Calcium Influx Inhibition is Involved in the Hypotensive and Vasorelaxant Effects Induced by Yangambin
Abstract
:1. Introduction
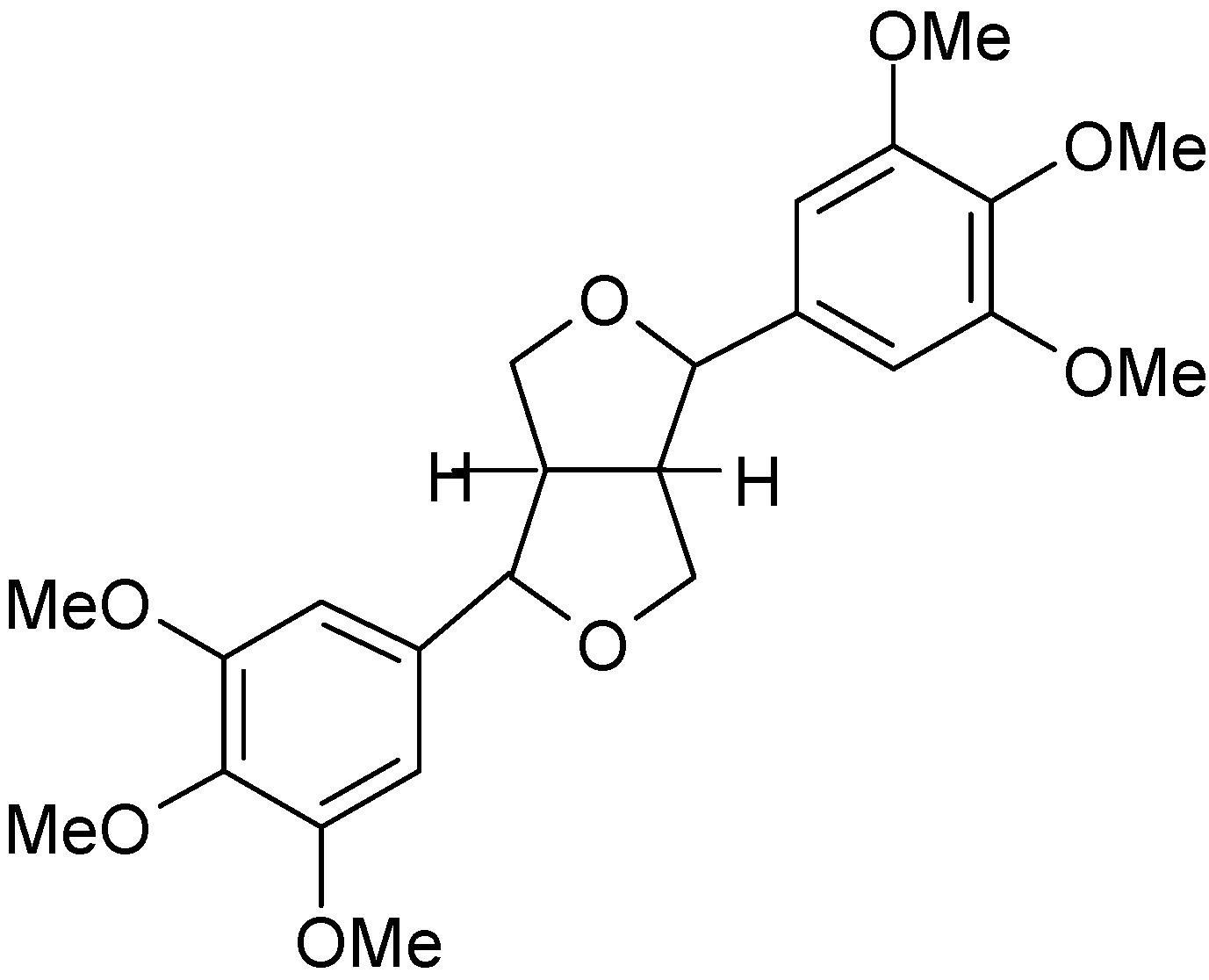
2. Results and Discussion
2.1. Effect of Yangambin on Blood Pressure

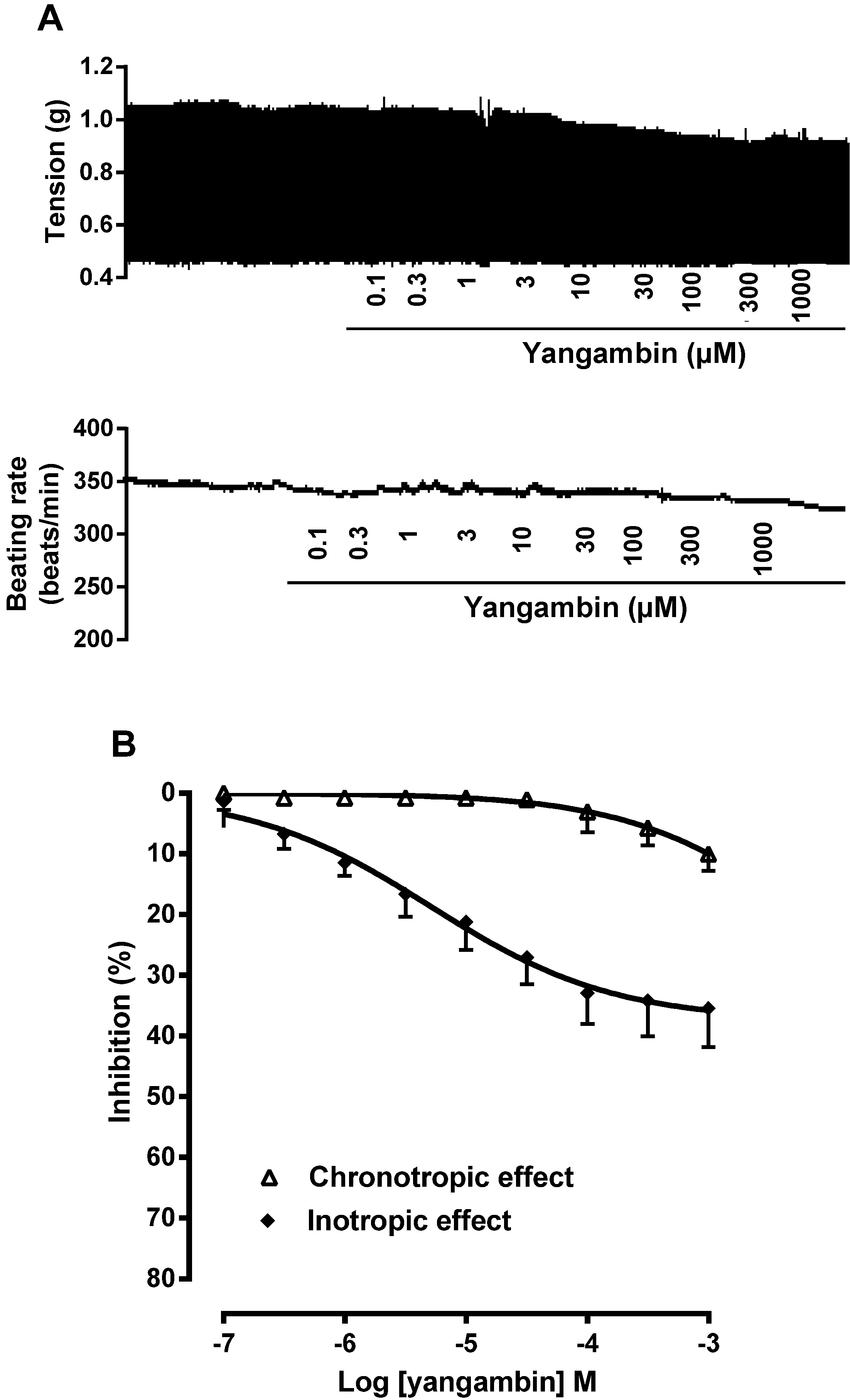
2.2. Effect of Yangambin on Isolated Rat Atria
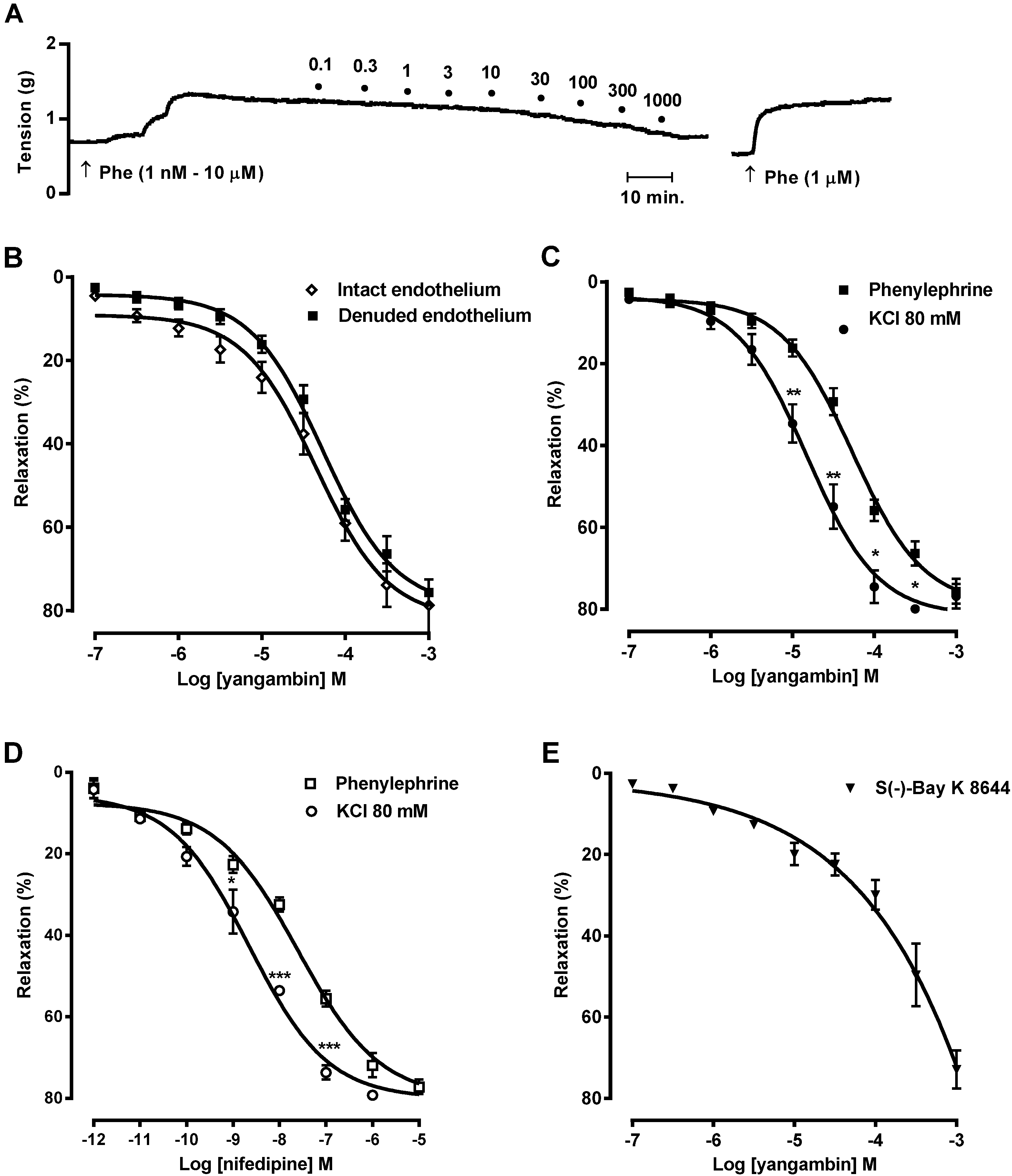
2.3. Effect of Yangambin on Rat Superior Mesenteric Arteries

2.4. Effect of Yangambin on KCl (60 mM)-Induced [Ca2+]i Increases in Fura-2/AM-Loaded Mesenteric Smooth Muscle Cells

3. Experimental
3.1. Animals
3.2. Yangambin
3.3. Direct Blood Pressure Measurements in Normotensive Non-Anaesthetized Rats
3.4. Preparation of Rat Atrial Muscle
3.5. Vascular Tension study in Rat Superior Mesenteric Artery Rings
3.6. Preparation of Mesenteric Smooth Muscle Cells
3.7. Loading of Vascular Smooth Muscle Cells with Fura-2/AM
3.8. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ghisalberti, E.L. Cardiovascular activity of naturally occurring lignans. Phytomedicine 1997, 4, 151–166. [Google Scholar] [CrossRef]
- Tibirica, E. Cardiovascular properties of yangambin, a lignan isolated from Brazilian plants. Cardiovasc. Drug 2001, 19, 313–328. [Google Scholar]
- Park, J.Y.; Choi, Y.W.; Yun, J.W.; Bae, J.U.; Seo, K.W.; Lee, S.J.; Kim, C.D. Gomisin J from Schisandra. chinensis induces vascular relaxation via activation of endothelial nitric oxide synthase. Vascul. Pharmacol. 2012, 57, 124–130. [Google Scholar] [CrossRef]
