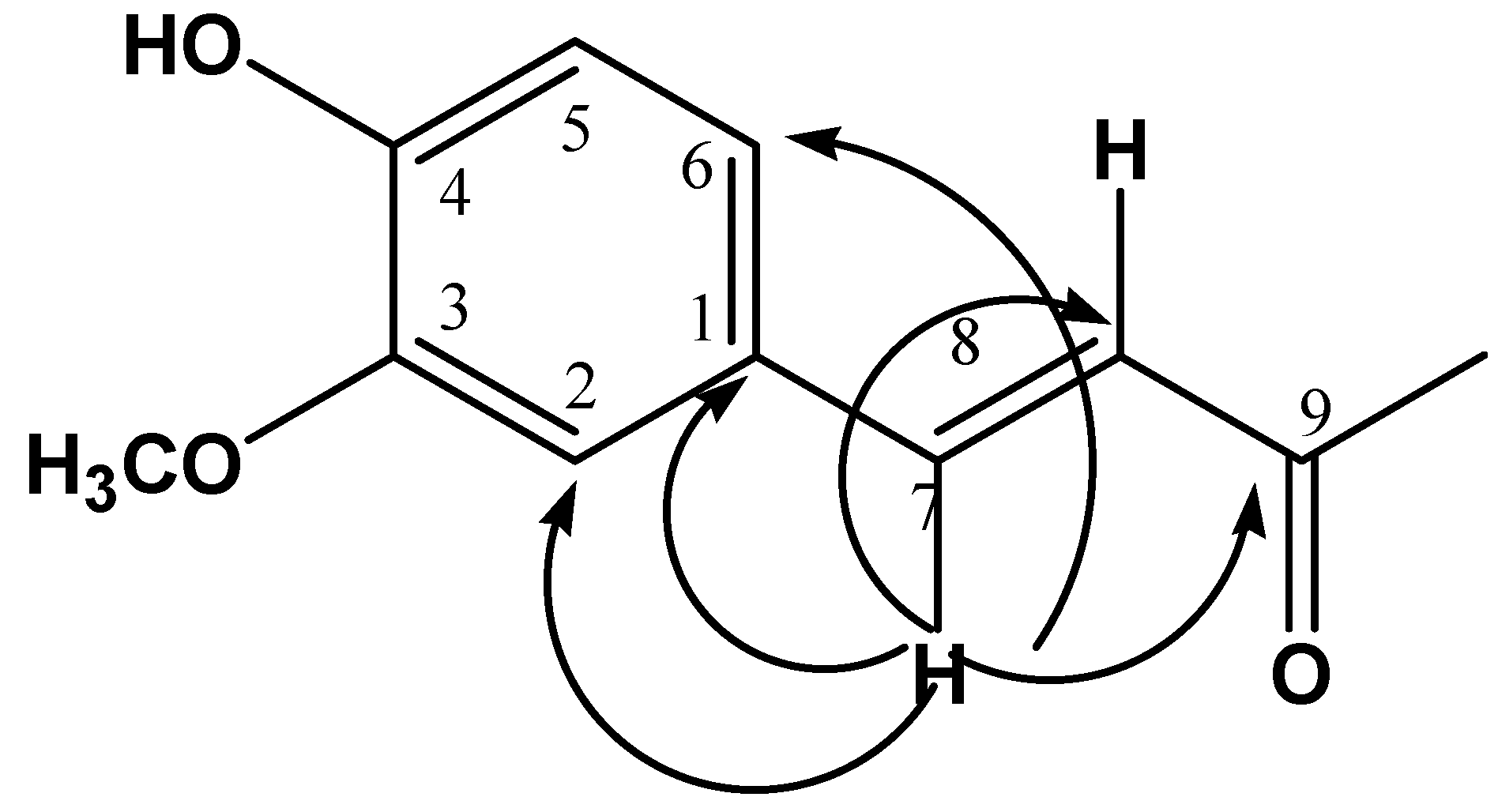
J = 7.2, 6.5 Hz)] assigned a H-7' and H-8' respectively. According to a HMBC experiment, H-7 (δ 7.43) correlated at two and three bonds with five carbon signals at δ 128.39, 111.6, 123.33, 118.8 and 169.28 which are assigned to C-1, C-2, C-6, C-8 and C-9 respectively, indicating the presence of a
trans-feruloyl group (see
Figure 1).
Figure 1.
HMBC correlations of H-7.
Figure 1.
HMBC correlations of H-7.
