Superhydrophobic Surfaces Developed by Mimicking Hierarchical Surface Morphology of Lotus Leaf
Abstract
:1. Introduction
2. Wetting Properties of a Solid Surface

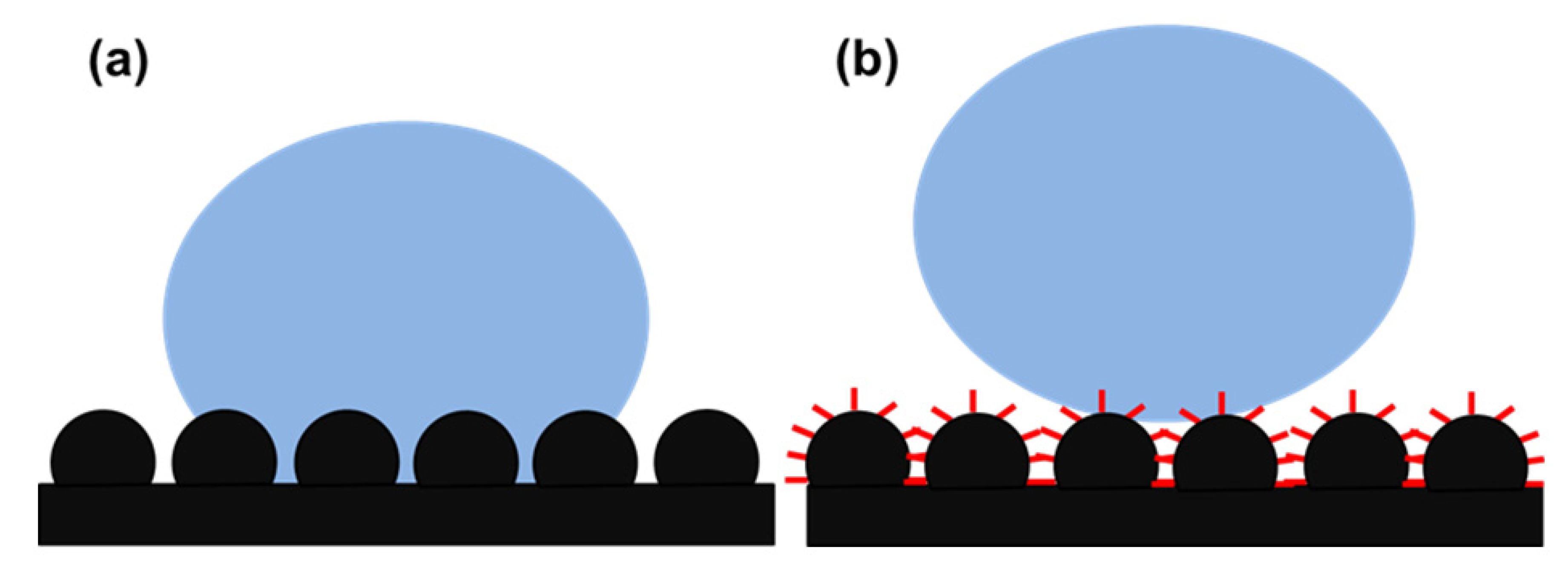
3. Different Wetting Properties of Lotus Leaf




4. Development of Lotus Leaf-Like Surface Morphology to Achieve Superhydrophobicity
4.1. Development of Lotus Leaf-Like Surface Structure Using Lotus Leaf Itself as a Template


4.2. Superhydrophobic Surfaces Developed by Mimicking Lotus-Leaf Morphology









5. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhao, Y.; Xie, Z.; Gu, H.; Zhu, C.; Gu, Z. Bio-inspired variable structural color materials. Chem. Soc. Rev. 2012, 41, 3297–3317. [Google Scholar] [CrossRef]
- Xia, F.; Jiang, L. Bio-inspired, smart, multiscale interfacial materials. Adv. Mater. 2008, 20, 2842–2858. [Google Scholar] [CrossRef]
- Huebsch, N.; Mooney, D.J. Inspiration and application in the evolution of biomaterials. Nature 2009, 462, 426–432. [Google Scholar] [CrossRef]
- Hou, X.; Guo, W.; Jiang, L. Biomimetic smart nanopores and nanochannels. Chem. Soc. Rev. 2011, 40, 2385–2401. [Google Scholar] [CrossRef]
- Quéré, D.; Reyssat, M. Non-adhesive lotus and other hydrophobic materials. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2008, 366, 1539–1556. [Google Scholar] [CrossRef]
- Liu, K.; Yao, X.; Jiang, L. Recent developments in bio-inspired special wettability. Chem. Soc. Rev. 2010, 39, 3240–3255. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Barthlott, W. Multifunctional surface structures of plants: An inspiration for biomimetics. Progr. Mater. Sci. 2009, 54, 137–178. [Google Scholar] [CrossRef]
- Shirtcliffe, J.N.; McHale, G.; Newton, M.I. Learning from superhydrophobic plants: The use of hydrophilic areas on superhydrophobic surfaces for droplet control part of the “Langmuir 25th Year: Wetting and superhydrophobicity” special issue. Langmuir 2009, 25, 14121–14128. [Google Scholar] [CrossRef]
- Marmur, A. The lotus effect: Superhydrophobicity and metastability. Langmuir 2004, 20, 3517–3519. [Google Scholar] [CrossRef]
- Neinhuis, C.; Koch, K.; Barthlott, W. Movement and regeneration of epicuticular waxes through plant cuticles. Planta 2001, 213, 427–434. [Google Scholar] [CrossRef]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Baxter, S.; Cassie, A. 8—The water repellency of fabrics and a new water repellency test. J. Textile Inst. Trans. 1945, 36, T67–T90. [Google Scholar] [CrossRef]
- Cassie, A. Contact angles. Discuss. Faraday Soc. 1948, 3, 11–16. [Google Scholar] [CrossRef]
- Onda, T.; Shibuichi, S.; Satoh, N.; Tsujii, K. Super-water-repellent fractal surfaces. Langmuir 1996, 12, 2125–2127. [Google Scholar] [CrossRef]
