Aqueous Solubility and Degradation Kinetics of the Phytochemical Anticancer Thymoquinone; Probing the Effects of Solvents, pH and Light
Abstract
:1. Introduction
2. Results and Discussion
2.1. Qualitative UV/VIS Spectrophotometric Analysis
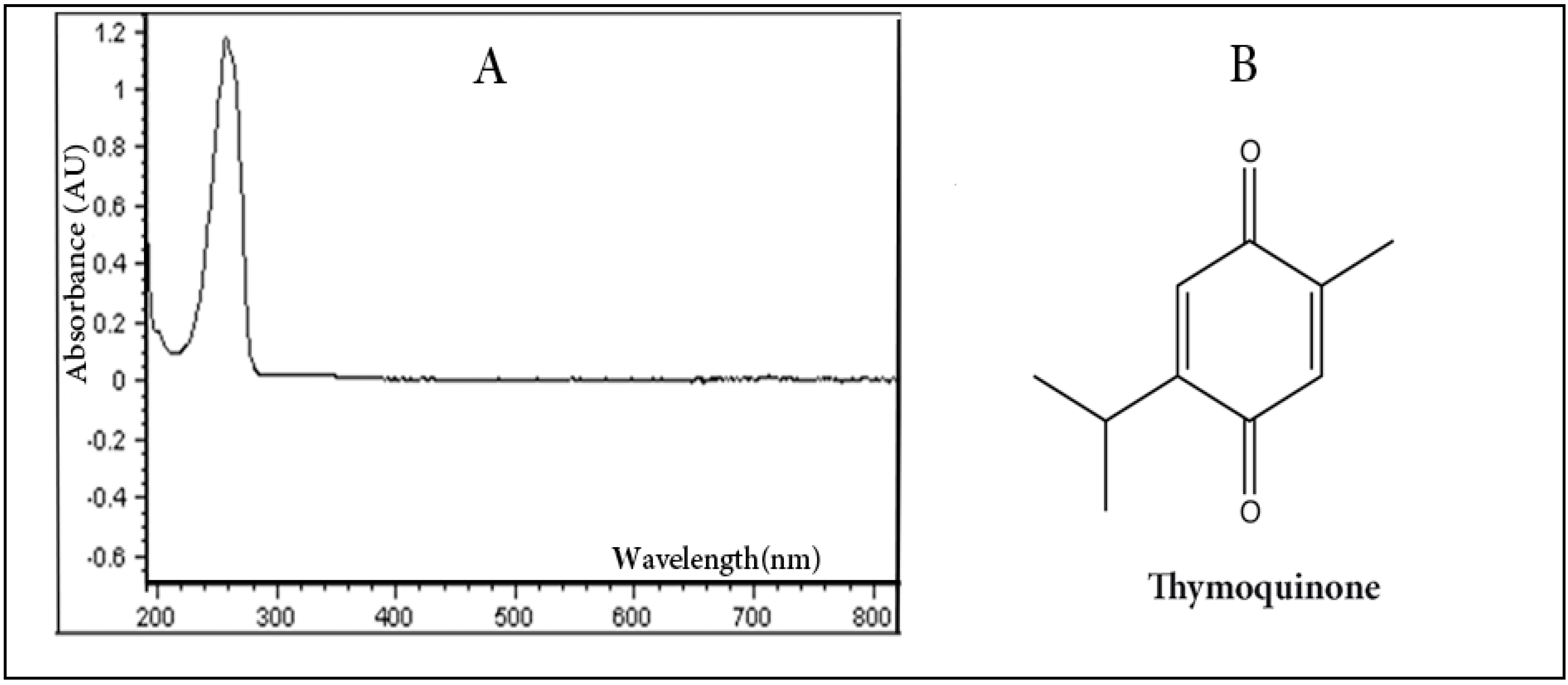
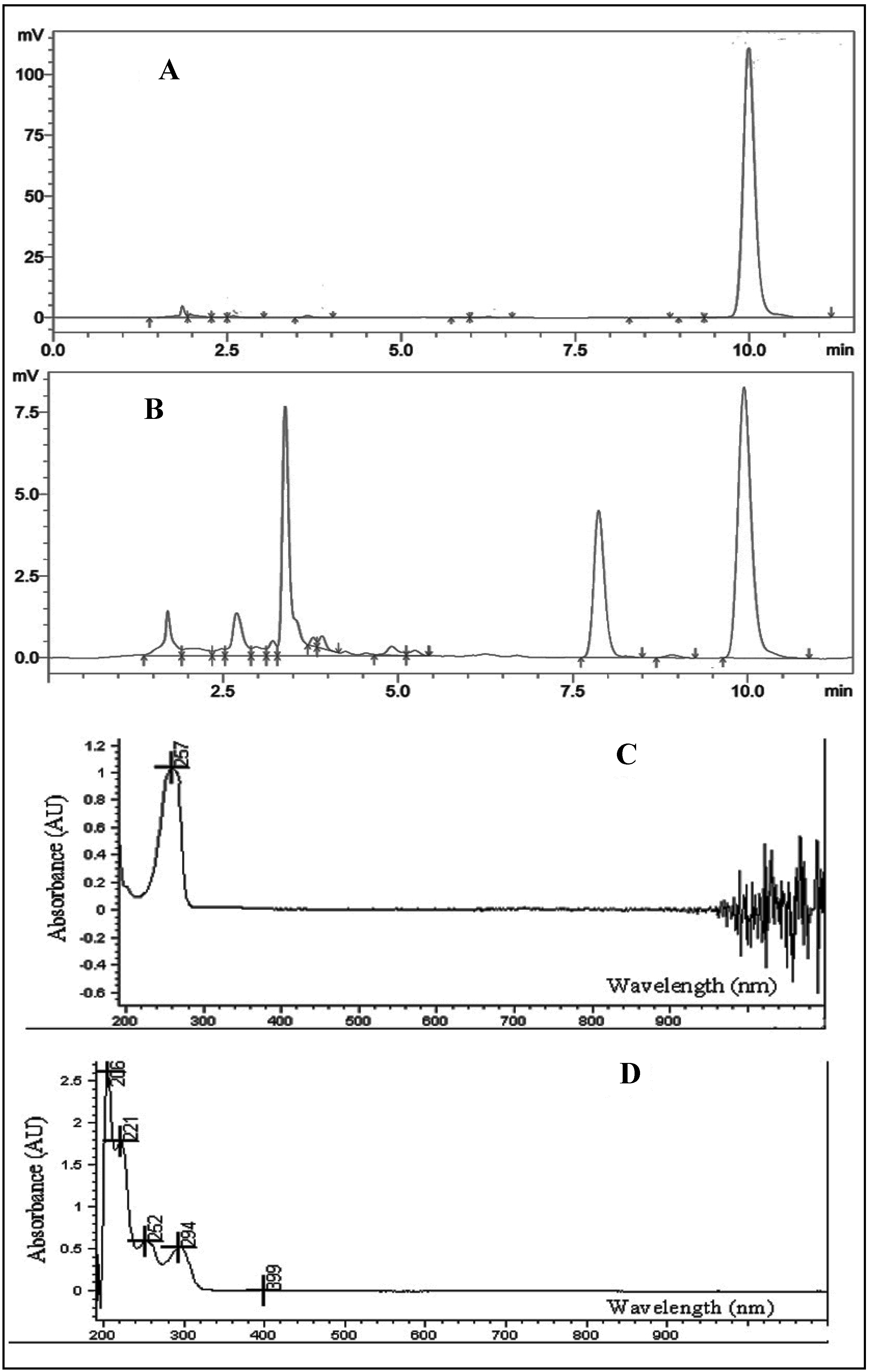
2.2. Quantitative Analysis by HPLC
| Conc. μg/mL | Intra-day | Inter-day | ||||
|---|---|---|---|---|---|---|
| Mean | SD | RSD% | Mean | SD | RSD% | |
| 0.5 | 0.5116 | 0.0368 | 7.2007 | 0.4672 | 0.0365 | 7.8063 |
| 1 | 1.0392 | 0.0837 | 8.0586 | 0.9736 | 0.0464 | 4.7630 |
| 2 | 2.0381 | 0.0342 | 1.6775 | 1.9635 | 0.0633 | 3.2219 |
| 4 | 4.0056 | 0.0485 | 1.2096 | 3.9289 | 0.1201 | 3.0579 |
| 6 | 5.9810 | 0.0523 | 0.8740 | 5.9191 | 0.1263 | 2.1337 |
| 8 | 7.8856 | 0.0438 | 0.5557 | 7.7934 | 0.1990 | 2.5530 |
| 10 | 9.9475 | 0.1498 | 1.5058 | 9.7306 | 0.1849 | 1.9001 |
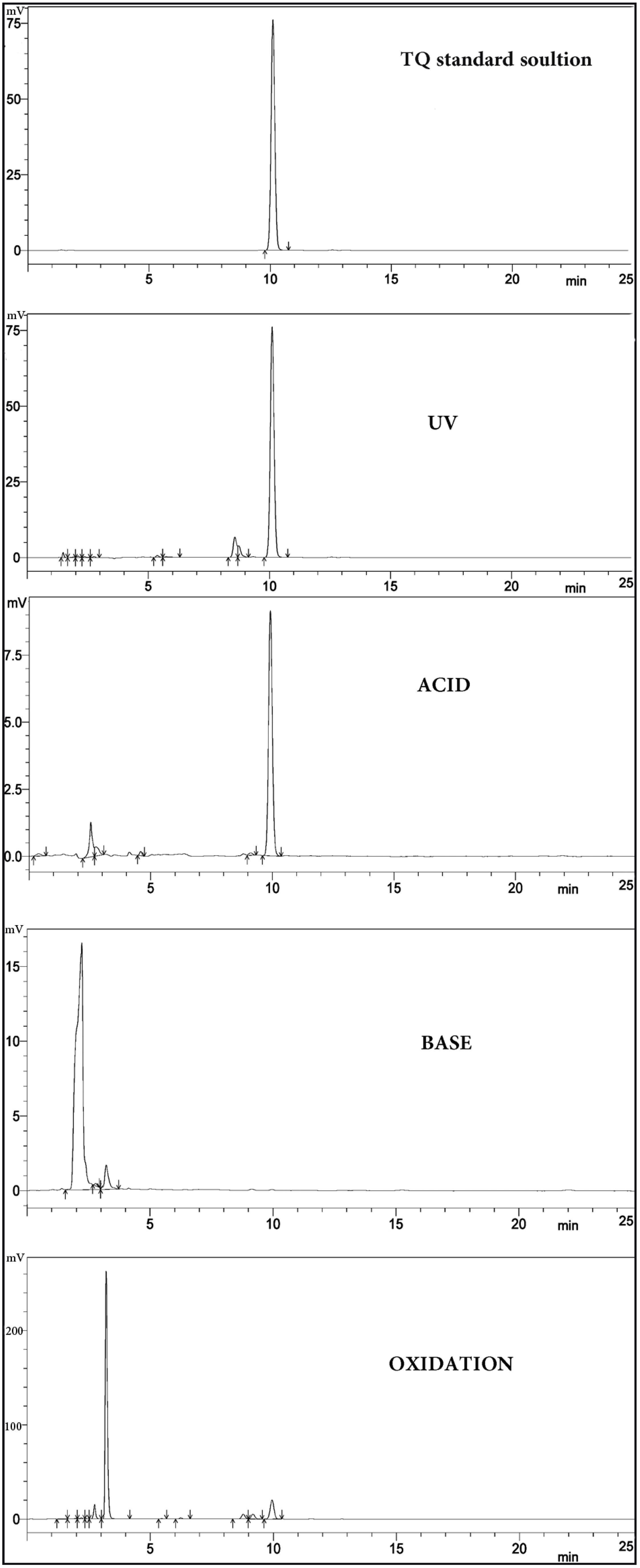
2.3. Forced Degradation Study
2.4. The Solubility Study
| Time (H) | Water | 0.1 N HCl | P.B pH 5 | P.B pH 7.4 | P.B pH 9 |
|---|---|---|---|---|---|
| 24 h | 669.13 ± 5.4 | 653.64 ± 1.9 | 619 ± 7.6 | 566.43 ± 0.19 | 549.19 ± 0.46 |
| 48 h | 698.16 ± 1.4 | 638.4 ± 2.93 | 720.1 ± 1.57 | 550.12 ± 1.87 | 727.57 ± 4.69 |