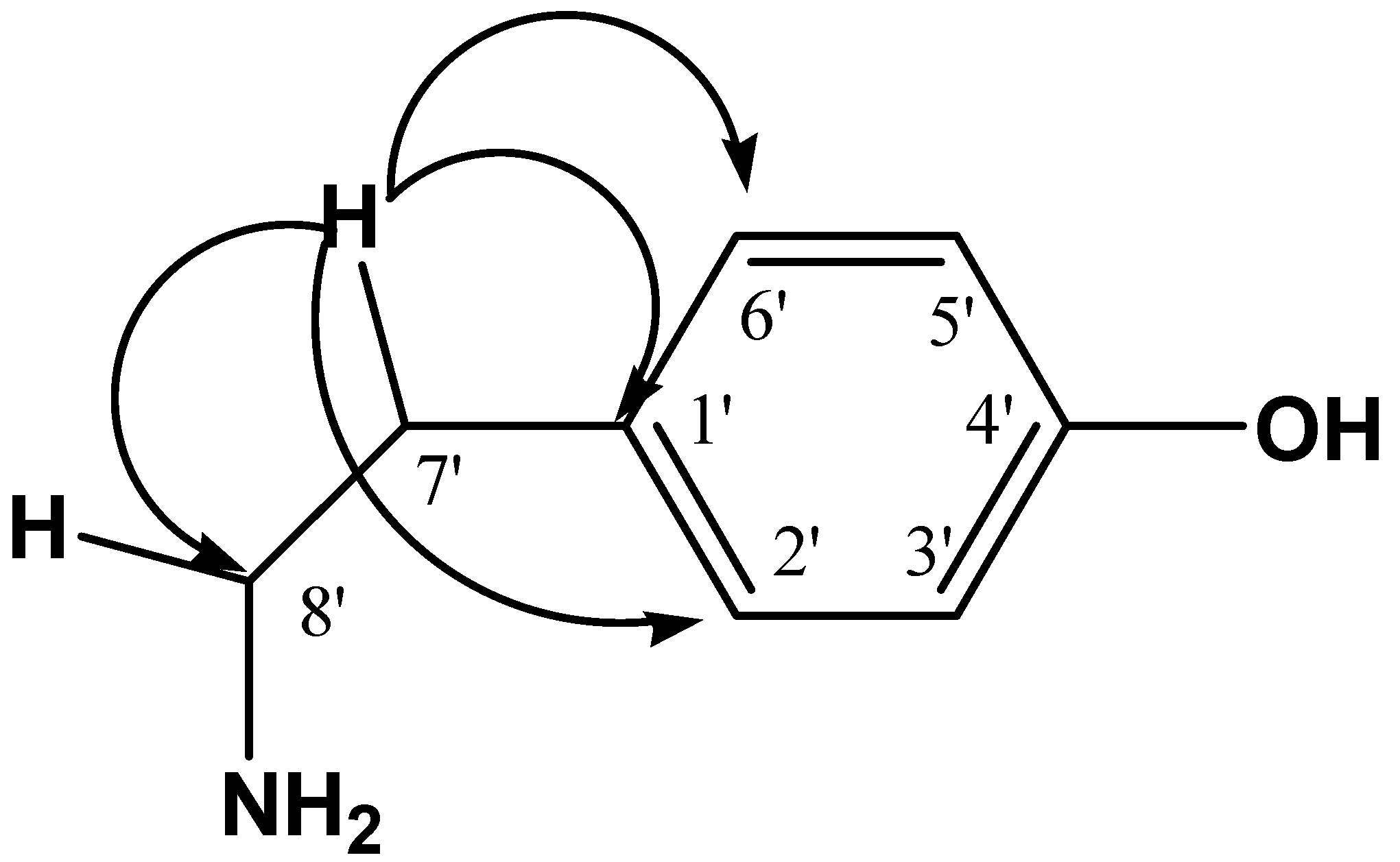
On the other hand the signal of H-7' (δ 2.75) correlated to J
2-3 with four signals
13C in δ 131.41, 130.85, 130.85 and 42.66 assigned to C-1', C-2', C-6' and C-8' respectively, indicates the presence of ethyl benzene (see
Figure 2).
Figure 2.
HMBC correlations of H-7'.
Figure 2.
HMBC correlations of H-7'.
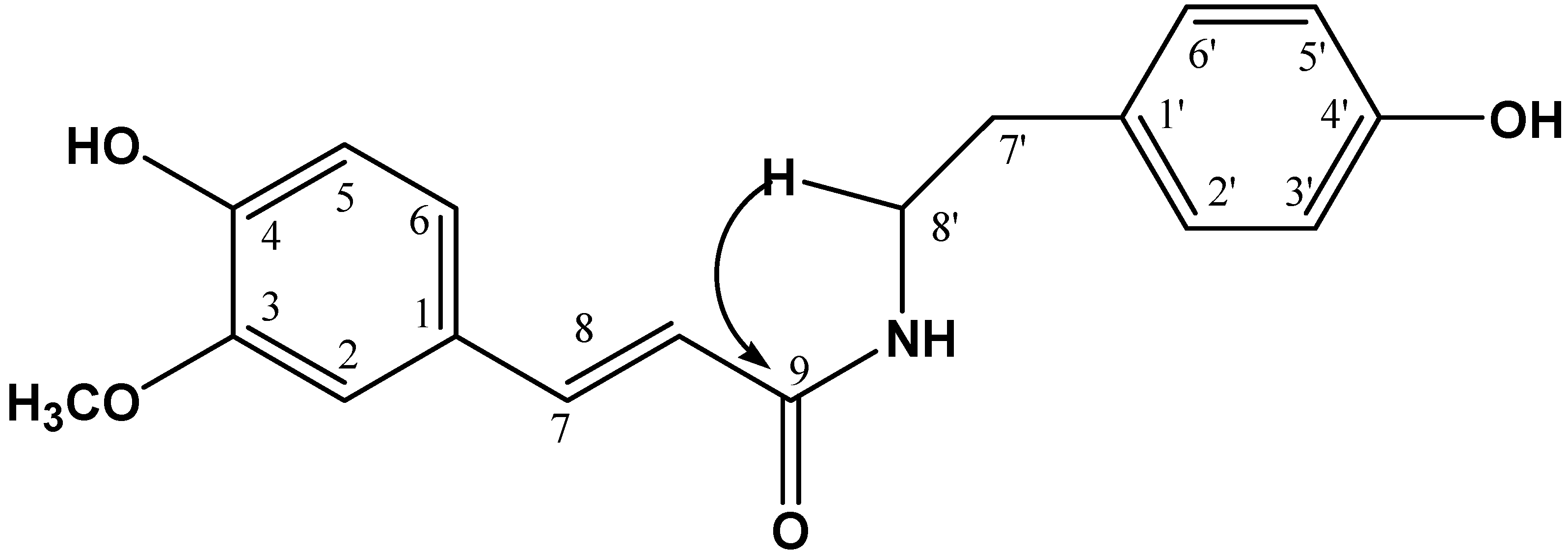
Additionally, the signal H-8' (δ 3.46) is attached to a group containing a nitrogen atom for its chemical shift. This allowed us to determine the presence of a terminal amine group, forming the skeleton of tyramine. This group is attached to the N-
trans-feruloyl moiety, according to the observed coupling of H-8' to J
2-3 with the signal of the C-9 carbonyl group (169.28) of an amide ester (see
Figure 3).
Figure 3.
HMBC correlations of H-8'.
Figure 3.
HMBC correlations of H-8'.
