Influence of Temperature and Preserving Agents on the Stability of Cornelian Cherries Anthocyanins
Abstract
:1. Introduction
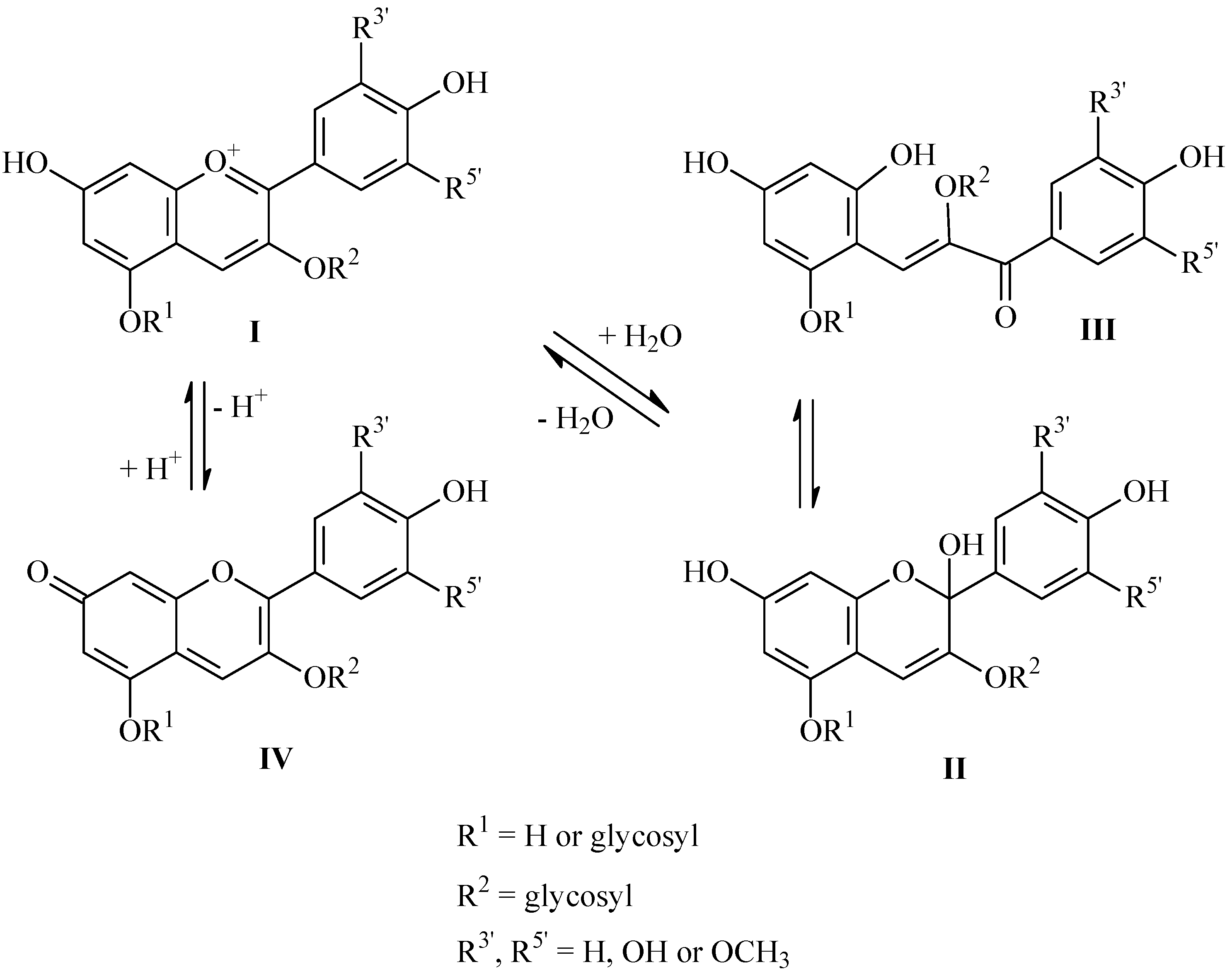
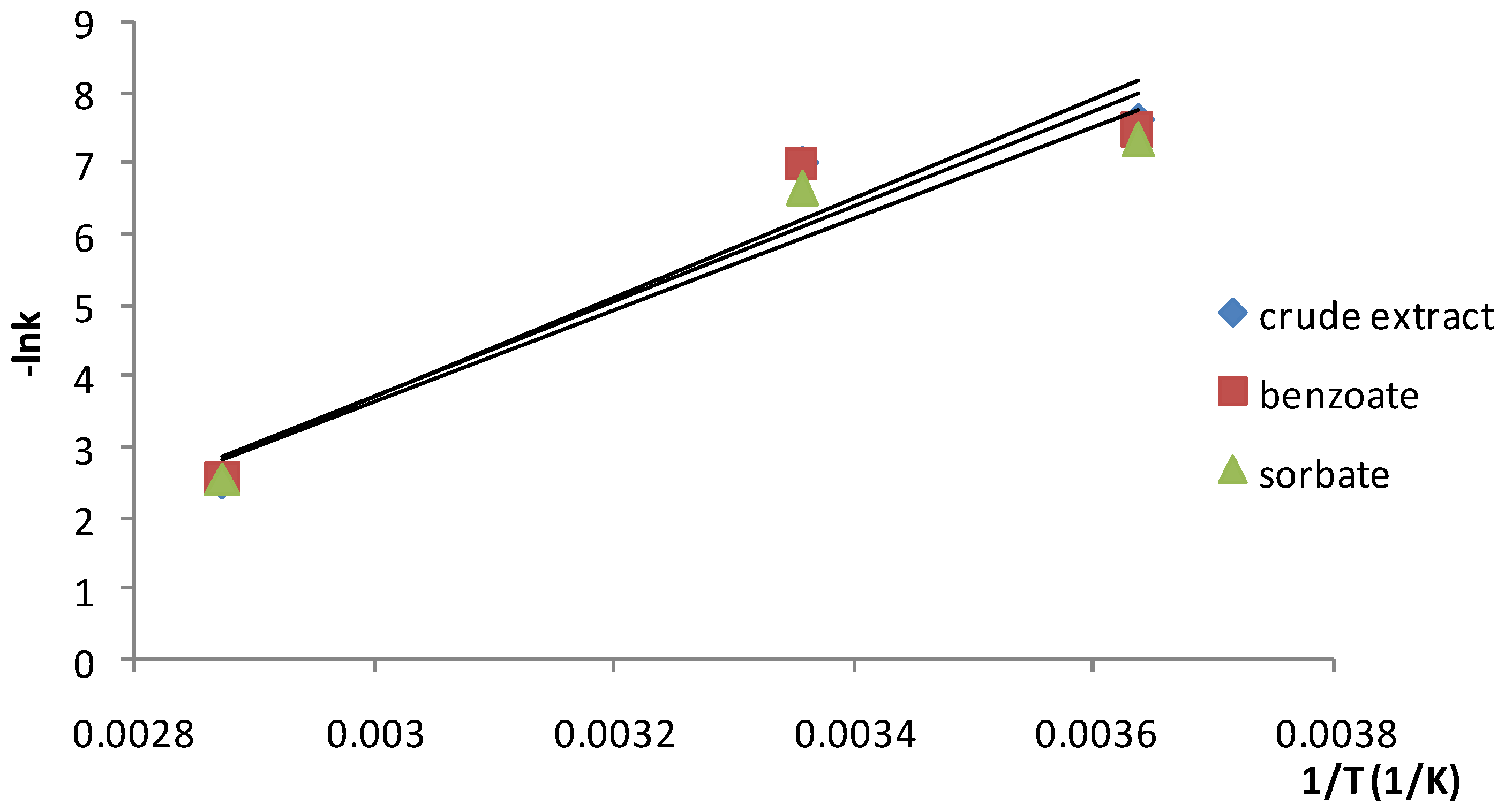
2. Results and Discussion
| Sample | Temp. (°C) | k∙10−3 (h−1) 1 | t1/2 (h) 2 |
|---|---|---|---|
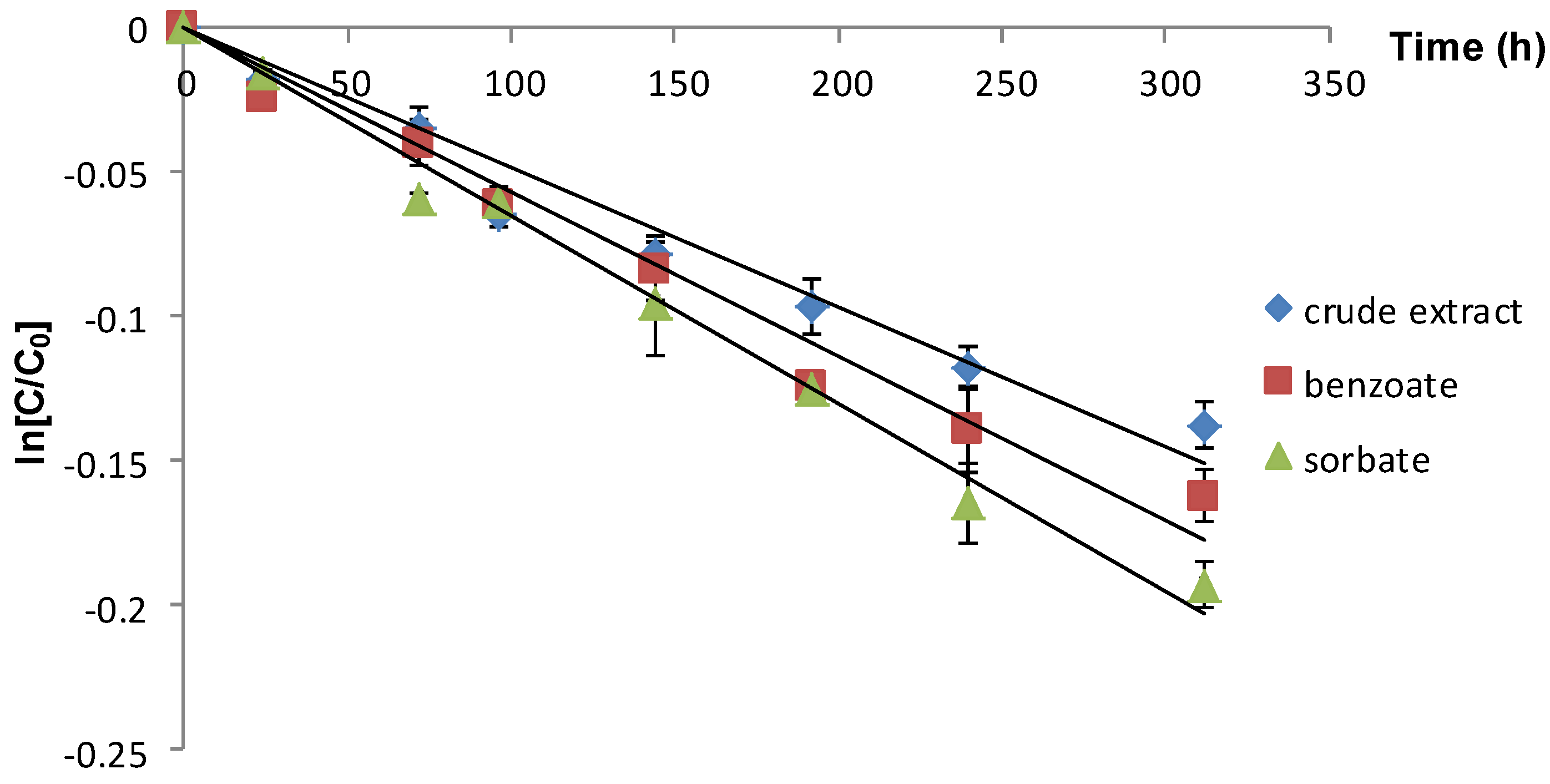
| Crude extract | 2 | 0.48 (0.9632) | 1443.75 a |
| Extract+sodium benzoate | 2 | 0.57 (0.9733) | 1215.78 b |
| Extract+potassium sorbate | 2 | 0.65 (0.9903) | 1066.15 c |
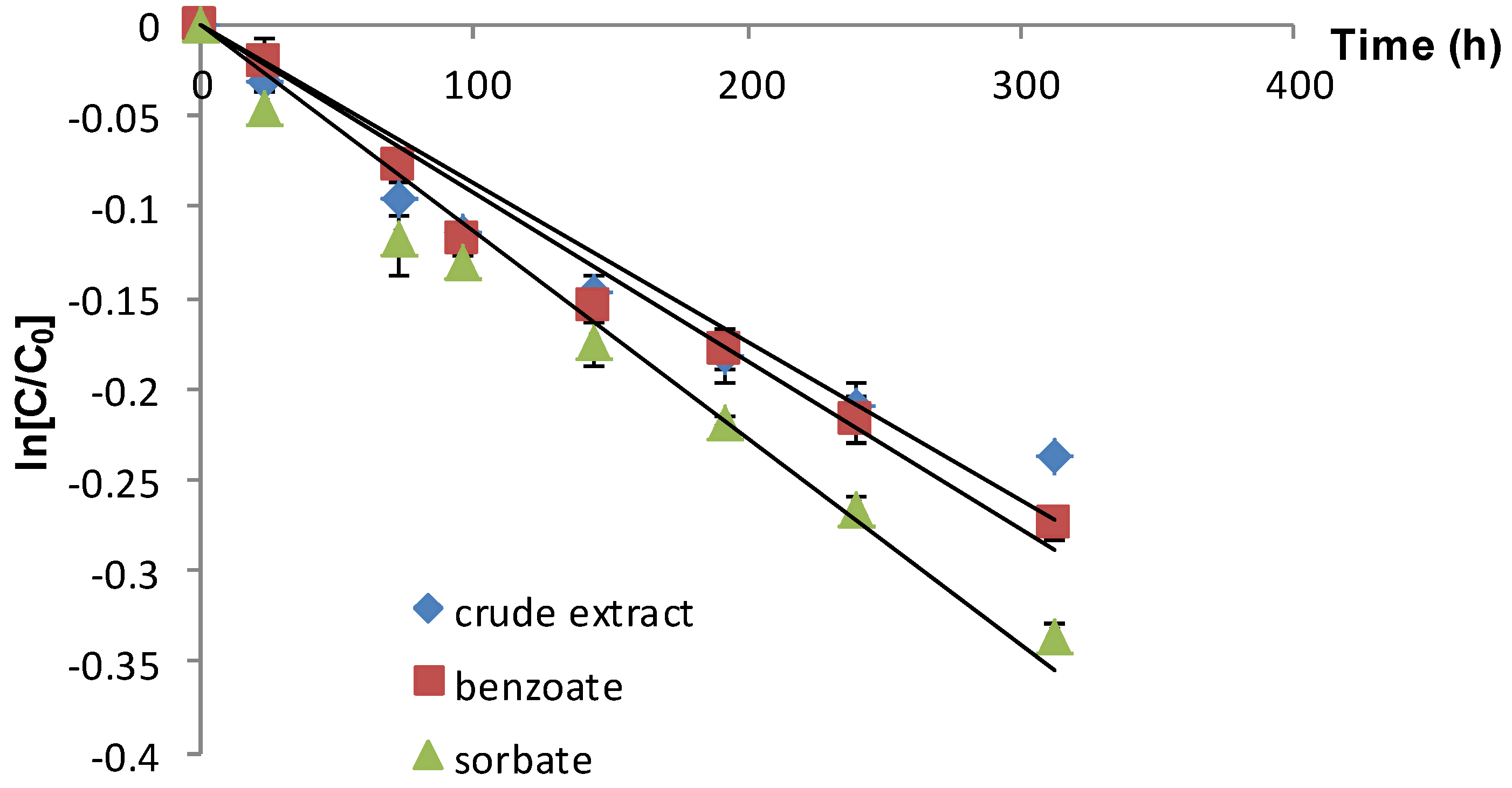
| Crude extract | 22 | 0.87 (0.9188) | 796.55 a |
| Extract+sodium benzoate | 22 | 0.93 (0.9739) | 745.16 b |
| Extract+potassium sorbate | 22 | 1.13 (0.9703) | 613.27 c |
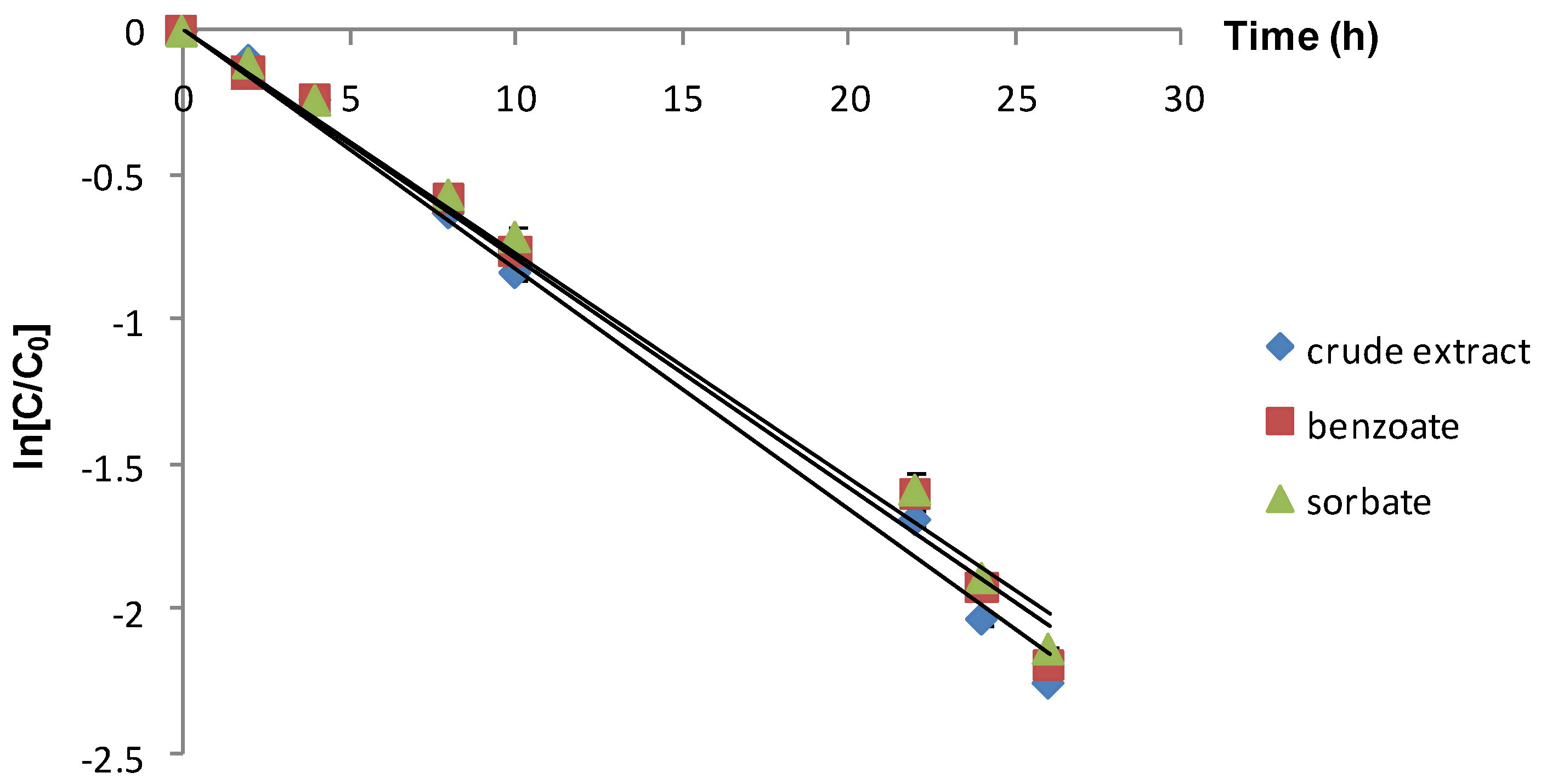
| Crude extract | 75 | 82.61 (0.9922) | 8.38 a |
| Extract+sodium benzoate | 75 | 78.84 (0.9908) | 8.78 a |
| Extract+potassium sorbate | 75 | 77.35 (0.9912) | 8.95 a |
| Solvent | Ea (kJ/mol) a | Ko (h−1) | Q10 | |
|---|---|---|---|---|
| 2–22 °C | 22–75 °C | |||
| Crude extract | 58.55 (0.9307) | 3.72 × 107 | 1.346 | 2.361 |
| Extract+sodium benzoate | 56.21 (0.9226) | 1.57 × 107 | 1.277 | 2.311 |
| Extract+potassium sorbate | 54.09 (0.9446) | 7.9 × 106 | 1.318 | 2.219 |