- Barthlott, W.; Neinhuis, C. Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta 1997, 202, 1–8. [Google Scholar] [CrossRef]
- Yabu, H.; Shimomura, M. Single-step fabrication of transparent superhydrophobic porous polymer films. Chem. Mater. 2005, 17, 5231–5234. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, C.; Han, Y. Low-density polyethylene superhydrophobic surface by control of its crystallization behavior. Macromol. Rapid Commun. 2004, 25, 1606–1610. [Google Scholar] [CrossRef]
- Khorasani, M.T.; Mirzadeh, H.; Kermani, Z. Wettability of porous polydimethylsiloxane surface: Morphology study. Appl. Surf. Sci. 2005, 242, 339–345. [Google Scholar] [CrossRef]
- Feng, X.; Feng, L.; Jin, M.; Zhai, J.; Jiang, L.; Zhu, D. Reversible super-hydrophobicity to super-hydrophilicity transition of aligned ZnO nanorod films. J. Am. Chem. Soc. 2003, 126, 62–63. [Google Scholar]
- Teshima, K.; Sugimura, H.; Inoue, Y.; Takai, O.; Takano, A. Transparent ultra water-repellent poly(ethylene terephthalate) substrates fabricated by oxygen plasma treatment and subsequent hydrophobic coating. Appl. Surf. Sci. 2005, 244, 619–622. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, F.; Yu, X.; Liu, H.; Fu, Y.; Wang, Z.; Jiang, L.; Li, X. Polyelectrolyte multilayer as matrix for electrochemical deposition of gold clusters: Toward super-hydrophobic surface. J. Am. Chem. Soc. 2004, 126, 3064–3065. [Google Scholar]
- Qian, B.; Shen, Z. Fabrication of superhydrophobic surfaces by dislocation-selective chemical etching on aluminum, copper, and zinc substrates. Langmuir 2005, 21, 9007–9009. [Google Scholar] [CrossRef]
- Shang, H.M.; Wang, Y.; Limmer, S.J.; Chou, T.P.; Takahashi, K.; Cao, G.Z. Optically transparent superhydrophobic silica-based films. Thin Solid Films 2005, 472, 37–43. [Google Scholar] [CrossRef]
- Wu, X.; Zheng, L.; Wu, D. Fabrication of superhydrophobic surfaces from microstructured ZnO-based surfaces via a wet-chemical route. Langmuir 2005, 21, 2665–2667. [Google Scholar]
- Li, Y.; Liu, F.; Sun, J. A facile layer-by-layer deposition process for the fabrication of highly transparent superhydrophobic coatings. Chem. Commun. 2009, 2009, 2730–2732. [Google Scholar]
- Rao, A.V.; Latthe, S.S.; Mahadik, S.A.; Kappenstein, C. Mechanically stable and corrosion resistant superhydrophobic sol–gel coatings on copper substrate. Appl. Surf. Sci. 2011, 257, 5772–5776. [Google Scholar] [CrossRef]
- Boinovich, L.B.; Emelyanenko, A.M. Anti-icing potential of superhydrophobic coatings. Mendeleev Commun. 2013, 23, 3–10. [Google Scholar] [CrossRef]
- Latthe, S.S.; Demirel, A.L. Polystyrene/octadecyltrichlorosilane superhydrophobic coatings with hierarchical morphology. Polym. Chem. 2013, 4, 246–249. [Google Scholar] [CrossRef]