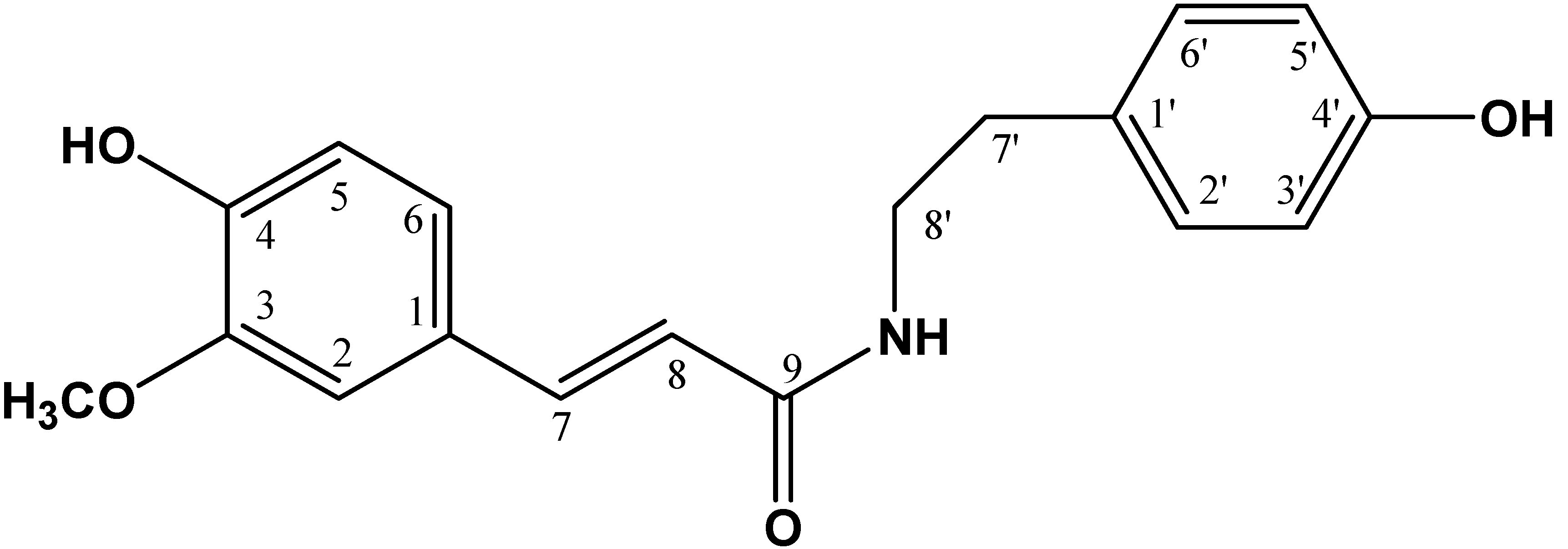
In conformity with NMR analysis data and comparison of the literature data [
14], the chemical structure was established as N-
trans-feruloyltyramine or NTF (
1) (see
Figure 4).
Figure 4.
Chemical structure of N-trans-feruloyltyramine.
Figure 4.
Chemical structure of N-trans-feruloyltyramine.
The mass spectrum gave a molecular ion of m/z 314 [M + H]
+, 192 [M-ethylbenzene], 177 [M-ethylaminobenzene]. This compound has not been reported previously for
S. aristolochiifolia. However, phenylethylcinnamide has been isolated from other species, and possesses activities such as antioxidative [
15,
16], α-glucosidase inhibitor [
17] and antiarrhythmic [
18]. It was used for the standardization of the extract of
S. aristolochiifolia by HPLC and the content of NTF in each sample of mg/g of extract was: acetone extract (AceSa, 2.77), F1 (2.63), F2 (60.22), F3 (8.76).
2.3. Pharmacological Characterization
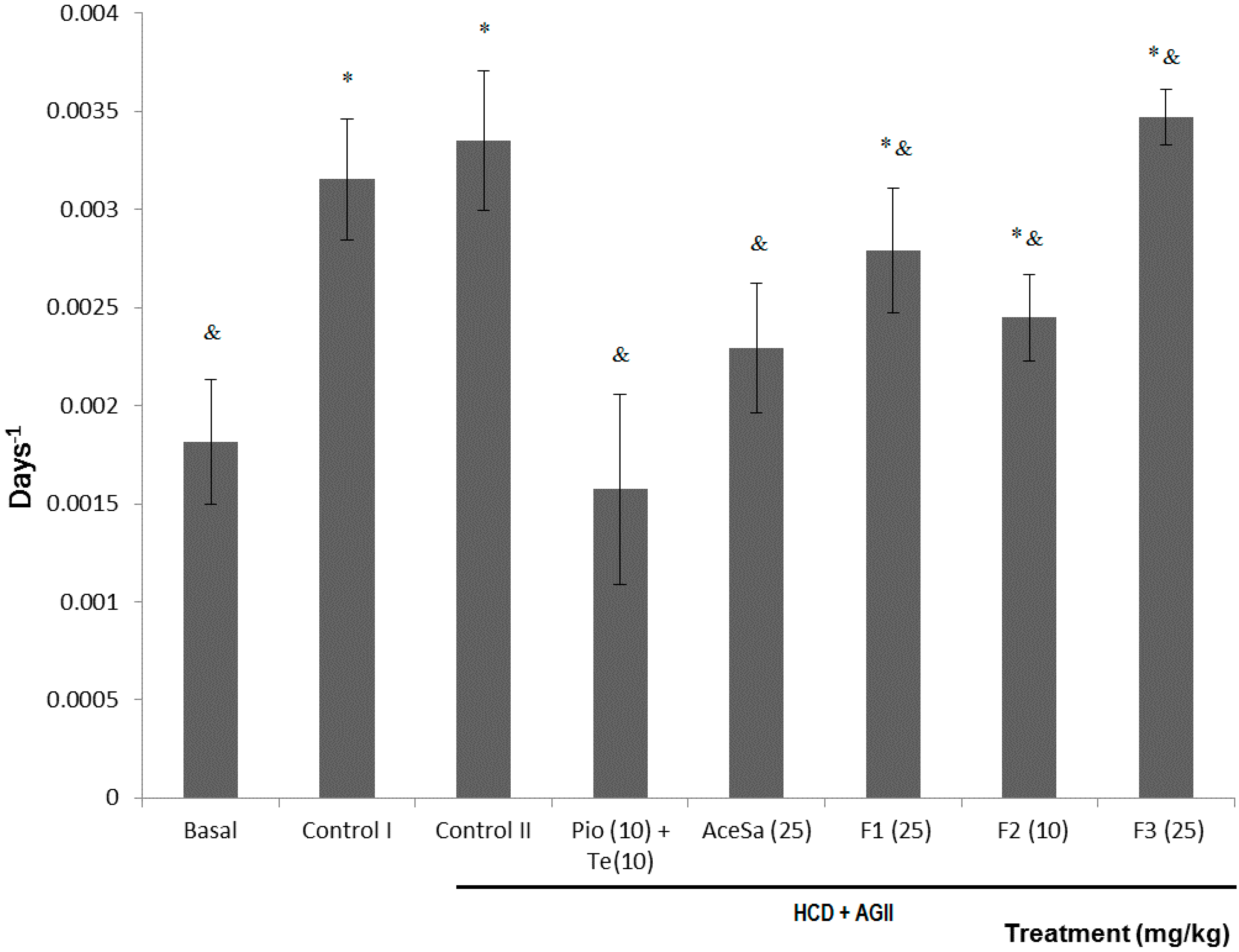
The effect of the administration of a high-caloric diet (HCD, control I group) for 20 weeks was compared against the effect of a regular diet (basal group) and against the negative control of the experiment (HCD + AGII, control II group). In both cases, a twofold increase was observed in growth rate values (which corresponds to the slope of the logarithmic phase of the change of weight with respect to time in days) compared to the values observed in the basal group; these data were significantly different from those of the basal group (*
p < 0.05) (see
Figure 6).
