1. Introduction
Dengue fever (DF) and dengue hemorrhagic fever (DHF) are acute febrile diseases transmitted by mosquitoes. Nowadays, they are the most rapidly spreading mosquito-borne diseases in the world. About 2.5 billion people, two-fifths of the world’s population, are now at risk of infection and 50 million cases of DF are reported worldwide every year [
1]. In recent decades, these diseases have spread to over more than 100 countries [
2]. The World Health Organization (WHO) estimates that the annual global incidence of dengue is close to 390 million, a number nearly three times higher than the number of cases estimated by the same organization for 2009 [
3].
Increased urbanization along with substandard housing, unreliable water supply, and poor sanitation provide a suitable environment for vector proliferation in close proximity to human hosts. Dengue proliferation became even more problematic in the Americas due to the collapse of vector eradication programs in the 1970s [
4].
The dengue virus particle is about 50 nm in diameter. The 10,723-nucleotide RNA genome encodes an uninterrupted open reading frame (ORF), directing the synthesis of a polyprotein precursor in the order NH
2-C-prM-E-NS1-NS2A-NS2B-NS3-NS4A-NS4B-NS5-COOH, where C is the capsid protein, M is the membrane-associated protein, E is the envelope protein, and NS1 through NS5 are nonstructural proteins [
5].
The disease has four viral serotypes (DENV 1–4), and its spectrum ranges from asymptomatic infection to dengue fever (DF), dengue hemorrhagic fever (DHF), and dengue shock syndrome (DSS), and may lead to patient death [
6]. All four serotypes of dengue virus are transmitted to humans by the
Aedes aegypti and
Aedes albopictus mosquitoes [
7].
During a dengue outbreak that struck Malaysia’s Sarawak state, on Borneo, blood and serum samples from a severe case labeled “dengue 4” were collected, and later, the sequence of the genome showed that the virus occupies a new branch on the dengue family tree, suggesting the serotype DENV-5 [
3]. Provided that this new serotype is transmissible as DENV 1–4, it might follow a similar pattern of geographical spread as described by Messina and co-workers. By using several maps, the authors demonstrated the expansion of the serotypes throughout the world, the growth of hyperendemicity (coexistence of multiple serotypes), and the establishment of dengue as an important infectious disease of global public health importance [
8].
To date, there is no clinically approved dengue vaccine or antiviral for humans, even though there have been great efforts towards this end. The treatment of the disease is limited to supportive care [
9,
10] with analgesics, fluid replacement and bed rest [
11]. Aspirin, non-steroidal anti-inflammatory drugs (NSAIDs), and corticosteroids should be avoided. Special attention should be given to severe cases of dengue in terms of fluid administration and treatment of hemorrhage. A placebo-controlled, double-blind investigation was conducted with sixty-three children having severe dengue shock syndrome in two hospitals in Thailand. The children were completely randomized into two groups. One of the groups was treated with a single dose of steroidal drug methylprednisolone and a placebo was administered to the other group. The study revealed that there was no significant difference in mortality between the groups [
12].
The cost of dengue to society is considerable, from lost wages and diminished productivity to costs related to care-giving and direct medical expenses. The cost of dengue in the Western Hemisphere alone is estimated to be US$2.1 billion per year [
13]. In view of these problems, an efficient dengue vaccine or antiviral is highly desirable.
Different approaches have been used in the search for dengue antivirals, such as screening of compounds against dengue enzymes [
14] and structure-based computational discovery [
15]. During the last decades, researchers have turned their attention to nature, trying to identify compounds that can be used as dengue antivirals. In fact, nature is a fantastic reservoir of substances that can be used directly as pharmaceuticals or can serve as lead structures that can be optimized towards the development of new therapeutic agents [
16,
17,
18,
19,
20,
21,
22,
23].
Several plants around the world present potential dengue antiviral activity. Recently, Kadir and co-workers reviewed sixty nine studies from 1997 to 2012 related to plants presenting potential antidengue activity [
24]. It should be mentioned that according to a WHO factsheet, 80% of the population in some Asian and African countries depends on traditional medicine as their primary health care due to economic and geographical constraints [
25,
26,
27]. In view of their few (or lack of) adverse effetcts, the world-wide use of medicinal plants or herbal-based medicine is steadily growing.
Even though a number of plants are known for their antidengue activity, few investigations have been published related to isolation (identification) of compounds from plants and subsequent evaluation of their dengue antiviral activities. We describe in this review several investigations which resulted in the isolation (or identification) of compounds endowed with antiviral dengue activity, most of them isolated from plants. The review also covers metabolites isolated from other natural sources.
2. Polysaccharides
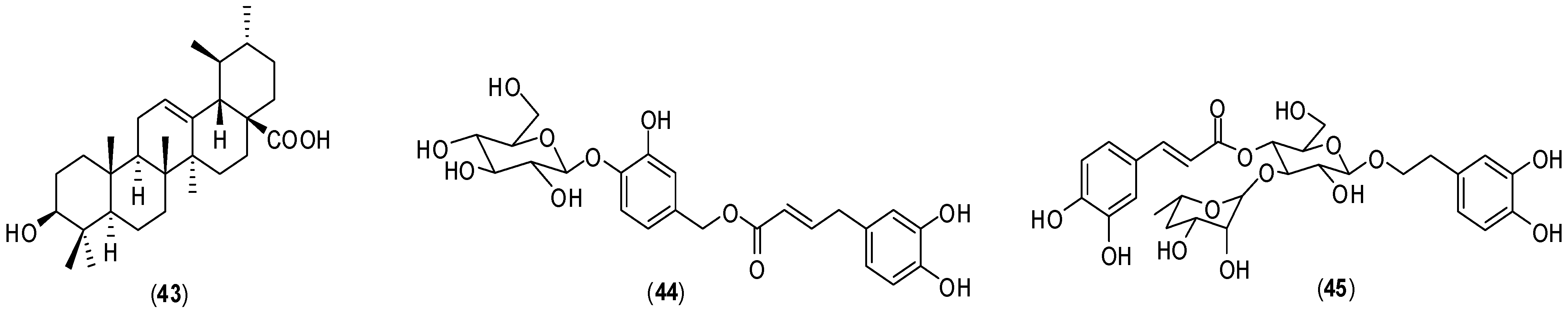
Fucoidans are a group of polysaccharides which contain considerable percentages of L-fucose and sulfate ester groups. These compounds are mainly derived from brown seaweed, and several bioactivities have been described for them, including antiviral ones [
28,
29,
30]. Fucoidan (
1,
Figure 1), is a polysaccharide isolated from the marine alga brown seaweed
Cladosiphon okamuranus. Its structure is composed of repeating units of sulphated fucose and glucuronic acid residues. The investigation conducted with
Cladosiphon fucoidan (
1) demonstrated that this polysaccharide inhibits dengue virus type 2 (DENV-2) infection [
31]. The biological assays to evaluate antiviral activity were conducted
in vitro by focus-forming assay using BHK-21 cells. Compound
1 inhibited virus infection in a concentration-dependent matter. When the virus was treated with 10µg/mL of fucoidan (
1), infectivity by dengue virus serotype 2 was reduced by 80% compared with that in untreated cells and the determined IC
50 corresponded to 4.7 µg/mL. Dengue virus serotypes 3 and 4 were moderately susceptible to
1. For serotype 1, fucoidan (
1) did not present an effect on the infection. Fucoidan derivatives (
2–
4) (
Figure 1) were also examined for their effects on infection of BHK-21 cells by dengue virus serotype 2. Polysaccharide
2 is a derivative obtained by removal of the sulfated groups in fucoidan (
1); derivative
3 was prepared by reduction of fucoidan (
1). Fucan (
4) is a fucose polymer.
Figure 1.
Fucoidan (1) and derivatives 2–4.
Figure 1.
Fucoidan (1) and derivatives 2–4.
The desulfation of fucoidan (
1) led to derivatives
2 and
4 which showed remarkable suppression of inhibitory activity. This finding is in agreement with previous investigations which demonstrated that sulfation is required for the antiviral activity of glycosaminoglycans [
32]. Even though compound
3 is a sulfated derivative, the reduction of carboxylic acid functionality in fucoidan (
1) to the corresponding alcohol also resulted in a decrease of the capability of
3 in preventing serotype 2 virus infection. Therefore, it was concluded that the glucuronic acid residue as well as the sulphate groups are fundamental for the inhibitory activity of fucoidan (
1) against DENV-2 [
31]. It was also reported that glucuronic acid and sulfated fucose residues of the
Cladosiphon fucoidan appear to critically affect the interaction of DENV-2 with cellular receptors, but the precise molecular mechanism of the inhibitory effects of this compound has not been elucidated.