- Jeong, C.; Choi, C.-H. Single-step direct fabrication of pillar-on-pore hybrid nanostructures in anodizing aluminum for superior superhydrophobic efficiency. ACS Appl. Mater. Interf. 2011, 4, 842–848. [Google Scholar] [CrossRef]
- Parkin, I.P.; Palgrave, R.G. Self-cleaning coatings. J. Mater. Chem. 2005, 15, 1689–1695. [Google Scholar] [CrossRef]
- Ma, M.; Hill, R.M. Superhydrophobic surfaces. Curr. Opin. Colloid Interf. Sci. 2006, 11, 193–202. [Google Scholar] [CrossRef]
- Li, X.-M.; Reinhoudt, D.; Crego-Calama, M. What do we need for a superhydrophobic surface? A review on the recent progress in the preparation of superhydrophobic surfaces. Chem. Soc. Rev. 2007, 36, 1350–1368. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, F.; Niu, J.; Jiang, Y.; Wang, Z. Superhydrophobic surfaces: From structural control to functional application. J. Mater. Chem. 2008, 18, 621–633. [Google Scholar] [CrossRef]
- Ganesh, V.A.; Raut, H.K.; Nair, A.S.; Ramakrishna, S. A review on self-cleaning coatings. J. Mater. Chem. 2011, 21, 16304–16322. [Google Scholar]
- Guo, Z.; Liu, W.; Su, B.-L. Superhydrophobic surfaces: From natural to biomimetic to functional. J. Colloid Interf. Sci. 2011, 353, 335–355. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Xia, H.; Kim, E.; Sun, H.-B. Recent developments in superhydrophobic surfaces with unique structural and functional properties. Soft Matter 2012, 8, 11217–11231. [Google Scholar] [CrossRef]
- Nishimoto, S.; Bhushan, B. Bioinspired self-cleaning surfaces with superhydrophobicity, superoleophobicity, and superhydrophilicity. Rsc Adv. 2013, 3, 671–690. [Google Scholar] [CrossRef]
- Latthe, S.S.; Gurav, A.B.; Maruti, C.S.; Vhatkar, R.S. Recent progress in preparation of superhydrophobic surfaces: A review. J. Surf. Eng. Mater. Adv. Technol. 2012, 2, 76–94. [Google Scholar]
- Celia, E.; Darmanin, T.; Taffin de Givenchy, E.; Amigoni, S.; Guittard, F. Recent advances in designing superhydrophobic surfaces. J. Colloid Interf. Sci. 2013, 402, 1–18. [Google Scholar]
- Venkateswara Rao, A.; Latthe, S.S.; Nadargi, D.Y.; Hirashima, H.; Ganesan, V. Preparation of MTMS based transparent superhydrophobic silica films by sol–gel method. J. Colloid Interf. Sci. 2009, 332, 484–490. [Google Scholar] [CrossRef]
- Dhere, S.L.; Latthe, S.S.; Kappenstein, C.; Pajonk, G.M.; Ganesan, V.; Rao, A.V.; Wagh, P.B.; Gupta, S.C. Transparent water repellent silica films by sol–gel process. Appl. Surf. Sci. 2010, 256, 3624–3629. [Google Scholar] [CrossRef]
- Latthe, S.S.; Imai, H.; Ganesan, V.; Venkateswara Rao, A. Ultrahydrophobic silica films by sol–gel process. J. Porous Mater. 2010, 17, 565–571. [Google Scholar] [CrossRef]