| 72 h | 740.63 ± 5 | 478.48 ± 0.96 | 739.20 ± 12 | 610.88 ± 4.5 | 665.66 ± 17.5 |
2.5. Aqueous Stability Study

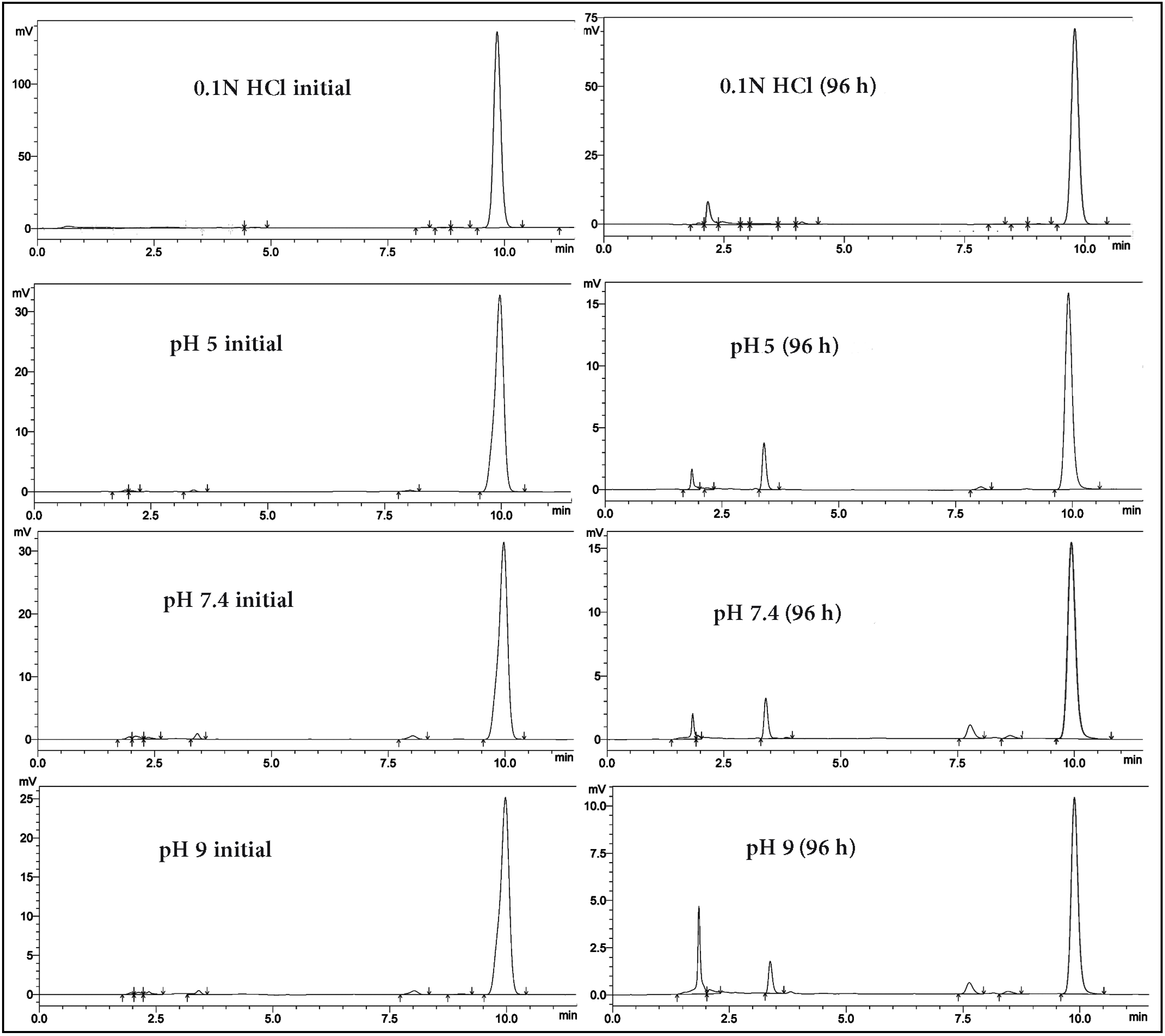
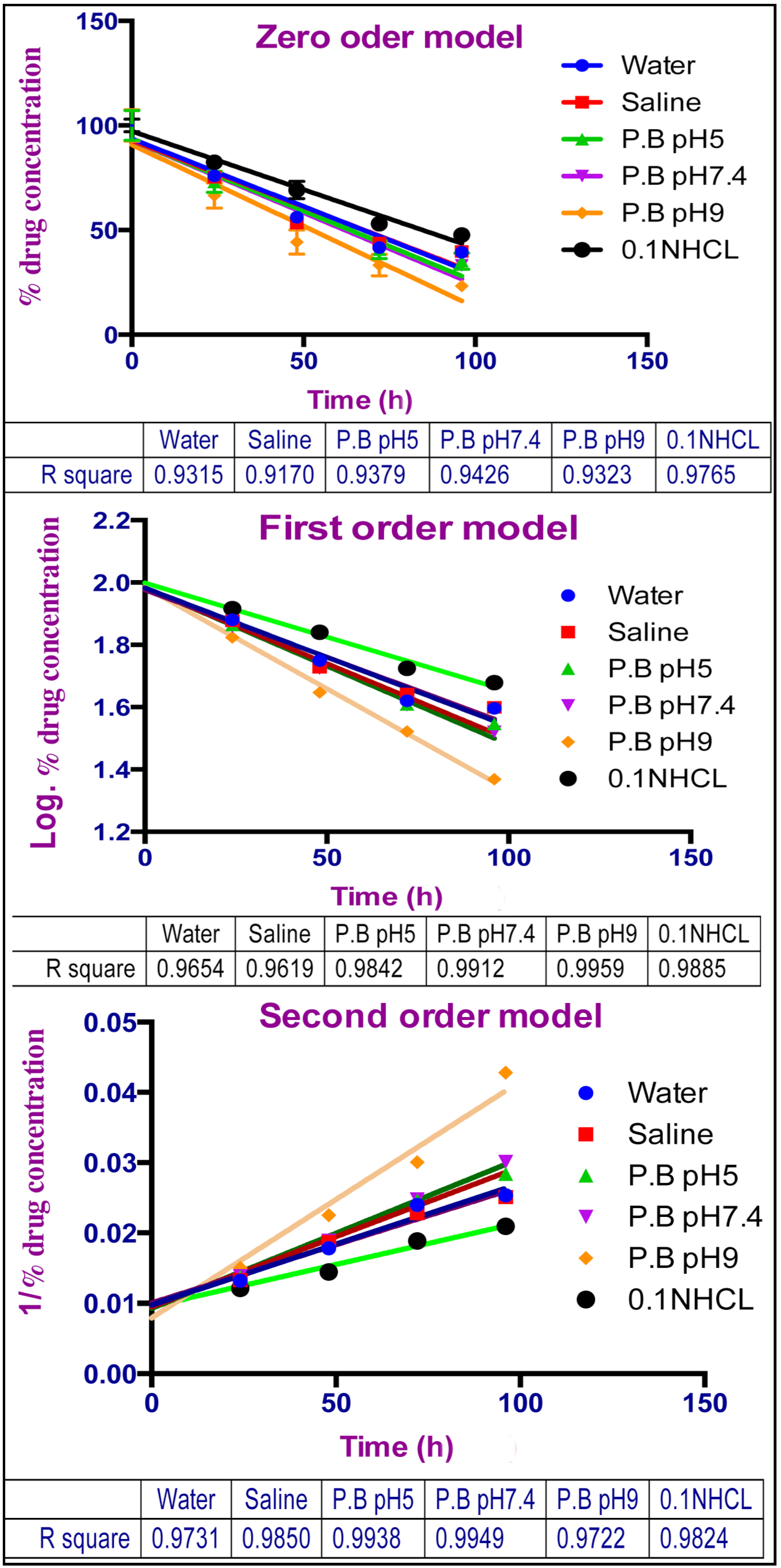
2.6. Effect of Light and Solvent Type on the Degradation of TQ

3. Experimental
3.1. Qualitative Analysis of Thymoquinone
3.2. Quantitative Analysis of Thymoquinone
3.3. Solubility Study
3.4. Aqueous Stability Study
3.5. Effect of Light and Solvent Type on the Degradation of TQ
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Woo, C.C.; Kumar, A.P.; Sethi, G.; Tan, K.H.B. Thymoquinone: Potential cure for inflammatory disorders and cancer. Biochem. Pharmacol. 2012, 83, 443–451. [Google Scholar] [CrossRef]
- Banerjee, S.; Padhye, S.; Azmi, A.; Wang, Z.; Philip, P.A.; Kucuk, O.; Sarkar, F.H.; Mohammad, R.M. Review on molecular and therapeutic potential of thymoquinone in cancer. Nutr. Cancer 2010, 62, 938–946. [Google Scholar] [CrossRef]
- Severina, I.I.; Severin, F.F.; Korshunova, G.A.; Sumbatyan, N.V.; Ilyasova, T.M.; Simonyan, R.A.; Rogov, A.G.; Trendeleva, T.A.; Zvyagilskaya, R.A.; Dugina, V.B.; et al. In search of novel highly active mitochondria-targeted antioxidants: Thymoquinone and its cationic derivatives. FEBS Lett. 2013, 587, 2018–2024. [Google Scholar] [CrossRef]