3. Experimental
3.1. Materials
3.1.1. Plant Material
3.1.2. Chemicals and Reagents
3.2. Methods
3.2.1. Preparation of Anthocyanin Extract
3.2.2. Determination of Anthocyanin Content
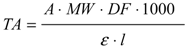
3.2.3. Degradation Studies
3.2.4. Degradation Kinetics
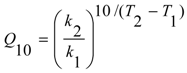
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Seeram, N.P.; Schutzki, R.; Chandra, A.; Nair, M.G. Characterization, quantification and bioactivities of anthocyanins in Cornus species. J. Agric. Food Chem. 2002, 50, 2519–2523. [Google Scholar]
- Celik, S.; Bakirci, I.; Sat, I.G. Physico-chemical and organoleptic properties of yogurt with cornelian cherry paste. Int. J. Food Prop. 2006, 9, 401–408. [Google Scholar] [CrossRef]
- Vareed, S.K.; Reddy, M.K.; Schutzki, R.E.; Nair, M.G. Anthocyanins in Cornus alternifolia, Cornus controversa, Cornus kousa and Cornus florida fruits with health benefits. Life Sci. 2006, 78, 777–784. [Google Scholar] [CrossRef]
- Polinicencu, C.; Popescu, H.; Nistor, C. Vegetal extracts for cosmetic use: Extracts from fruits of Cornus mas. Preparation and characterization. Clujul Med. 1980, 53, 160–163. [Google Scholar]