- Gurav, A.B.; Latthe, S.S.; Vhatkar, R.S.; Lee, J.-G.; Kim, D.-Y.; Park, J.-J.; Yoon, S.S. Superhydrophobic surface decorated with vertical ZnO nanorods modified by stearic acid. Ceram. Int. 2014, 40, 7151–7160. [Google Scholar] [CrossRef]
- Il’darkhanova, F.I.; Mironova, G.A.; Bogoslovsky, K.G.; Men’shikov, V.V.; Bykov, E.D. Development of paint coatings with superhydrophobic properties. Protect. Metals Phys. Chem. Surf. 2012, 48, 796–802. [Google Scholar] [CrossRef]
- Choi, C.-H.; Kim, C.-J. Large slip of aqueous liquid flow over a nanoengineered superhydrophobic surface. Phys. Rev. Lett. 2006, 96, 066001. [Google Scholar] [CrossRef]
- Aljallis, E.; Sarshar, M.A.; Datla, R.; Sikka, V.; Jones, A.; Choi, C.-H. Experimental study of skin friction drag reduction on superhydrophobic flat plates in high Reynolds number boundary layer flow. Phys. Fluids 2013, 25, 025103. [Google Scholar] [CrossRef]
- Sarshar, M.; Swarctz, C.; Hunter, S.; Simpson, J.; Choi, C.-H. Effects of contact angle hysteresis on ice adhesion and growth on superhydrophobic surfaces under dynamic flow conditions. Colloid Polym. Sci. 2013, 291, 427–435. [Google Scholar] [CrossRef]
- Liu, Y.; Xin, J.H.; Choi, C.-H. Cotton fabrics with single-faced superhydrophobicity. Langmuir 2012, 28, 17426–17434. [Google Scholar] [CrossRef]
- Drelich, J.; Chibowski, E.; Meng, D.D.; Terpilowski, K. Hydrophilic and superhydrophilic surfaces and materials. Soft Matter 2011, 7, 9804–9828. [Google Scholar] [CrossRef]
- Hsu, S.-H.; Woan, K.; Sigmund, W. Biologically inspired hairy structures for superhydrophobicity. Mater. Sci. Eng. 2011, 72, 189–201. [Google Scholar]
- Ensikat, H.J.; Ditsche-Kuru, P.; Neinhuis, C.; Barthlott, W. Superhydrophobicity in perfection: The outstanding properties of the lotus leaf. Beilstein J. Nanotechnol. 2011, 2, 152–161. [Google Scholar] [CrossRef]
- Cheng, Y.-T.; Rodak, D.E. Is the lotus leaf superhydrophobic? Appl. Phys. Lett. 2005, 86, 144101:1–144101:3. [Google Scholar]
- Cheng, Y.-T.; Rodak, D.E.; Angelopoulos, A.; Gacek, T. Microscopic observations of condensation of water on lotus leaves. Appl. Phys. Lett. 2005, 87, 194112:1–194112:3. [Google Scholar]
- Xu, W.; Choi, C.-H. From sticky to slippery droplets: Dynamics of contact line depinning on superhydrophobic surfaces. Phys. Rev. Lett. 2012, 109, 024504. [Google Scholar] [CrossRef]
- Xu, W.; Leeladhar, R.; Kang, Y.T.; Choi, C.-H. Evaporation kinetics of sessile water droplets on micropillared superhydrophobic surfaces. Langmuir 2013, 29, 6032–6041. [Google Scholar] [CrossRef]
- Mockenhaupt, B.; Ensikat, H.; Spaeth, M.; Barthlott, W. Superhydrophobicity of biological and technical surfaces under moisture condensation: Stability in relation to surface structure. Langmuir 2008, 24, 13591–13597. [Google Scholar] [CrossRef]