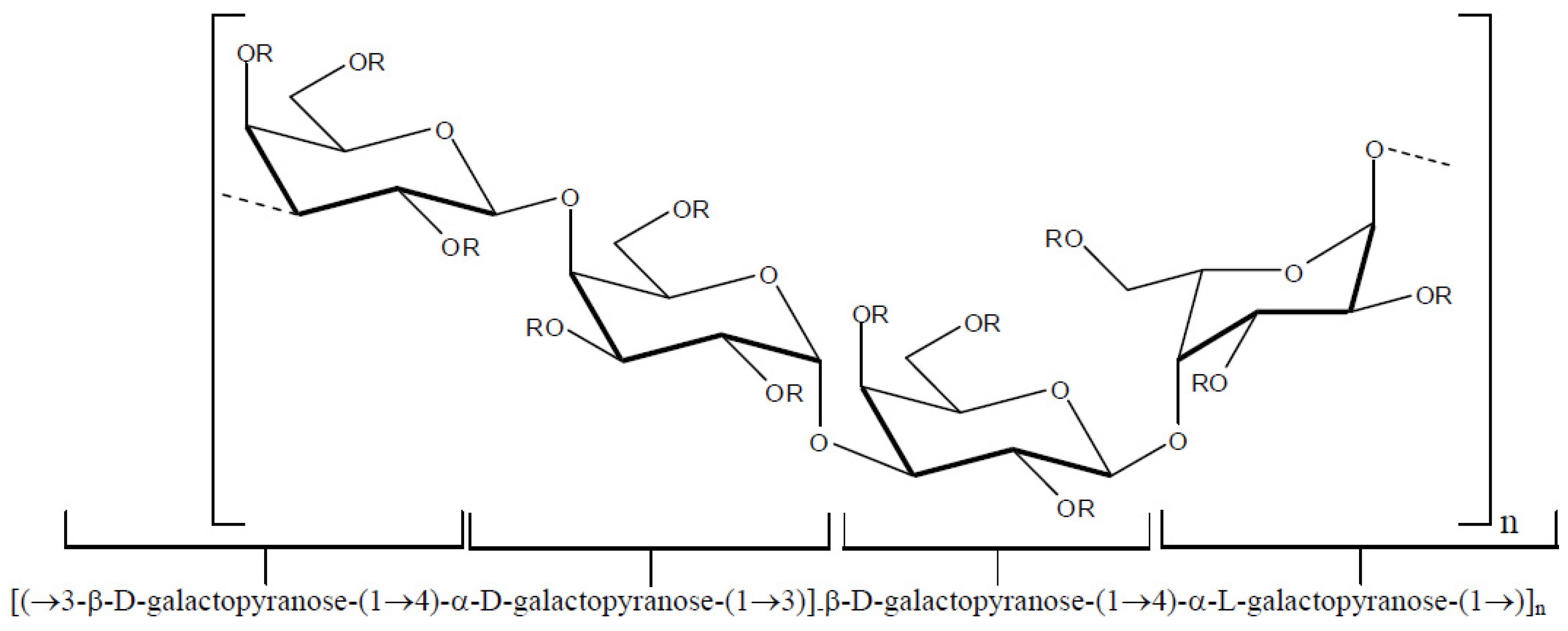
Several polysaccharides known as galactans have been isolated from red seaweeds [
33,
34,
35,
36]. The structures of these compounds correspond to a linear chain of β-
d-galactopyranose residues linked by positions 1 and 3 (
Figure 2, unit A) and residues of α-galactopyranose linked by positions 1 and 4 (
Figure 2, unit B) resulting in an arrangement in which units A and B are alternating. Natural chemical modifications in these structures include the presence of sulfate esters groups, pyruvate acetal and/or methyl ethers. In addition, unit B can exist in the 3,6-anhydro-α-galactopyranose form.
Figure 2.
Repeating basic structure of galactans.
Figure 2.
Repeating basic structure of galactans.
Depending on the stereochemistry of unit B (
Figure 2), the galactans can be classified as follows: (i) carrageenans—these correspond to sulfated polysaccharides with 4-linked α-galactose residues of the
d-series or their 3,6-anhydro derivatives (
Figure 3). Carrageenans are typically classified according to their structural features, including their sulfation patterns and the presence or absence of 3,6-anhydro-α-galactopyranose on
d-units. There are at least 15 different carrageenan structures, the most relevant being
iota,
kappa [
Figure 3, (I)],
lambda,
mu, and
nu [
Figure 3, (II)] [
37]. It is important to mention that natural carrageenans typically occur as mixtures of different hybrids. Moreover, methyl or pyruvic acid acetal moieties and the presence of small amounts of other sugars can add to the structures of these polysaccharides [
37].
Figure 3.
Repeating structures of carrageenans.
Figure 3.
Repeating structures of carrageenans.
(ii) agarans—this group of galactans differs from carrageenans in terms of the stereochemistry of unit B. For the agarans, the 4-linked α-galactose residues unit b correspond to the l-series.
(iii)
dl-hybrids—these galactans are characterized by the presence of 4-linked α-galactose residues of the
d and
l series in the same molecule.
Figure 4 depicts the basic repeating structure of
dl-hybrid galactans.
Figure 4.
Basic repeating unit of dl-hybrid galactans.
Figure 4.
Basic repeating unit of dl-hybrid galactans.
Commercially available
iota,
kappa and
lambda carrageenans (
Figure 3) were evaluated against DENV 1–4 serotypes [
38]. The assays were conducted
in vitro, using Vero and HepG2 cells and the activity of carrageenans was initially tested by plaque reduction assays, necessarily performed at a low multiplicity of infection (m.o.i.), but then confirmed by inhibition of virus yield and antigen expression assays, accomplished at high m.o.i. The polysaccharides were more effective on DENV-2 and DENV-3 serotypes. It was also determined that the carrageenans
lambda and
iota are potent inhibitors of DENV-2 and DENV-3 multiplication in Vero and HepG2 cells with EC
50 (effective concentration 50%) ranging from 0.14 to 4.1 µg/mL (
Table 1). The results showed that the lack of dependence of the antiviral potency of carrageenans on the infecting virus inoculum was even more evident when the assays were performed simultaneously at a wide range of multiplicities. This important property represents a clear advantage for those compounds able to block infection even in the presence of high initial virus doses.
Table 1.
EC50 values for inhibition of DEN-2 and DENV-3 multiplication in Vero and HepG2 cells.
Table 1.
EC50 values for inhibition of DEN-2 and DENV-3 multiplication in Vero and HepG2 cells.
| Polysaccharide | EC50 (µg/mL) |
|---|
| Vero Cells | HepG2 cells |
|---|
| DENV-2 | | |
| iota-carrageenan | 0.4 ± 0.1 | 0.14 ± 0.01 |
| lambda-carrageenan | 0.22 ± 0.02 | 0.17 ± 0.01 |
| DENV-3 | | |
| iota -carrageenan | 1.1 ± 0.1 | 0.63 ± 0.01 |
| lambda -carrageenan | 0.6 ± 0.1 | 0.63 ± 0.01 |
This study was able to demonstrate that a heparin sulfate (HS) imitative compound lambda had the ability to interfere with DENV-2 replication when added after virus adsorption, and even under these conditions, the antiviral potential of lambda-carrageenan was higher than its ability to affect virus adsorption. The mechanism of the inhibitory multiplication effect of the iota carrageenan was not described.