- Pantelidis, G.E.; Vasilakakis, M.; Manganaris, G.A.; Diamantidis, G. Antioxidant capacity, phenol, anthocyanin and ascorbic acid contents in raspberries, blackberries, red currants, gooseberries and Cornelian cherries. Food Chem. 2007, 102, 777–783. [Google Scholar] [CrossRef]
- Mazza, G. Natural Food Colorants: Science and Technology; Marcel Decker: New York, NY, USA, 2000; pp. 289–314. [Google Scholar]
- Reyes, L.F.; Cisneros-Zevallos, L. Degradation kinetics and colour of anthocyanins in aqueous extracts of purple- and red-flesh potatoes (Solanum tuberosum L.). Food Chem. 2007, 100, 885–894. [Google Scholar] [CrossRef]
- Prodanov, M.P.; Dominguez, J.A.; Blazquez, I.; Salinas, M.R.; Alonso, G.L. Some aspects of the quantitative/qualitative assessment of commercial anthocyanin-rich extracts. Food Chem. 2005, 90, 585–596. [Google Scholar] [CrossRef]
- Yoshimoto, M.; Okuno, S.; Yamaguchi, M.; Yamakawa, O. Antimutagenicity of Deacylated Anthocyanins in Purple-fleshed Sweetpotato. Biosci. Biotechnol. Biochem. 2001, 65, 1652–1655. [Google Scholar] [CrossRef]
- Tsuda, T.; Horio, F.; Uchida, K.; Aoki, H.; Osawa, T. Dietary cyanidin 3-O-β-d-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J. Nutr. 2003, 133, 2125–2130. [Google Scholar]