- Yin, L.; Wang, Q.; Xue, J.; Ding, J.; Chen, Q. Stability of superhydrophobicity of lotus leaf under extreme humidity. Chem. Lett. 2010, 39, 816–817. [Google Scholar] [CrossRef]
- Boreyko, J.B.; Chen, C.-H. Restoring superhydrophobicity of lotus leaves with vibration-induced dewetting. Phys. Rev. Lett. 2009, 103, 174502. [Google Scholar] [CrossRef]
- Liu, Y.; Choi, C.-H. Condensation-induced wetting state and contact angle hysteresis on superhydrophobic lotus leaves. Colloid Polym. Sci. 2013, 291, 437–445. [Google Scholar] [CrossRef]
- Zhang, J.; Sheng, X.; Jiang, L. The dewetting properties of lotus leaves. Langmuir 2008, 25, 1371–1376. [Google Scholar] [CrossRef]
- Sheng, X.; Zhang, J. Air layer on superhydrophobic surface underwater. Colloids Surf. A: Physicochem. Eng. Asp. 2011, 377, 374–378. [Google Scholar] [CrossRef]
- Shirtcliffe, N.J.; McHale, G.; Newton, M.I.; Perry, C.C.; Pyatt, F.B. Plastron properties of a superhydrophobic surface. Appl. Phys. Lett. 2006, 89, 104106:1–104106:2. [Google Scholar]
- Zhao, Y.; Tang, Y.; Wang, X.; Lin, T. Superhydrophobic cotton fabric fabricated by electrostatic assembly of silica nanoparticles and its remarkable buoyancy. Appl. Surf. Sci. 2010, 256, 6736–6742. [Google Scholar] [CrossRef]
- Poetes, R.; Holtzmann, K.; Franze, K.; Steiner, U. Metastable underwater superhydrophobicity. Phys. Rev. Lett. 2010, 105, 166104. [Google Scholar] [CrossRef]
- McHale, G.; Newton, M.I.; Shirtcliffe, N.J. Immersed superhydrophobic surfaces: Gas exchange, slip and drag reduction properties. Soft Matter 2010, 6, 714–719. [Google Scholar] [CrossRef]
- Flynn, M.; Bush, J.W. Underwater breathing: The mechanics of plastron respiration. J. Fluid Mech. 2008, 608, 275–296. [Google Scholar]
- Cheng, Y.T.; Rodak, D.; Wong, C.; Hayden, C. Effects of micro-and nano-structures on the self-cleaning behaviour of lotus leaves. Nanotechnology 2006, 17, 1359. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, X.; Xin, J. Can superhydrophobic surfaces repel hot water? J. Mater. Chem. 2009, 19, 5602–5611. [Google Scholar] [CrossRef]
- Cheng, Q.; Li, M.; Zheng, Y.; Su, B.; Wang, S.; Jiang, L. Janus interface materials: Superhydrophobic air/solid interface and superoleophobic water/solid interface inspired by a lotus leaf. Soft Matter 2011, 7, 5948–5951. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, Z.; Cheng, B.; de Simone, J.M.; Samulski, E.T. Superhydrophobic behavior of a perfluoropolyether lotus-leaf-like topography. Langmuir 2006, 22, 8576–8580. [Google Scholar] [CrossRef]
- Xia, Y.; Whitesides, G.M. Soft Lithography. Angew. Chem. Int. Ed. 1998, 37, 550–575. [Google Scholar] [CrossRef]
- Sun, M.; Luo, C.; Xu, L.; Ji, H.; Ouyang, Q.; Yu, D.; Chen, Y. Artificial lotus leaf by nanocasting. Langmuir 2005, 21, 8978–8981. [Google Scholar] [CrossRef]
