Seasonal Variation and Resin Composition in the Andean Tree Austrocedrus chilensis
Abstract
:1. Introduction
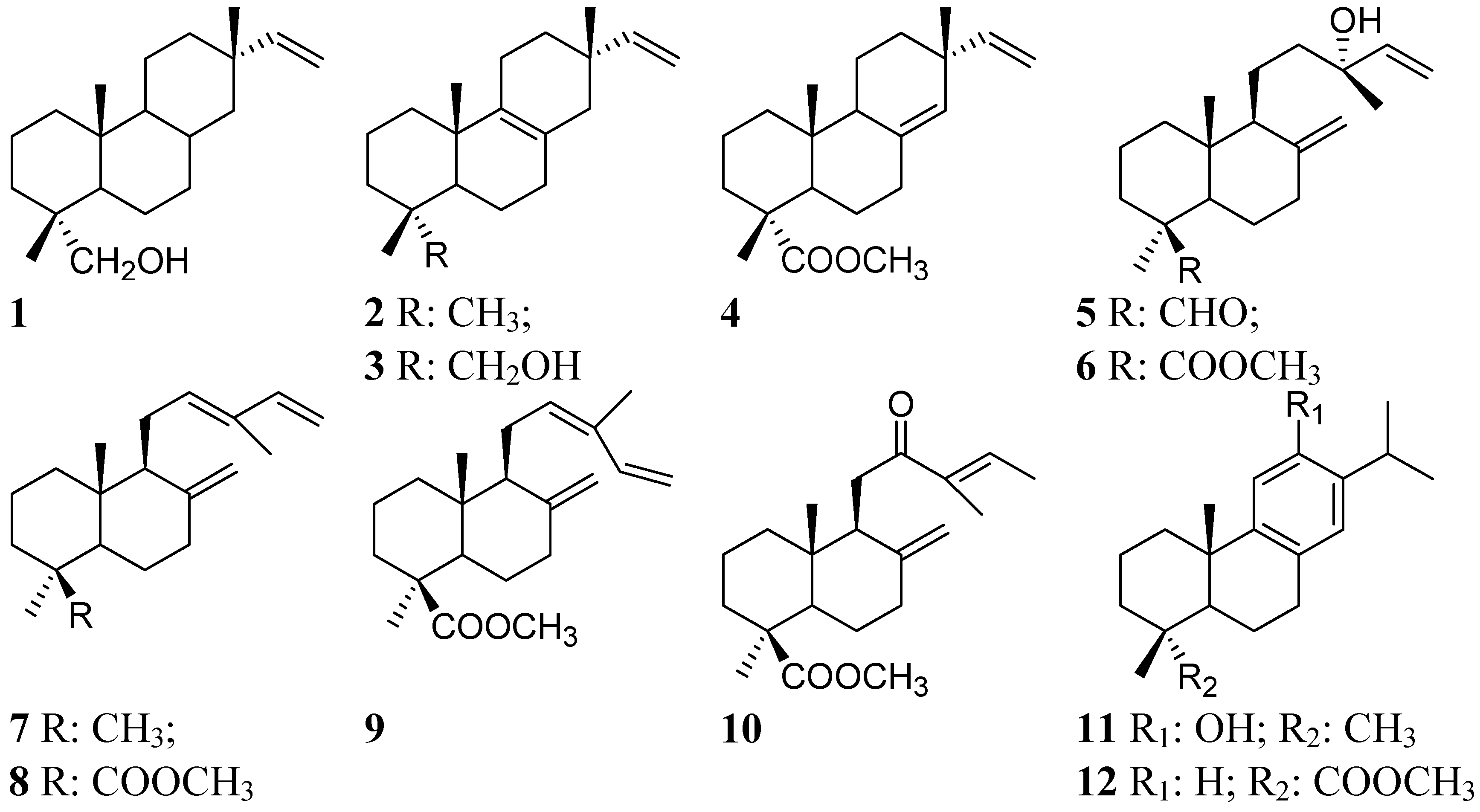
2. Results and Discussion
| Tree | Sex | Sample number | ||
|---|---|---|---|---|
| February 2011 | June 2011 | November 2010 | ||
| 1 | Female | 101, 102, 103, 104 | 201, 202, 203 | 01, 02, 03, 04 |
| 2 | Male | 105, 106 | 204, 205 | NC |
| 3 | Female | 109, 110 | 209 | 05, 06, 07 |
| 4 | Female | 111, 112 | 210 | 08, 09, 10 |
| 5 | Male | 113, 114 | 211, 212 | 11, 12 |
| 6 | Male | 115 | 213, 214 | 13, 14, 15 |
| 7 | Male | 107, 108 | 206, 207, 208 | NC |
| Compound number | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | |
| Sex | ||||||||||||
| Female | ||||||||||||
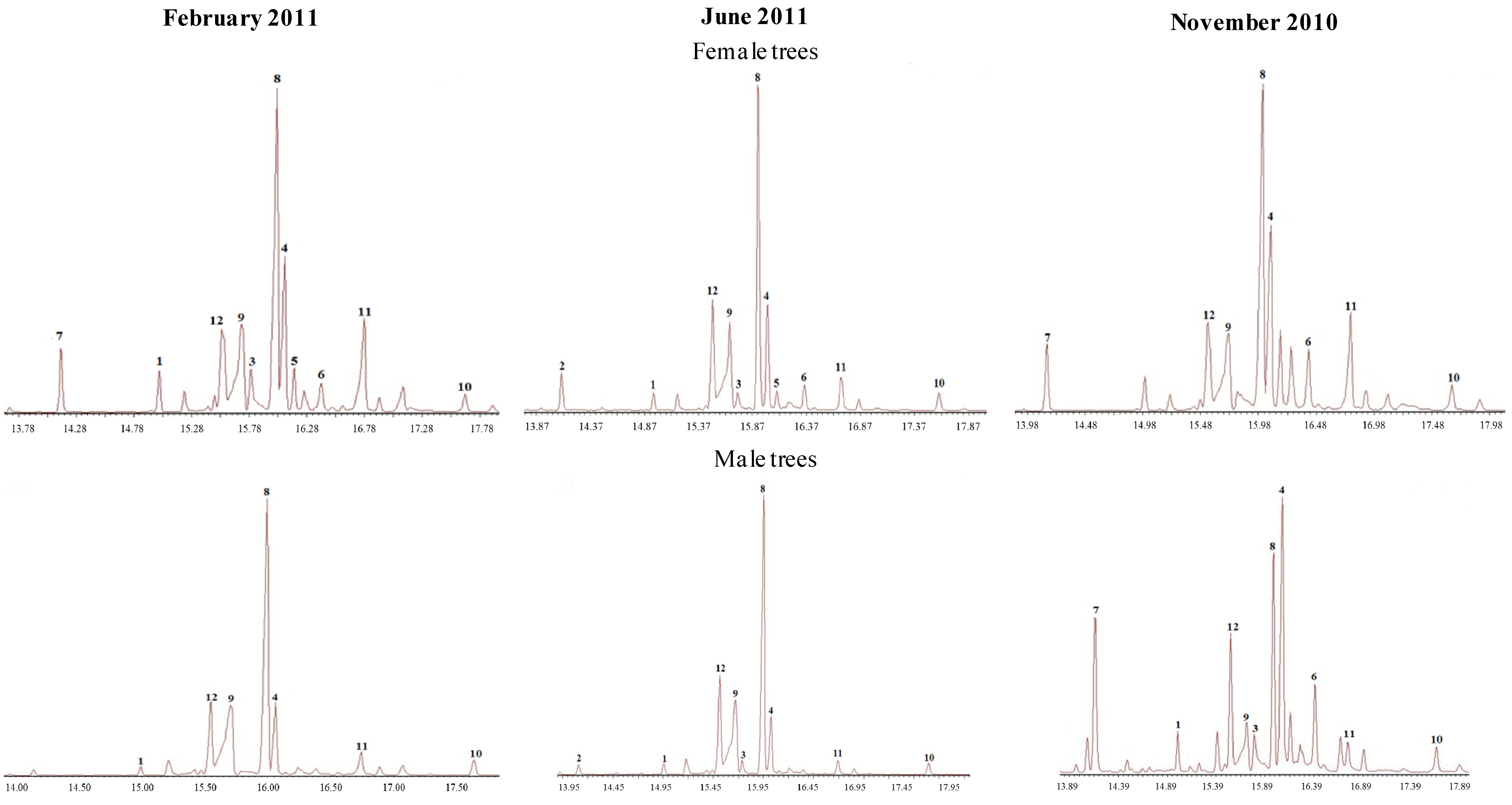
| 101 | 2.8 | Nd | 4.1 | 9.8 | 3.3 | 6.2 | 7.3 | 27.8 | 9.9 | Nd | 9.5 | 9.2 |
| 102 | 3.3 | Nd | 3.3 | 10.8 | 2.5 | 2.7 | 4.9 | 35.7 | 11.6 | 1.9 | 10.8 | 7.8 |
| 103 | 1.9 | Nd | Nd | 10.9 | Nd | Nd | Nd | 42.3 | 20.0 | Nd | 10.0 | 12.1 |