The treatments with AceSa extract and positive control (Pio + Tel) showed no significant change (
p > 0.05) compared to baseline conditions. But when these groups were compared with the damage group (control II) it is showed a significant decrease (
& p < 0.05,
Figure 6). Although the effect of F1, F2 and F3 was lower than AceSa and it was significantly different from the basal group (*
p < 0.05), these treatments also induced a statistically decrement of the growth velocity curve with respect to the control II group (
& p < 0.05).
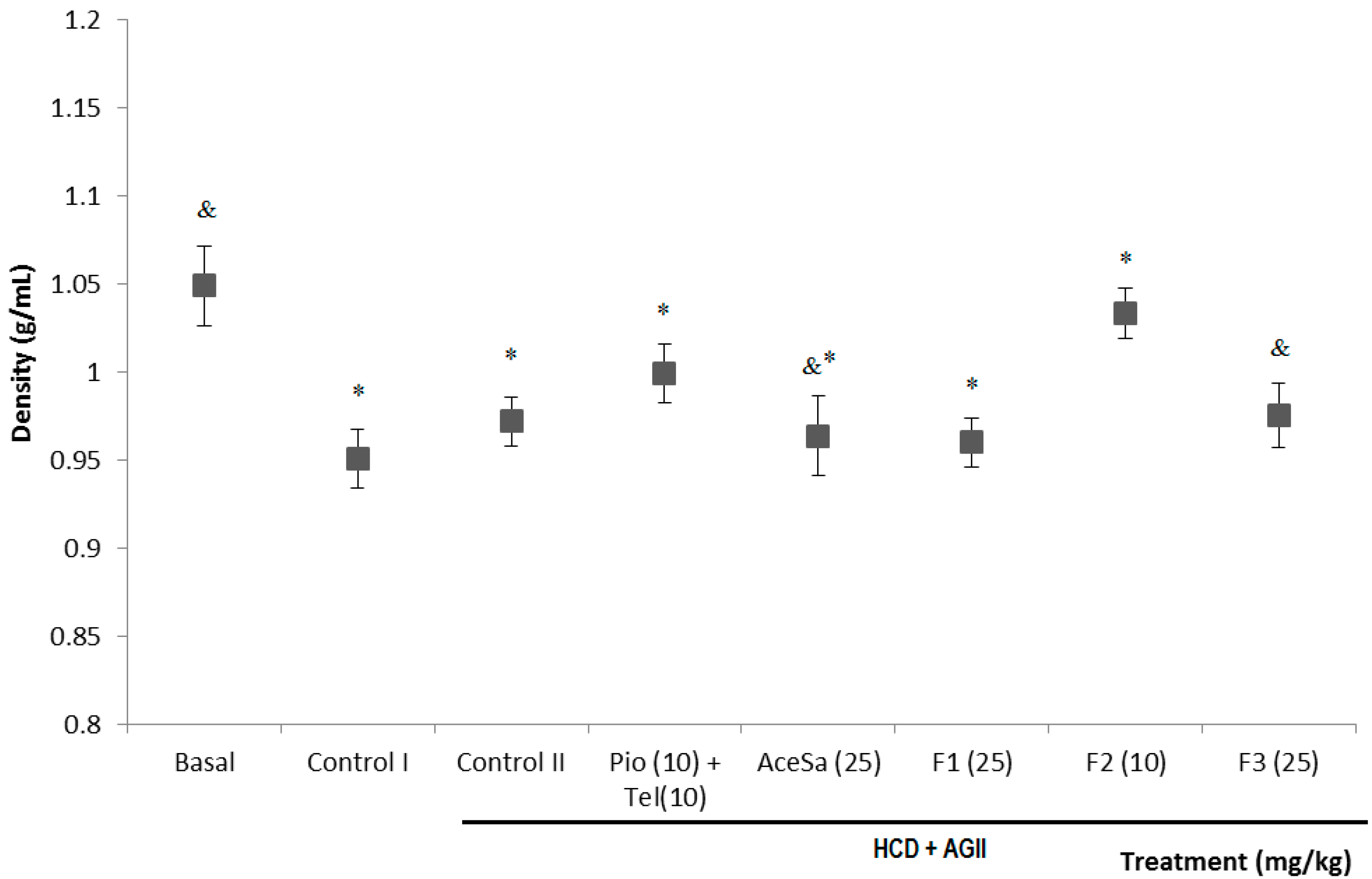
Mice fed with a regular diet (basal) showed an increment dependent on the increase of lean tissue, reaching a value of body density of 1.05 ± 0.02 g/mL (
Figure 7). In the groups with HCD or HCD + AGII, control group I and II respectively, was observed a significant decrement of the body density with respect to the basal group (*
p < 0.05); this effect results from the increase in weight that can be calculated from the values in the growth rate (
Figure 6).