- Smith, M.A.L.; Marley, K.A.; Seigler, D.; Singletary, K.W.; Meline, B. Bioactive Properties of Wild Blueberry Fruits. J. Food Sci. 2000, 65, 352–356. [Google Scholar] [CrossRef]
- Crisan, M.; David, L.; Moldovan, B.; Vulcu, A.; Dreve, S.; Perde-Schrepler, M.; Tatomir, C.; Filip, A.G.; Bolfa, P.; Achim, M.; et al. New nanomaterials for the improvement of psoriatic lesions. J. Mater. Chem. B 2013, 1, 3152–3158. [Google Scholar] [CrossRef]
- Cevallos-Casalas, B.A.; Cisneros-Zevallos, L. Stability of anthocyanin-based aqueous extracts of Andean purple corn and red-fleshed sweet potato compared to synthetic and natural colorants. Food Chem. 2004, 86, 69–77. [Google Scholar] [CrossRef]
- Özkan, M.; Yemenicioğlu, A.; Asefi, N.; Cemeroğlu, B. Degradation kinetics of anthocyanins from sour cherry, pomegranate, and strawberry juices by hydrogen peroxide. J. Food Sci. 2002, 67, 525–529. [Google Scholar] [CrossRef]
- Hernández-Herrero, J.A.; Frutos, M.J. Degradation kinetics of pigment, colour and stability of the antioxidant capacity in juice model systems from six anthocyanin sources. Int. J. Food Sci. Technol. 2011, 46, 2550–2557. [Google Scholar] [CrossRef]
- Garzon, G.A.; Wrolstad, R.E. Comparison of the stability of pelargonidin-based anthocyanins in strawberry juice and concentrate. J. Food Sci. 2002, 67, 1288–1299. [Google Scholar] [CrossRef]