| 104 | 2.7 | Nd | 1.7 | 13.5 | 1.2 | 2.5 | 3.3 | 39.6 | 12.4 | 1.9 | 10.0 | 7.5 |
| 109 | 6.3 | Nd | 7.8 | 8.4 | 4.5 | Nd | 29.5 | 17.6 | 7.2 | Nd | 3.4 | 8.8 |
| 110 | 3.9 | Nd | 2.2 | 14.0 | Nd | 1.1 | 1.4 | 32.5 | 17.2 | 2.4 | 3.9 | 12.5 |
| 111 | 5.9 | Nd | 3.7 | 8.5 | 5.0 | 5.5 | 13.3 | 23.8 | 9.1 | 1.2 | 3.7 | 10.5 |
| 112 | 4.7 | Nd | 5.5 | 9.1 | 3.8 | 5.0 | 14.1 | 22.3 | 10.4 | 1.0 | 3.1 | 9.3 |
| Male | ||||||||||||
| 105 | 1.2 | Nd | Nd | 7.8 | Nd | Nd | Nd | 45.0 | 23.4 | 2.5 | 3.9 | 11.9 |
| 106 | Nd | Nd | Nd | 6.6 | Nd | Nd | Nd | 44.5 | 26.2 | 3.6 | 4.3 | 12.7 |
| 107 | 4.2 | Nd | Nd | 10.4 | 1.9 | 3.3 | 21.5 | 24.5 | 12.4 | 1.8 | 5.2 | 9.3 |
| 108 | 3.8 | Nd | Nd | 10.4 | Nd | 4.4 | 17.2 | 23.6 | 11.9 | 1.9 | 4.8 | 8.9 |
| 113 | 6.2 | Nd | 6.5 | 11.5 | Nd | Nd | Nd | 31.5 | 12.5 | Nd | 5.8 | 9.5 |
| 114 | 8.5 | Nd | 7.9 | 10.6 | Nd | Nd | Nd | 28.9 | 8.9 | Nd | 5.9 | 10.9 |
| 115 | 0.9 | Nd | Nd | 9.6 | Nd | Nd | Nd | 33.1 | 30.2 | 2.9 | 4.5 | 14.2 |