Figure 6.
Effect of different treatments on the growth rate of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSa, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post‑test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 6.
Effect of different treatments on the growth rate of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSa, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post‑test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
The treatments AceSa, F1 and F3 from
S. aristolochiifolia did not prevent the effect of HCD and AGII on the body density. Data from these groups were significantly different from the basal (
Figure 7, *
p < 0.05), but not from the damage group (
Figure 7, *
p > 0.05). However, the fraction F2 and the positive control group (Pio + Tel) were able to inhibit the decrement of body density induced with HCD and AGII. This effect was different to the damage group (HCD + AGII,
& p <0.05), but statistically similar to the basal (
p > 0.05).
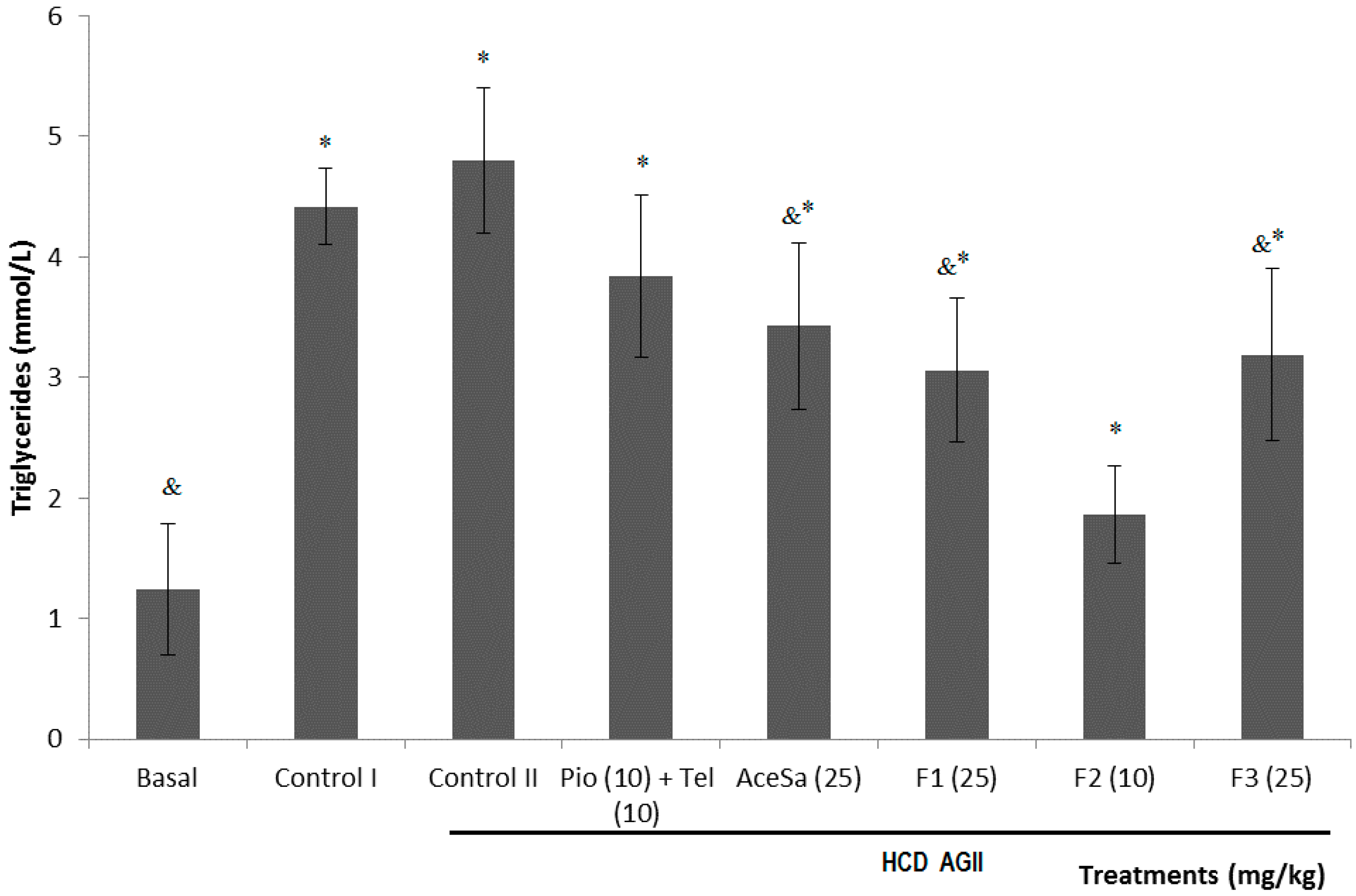
Hypertriglyceridemia is a cardinal alteration of MS associated with dyslipidemia. In this work, the administration of HCD and HCD + AGII caused a significant increase (*
p < 0.05) in the concentration of plasma triglycerides, which increased in respect to the basal group up to 3 and 4 times, respectively. Although the treatments from
S. aristolochiifolia were higher and statistically different to basal group (*
p < 0.05), they provoked a statistical significant diminish of the triglyceride serum concentration in regard to the damage group with HCD + AG II (
& p < 0.05), and it was worth noting that the group of mice treated with F2 responded better, given that the triglycerides were lower than HCD + AG II but statistically equal to basal (
Figure 8), while the group with Pio + Tel, did not diminish this parameter in respect to the control II group (
p > 0.05,
Figure 8).
Figure 7.
Effect of different treatments on the body density of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSa, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post‑test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 7.
Effect of different treatments on the body density of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSa, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post‑test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 8.
Effect of different treatments on the serum concentration of triglycerides of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 8.