- Mollataghi, A.; Hadi, A.H.; Awang, K.; Mohamad, J.; Litaudon, M.; Mukhtar, M.R. (+)-Kunstlerone, a new antioxidant neolignan from the Leaves of Beilschmiedia. kunstleri gamble. Molecules 2011, 16, 6582–6590. [Google Scholar] [CrossRef]
- Mesquita, L.M.; Roque, N.F.; Quintana, L.M.B.; Paulo, M.Q.; Barbosa-Filho, J.M. Lignans from Rollinia. species. Biochem. Syst. Ecol. 1988, 16, 379–380. [Google Scholar] [CrossRef]
- Figueiredo, S.F.L.; Viana, V.R.C.; Simões, C.; Albarello, N.; Trugo, L.C.; Kaplan, M.A.C.; Krul, W.R. Lignans from leaves, seed lings and micropropagated plants of Rollinia. mucosa (Jacq.) Baill. – Annonaceae. Plant Cell Tissue Organ Cult. 1999, 56, 121–124. [Google Scholar]
- Morais, L.C.S.L.; Almeida, R.N.; Cunha, E.V.L.; Silva, M.S.; Barbosa-Filho, J.M.; Gray, A.I. Further Lignans from Ocotea Duckei. Pharm. Biol. 1999, 37, 144–147. [Google Scholar]
- Castro-Faria-Neto, H.C.; Araujo, C.V.; Moreira, S.; Bozza, P.T.; Thomas, G.; Barbosa-Filho, J.M.; Cordeiro, R.S.; Tibirica, E.V. Yangambin: A new naturally-occurring platelet-activating factor receptor antagonist: In vivo pharmacological studies. Planta Med. 1995, 61, 106–112. [Google Scholar]
- Castro-Faria-Neto, H.C.; Bozza, P.T.; Cruz, H.N.; Silva, C.L.; Violante, F.A.; Barbosa-Filho, J.M.; Thomas, G.; Martins, M.A.; Tibirica, E.V.; Noel, F.; et al. Yangambin: A new naturally-occurring platelet-activating factor receptor antagonist: Binding and in vitro functional studies. Planta Med. 1995, 61, 101–105. [Google Scholar] [CrossRef]
- Tibiriçá, E.V.; Mosquera, K.; Abreu, M.; Ribeiro, R.; Carvalho, F.A.; Barbosa-Filho, J.M.; Cordeiro, R.S. Antagonistic effect of yangambin on platelet-activating factor (PAF)-induced cardiovascular collapse. Phytomedicine 1996, 2, 235–242. [Google Scholar] [CrossRef]