- Moldovan, B.; David, L.; Chisbora, C.; Cimpoiu, C. Degradation kinetics of anthocyanins from European Cranberrybush (Viburnum opulus L.) fruits extracts. Effects of temperature, pH and storage solvent. Molecules 2012, 17, 11655–11666. [Google Scholar] [CrossRef]
- Moldovan, B.; David, L.; Donca, R.; Chisbora, C. Degradation kinetics of anthocyanins from crude ethanolic extract from sour cherries. Stud. Univ. Babes-Bolyai Chem. 2011, 56, 189–194. [Google Scholar]
- Gurtler, J.B.; Bailey, R.B.; Geveke, J.D.; Zhang, H.Q. Pulsed electric field inactivation of E. coli O157:H7 and non-pathogenic surrogate E. coli in strawberry juice as influenced by sodium benzoate, potassium sorbate, and citric acid. Food Control. 2011, 22, 1689–1694. [Google Scholar] [CrossRef]
- Thakur, B.R.; Arya, S.S. Studies on stability of blue grape anthocyanins. Int. J. Food Sci. Technol. 1989, 24, 321–326. [Google Scholar] [CrossRef]
- Chisté, R.C.; Lopes, A.S.; DeFaria, L.J.G. Thermal and light degradation kinetics of anthocyanin extracts from mangosteen peel (Garcinia mangostana L.). Int. J. Food Sci. Technol. 2010, 45, 1902–1908. [Google Scholar] [CrossRef]