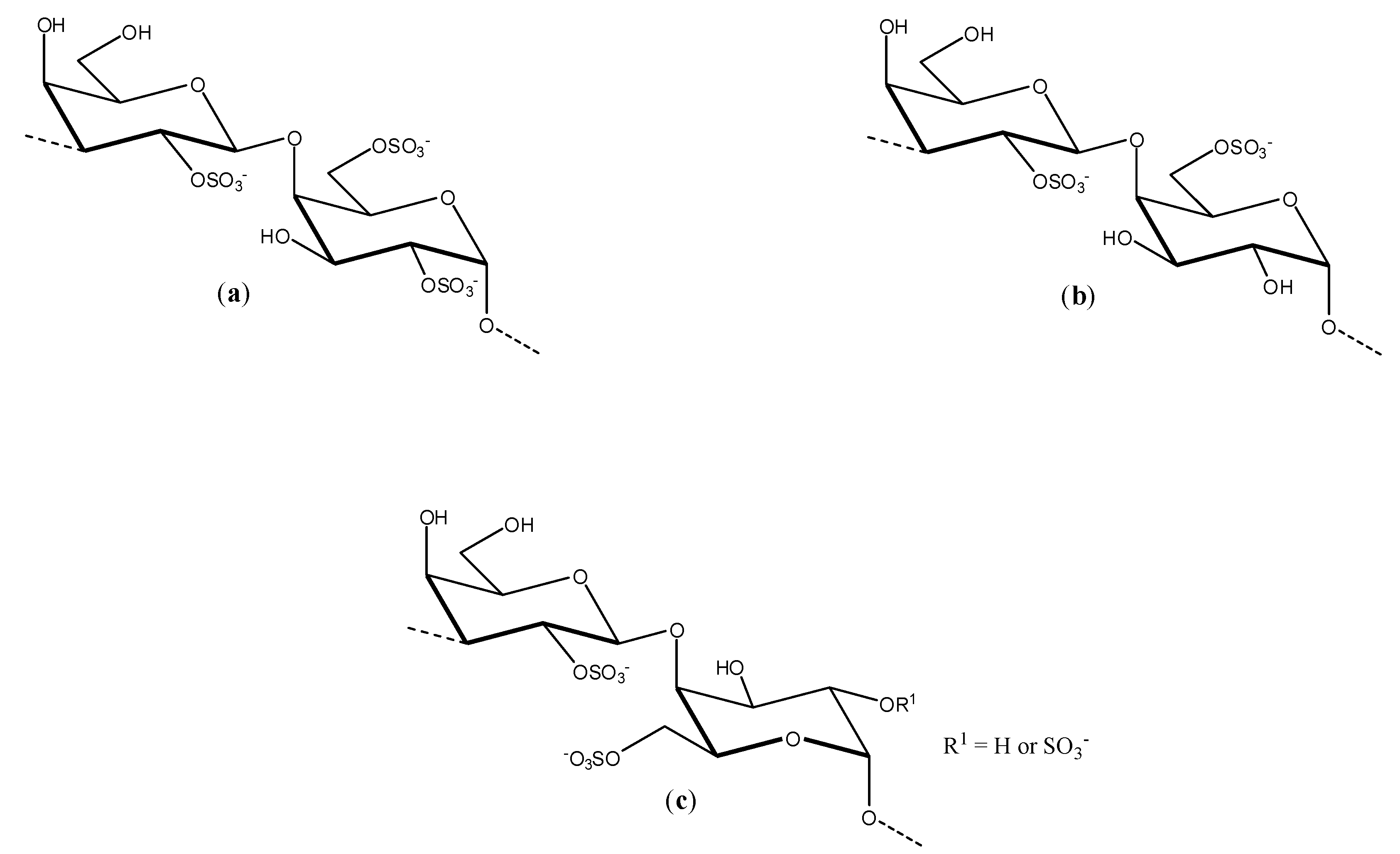
Talarico and co-workers investigated the dengue antiviral activity of two sulfated polysaccharides obtained from the read seaweeds
Gymnogongrus griffithsiae and
Cryptonemia crenulata [
39]. The
G3d compound obtained from
G. griffithsiae is a carrageenan composed of
kappa/
iota/
nu repeating units (see
Figure 3). The DL-galactan hybrid
C2S-3 obtained from
C. crenulata is made of the units disaccharide units (
a), (
b), and (
c) shown in
Figure 5.
Figure 5.
Disaccharides units present in the structure of C2S-3.
Figure 5.
Disaccharides units present in the structure of C2S-3.
The antiviral activity evaluation of
G3d and
C2S-3 was conducted
in vitro and it was carried out by two methods: plaque reduction and virus yield inhibition assays. It was found that the dengue antiviral activity of sulfated
G3d and
C2S-3 polysaccharides depends on both virus serotype and the host cell. Both compounds were capable of inhibiting DENV-2 in Vero cells with IC
50 values for the inhibitory activity close to 1 μg/mL and selectivity indexes superior to 1,000. The compounds were completely ineffective against DENV-1 and their inhibitory activity on DENV-3 (IC
50 ranging from 13.9–14.2 μg/mL) and DENV-4 (IC
50 ranging from 29.3 to > 50 μg/mL) were significantly lower. The inhibitory effects of
G3d and
C2S-3 against DENV-2 and DENV-3 were also evaluated with respect to human HepG2 and foreskin PH cells as well as mosquito C6/36 HT cells. While the polysaccharides were totally ineffective against mosquito cells, the effectiveness of the substances with respect HepG2 and PH cells were similar to that observed with Vero cells. From the mechanism of action standpoint, it was determined that
G3d and
C2S-3 present inhibitory effects on DENV-2 serotype only when they are added together with the virus or early after infection. Therefore, the processes of virus adsorption and internalization are the main targets of these compounds. The results suggest that these compounds act on virus binding. These polysaccharides may be useful tools to elucidate the mechanisms of binding and internalization of DENV serotypes to vertebrate and invertebrate cells, and also to establish structure-activity relationships [
39].
Another study involving sulfated polysaccharides showed that the extracts and carrageenans derived from
Meristiella gelidium were more effective inhibitors of DENV-2 compared to those derived from
G. griffithsiae. The antiviral property evaluation was performed
in vitro using Vero cells and virus plaque reduction assay. In this investigation, no description on the mechanism of action for DENV inhibition was reported [
40].
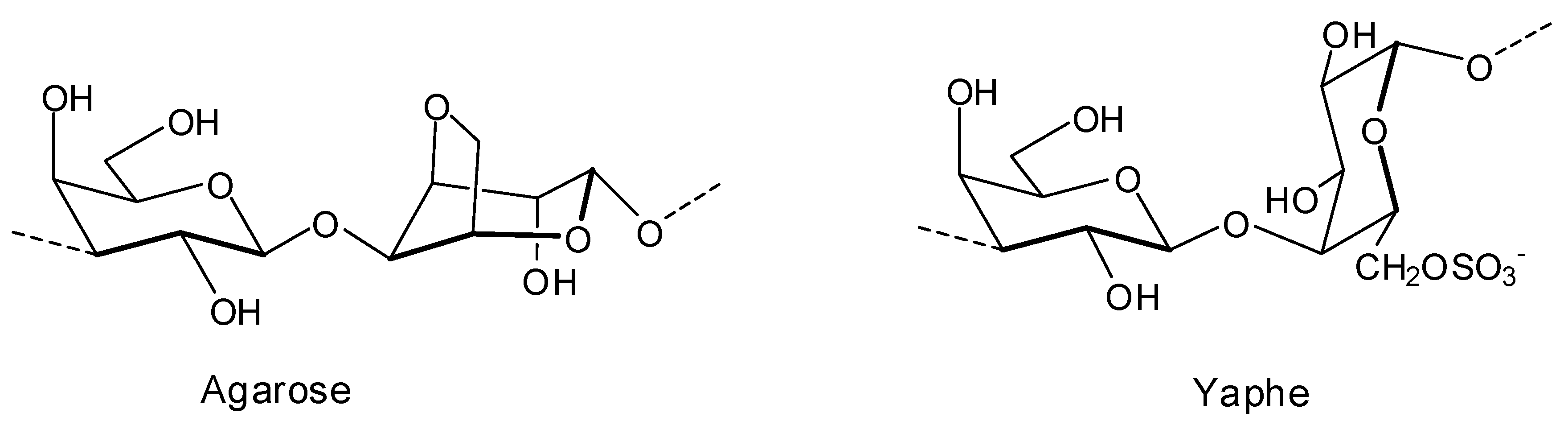
A series of DL-galactan hybrids isolated from the red seaweed
Gymnogongrus torulosus was assessed
in vitro against DENV-2 serotype by virus reduction assay in Vero cells [
41]. The repeating disaccharide units and the percentage of them found in the composition of the polysaccharides are presented in
Table 2. Structures of agarose and Yaphe repeating units are shown in
Figure 6.
Table 2.
.dl-galactan hybrids from G. torulosus and their IC50 values when the galactans were evaluated against DENV-2 virus serotype.
Table 2.
.dl-galactan hybrids from G. torulosus and their IC50 values when the galactans were evaluated against DENV-2 virus serotype.
| Polyssacharide | MW(kDa) a | Galactan Structure | IC50(μg/mL) c |
|---|
| Kappa/iota b | mu/nu b | Agarose b | Yaphe b |
|---|
| C1 | 44 | 57 | 10 | 19 | 12 | 1.1 ± 0.2 |
| C2 | 56 | 1845 | 18 | 22 | 12 | 0.7 ± 0.2 |
| C3 | 77 | 28 | 21 | 31 | 14 | 0.34 ± 0.06 |
| C4 | 18 | 54 | 6 | 7 | 17 | 0.19 ± 0.03 |
| F1 | 56 | 76 | - | 19 | 12 | 0.5 ± 0.2 |
| F2 | 77 | 73 | 12 | 12 | - | 0.8 ± 0.1 |
| F3 | 18 | 34 | - | 12 | 51 | 0.9 ± 0.2 |
| F3T2 | 35 | 62 | - | 32 | 20 | 0.25 ± 0.09 |
| F3T4 | 45 | 38 | 12 | 40 | 9 | 0.34 ± 0.02 |
| F3T6 | 22 | 17 | 8 | 8 | 59 | 1.7 ± 0.1 |
Figure 6.
Agarose and Yaphe repeating units found in dl-galactan hybrids isolated from the red seaweed G. torulosus.
Figure 6.
Agarose and Yaphe repeating units found in dl-galactan hybrids isolated from the red seaweed G. torulosus.
As can be seen in
Table 2, the evaluated polysaccharides were very active against DENV-2 serotype with IC
50 ranging from 0.19–1.7 μg/mL. In addition to their inhibitory activity, the compounds did not present cytotoxic effects on stationary or on actively dividing cells, and they presented anticoagulant properties. It is suggested that the mechanism of action of these compounds corresponds to interference in the binding of the surface glycoprotein with the cell receptor [
41].