| Sex | Compound number | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | |
| Female | ||||||||||||
| 201 | 2.4 | 5.9 | 2.1 | 12.3 | 1.8 | 3.1 | Nd | 30.2 | 14.9 | 2.9 | 4.5 | 15.9 |
| 202 | 2.5 | 8.5 | 2.6 | 13.9 | 1.5 | 4.3 | Nd | 24.4 | 14.8 | 2.4 | 5.6 | 16.7 |
| 203 | 1.7 | 4.2 | 1.8 | 11.0 | 1.6 | 2.4 | Nd | 34.1 | 18.6 | 2.4 | 4.4 | 14.9 |
| 209 | 3.8 | 27.8 | 7.2 | 7.3 | 2.7 | 2.1 | Nd | 16.9 | 9.1 | 0.3 | 2.7 | 7.8 |
| 210 | 2.7 | 3.3 | 1.4 | 12.6 | 1.3 | 2.9 | Nd | 32.3 | 15.3 | 2.6 | 3.4 | 14.9 |
| Male | ||||||||||||
| 204 | Nd | 1.7 | Nd | 12.1 | Nd | Nd | Nd | 42.8 | 23.5 | 1.9 | 1.4 | 14.2 |
| 205 | 1.6 | 1.5 | 0.7 | 9.4 | Nd | Nd | Nd | 48.4 | 17.4 | 2.0 | 2.5 | 12.9 |
| 206 | 2.7 | 27.3 | 2.7 | 8.6 | 0.9 | Nd | Nd | 21.2 | 11.6 | 1.9 | 2.9 | 9.0 |
| 207 | 3.2 | 31.8 | 3.3 | 6.4 | 2.9 | Nd | Nd | 17.9 | 11.0 | 0.0 | 2.9 | 8.7 |
| 208 | 2.4 | 23.7 | 3.2 | 8.4 | 1.5 | 1.7 | Nd | 21.5 | 11.1 | 0.3 | 3.9 | 8.3 |