Effect of different treatments on the serum concentration of triglycerides of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
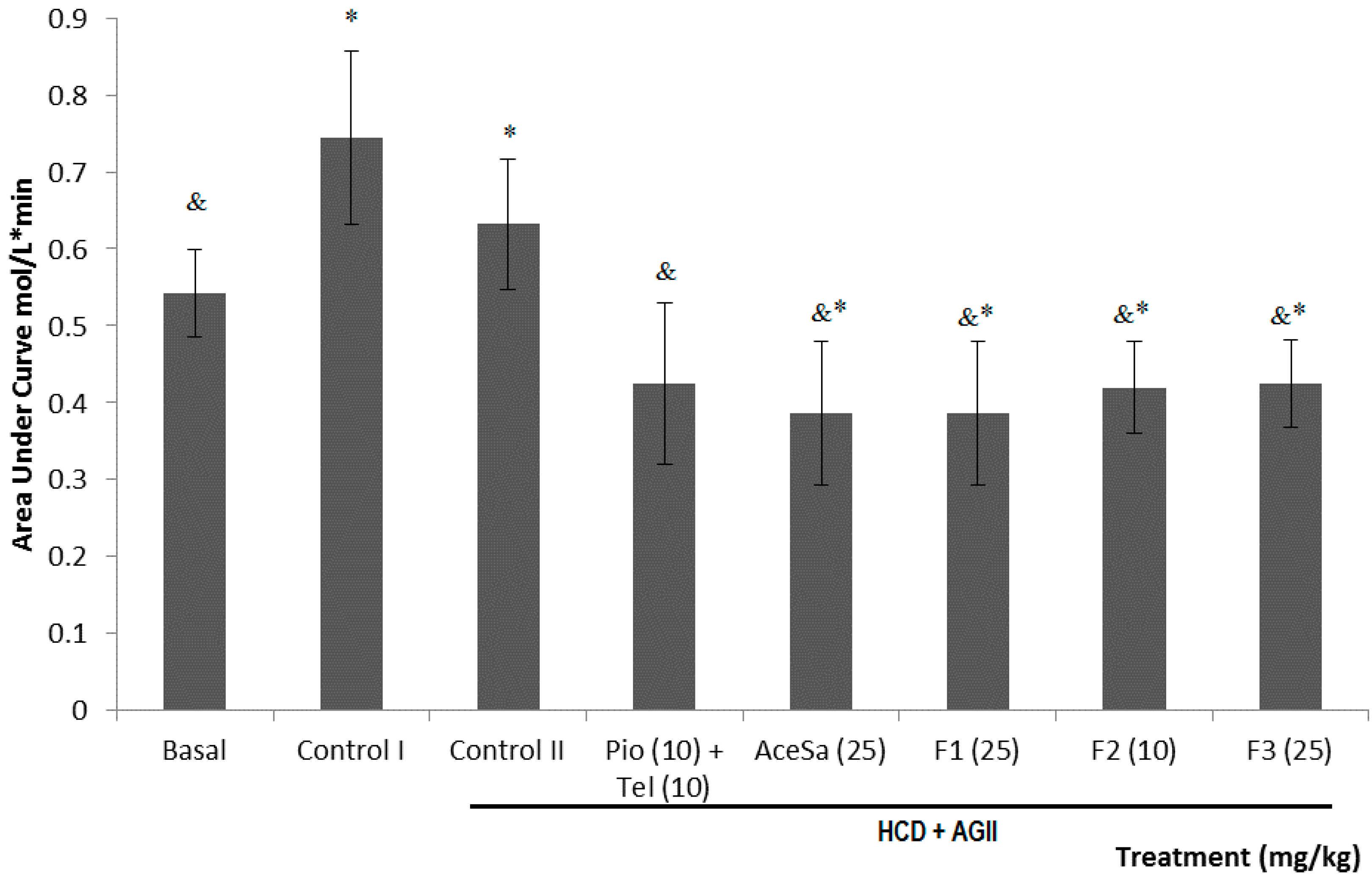
Another component of MS is the insulin resistance that leads to type 2 diabetes mellitus. This parameter is shown as the area under the curve (AUC) of plasma glucose concentration with the administration of a dose of insulin. The animals exposed to both HCD and HCD + AGII showed a significant increment value of the AUC respect to basal (*
p < 0.05,
Figure 9). All treatments from
S. aristolochiaefolia decreased the insulin resistance parameter by decreasing the value of AUC data, that were statistically different to the group with damage (
& p < 0.05,
Figure 9). The positive control treatment (Pio + Tel) was the best to cause a decrement of this parameter (
& p < 0.05), even with a greater reduction than the observed for the basal (*
p < 0.05).
Figure 9.
Effect of different treatments on the insulin resistance (measured as the area under the curve) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 9.
Effect of different treatments on the insulin resistance (measured as the area under the curve) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
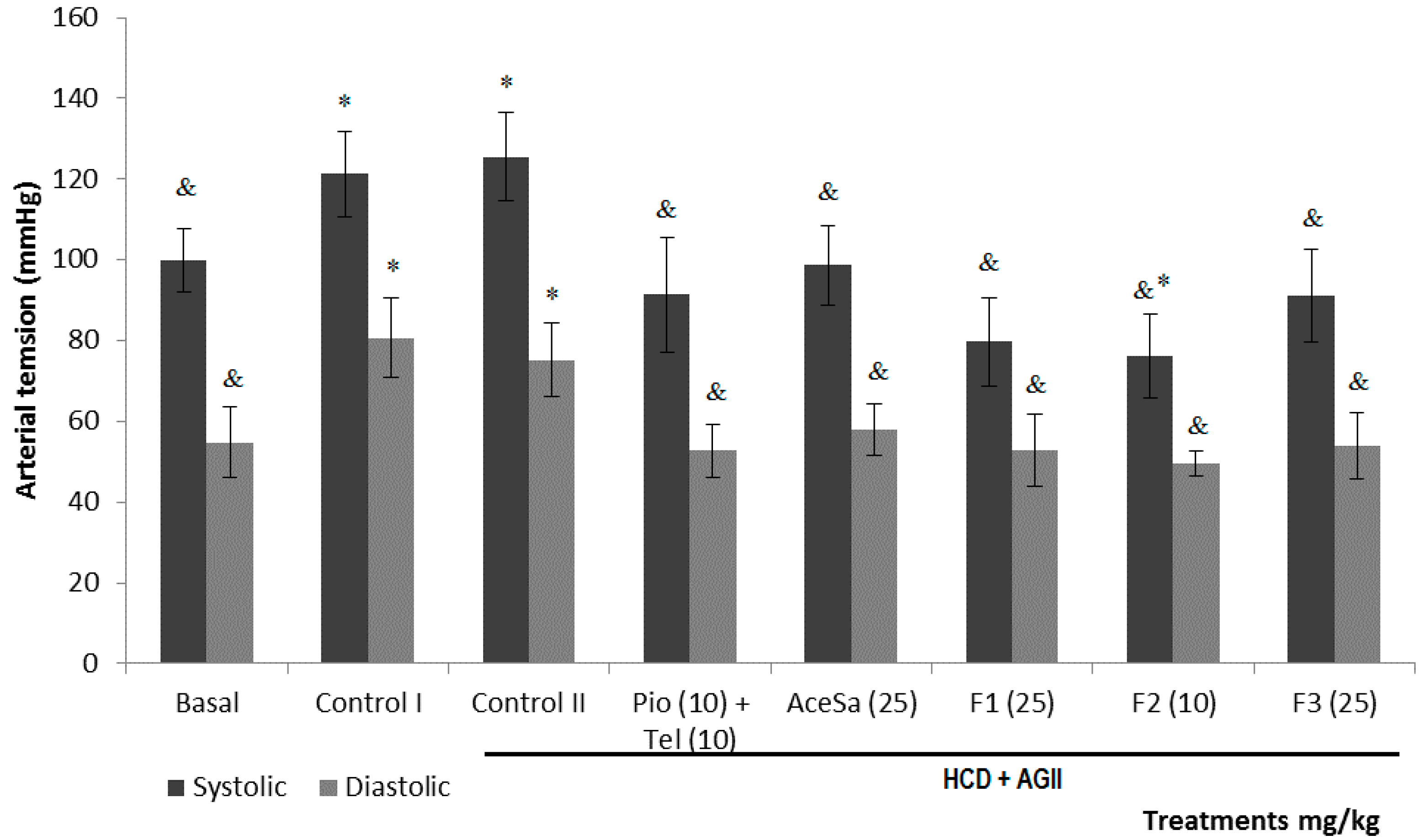
When the diastolic (DBP) and systolic (SBP) blood pressure were measured, it was observed that groups of mice with HCD or HCD + AGII showed a substantial and statistically significant increase in both, in comparison to the basal situation (*
p < 0.05,
Figure 10). This increment was controlled with the administration of extract (AceSa) and fractions (F1, F2 and F3) from
S. aristolichiifolia to different animal groups; therefore a decrease of SBP and DBP could be observed (
& p < 0.05,
Figure 10).
The levels of cytokines were quantified in kidney and adipose tissue from animals with different experimental conditions associated with MS. The kidney administration of treatments and HCD or HCD + AGII established an inflammatory environment, in comparison to the basal conditions (
Figure 11a). When this inflammatory process was evaluated in the groups of mice with extract or fractions from
S. aristolochiifolia (
Figure 11a), a decrease of the pro-inflammatory conditions was generally observed, in the same way as it happened with the positive control treatment (Pio + Tel).
Figure 10.
Effect of different treatments on systolic and diastolic blood pressure (SBP and DBP) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (controlII): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
Figure 10.
Effect of different treatments on systolic and diastolic blood pressure (SBP and DBP) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (controlII): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet. ANOVA followed by Tukey post-test (mean ± SD, n = 10); * p < 0.05 (in comparison with the basal group), & p < 0.05 (in comparison with control II).
In regard to the response of the adipose tissue that can be observed in
Figure 11b, both treatments HCD and HCD + AGII had a similar effect. Positive control treatment (Pio + Tel) was not able to decrease the pro-inflammatory response in adipose tissue (
Figure 11b), in contrast to mice with treatments from
S. aristolochiifolia which generally showed decreased tissue pro-inflammatory responses, although fraction F3 did so to a lesser extent (
Figure 11b).