One aspect deserves comment at this point. Liang and co-workers prepared sulfated agarose, sulfated
kappa-carrageenan, desulfated
kappa-carrageenan, and
kappa-carrageenan oligosaccharides to investigate their anticoagulant and cytotoxic activities. They found that anticoagulant activity and effects on cell proliferation are both dependent on the substitution position rather than the degree of sulfate group substitution. Moreover, these activities are dependent on the secondary structures of polysaccharides. The investigation led to the conclusion that carrageenan and agarose can be considered for biomedical applications after careful tailoring of sulfate groups [
42].
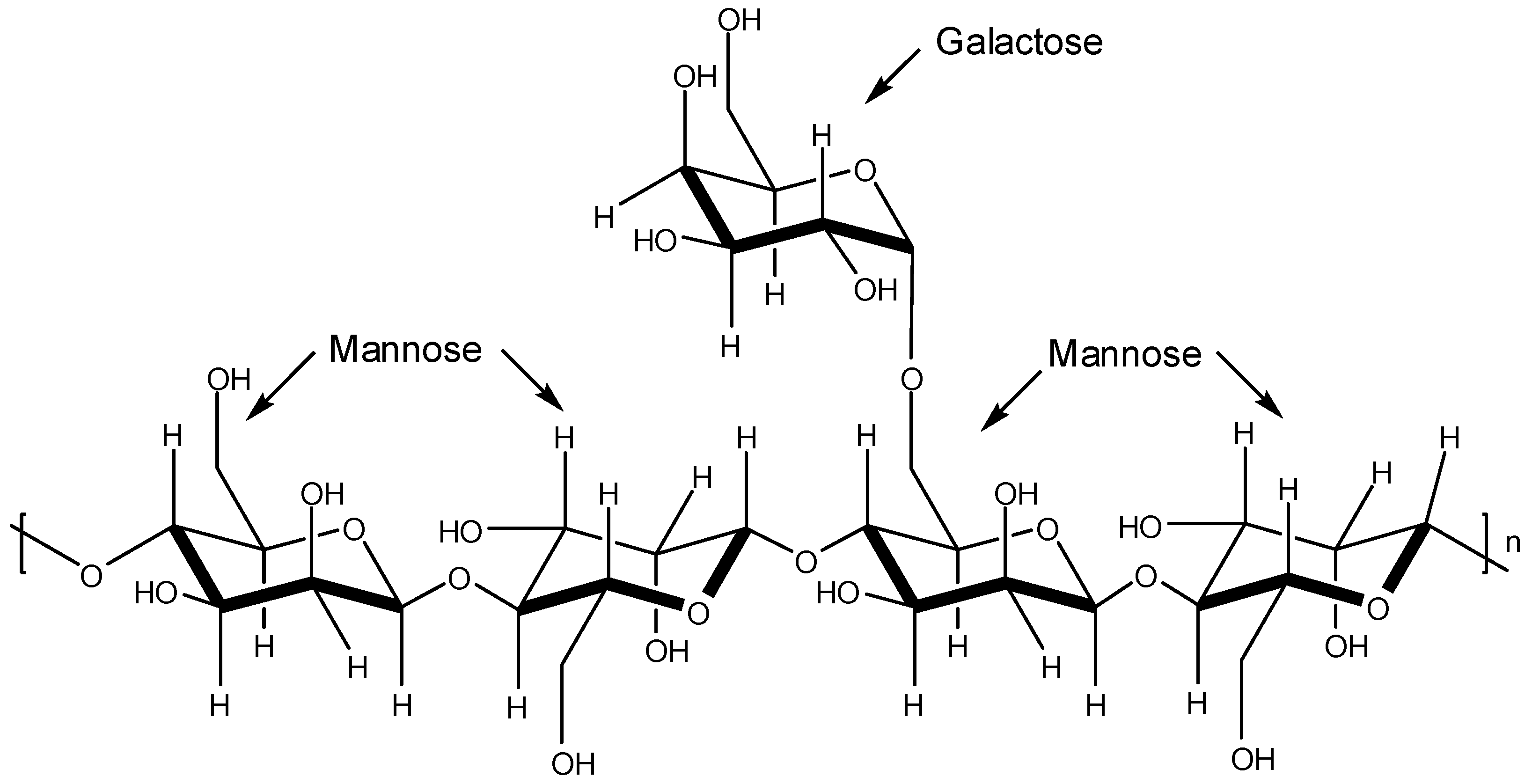
The plant storage polysaccharides known as galactomannans are characterized by a main chain of β-
d-mannopyranosyl residues linked by positions 1 and 4 with single unit α-
d-galactopyranosyl side-chain-residues (
Figure 7). The mannose/galactose ratio depends on either the plant source or the extraction method used [
43,
44].
Figure 7.
Basic structure of galactomannans.
Figure 7.
Basic structure of galactomannans.
Ono and co-workers isolated two sulfated galactomannans [
45]. One was isolated from the seeds of
Mimosa scabrella presenting a mannose/galactose ratio of 1.1 and the other was obtained from the seeds of
Leucaena leucocephala (mannose/galactose ratio of 1.4). These sulfated polysaccharides named by the authors as BRS (from
M. scabrella) and LLS (from
L. leucocephala) were submitted to
in vitro and
in vivo assays to assess their effects on DENV-1 (Hawaii strain) virus. The
in vivo assay was carried out with female mice to determine antiviral activity. The
in vitro evaluation was conducted with C6/36 cells by virus plaque reduction. Death of DENV-1-infected mice was not noticed. For the
in vitro assay, it was found that the concentrations that produced a 100-fold decrease in virus titer were 347 mg/L and 37 mg/L for BRS and LLS, respectively [
45].
3. Flavonoids
Several flavonoids have been screened for dengue antiviral activity.
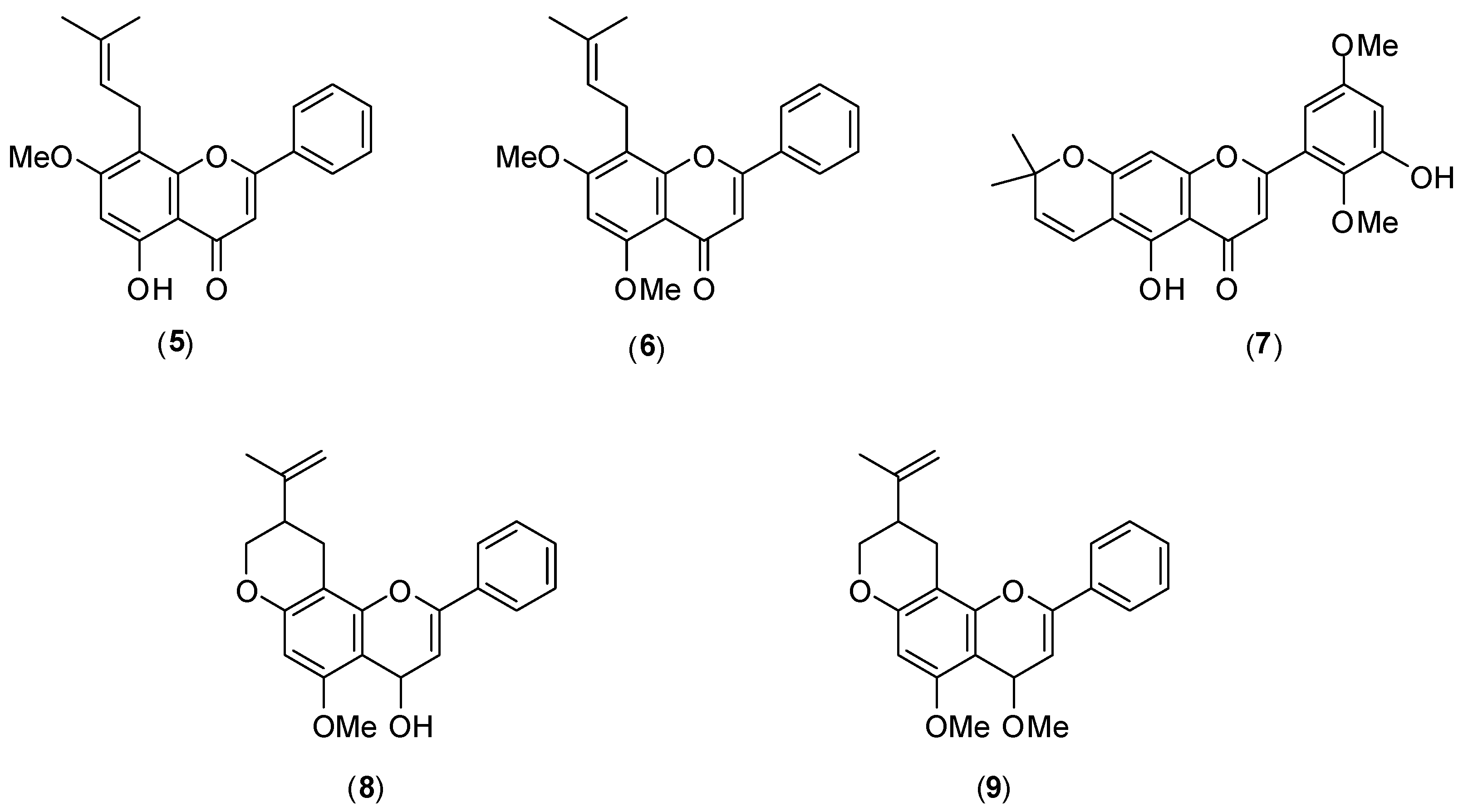
Figure 8 shows the structures of members of this class of compounds
5–
9, isolated from Mexican
Tephrosia species. Their antiviral activity was evaluated
in vitro by the plaque assay using LLC-MK
2 cells and DENV-2 serotype. Of the five flavonoids tested, only glabranine (
5) and 7-
O-methylglabranine (
6) showed significant inhibitory activity (they presented 70% virus infection inhibition at 25 µmol/L). No IC
50 values were reported. The other flavonoids
7–
9 had no antiviral effect. Considering the structures of compounds
5–
9, it is evident that a relationship exists between the structure and antiviral activity of the investigated flavonoids since glabranine (
5) and 7-
O-methylglabranine (
6), which both contain a prenyl side-chain at C-8, were active as replication inhibitors [
46].