| 211 | 5.8 | 1.0 | 8.3 | 15.2 | Nd | Nd | Nd | 24.6 | 9.5 | 1.0 | 4.3 | 17.3 |
| 212 | Nd | Nd | 12.6 | 10.3 | Nd | Nd | Nd | 25.8 | 9.2 | 1.1 | 4.1 | 13.8 |
| 213 | 2.6 | 4.8 | 2.0 | 13.7 | 1.5 | 1.7 | Nd | 39.9 | 14.4 | 0.6 | 2.7 | 10.8 |
| 214 | 2.6 | 3.1 | 3.2 | 12.9 | 1.5 | 2.4 | Nd | 36.8 | 15.1 | 2.8 | 3.1 | 11.2 |
| Sex | Compound number | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | |
| Female | ||||||||||||
| 01 | Nd | Nd | Nd | 4.0 | Nd | 1.0 | 0.1 | 85.0 | 3.2 | 0.7 | 2.7 | 3.0 |
| 02 | 2.4 | Nd | Nd | 22.5 | Nd | Nd | Nd | 35.5 | 11.4 | 1.9 | 17.3 | Nd |
| 03 | Nd | 4.2 | Nd | 17.6 | Nd | 4.7 | Nd | 28.3 | 11.9 | 3.0 | 4.9 | 12.0 |
| 04 | Nd | 4.1 | Nd | 8.8 | Nd | Nd | Nd | 51.7 | 12.1 | 1.1 | 9.7 | 11.6 |
| 05 | 1.7 | Nd | Nd | 20.9 | Nd | Nd | Nd | 36.4 | 16.5 | 1.9 | 8.0 | 10.3 |
| 06 | 3.5 | Nd | Nd | 19.4 | Nd | Nd | Nd | 38.7 | 18.6 | 1.8 | 5.1 | 11.2 |
| 07 | Nd | Nd | Nd | 25.9 | Nd | Nd | Nd | 31.6 | 23.4 | 2.1 | 3.3 | 13.7 |