- Saison, T.; Peroz, C.; Chauveau, V.; Berthier, S.; Sondergard, E.; Arribart, H. Replication of butterfly wing and natural lotus leaf structures by nanoimprint on silica sol–gel films. Bioinspir. Biomimet. 2008, 3, 046004. [Google Scholar] [CrossRef]
- Shafiei, M.; Alpas, A.T. Nanocrystalline nickel films with lotus leaf texture for superhydrophobic and low friction surfaces. Appl. Surf. Sci. 2009, 256, 710–719. [Google Scholar] [CrossRef]
- Yang, J.; Yan, X.; Wang, Y.; Luo, B.; Wang, L.; Xue, Q. Deposition of bio-mimicking graphene sheets with lotus leaf-like and cell-like structures on the nickel substrate. Chin. Sci. Bull. 2012, 57, 3036–3039. [Google Scholar] [CrossRef]
- Szilágyi, I.M.; Teucher, G.; Härkönen, E.; Färm, E.; Hatanpää, T.; Nikitin, T.; Khriachtchev, L.; Räsänen, M.; Ritala, M.; Leskelä, M. Programming nanostructured soft biological surfaces by atomic layer deposition. Nanotechnology 2013, 24, 245701. [Google Scholar] [CrossRef]
- Fürstner, R.; Barthlott, W.; Neinhuis, C.; Walzel, P. Wetting and self-cleaning properties of artificial superhydrophobic surfaces. Langmuir 2005, 21, 956–961. [Google Scholar] [CrossRef]
- Lee, S.M.; Kwon, T.H. Effects of intrinsic hydrophobicity on wettability of polymer replicas of a superhydrophobic lotus leaf. J. Micromechan. Microeng. 2007, 17, 687–692. [Google Scholar]
- Liu, B.; He, Y.; Fan, Y.; Wang, X. Fabricating super-hydrophobic lotus-leaf-like surfaces through soft-lithographic imprinting. Macromol. Rapid Commun. 2006, 27, 1859–1864. [Google Scholar] [CrossRef]
- Yuan, Z.; Chen, H.; Zhang, J. Facile method to prepare lotus-leaf-like super-hydrophobic poly (vinyl chloride) film. Appl. Surf. Sci. 2008, 254, 1593–1598. [Google Scholar] [CrossRef]
- Guan, Z. Preparation of material surface structure similar to hydrophobic structure of lotus leaf. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2008, 23, 513–517. [Google Scholar] [CrossRef]
- Liu, C. Rapid fabrication of microfluidic chip with three-dimensional structures using natural lotus leaf template. Microfluid. Nanofluid. 2010, 9, 923–931. [Google Scholar] [CrossRef]
- Jin, G.; Kim, G. Pressure/electric-field-assisted micro/nanocasting method for replicating a lotus leaf. Langmuir 2010, 27, 828–834. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, D.; Fan, T.; Ding, J.; Gu, J.; Guo, Q.; Ogawa, H. Biomimetic zinc oxide replica with structural color using butterfly (Ideopsis similis) wings as templates. Bioinspir. Biomimet. 2006, 1, 89. [Google Scholar] [CrossRef]
- Parker, A.R.; Townley, H.E. Biomimetics of photonic nanostructures. Nat. Nanotechnol. 2007, 2, 347–353. [Google Scholar] [CrossRef]