Figure 8.
Structures of flanovoids from Mexican Tephrosia species
Figure 8.
Structures of flanovoids from Mexican Tephrosia species
The aqueous extract of
Houttuynia cordata, a popular side dish vegetable consumed in Northern and Eastern regions of Thailand, was tested against DENV-2 [
47]. The assays to evaluate antiviral aqueous activity were performed
in vitro in three different modes: (i) protective, (ii) treatment, and (iii) direct blocking in HepG2 and LLC-MK
2 cells. For (i) and (ii) the experiments were conducted using plaque titration assay, and for (iii) it was performed using plaque reduction assay. Considering HepG2 cells,
H. cordata aqueous extract displayed an inhibitory effect to DENV-2 RNA production in all experimental modes. The experiments were conducted at 10 μg/mL and 100 μg/mL. The higher concentration was effective to: (a) protect HepG2 cells from DENV-2 infection (protective mode); (b) decrease the intracellular viral RNA synthesis (treatment mode); (c) to inactivate the virus (direct blocking). The greatest inhibitory effect was observed in the protective mode. For LLC-MK
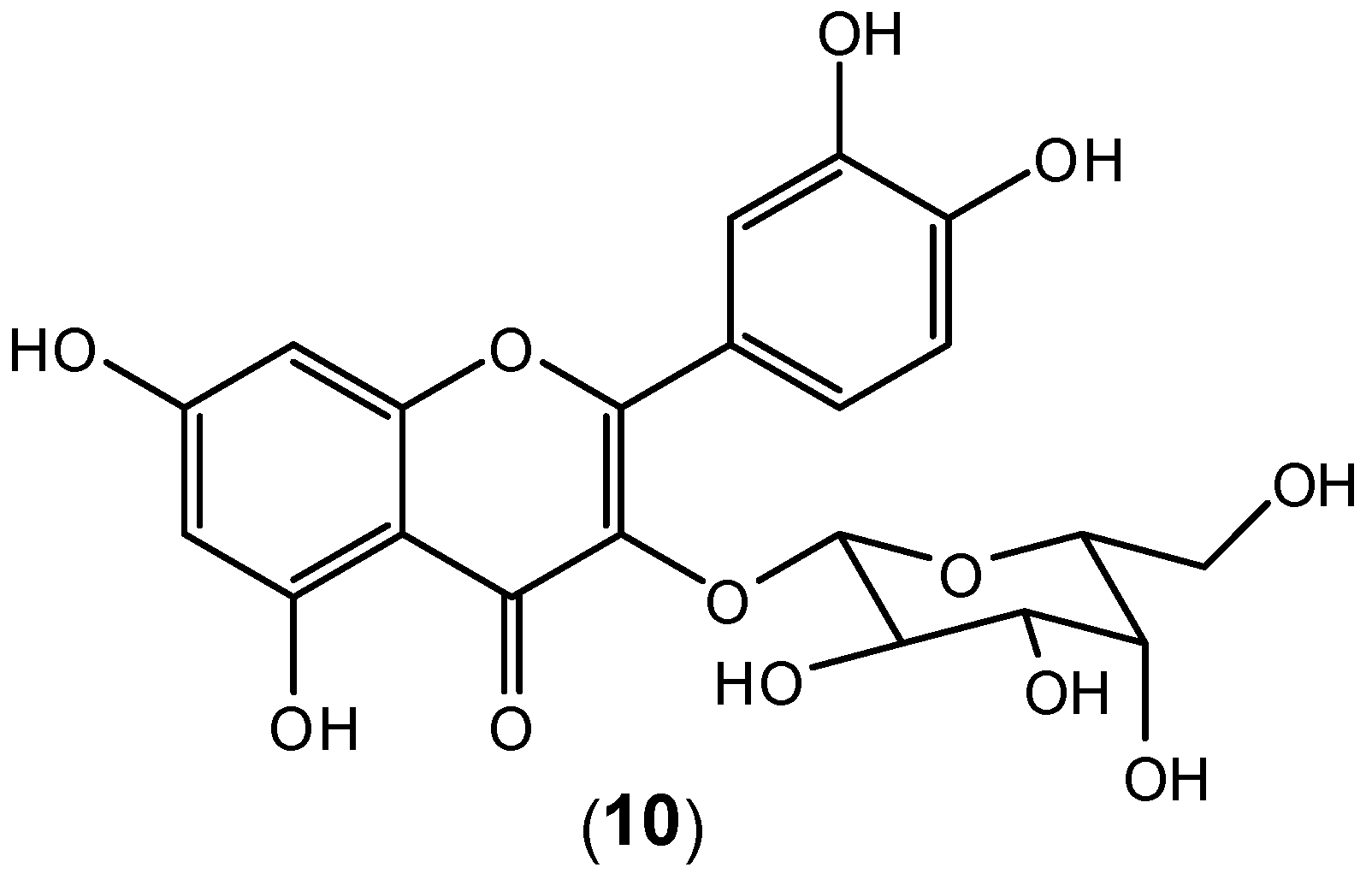
2 cells, the experiments were conducted within 10–40 μg/mL concentration range, and the aqueous extract also exhibited a protective effect on virion release. High Performance Liquid Chromatography (HPLC) analysis identified the flavonoid hyperoside (
10) as the major component of the extract (
Figure 9). It is very likely that the observed dengue antiviral activity is associated with the presence of this flavonoid as the major component in the aqueous extract. The extract also protects the cells from viral entry and inhibits virus activities after adsorption. In the treatment mode, hyperoside (
10) in the extract probably inhibited intracellular RNA synthesis by interacting with enzymes or proteins in the viral replication complex [
47]. Apparently pure hyperoside-even though quite readily available from many plants- has not been tested as an isolated compound.
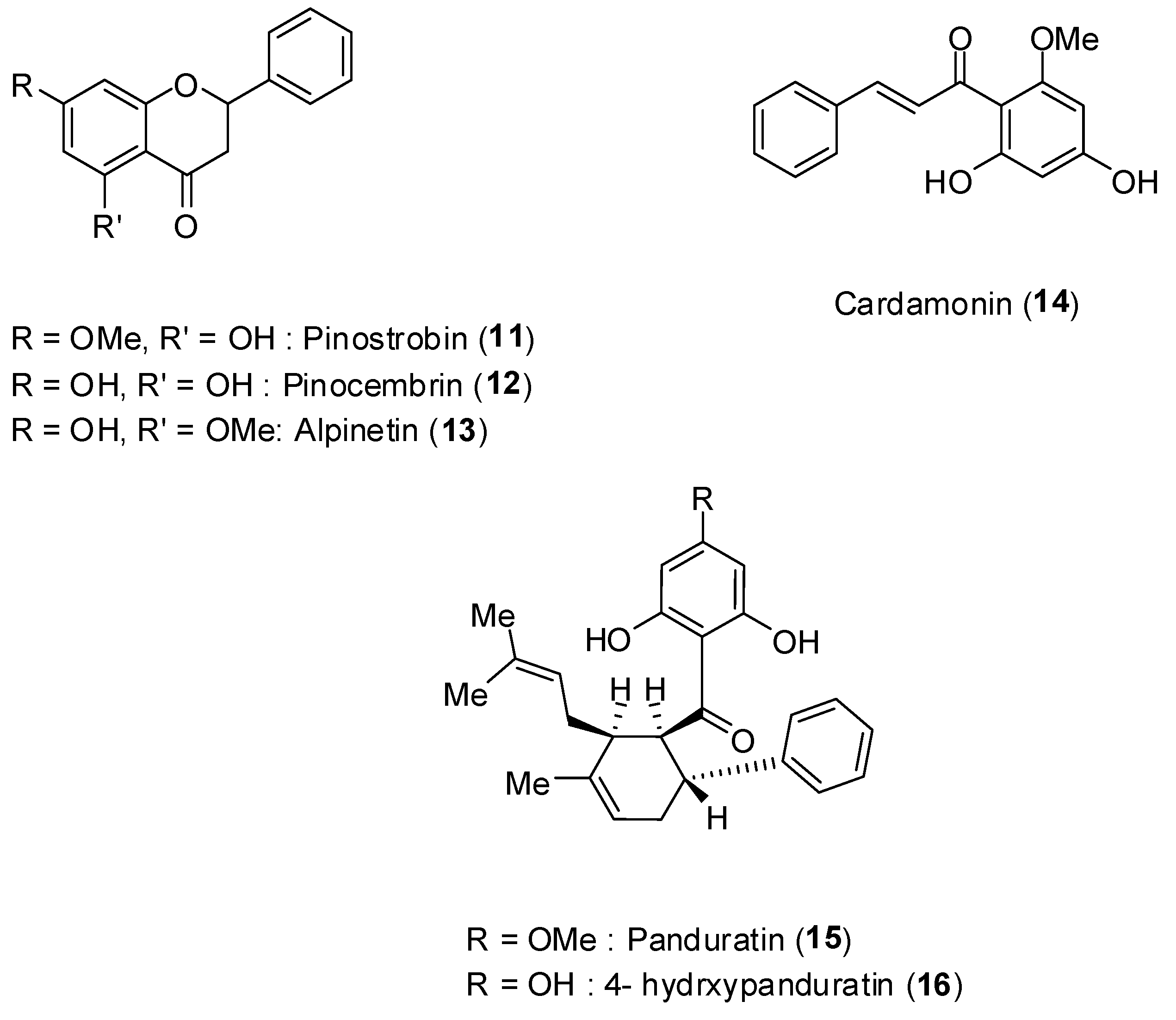
Boesenbergia rotunda (L.) is a common spice belonging to the ginger family (Zingiberaceae).