| 08 | 4.9 | Nd | Nd | 10.3 | 3.2 | 4.5 | 19.8 | 20.7 | 11.0 | Nd | 2.3 | 11.4 |
| 09 | 5.3 | Nd | 7.7 | 12.8 | 4.8 | 11.4 | 21.2 | 13.6 | 4.9 | 2.1 | 3.0 | 13.7 |
| 10 | Nd | 7.5 | 5.5 | 34.3 | Nd | 7.8 | Nd | 6.8 | 0.1 | 9.1 | 7.2 | 17.7 |
| Male | ||||||||||||
| 11 | 2.8 | Nd | 5.6 | 16.9 | Nd | 3.2 | Nd | 31.5 | 8.4 | Nd | 2.0 | 14.4 |
| 12 | 8.1 | Nd | 15.4 | 9.4 | Nd | Nd | Nd | 12.7 | 6.0 | Nd | 20.4 | 5.9 |
| 13 | 3.5 | Nd | 3.1 | 27.3 | Nd | 7.9 | 14.1 | 17.9 | 3.9 | 2.8 | 3.0 | 10.8 |
| 14 | 4.7 | Nd | 3.8 | 17.1 | 4.0 | 3.4 | 11.0 | 24.7 | 6.7 | 2.0 | 8.2 | 7.8 |
| 15 | 5.2 | Nd | Nd | 16.2 | 4.2 | 3.1 | 14.4 | 31.0 | 10.1 | 1.2 | 3.9 | 10.8 |
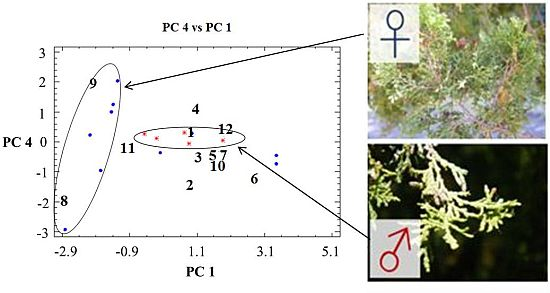
2.1. Percent Diterpene Composition According to Collection Time (Month) and Tree Sex
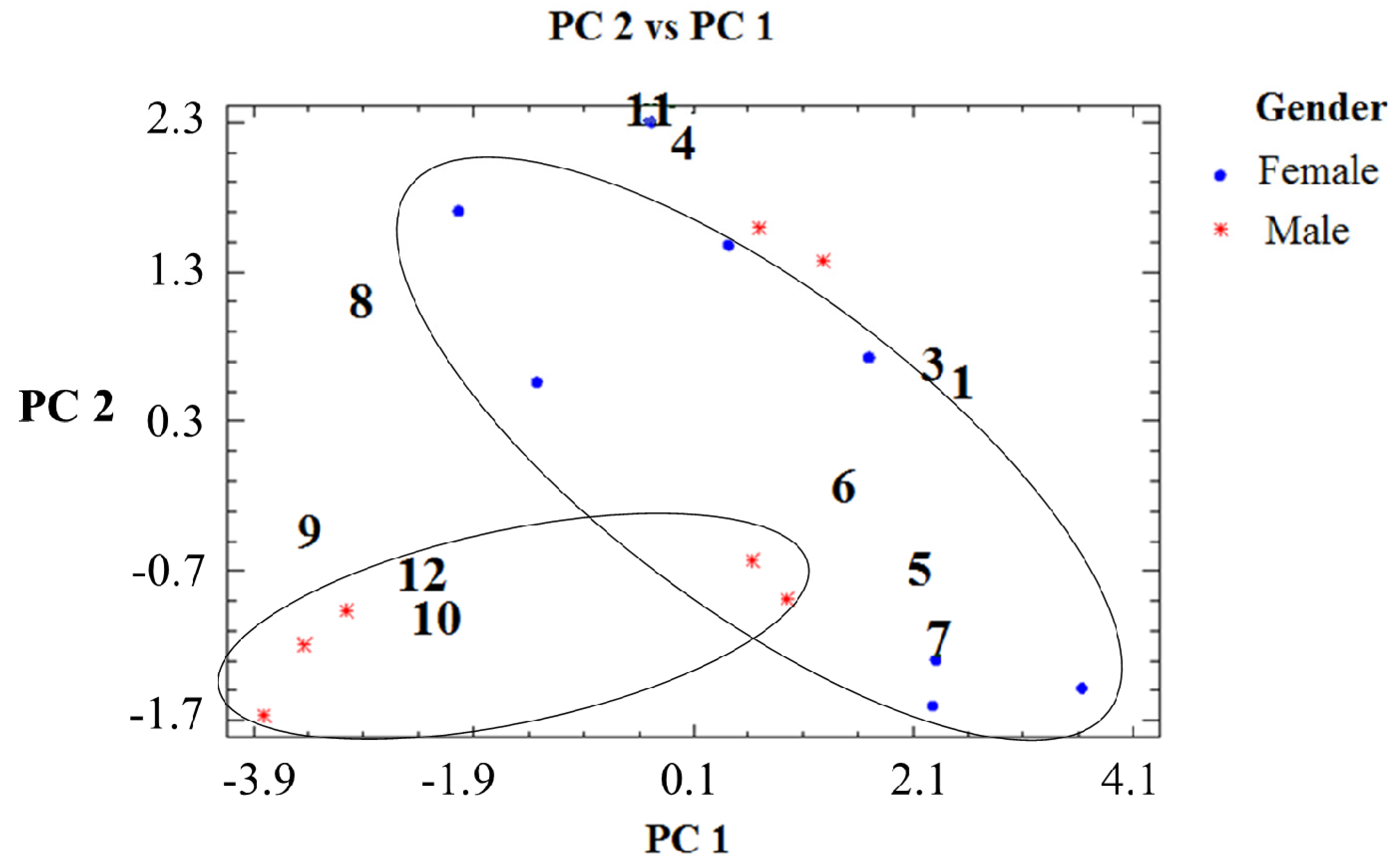
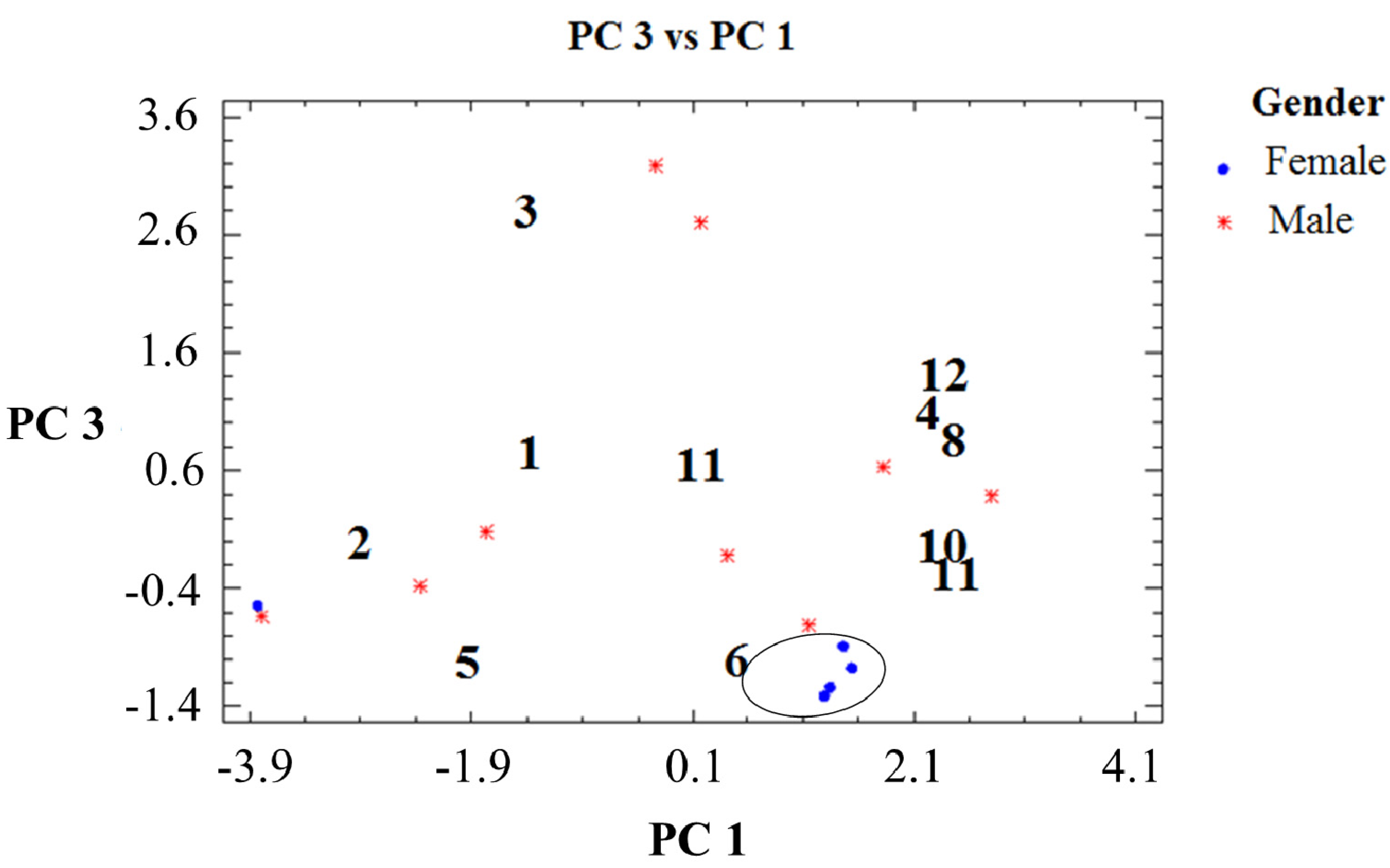

2.2. Percent Diterpene Composition and Collection Time (Month/Season)
3. Experimental
3.1. Plant Material
3.2. GC-MS and NMR Analysis
3.3. Description of the Parameters System
3.4. Principal Component Analysis (PCA)
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dodd, R.S.; Rafii, Z.A.; Power, A.B. Ecotypic adaptation in Austrocedrus chilensis in cuticular hydrocarbon composition. New Phytol. 1998, 138, 699–708. [Google Scholar]
- Pastorino, M.J.; Gallo, L.A. Quaternary evolutionary history of Austrocedrus chilensis, a cypress native to the Andean-Patagonian forest. J. Biogeogr. 2002, 29, 1167–1178. [Google Scholar] [CrossRef]
- Arana, M.V.; Gallo, L.A.; Vendramin, G.G.; Pastorino, M.J.; Sebastiani, F.; Marchelli, P. High genetic variation in marginal fragmented populations at extreme climatic conditions of the Patagonian cypress Austrocedrus chilensis. Mol. Phylogenet. Evol. 2010, 54, 941–949. [Google Scholar] [CrossRef]
- CONAF. Catastro de Los Recursos Vegetacionales Nativos de Chile. Monitoreo de Cambios y Actualizaciones. Período 1997-2011. 2011. Available online: http://sit.conaf.cl/ (accessed on 19 May 2014).