- Ribeiro, R.; Carvalho, F.A.S.; Barbosa-Filho, J.M.; Cordeiro, R.S.B.; Tibiriçá, E.V. Protective effects of yangambin, a natural platelet-activating factor (PAF) receptor antagonist - on the anaphylactic shock in rats. Phytomedicine 1996, 3, 249–256. [Google Scholar] [CrossRef]
- Serra, M.F.; Diaz, B.L.; Barreto, E.O.; Pereira, A.P.; Lima, M.C.; Barbosa-Filho, J.M.; Cordeiro, R.S.; Martins, M.A.; de Silva, P.M. Anti-allergic properties of the natural PAF antagonist yangambin. Planta Med. 1997, 63, 207–212. [Google Scholar] [CrossRef]
- Sousa, F.C.; Pereira, B.A.; Lima, V.T.; Lacerda, C.D.; Melo, C.T.; Barbosa-Filho, J.M.; Vasconcelos, S.M.; Viana, G.S. Central nervous system activity of yangambin from Ocotea duckei Vattimo (Lauraceae) in mice. Phytother. Res. 2005, 19, 282–286. [Google Scholar] [CrossRef]
- Monte, R.L.; Barbosa, J.M.; Sousa, L.M.; Athayde, P.F.; Dias, C.S.; Oliveira, M.R. Crude ethanolic extract, lignoid fraction and yangambin from Ocotea. duckei (Lauraceae) show antileishmanial activity. Z. Naturforsch C 2007, 62, 348–352. [Google Scholar]
- Lami, N.; Kadota, S.; Kikuchi, T.; Momose, Y. Constituents of the roots of Boerhaavia. diffusa L. III. Identification of Ca2+ channel antagonistic compound from the methanol extract. Chem. Pharm. Bull (Tokyo) 1991, 39, 1551–1555. [Google Scholar] [CrossRef]
- Abe, M.; Morikawa, M.; Inoue, M.; Tsuboi, M. Inhibitory effect of 2,3-dibenzylbutane-1,4-diol; a mammalian lignan, on the Ca2+ channel in rabbit femoral artery. Res. Commun. Chem. Pathol. Pharmacol. 1990, 69, 271–284. [Google Scholar]