In this work, a high-caloric diet was administered to C57BL6 mice, control group I, increasing their weight gain and generating damage associated with MS: overweight, hyperglycemia, hypertension, and hypertriglyceridemia. These alterations may eventually cause vascular damage, but not necessarily because of the short period of exposure time. Due to the above control group II that was used, potential vascular disorders associated with SM ailments were exacerbated with the chronic administration of the potent vasoconstrictor AG II. The latter caused the establishment of organic damage to vasculature, through the oxidative stress and inflammatory processes in endothelial cells.
The increase in glucose concentration, triglycerides and chronic hypertension associated with obesity and diabetes is the major pathological factor for endothelial dysfunction, which is a key element of the mechanisms underlying vascular disease [
19]. It is also a predictor of clinical events that have a high rate of comorbidity, such as cerebral vascular accidents [
20,
21], mainly due to the fact that endothelial cells play an important role in regulating vascular structure and function [
19].
Obesity is a risk factor for the development of hypertension and dyslipidemia. Weight gain induced by HCD produces an increase in the plasma concentrations of AG II, which raises systolic blood pressure in male mice [
22]. Adipocytes, the main cells of adipose tissue, are capable of expressing angiotensinogen and angiotensin peptides [
22].
Figure 11.
Effect of different treatments on concentration of cytokines IL-1β, TNF-α and IL-6 with respect to the concentration of IL-10, in kidney (a) and fat tissue (b) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet.
Figure 11.
Effect of different treatments on concentration of cytokines IL-1β, TNF-α and IL-6 with respect to the concentration of IL-10, in kidney (a) and fat tissue (b) of C57/BL-6 mice exposed to hypercaloric diet and chronic angiotensin II (control II): acetone extract of Smilax aristolochiifolia (AceSa), fractions F1, F2 and F3 from AceSA, pioglitazone and telmisartan (Pio + Tel). Control I = hypercaloric diet.
In humans, AG II produced by adipocytes seems to inhibit the differentiation of adipocyte precursors, decreasing the percentage of small adipocytes sensitive to insulin. Thus, the storage capacity of lipids in the adipose tissue is reduced, and the triglycerides can accumulate in the liver and in the skeletal muscle, contributing to insulin resistance [
23]. Besides, the steady increase in the level of AGII producing a prolonged vascular constriction induces release of aldosterone, retention of sodium and water and increase of fluids, ultimately leading to a state of hypertension [
24]. Thus, a chronic imbalance of AG II regulation induces chronic hypertension and vascular damage, as well as dyslipidemia and hyperglycemia; this was reproduced in the model used in this work with an HCD and AG II diet. Prolonged hyperglycemia and insulin resistance result in hyperinsulinemia; it up-regulates the components of the renin-angiotensin system and sharpens the pro-inflammatory and pro-fibrotic condition mediated by AG II, which is associated with macrovascular complications of diabetes. The imbalance of RAS due to the increase of AG II in tissues such as kidney, liver and adipose tissue by hyperglycemia may contribute to hypertension associated with MS and to the organ damage observed in diabetes mellitus II [
23].