- Cho, W.K.; Choi, I.S. Fabrication of hairy polymeric films inspired by geckos: Wetting and high adhesion properties. Adv. Funct. Mater. 2008, 18, 1089–1096. [Google Scholar]
- Feng, L.; Zhang, Y.; Li, M.; Zheng, Y.; Shen, W.; Jiang, L. The structural color of red rose petals and their duplicates. Langmuir 2010, 26, 14885–14888. [Google Scholar]
- Koch, K.; Bhushan, B.; Jung, Y.C.; Barthlott, W. Fabrication of artificial Lotus leaves and significance of hierarchical structure for superhydrophobicity and low adhesion. Soft Matter 2009, 5, 1386–1393. [Google Scholar] [CrossRef]
- Pacifico, J.; Endo, K.; Morgan, S.; Mulvaney, P. Superhydrophobic effects of self-assembled monolayers on micropatterned surfaces: 3-D arrays mimicking the lotus leaf. Langmuir 2006, 22, 11072–11076. [Google Scholar] [CrossRef]
- Wang, S.; Feng, L.; Jiang, L. One-Step Solution-Immersion Process for the Fabrication of Stable Bionic Superhydrophobic Surfaces. Adv. Mater. 2006, 18, 767–770. [Google Scholar] [CrossRef]
- Ji, J.; Fu, J.; Shen, J. Fabrication of a superhydrophobic surface from the amplified exponential growth of a multilayer. Adv. Mater. 2006, 18, 1441–1444. [Google Scholar] [CrossRef]
- Yuan, Z.Q.; Chen, H.; Tang, J.X.; Gong, H.F.; Liu, Y.J.; Wang, Z.X.; Shi, P.; Zhang, J.D.; Chen, X. A novel preparation of polystyrene film with a superhydrophobic surface using a template method. J. Phys. D: Appl. Phys. 2007, 40, 3485–3489. [Google Scholar] [CrossRef]
- Xiu, Y.; Zhu, L.; Hess, D.W.; Wong, C. Hierarchical silicon etched structures for controlled hydrophobicity/superhydrophobicity. Nano Lett. 2007, 7, 3388–3393. [Google Scholar] [CrossRef]
- Li, Y.; Li, C.; Cho, S.O.; Duan, G.; Cai, W. Silver hierarchical bowl-like array: Synthesis, superhydrophobicity, and optical properties. Langmuir 2007, 23, 9802–9807. [Google Scholar] [CrossRef]
- Larmour, I.A.; Bell, S.E.; Saunders, G.C. Remarkably simple fabrication of superhydrophobic surfaces using electroless galvanic deposition. Angew. Chem. 2007, 119, 1740–1742. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, M.; Zhai, J.; Wang, J.; Jiang, L. Bioinspired construction of Mg-Li alloys surfaces with stable superhydrophobicity and improved corrosion resistance. Appl. Phys. Lett. 2008, 92, 183103:1–183103:3. [Google Scholar]
- Kim, D.; Kim, J.; Park, H.C.; Lee, K.-H.; Hwang, W. A superhydrophobic dual-scale engineered lotus leaf. J. Micromechan. Microeng. 2008, 18, 015019. [Google Scholar]
- Crick, C.R.; Parkin, I.P. A single step route to superhydrophobic surfaces through aerosol assisted deposition of rough polymer surfaces: Duplicating the lotus effect. J. Mater. Chem. 2009, 19, 1074–1076. [Google Scholar] [CrossRef]