- Zubair, H.; Khan, H.Y.; Sohail, A.; Azim, S.; Ullah, M.F.; Ahmad, A.; Sarkar, F.H.; Hadi, S.M. Redox cycling of endogenous copper by thymoquinone leads to ROS-mediated DNA breakage and consequent cell death: Putative anticancer mechanism of antioxidants. Cell Death Dis. 2013, 4, e660. [Google Scholar] [CrossRef]
- Badary, O.A.; Taha, R.A.; Gamal El-Din, A.M.; Abdel-Wahab, M.H. Thymoquinone is a potent superoxide anion scavenger. Drug Chem. Toxicol. 2003, 26, 87–98. [Google Scholar] [CrossRef]
- Gali-Muhtasib, H.; Kuester, D.; Mawrin, C.; Bajbouj, K.; Diestel, A.; Ocker, M.; Haboldl, C.; Foltzer-Jourdainne, C.; Schoenfeld, P.; Peters, B.; et al. Thymoquinone Triggers Inactivation of the Stress Response Pathway Sensor CHEK1 and Contributes to Apoptosis in Colorectal Cancer Cells. Cancer Res. 2008, 68, 5609–5618. [Google Scholar] [CrossRef]
- Dergarabetian, E.M.; Ghattass, K.I.; El-Sitt, S.B.; Al-Mismar, R.M.; El-Baba, C.O.; Itani, W.S.; Melhem, N.M.; El-Hajj, H.A.; Bazarbachi, A.A.; Schneider-Stock, R.; et al. Thymoquinone induces apoptosis in malignant T-cells via generation of ROS. Front. Biosci. (Elite Ed.) 2013, 5, 706–719. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Schneider-Stock, R.; Fakhoury, I.H.; Zaki, A.M.; El-Baba1, C.O.; Gali-Muhtasib, H.U. Thymoquinone: Fifty years of success in the battle against cancer models. Drug Discov. Today 2014, 19, 18–30. [Google Scholar] [CrossRef]
- Ganea, G.M.; Fakayode, S.O.; Losso, J.N.; van Nostrum, C.F.; Sabliov, C.M.; Warner, I.M. Delivery of phytochemical thymoquinone using molecular micelle modified poly(D, L lactide-co-glycolide) (PLGA) nanoparticles. Nanotechnology 2010, 21, 1–10. [Google Scholar]
- Alam, S.; Khan, Z.I.; Mustafa, G.; Kumar, M.; Islam, F.; Bhatnagar, A.; Ahmad, F.J. Development and evaluation of thymoquinone-encapsulated chitosan nanoparticles for nose-to-brain targeting: A pharmacoscintigraphic study. Int. J. Nanomed. 2012, 7, 5705–5718. [Google Scholar]
- Singh, A.; Ahmad, I.; Akhter, S.; Jain, G.K.; Iqbal, Z.; Talegaonkar, S.; Ahmad, F.J. Nanocarrier based formulation of Thymoquinone improves oral delivery: Stability assessment, in-vitro and in-vivo studies. Colloids Surf. B 2013, 102, 822–832. [Google Scholar] [CrossRef]
- Odeh, F.; Ismail, S.I.; Abu-Dahab, R.; Mahmoud, I.S.; Bawab, A.A. Thymoquinone in liposomes: A study of loading efficiency and biological activity towards breast cancer. Drug Deliv. 2012, 19, 371–317. [Google Scholar] [CrossRef]