- Kirca, A.; Özkan, M.; Cemeroğlu, B. Effects of temperature, solid content and pH on the stability of black carrot anthocyanins. Food Chem. 2007, 101, 212–218. [Google Scholar] [CrossRef]
- Wang, W.D.; Xu, S.Y. Degradation kinetics of anthocyanins in blackberry juice and concentrate. J. Food Eng. 2007, 82, 271–275. [Google Scholar] [CrossRef]
- Amaro, L.F.; Soares, M.T.; Pinho, C.; Almeida, I.F.; Pinho, O.; Ferreira, I.M. Processing and storage effects on anthocyanin composition and antioxidant activity of jams produced with Camarosa strawberry. Int. J. Food Sci. Technol. 2013, 48, 2071–2077. [Google Scholar]
- Jackman, R.L.; Yada, R.Y.; Tung, M.A. A review: Separation and chemical properties of anthocyanins used for their qualitative and quantitative analysis. J. Food Biochem. 1987, 11, 279–308. [Google Scholar] [CrossRef]
- Kirca, A.; Cemeroğlu, B. Thermal degradation of blood orange anthocyanins. Food Chem. 2003, 81, 583–587. [Google Scholar] [CrossRef]
- Fang-Chiang, H.J.; Wrolstad, R.E. Anthocyanins pigment composition of blackberries. J. Food Sci. 2005, 70, 198–202. [Google Scholar] [CrossRef]
- Rommel, A.; Wrolstad, R.E. Blackberry juice and wine: Processing and storage effects on anthocyanin, color and appearance. J. Food Sci. 1992, 57, 385–391. [Google Scholar] [CrossRef]