- Castro, M.A.; Gordaliza, M.; del Corral, J.M.M.; San Feliciano, A. The distribution of lignanoids in the order Coniferae. Phytochemistry 1996, 41, 995–1011. [Google Scholar] [CrossRef]
- Guerra, P.E.; González, S.B.; Elabum, J.H. Caraacterísticas botánicas y determinación de aceites esenciales en Cupresáceas nativas y exóticas cultivadas en el oeste del Chubut; Publicación Técnica no 32; Centro de Investigación y Extensión Forestal Andino Patagónico-CIEFAP; Universidad Nacional de la Patagonia San Juan Bosco: Tierra del Fuego, Argentina, 2006; pp. 37–41. [Google Scholar]
- Olate, V.R.; Goikoetxeaundia, O.; Schmeda-Hirschmann, G. Resin diterpenes from Austrocedrus chilensis. Molecules 2011, 16, 10653–10667. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chen, Y.-J.; Cheng, S.-S.; Chang, S.-T. Rapid differentiation of three Chamaecyparis species (Cupressaceae) grown in Taiwan using solid-phase microextraction-gas chromatography/mass spectrometry, cluster analysis, and principal component analysis. J. Agric. Food Chem. 2011, 59, 10854–10859. [Google Scholar] [CrossRef]
- Flanigan, P.M.; Radell, L.L.; Brady, J.J.; Levi, R.J. Differentiation of eight phenotypes and discovery of potential biomarkers for a single plant organ class using laser electrospray mass spectrometry and multivariate statistical analysis. Anal. Chem. 2012, 84, 6225–6232. [Google Scholar]
- Brus, R.; Ballian, D.; Zhelev, P.; Pandza, M.; Bobinac, M.; Acevski, J.; Raftoyannis, Y.; Jarrín, K. Absence of geographical structure of morphological variation in Juniperus oxycedrus L. subsp. oxycedrus in the Balkan Peninsula. Eur. J. For. Res. 2011, 130, 657–670. [Google Scholar] [CrossRef]
- Suarez, E.; Nguyen, H.P.; Ortiz, I.P.; Lee, K.J.; Kim, S.B.; Krzywinski, J.; Schug, K.A. MALDI-MS Cuticular lipid profiles can differentiate sex, age, and mating status of Anopheles gambiae mosquitoes. Anal. Chim. Acta 2011, 706, 157–163. [Google Scholar] [CrossRef]
- Pi, Z.; Yue, H.; Ma, L.; Ding, L.; Liu, Z.; Liu, S. Differentiation of various kinds of Fructus schisandrae by surface desorption atmospheric pressure chemical ionization. Anal. Chim. Acta 2011, 706, 285–290. [Google Scholar] [CrossRef]
- Vieillescazes, C.; Larice, J.-L.; Coen, S. Ancient resinous substances characterization, an approach by principal component analysis. Anal. Chim. Acta 1995, 317, 65–73. [Google Scholar] [CrossRef]
- Lambert, J.B.; Kozminski, M.A.; Santiago-Blay, J.A. Distinctions among conifer exudates by proton magnetic resonance spectroscopy. J. Nat. Prod. 2007, 70, 1283–1294. [Google Scholar] [CrossRef]
- Longobardi, F.; Ventrella, A.; Napoli, C.; Humpfer, E.; Schütz, B.; Schäfer, H.; Kontominas, M.G.; Sacco, A. Classification of olive oils according to geographical origin by using H-1 NMR fingerprinting combined with multivariate analysis. Food Chem. 2012, 130, 177–183. [Google Scholar] [CrossRef]
- Farag, M.A.; Porzel, A.; Wessjohann, L.A. Comparative metabolite profiling and fingerprinting of medicinal licorice roots using a multiplex approach of GC-MS, LC-MS and 1D NMR techniques. Phytochemistry 2012, 76, 60–72. [Google Scholar] [CrossRef]
- Ward, J.L.; Harris, C.; Lewis, J.; Beale, M.H. Assessment of 1H NMR spectroscopy and multivariate analysis as a technique for metabolite fingerprinting of Arabidopsis thaliana. Phytochemistry 2003, 62, 949–957. [Google Scholar]