Figure 10 depicts the structures of the flavonoids pinostrobin (
11), pinocembrin (
12), alpinetin (
13), the phenylpropanoid cardamonin (
14), and the cyclohexenylchalcone derivatives pandurantin A (
15) and 4-hydroxypanduratin A (
16), all of them isolated from the aforementioned vegetal species [
48].
Figure 9.
Structure of hyperoside (10).
Figure 9.
Structure of hyperoside (10).
Figure 10.
Structures of compounds 11–16.
Figure 10.
Structures of compounds 11–16.
These natural products were screened against DENV-2 virus NS2B-NS3 protease by enzyme assay. The assay was performed with the purified protease [
48]. Increased concentration of the compounds led to increase inhibition of enzyme activity. Among the compounds, pinocembrin (
12) was the least active inhibiting by about 60% at 400 ppm concentration. The most active substances corresponded to panduratin A (
15) and 4-hydroxypanduratin B (
16), and their inhibitory protease activities are presented in
Table 3.
Table 3.
Percentage inhibition of DENV-2 NS2B-NS3 virus protease cleavage a of compounds and 15 and 16.
Table 3.
Percentage inhibition of DENV-2 NS2B-NS3 virus protease cleavage a of compounds and 15 and 16.
| Compound | Percentage Inhibition of DENV-2 NS2B-NS3 Protease; Concentration Used (ppm) |
|---|
| 40 | 80 | 120 | 160 | 240 | 400 |
|---|
| 15 | 27.1 ± 4.8 | 66.7 ± 0.1 | 87.7 ± 0.6 | 93.7 ± 0.5 | 92.12 ± 1.2 | 99.8 ± 1.1 |
| 16 | 52.0 ± 1.1 | 78.1 ± 0.1 | 87.6 ± 0.4 | 96.0 ± 0.5 | 97.3 ± 0.3 | 99.8 ± 0.3 |
Interestingly, although the compounds
12 and
14 had low inhibitory activity on NS2B-NS3 protease, some synergistic effect was noticed when these compounds were mixed (
Table 4).
Table 4.
Percentage inhibition of DENV-2 NS2B-NS3 virus protease cleavage a of compounds and 12 and 14.
Table 4.
Percentage inhibition of DENV-2 NS2B-NS3 virus protease cleavage a of compounds and 12 and 14.
| Compound | Percentage Inhibition of DENV-2 NS2B-NS3 Protease;
Concentration Used (ppm) |
|---|
| 120 | 240 | 400 |
|---|
| 12 | 30.1 ± 0.5 | 47.3 ± 0.5 | 56.1 ± 0.4 |
| 14 | 39.4 ± 0.6 | 50.1 ± 0.4 | 71.3 ± 0.3 |
| 12 + 14 | 52.6 ±0.4 | 63.5 ± 0.5 | 81.8 ± 0.3 |
For the most active compounds 15 and 16, it was determined that they present competitive inhibitory activity on NS2B-NS3 displaying inhibitory constants (Ki) of 21 and 25 μmol/L, respectively.
The flavonoids quercetin (
17), naringenin (
18) daidzein (
19), and hesperetin (
20) (
Figure 11) were evaluated against DENV-2 serotype by Zandi and co-workers. The assessment of antiviral activity was conducted
in vitro utilizing Vero cells. DENV replication was measured by Foci Forming Unit Reduction Assay (FFURA) and quantitative real time polymerase chain amplification (qRT-PCR). [
49]. Of these compounds, only quercetin (
17) presented significant inhibitory activity (IC
50 was 35.7 μg/mL) against DENV-2 infection in Vero cells. The selective index for quercetin when the infected cells were treated or when uninfected cells were treated continuously 5h before infection until 4 days post infection were 7.07 and 8.74, respectively. The mechanism by which quercetin exerts its antiviral effect remains unknown. However, it is believed that DENV antiviral activity of
17 could be similar to that presented by others flavonoids that act against cellular RNA polymerases and formation of the complex with RNA [
49].
Figure 11.
Structures of compounds 17–20.
Figure 11.
Structures of compounds 17–20.
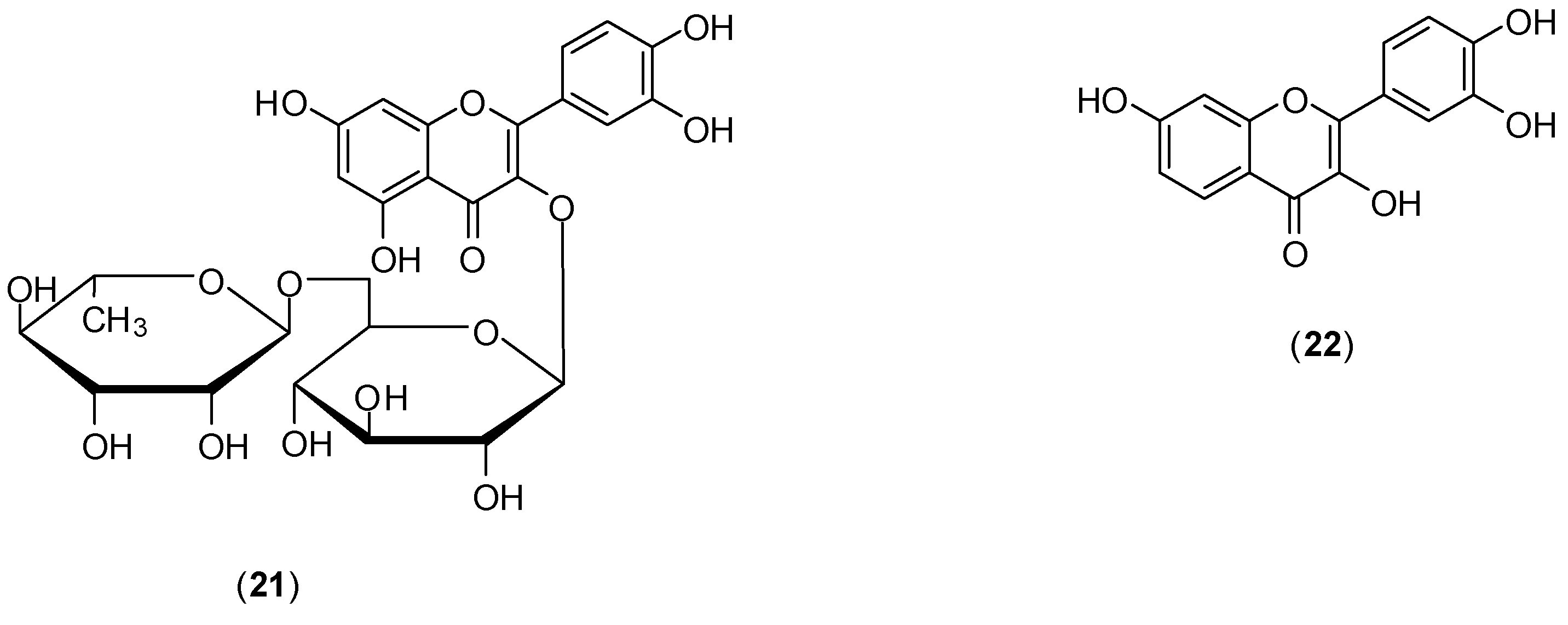
The study of Zandi and co-workers evaluated the effects of naringenin (
18) (
Figure 11), rutin (
21), and fisetin (
22) (
Figure 12) against DENV-2 (NGC strain). Antiviral effects of each compound at the different stages of DENV-2 infection were examined
in vitro using FFURA and qRT-PCR. The authors found that among the flavonoids studied only
22 presented significant
in vitro activity on dengue virus replication activity [
50]. Compound
22 did inhibit virus replication (IC
50 of 55 µg/mL and selectivity index of 4.49) after virus adsorption on Vero cells. When Vero cells were continuously treated for 5 h before virus infection and continuously up to 4 days post-infection, IC
50 corresponded to 43.12 µg/mL and selective index of 5.72. The authors report that no virucidal or prophylactic activity was noticed for fisetin (
22). Even though compounds
18 and
21 did not inhibit DENV-2 replication, flavonoid
18 displayed virucidal activity (IC
50 of 52.64 µg/mL), albeit with a low selectivity index (<1). The mechanism of how fisetin affects DENV virus replication is unclear. For the authors, it is not likely to act directly on the virus, because it does not affect DENV-2 binding to cells. They believe that fisetin (
22) could affect DENV genome copy number by interference in DENV-2 replication (by binding directly to virus RNA or forming a flavonoid-RNA complex) or by affecting the RNA polymerases resulting in inhibition of virus replication [
50].