- Ravindran, J.; Nairb, H.B.; Sunga, B.; Prasada, S.; Tekmalb, R.R.; Aggarwala, B.B. Thymoquinone poly (lactide-co-glycolide) nanoparticles exhibit enhanced anti-proliferative, anti-inflammatory, and chemosensitization potential. Biochem. Pharmacol. 2010, 79, 1640–1647. [Google Scholar]
- Cardoso, T.; Galhano, C.I.C.; Ferreira Marques, M.F.; da Silva, A.M. Thymoquinone β-cyclodextrin nanoparticles system: A preliminary study. Spectroscopy 2012, 27, 329–336. [Google Scholar] [CrossRef]
- Abdelwahab, S.I.; Sheikh, B.Y.; Taha, M.M.; How, C.W.; Abdullah, R.; Yagoub, U.; El-Sunousi, R.; Eid, E.E. Thymoquinone-loaded nanostructured lipid carriers: Preparation, gastroprotection, in vitro toxicity, and pharmacokinetic properties after extravascular administration. Int. J. Nanomed. 2013, 8, 2163–2172. [Google Scholar]
- Raschi, A.B.; Romano, E.; Benavente, A.M.; Ben Altabef, A.; Tuttolomondo, M.E. Structural and vibrational analysis of thymoquinone. Spectrochim Acta. Part A Mol. Biomol. Spectrosc. 2010, 77, 497–505. [Google Scholar] [CrossRef]
- Petrucci, R.; Marrosu, G.; Astolfi, P.; Lupidi, G.; Greci, L. Cyclic voltammetry, spectroelectrochemistry and electron spin resonance as combined tools to study thymoquinone in aprotic medium. Electrochim. Acta 2012, 60, 230–238. [Google Scholar] [CrossRef]