- Winning, H.; Larsen, F.H.; Bro, R.; Engelsen, S.B. Quantitative analysis of NMR spectra with chemometrics. J. Magn. Reson. 2008, 190, 26–32. [Google Scholar] [CrossRef]
- Von Rudloff, E.; Lapp, M.S.; Yeh, F.C. Chemosystematic study of Thuja plicata-multivariate analysis of leaf oil terpene composition. Biochem. Syst. Ecol. 1988, 16, 119–125. [Google Scholar] [CrossRef]
- Naydenov, K.D.; Tremblay, F.M.; Bergeron, Y.; Alexandrov, A.; Fenton, N. Dissimilar patterns of Pinus heldreichii Christ. populations in Bulgaria revealed by chloroplast microsatellites and terpenes analysis. Biochem. Syst. Ecol. 2005, 33, 133–148. [Google Scholar] [CrossRef]
- Iszkulo, G.; Boratynski, A. Initial period of sexual maturity determines the greater growth rate of male over female in the dioecious tree Juniperus communis subsp. communis. Acta Oecol. 2011, 37, 99–102. [Google Scholar] [CrossRef]
- Vasiliauskas, S.A.; Aarsen, L.W. Sex ratio and neighbor effects in monospecific stand of Juniperus virginiana. Ecology 1992, 73, 622–632. [Google Scholar] [CrossRef]
- Massei, G.; Watkins, R.; Hartler, S.E. Sex-related growth and secondary compounds in Juniperus oxycedrus macrocarpa. Acta Oecol. 2006, 29, 135–140. [Google Scholar] [CrossRef]
- Cool, L.G.; Power, A.B.; Zavarin, E. Variability of foliage terpenes of Fitzroya cupressoides. Biochem. Syst. Ecol. 1991, 19, 421–432. [Google Scholar] [CrossRef]
- Cool, L.G. Sesquiterpene alcohols from foliage of Fitzroya cupressoides. Phytochemistry 1996, 42, 1015–1019. [Google Scholar] [CrossRef]
- Machado, L.B.; Zoghbi, M.G.B.; Andrade, E.H.A. Seasonal variation in the composition of the essential oils from the leaves, thin branches and resin of Protium spruceanum (Benth.) Engl. Flavour Fragr. J. 2003, 18, 338–341. [Google Scholar] [CrossRef]
- Arrabal, C.; García-Vallejo, M.C.; Cadahia, E.; Cortijo, M.; Fernández de Simón, B. Seasonal variations of lipophilic compounds in needles of two chemotypes of Pinus pinaster Ait. Plant Syst. Evol. 2014, 300, 359–367. [Google Scholar] [CrossRef]
- Schloman, W.W.; Garrot, D.J.; Ray, D.T.; Bennet, D.J. Seasonal effects on guayule resin composition. J. Agric. Food Chem. 1986, 34, 177–179. [Google Scholar] [CrossRef]
- Simpson, B.S. Dioecy in plants—Is it an important factor for phytochemists to consider? Planta Med. 2013, 79, 613–615. [Google Scholar] [CrossRef]
- Nybakken, L.; Julkunen-Tiitto, R. Gender differences in Salix myrsinifolia at the pre-reproductive stage are little affected by simulated climatic change. Physiol. Plant. 2013, 147, 465–476. [Google Scholar] [CrossRef]
- Cox, R.E.; Yamamoto, S.; Otto, A.; Simoneit, B.R.T. Oxygenated di- and tricyclic diterpenoids of southern hemisphere conifers. Biochem. Syst. Ecol. 2007, 35, 342–362. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the resin are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Olate, V.R.; Soto, A.; Schmeda-Hirschmann, G. Seasonal Variation and Resin Composition in the Andean Tree Austrocedrus chilensis. Molecules 2014, 19, 6489-6503. https://doi.org/10.3390/molecules19056489
Olate VR, Soto A, Schmeda-Hirschmann G. Seasonal Variation and Resin Composition in the Andean Tree Austrocedrus chilensis. Molecules. 2014; 19(5):6489-6503. https://doi.org/10.3390/molecules19056489
Chicago/Turabian StyleOlate, Verónica Rachel, Alex Soto, and Guillermo Schmeda-Hirschmann. 2014. "Seasonal Variation and Resin Composition in the Andean Tree Austrocedrus chilensis" Molecules 19, no. 5: 6489-6503. https://doi.org/10.3390/molecules19056489