Figure 12.
Structures of rutin (21) and fisetin (22).
Figure 12.
Structures of rutin (21) and fisetin (22).
A biological screening conducted on 1,350 ethyl acetate extracts prepared from various parts of approximately 650 New Caledonian plants was carried out targeting the DENV-2 NS5 polymerase. Enzyme assays were performed. DENV-2 NS5 polymerase genes were tagged by six N-terminal histidine residues and expressed from the pQE30 vector (Qiagen, Venlo, Limburg, Netherlands) in E. coli Rosetta pLacI cells (Novagen, Darmstadt, Germany). The enzymes were produced and purified by Heparin-Sepharose Chromatography. Alternatively, gel filtration was used as a second purification step.
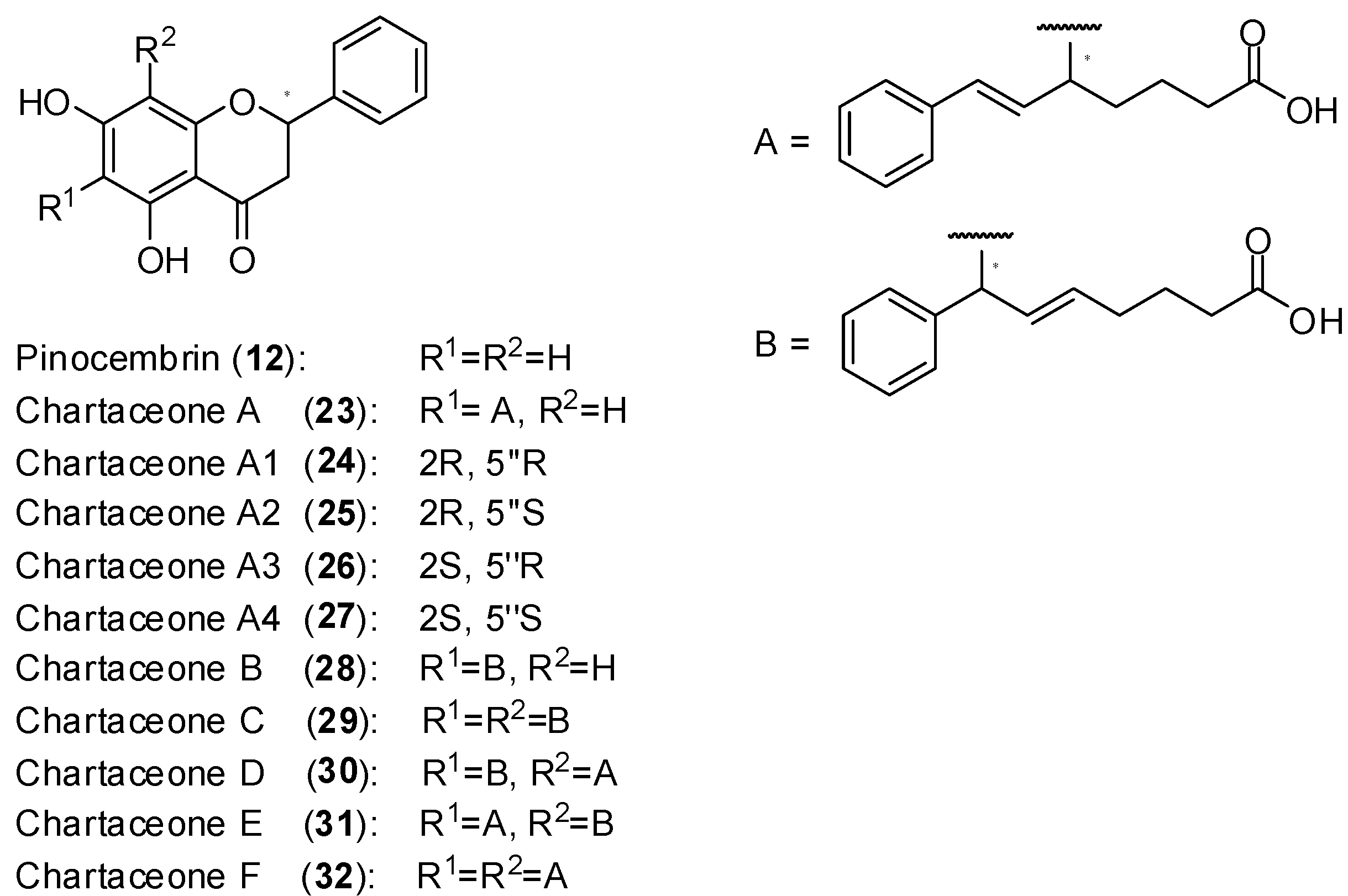
Polymerase activity was assayed by monitoring the incorporation of radiolabeled guanosine into a homopolymeric cytosine RNA template. Among several active extracts, the very active one from the bark of
Cryptocarya chartacea Kostern, a species belonging to the Lauraceae family [
51] was selected. From the selected extract, it was isolated the non-alkylated flavonoid pinocembrin (
12) as well as series of new mono and dialkylated ones named chartaceones A-F (
23–
32,
Figure 13). The screening of compounds
23–
32 against DENV-2 NS5 polymerase showed that the chartaceones C-F (compounds
29–
32) were the most active in inhibiting polymerase activity (IC
50 ranging from 1.8 to 4.2 µmol/L) while the other chartaceones were less effective. On the contrary, compound
12 was completely inactive. These findings suggest that the presence of alkylated chains in the structures of chartaceones C-F (
29–
32) play important role in terms of inhibitory activity on DENV-2 NS5 polymerase. The compounds
12,
23–
33 were also screened against bovine diarrhea virus (BVDV) NS5 polymerase and no activity was observed. Therefore, it seems that these natural substances present some selectivity towards DENV2-NS5 polymerase. Considering that the activity of compounds
29–
32 against DENV-2 NS5 polymerase was similar, it was concluded that side chains A and B (
Figure 13) play an equivalent role in terms of biological activity [
51].
Figure 14 shows the structures of the last two compounds discussed in this section.
Figure 13.
Structures of pinocembrin and chartaceones.
Figure 13.
Structures of pinocembrin and chartaceones.
Figure 14.
Structures of compounds 33–35.
Figure 14.
Structures of compounds 33–35.
Scutellaria baicalensis is a tradicional Chinese medicinal herb belonging to the Lamiaceae family. From the roots of this species, baicalein (
33) is usually extracted [
52]. The authors conducted
in vitro assay using Vero cells an FFURA to assess antiviral activity against DENV-2. This flavonoid inhibited DENV-2 serotype replication in Vero cells displaying an IC
50 of 6.46 µg/mL and a selectivity index of 17.8 when it was added after adsorption to the cells. The IC
50 against DENV-2 is 5.39 µg/mL and the selectivity index increased to 21.3 when Vero cells were treated before virus infection and continuously up to 4 days post-infection. Substance
33 displayed direct virucidal (IC
50 of 1.55 µg/mL) as well as anti-adsorption (IC
50 of 7.14 µg/mL) effects against DENV-2. The results suggest that a possible mechanism for the extracellular and intracellular activities of baicalein (33) against DENV-2 could be attributed to its ability to bind and/or to inactivate important structural and/or non-structural protein(s) of DENV-2 [
52].
A phytochemical investigation of the ethanol extracts from
Distictella elongate (Vahl) Urb led to isolation of petcolinarin (
34) from the leaf extract and a mixture of
34 and acacetin-7-
O-rutinoside (
35) from fruit extract.