- Cao, S.Q.; Liu, L.; Lu, Q.; Xu, Y.; Pan, S.Y.; Wang, K.X. Integrated effects of ascorbic acid, flavonoids and sugars on thermal degradation of anthocyanins in blood orange juice. Eur. Food Res. Technol 2009, 228, 975–983. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Current Protocols in Food Analytical Chemistry; Wiley: New York, NY, USA, 2001; pp. 1–13. [Google Scholar]
- Sample Availability: Samples of extracts are available for the next 2 months (the anthocyanins from the extracts totally degraded after this time period) from the authors.
© 2014 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moldovan, B.; David, L. Influence of Temperature and Preserving Agents on the Stability of Cornelian Cherries Anthocyanins. Molecules 2014, 19, 8177-8188. https://doi.org/10.3390/molecules19068177
Moldovan B, David L. Influence of Temperature and Preserving Agents on the Stability of Cornelian Cherries Anthocyanins. Molecules. 2014; 19(6):8177-8188. https://doi.org/10.3390/molecules19068177
Chicago/Turabian StyleMoldovan, Bianca, and Luminiţa David. 2014. "Influence of Temperature and Preserving Agents on the Stability of Cornelian Cherries Anthocyanins" Molecules 19, no. 6: 8177-8188. https://doi.org/10.3390/molecules19068177