The data obtained in this work showed for the first time the effect of
S. aristolochiifolia on an injury model associated with MS. The previous pharmacological use of this plant indicated only the diuretic effect [
14]. There are other works which have shown that species of the same genus induced a beneficial effect on different alterations associated to MS. For example,
S. officinalis, inhibits α-glucosidase enzymes and possesses an antioxidant effect [
25];
S. canariensis has a diuretic effect [
26];
S. aspera and
S. china Linn decrease the plantar inflammation induced by carrageenan [
27,
28]. The latter possesses the compound sieboldogenin that inhibits the activity of lipoxygenase [
28]. Glycosides (smilasides G-L) from
S. bracteata have scavenger activity against DPPH radicals [
29] and
S. glabra has shown a hypoglycemic effect in a test of insulin tolerance in mice [
30].
In the present study, the administration of the AceSa extract and fractions F1, F2 and F3 from
S. aristolichiifolia were an effective treatment to control the major alterations associated with the MS, in C57BL6 mice with damage induced by HCD and chronic administration of AG II. This is the case of the AceSa extract, which counteracted the effect of the HCD diet and AG II on the growth curve (corroborated by the decrease of body density), and kept the animals under conditions similar to those of the baseline group. This effect was also observed in the group that received the combination of drugs pioglitazone and telmisartan. This is likely due to the presence of phenolic compounds in AceSa extract, since they have the capacity of inhibiting α-glucosidase enzymes, which are a therapeutic target for the treatment of obesity in humans [
17]. Furthermore, triglyceride levels in mice with a HCD + AGII diet were higher than in animals that received only an HCD diet. This parameter decreased significantly in all groups that received some treatment from
S. aristolochiifolia (AceSa, Fi, F2 and F3), in comparison with the control II group. Although the results with F2 are worth noting, this treatment was better than all groups including the treatment with pioglitazone and telmisartan. Serum glucose levels, hypertension, and the local concentration of pro-inflammatory cytokines induced by HCD + AGII diet (control II) also decreased in the animals that received any treatment of
S. aristolochiifolia.
The chemical analysis indicated that all S. aristolochiifolia preparations used for the treatment of animals with MS contained different concentrations of isolated compound NTF; the highest level of this substance is in fraction F2. Then, all the pharmacological effects against MS in mice could be attributed mainly to the presence of NTF, because F2 possesses the major effect and the concentration of this compound is higher in it. This does not rule out the possibility of pharmacological interaction with other active principles, and that the pharmacological effect observed could be the result of different activities related to the complex composition of the fraction.
It is worth noting that the effects of different treatments of
S. aristolochiifolia could be due to NTF because it has been described as a noncompetitive inhibitor of α-glucosidases, which explains its anti‑hyperglycemic capacity [
17]. Also, it was shown that NTF has an antioxidant capacity through the sequestration of free radicals [
15]. On the other hand, NTF counteracts the inflammatory disorders of MS, and a dose-dependent regulation effect on the release of nitric oxide (respiratory burst) stimulated by LPS in a cell culture RAW 264.7 has been demonstrated [
31].
However, the most remarkable pharmacological effect of NTF, which might explain part of the effect of the extract of
S. aristolochiifolia as anti-MS, is that it acts as ligand for the gamma receptor activated by the peroxisome proliferator (PPAR-γ). Therefore, NTF is capable of improving the morbid state of visceral obesity and the conditions resulting from this disease. This occurs because PPAR-γ2 is expressed specifically in adipose tissue and is a master regulator in the differentiation and maturation of adipocytes [
32]. It has been shown that ligands for PPAR-γ2, improve the condition of insulin resistance. For all this, the administration of these substances can be an effective treatment for the prevention and/or improvement not only of type 2 diabetes mellitus, but also of the insulin resistance syndrome with hyper-insulinemia, abnormal lipid metabolism, obesity, hypertension and atherosclerotic disease [
33].
It is noteworthy that the administrations of synthetic drugs of the type of glitazones (which act on PPAR) have shown adverse side effects, especially cardio-toxicities such as congestive heart failure, arrhythmias and peripheral edema [
34]. Nevertheless, the widespread use of the extract of
Smilax aristoliochiifolia in soft drinks such as “
zarzaparilla” root beer allows us to assume that NFT might be free of any toxic effects. Furthermore, MS-associated inflammation was monitored by quantifying the concentration in kidney and in adipose tissue of pro-inflammatory cytokines such as IL-6, IL-1β and TNF-α, as well as the anti-inflammatory IL-10.
It was observed that the group with HCD diet had a non-significant increase in the concentration of pro-inflammatory cytokines with respect to the level of IL-10 in the kidney; even so, the injury model fed with HCD and AG II showed a significant increase of these molecules in the kidney, indicating that the chronic administration of AG II induces an exacerbated pro-inflammatory state.
Obesity and hypertension are recognized as diseases with an important inflammatory background; a study showed that hypertensive and obese patients had low levels of IL-10 and high serum concentrations of IL-8, compared with hypertensive subjects with normal weight [
35]. The mechanism of AG II-mediated chronic hypertension also activates T cells by AGII, which promotes vascular inflammation and elevated pressure [
36]. This explains the essential role described for TNF-α as a blood pressure regulator. Regarding the increase in blood pressure caused by the infusion of AG II in hypertensive rats fed with high salt diet, etanercept (TNF-α inhibitor) retards the progression of hypertension, an effect that is attributed to a reduction of kidney damage, due to a decrease of proteinuria and of the infiltration of macrophages/monocytes [
37]. Another study indicates that in knockout mice for TNF-α, the infusion of AG II for two weeks fails to cause hypertension [
38].