- Pagola, S.; Benavente, A.; Raschi, A.; Romano, E.; Molina, M.A.A.; Stephens, P.W. Crystal structure determination of thymoquinone by high-resolution X-ray powder diffraction. AAPS PharmSciTech 2003, 5, 1–8. [Google Scholar]
- Owsik, I.; Kolarz, B. The oxidation of hydroquinone to p-benzoquinone catalysed by Cu(II) ions immobilized on acrylic resins with aminoguanidyl groups: Part 1. J. Mol. Catal. A 2002, 178, 63–71. [Google Scholar] [CrossRef]
- Pathan, S.A.; Jain, G.K.; Zaidi, S.M.; Akhter, S.; Vohora, D.; Chander, P.; Kole, P.L.; Ahmad, F.J.; Khar, R.K. Stability-indicating ultra-performance liquid chromatography method for the estimation of thymoquinone and its application in biopharmaceutical studies. Biomed. Chromatogr. 2011, 25, 613–620. [Google Scholar] [CrossRef]
- Orlando, C.M.; Mark, H.; Bose, A.K.; Manhas, M.S. Photoreactions: Rearrangement of thymoquinone. Chem. Commun. (Lond.) 1966, 20, 714–715. [Google Scholar]
- El-Najjar, N.; Ketola, R.A.; Nissilä, T.; Mauriala, T.; Antopolsky, M.; Jänis, J.; Gali-Muhtasib, H.; Urtti, A.; Vuorela, H. Impact of protein binding on the analytical detectability and anticancer activity of thymoquinone. J. Chem. Biol. 2011, 4, 97–107. [Google Scholar]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Salmani, J.M.M.; Asghar, S.; Lv, H.; Zhou, J. Aqueous Solubility and Degradation Kinetics of the Phytochemical Anticancer Thymoquinone; Probing the Effects of Solvents, pH and Light. Molecules 2014, 19, 5925-5939. https://doi.org/10.3390/molecules19055925
Salmani JMM, Asghar S, Lv H, Zhou J. Aqueous Solubility and Degradation Kinetics of the Phytochemical Anticancer Thymoquinone; Probing the Effects of Solvents, pH and Light. Molecules. 2014; 19(5):5925-5939. https://doi.org/10.3390/molecules19055925
Chicago/Turabian StyleSalmani, Jumah Masoud M., Sajid Asghar, Huixia Lv, and Jianping Zhou. 2014. "Aqueous Solubility and Degradation Kinetics of the Phytochemical Anticancer Thymoquinone; Probing the Effects of Solvents, pH and Light" Molecules 19, no. 5: 5925-5939. https://doi.org/10.3390/molecules19055925
APA StyleSalmani, J. M. M., Asghar, S., Lv, H., & Zhou, J. (2014). Aqueous Solubility and Degradation Kinetics of the Phytochemical Anticancer Thymoquinone; Probing the Effects of Solvents, pH and Light. Molecules, 19(5), 5925-5939. https://doi.org/10.3390/molecules19055925