In vitro MTT colorimetric assays, using Vero and LLCMK2 cells, were conducted to assess antiviral activity against DENV-2. The mixture of
34 and
35 presented better anti-DENV-2 activity (EC
50 of 11.1 ± 1.6 µg/mL and selectivity index > 45) than pure petcolinarin (
34) (EC
50 of 86.4 ± 3.8 µg/mL and selectivity index of 4.6). The mechanism of inhibition of the compounds is unclear, but it is suggested that it may correspond to one of the putative mechanisms already described for flavonoids [
53].
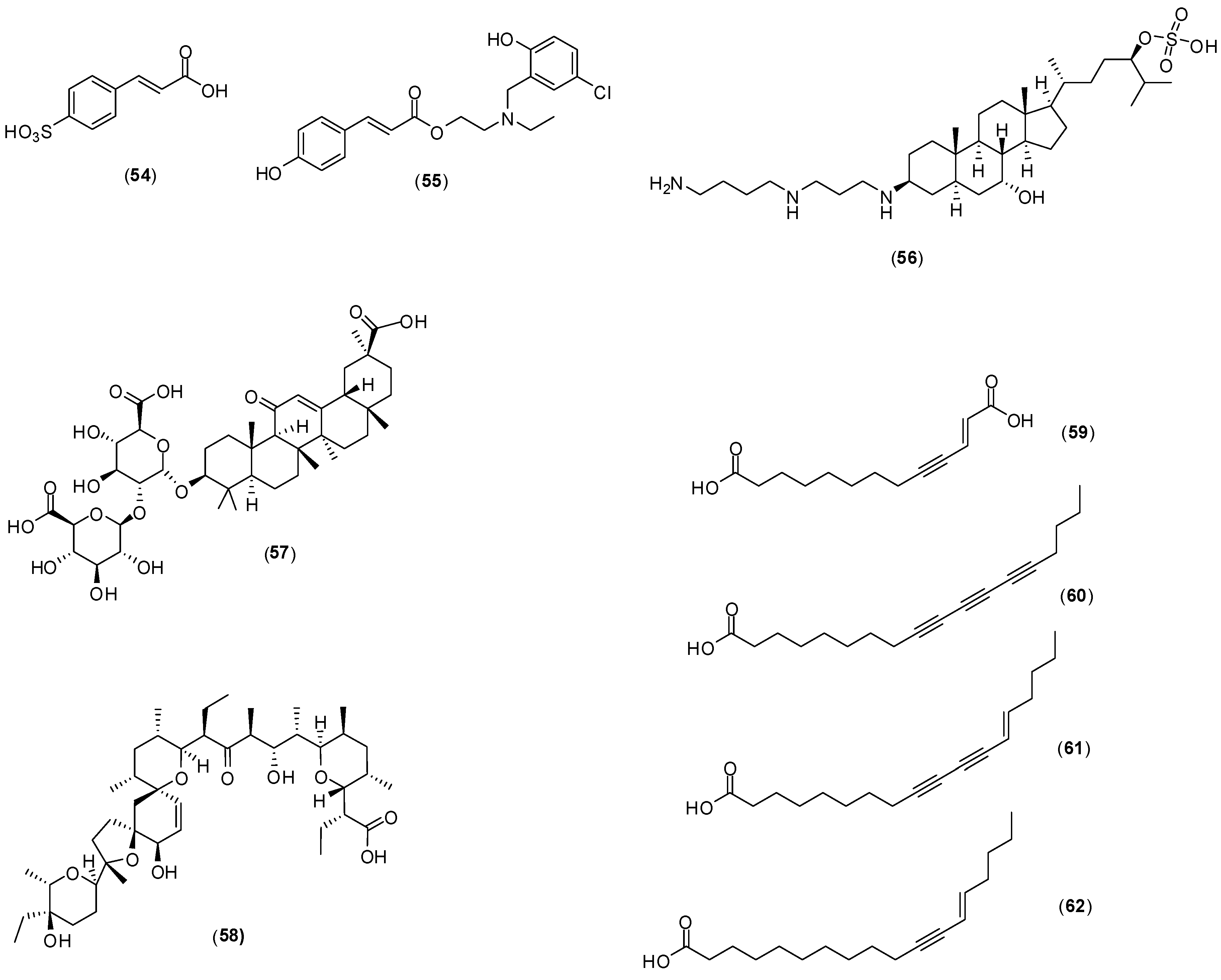
8. Miscellaneous
Several other compounds (
Figure 23) have been reported to possess antivirus activity. Zoasteric acid (
54) is obtained from the temperate marine eelgrass,
Zastera marina. Rees and collaborators conducted
in vitro experiments with LLCMK-2 cells by focus forming unit reduction assay and qRT-PCR to evaluate if compound
54 could inhibit all serotypes of dengue virus. Compound
54 presented a modest antiviral activity against serotype 2 with IC
50 of approximately 2.3 mmol/L .This investigation also identified the synthetic analogue of zoasteric acid (
55) as a more active compound concerning antidengue activity. It presented inhibitory effects against all DENV serotypes displaying IC
50 of 24, 46, 14 and 47 μmol/L against DENV-1, DENV-2, DENV-3 and DENV-4, respectively. The analogue
55 showed support of inhibition at an entry step in the viral life cycle and enhanced virus-cell binding as evidenced by a quantitative RT-PCR assay system. The idea that compound
55 interferes with entry by promoting inappropriate virus-cell contacts would lend support to the hypothesis that these compounds function through binding to attachment domains on adherent organisms and subsequent release from the protected surface [
65].
Squalamine (
56) was first discovered in the tissues of the dog fish shark (
Squalus acanthias) and later identified within the circulating white blood cells of the sea lamprey (
Petromyzon marinus). The
in vitro effect of squalamine (
56) on dengue virus infection of human endothelial cells (HMEC-1) was evaluated by the plaque assay. At the concentration of 40 µg/mL, dengue infection was inhibited by 60%. The infection was completely suppressed at 100 µg/mL. The proposed mechanism of action involves the capacity of squalamine (
56) to neutralize the negative electrostatic surface charge of intracellular membranes in a way that renders the cell less effective in supporting viral replication [
66].
Figure 23.
Structures of compounds 54–62.
Figure 23.
Structures of compounds 54–62.
Table 5.
Bioactivities determined for glycyrrhizin (58) against DENV-1-3.
Table 5.
Bioactivities determined for glycyrrhizin (58) against DENV-1-3.
| Virus Serotype | EC50 a (μg/mL) | EC90 b (μg/mL) | Selectivity Index c |
|---|
| Based on CC50 of Confluent Cells d | Based on CC50 for Growing Cells e |
|---|
| DENV-1 | 450.0 | 316 | >6 | 6 |
| DENV-2 | 174.2 | 317 | >6 | 5 |
| DENV-4 | 632.7 | 416 | >6 | 4 |
Crance and co-workers evaluated glycyrrhizin (
57), the major component responsible for the sweet-tasting constituent of
Glycyrrhiza glabra (liquorice) root, against eleven flaviviruses including DENV-1, DENV-2, and DENV-3 [
67]. The antiviral evaluation was performed
in vitro with Vero cells by plaque reduction assay. Several bioactivities were determined for this compound and they are summarized in
Table 5. This antiviral compound has already been used in patients in the treatment of other diseases. It should be further considered for use, either alone or in combination with another antiviral compounds tested in this work (interferon, ribavirin, 6-azauridine) for the treatment of flavivirus infections.
Narasin (
58) is a polyether antibiotic and antibacterial produced by fermentation of
Streptomyces aureofaciens. A dose-dependent study revealed that
58 has a 50% inhibitory concentration of less than 1 μmol/L against all four DENV serotypes [
68]. Minimal cytotoxicity was determined for this compound (50% cytotoxic concentration > 1,000 μmol/L).
In vitro assays with Huh-7 cells by plaque assay with different concentrations, qRT-PCR, western blotting and ultrastructural imaging were performed in this investigation. Narasin (
58) treatment of DENV-2 infected Huh-7 cells suggested that the compound is involved in inhibiting the post-entry stages of viral replication during DENV infection. The antiviral mechanism of narasin (
58) is likely to be associated with the disruption of viral protein synthesis. No differences of RNA levels were found between narasin (
58) treated and DENV-2 infected cells. The study indicated a characteristic disruption of viral protein synthesis by substance (
58) without affecting viral RNA replication. However, a more detailed investigation is required to understand the exact molecular mechanism of narasin (
58) in the inhibition of DENV protein synthesis and replication [
68].
The acetylenic compounds
59–
62 were isolated from an ethyl acetate extract of
Anacolosa pervilleana, a Madagascan plant. By using purified DENV RdRp polymerase in enzyme assay, it was found that acetylenic compounds
59–
62 give rise to IC
50 values around 3 μmol/L in the DENV RdRp assay. The results show that compounds possess some selectivity toward DENV RdRp. All compounds except
59 showed an overall antimetabolic effect in Vero cells (CC50s between 20 and 30 μmol/L). The presence of an additional acidic group in compound
59, probably prevents its penetration through the cell membrane, which may explain the absence of cytotoxicity [
69].