- Yang, J.M.; Ip, P.S.; Che, C.T.; Yeung, J.H. Relaxant effects of Schisandra chinensis and its major lignans on agonists-induced contraction in guinea pig ileum. Phytomedicine 2011, 18, 1153–1160. [Google Scholar] [CrossRef]
- Lee, H.B.; Blaufox, M.D. Blood volume in the rat. J. Nucl. Med. 1985, 26, 72–76. [Google Scholar]
- Queiroz, T.M.; Guimarães, D.D.; Mendes-Junior, L.G.; Braga, V.A. α-lipoic acid reduces hypertension and increases baroreflex sensitivity in renovascular hypertensive rats. Molecules 2012, 17, 13357–13367. [Google Scholar] [CrossRef]
- Nasa, Y.; Ichihara, K.; Yoschida, R.; Abiko, Y. Positive inotropic and negative chronotropic effects of (−)-cis-diltiazem in rat isolated atria. Br. J. Pharmacol. 1992, 105, 696–702. [Google Scholar] [CrossRef]
- Davie, C.S.; Kubo, M.; Standen, N.B. Potassium channel activation and relaxation by nicorandil in rat small mesenteric arteries. Br. J. Pharmacol. 1998, 125, 1715–1725. [Google Scholar] [CrossRef]
- Schramm, M.; Thomas, G.; Towart, R.; Franckowiak, G. Novel dihydropyridines with positive inotropic action through activation of Ca2+ channels. Nature 1983, 303, 535–537. [Google Scholar]
- Palmberg, L.; Thyberg, J. Uterine smooth muscle cells in primary culture. Alterations in fine structure, cytoskeletal organization and growth characteristics. Cell. Tissue Res. 1986, 246, 253–262. [Google Scholar] [CrossRef]
- Sample Availability:Samples of the yangambin are not available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Araújo, I.G.A.; Silva, D.F.; Do Carmo de Alustau, M.; Dias, K.L.G.; Cavalcante, K.V.M.; Veras, R.C.; Barbosa-Filho, J.M.; Neto, M.D.A.; Bendhack, L.M.; De Azevedo Correia, N.; et al. Calcium Influx Inhibition is Involved in the Hypotensive and Vasorelaxant Effects Induced by Yangambin. Molecules 2014, 19, 6863-6876. https://doi.org/10.3390/molecules19056863
Araújo IGA, Silva DF, Do Carmo de Alustau M, Dias KLG, Cavalcante KVM, Veras RC, Barbosa-Filho JM, Neto MDA, Bendhack LM, De Azevedo Correia N, et al. Calcium Influx Inhibition is Involved in the Hypotensive and Vasorelaxant Effects Induced by Yangambin. Molecules. 2014; 19(5):6863-6876. https://doi.org/10.3390/molecules19056863
Chicago/Turabian StyleAraújo, Islania Giselia Albuquerque, Darizy Flávia Silva, Maria Do Carmo de Alustau, Katy Lísias Gondim Dias, Karla Veruska Marques Cavalcante, Robson Cavalcante Veras, José Maria Barbosa-Filho, Mario Dos Anjos Neto, Lusiane Maria Bendhack, Nadja De Azevedo Correia, and et al. 2014. "Calcium Influx Inhibition is Involved in the Hypotensive and Vasorelaxant Effects Induced by Yangambin" Molecules 19, no. 5: 6863-6876. https://doi.org/10.3390/molecules19056863
APA StyleAraújo, I. G. A., Silva, D. F., Do Carmo de Alustau, M., Dias, K. L. G., Cavalcante, K. V. M., Veras, R. C., Barbosa-Filho, J. M., Neto, M. D. A., Bendhack, L. M., De Azevedo Correia, N., & Almeida de Medeiros, I. (2014). Calcium Influx Inhibition is Involved in the Hypotensive and Vasorelaxant Effects Induced by Yangambin. Molecules, 19(5), 6863-6876. https://doi.org/10.3390/molecules19056863