- Chen, X.H.; Yang, G.B.; Kong, L.H.; Dong, D.; Yu, L.G.; Chen, J.M.; Zhang, P.Y. Direct growth of hydroxy cupric phosphate heptahydrate monocrystal with honeycomb-like porous structures on copper surface mimicking lotus leaf. Cryst. Growth Design 2009, 9, 2656–2661. [Google Scholar] [CrossRef]
- Bayer, I.; Steele, A.; Martorana, P.; Loth, E. Fabrication of superhydrophobic polyurethane/organoclay nano-structured composites from cyclomethicone-in-water emulsions. Appl. Surf. Sci. 2010, 257, 823–826. [Google Scholar] [CrossRef]
- Uchida, K.; Nishikawa, N.; Izumi, N.; Yamazoe, S.; Mayama, H.; Kojima, Y.; Yokojima, S.; Nakamura, S.; Tsujii, K.; Irie, M. Phototunable diarylethene microcrystalline surfaces: Lotus and petal effects upon wetting. Angew. Chem. Int. Ed. 2010, 49, 5942–5944. [Google Scholar] [CrossRef]
- Myint, M.T.Z.; Kitsomboonloha, R.; Baruah, S.; Dutta, J. Superhydrophobic surfaces using selected zinc oxide microrod growth on ink-jetted patterns. J. Colloid Interf. Sci. 2011, 354, 810–815. [Google Scholar] [CrossRef]
- Lin, J.; Cai, Y.; Wang, X.; Ding, B.; Yu, J.; Wang, M. Fabrication of biomimetic superhydrophobic surfaces inspired by lotus leaf and silver ragwort leaf. Nanoscale 2011, 3, 1258–1262. [Google Scholar] [CrossRef]
- Qu, M.; Zhao, G.; Cao, X.; Zhang, J. Biomimetic fabrication of lotus-leaf-like structured polyaniline film with stable superhydrophobic and conductive properties. Langmuir 2008, 24, 4185–4189. [Google Scholar] [CrossRef]
- Lee, M.W.; An, S.; Latthe, S.S.; Lee, C.; Hong, S.; Yoon, S.S. Electrospun Polystyrene Nanofiber Membrane with Superhydrophobicity and Superoleophilicity for Selective Separation of Water and Low Viscous Oil. ACS Appl. Mater. Interfaces 2013, 5, 10597–10604. [Google Scholar] [CrossRef]
- Lee, M.W.; An, S.; Joshi, B.; Latthe, S.S.; Yoon, S.S. Highly efficient wettability control via three-dimensional (3D) suspension of titania nanoparticles in polystyrene nanofibers. ACS Appl. Mater. Interfaces 2013, 5, 1232–1239. [Google Scholar] [CrossRef]
- Jiang, L.; Zhao, Y.; Zhai, J. A Lotus-leaf-like superhydrophobic surface: A porous microsphere/nanofiber composite film prepared by electrohydrodynamics. Angew. Chem. 2004, 116, 4438–4441. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, J.; Zheng, Y.; Huang, Z.; Feng, L.; Jiang, L. Stable, superhydrophobic, and conductive polyaniline/polystyrene films for corrosive environments. Adv. Funct. Mater. 2006, 16, 568–574. [Google Scholar] [CrossRef]
- Mao, C.; Liang, C.; Luo, W.; Bao, J.; Shen, J.; Hou, X.; Zhao, W. Preparation of lotus-leaf-like polystyrene micro-and nanostructure films and its blood compatibility. J. Mater. Chem. 2009, 19, 9025–9029. [Google Scholar] [CrossRef]
- Hou, W.; Wang, Q. Stable polytetrafluoroethylene superhydrophobic surface with lotus-leaf structure. J. Colloid Interf. Sci. 2009, 333, 400–403. [Google Scholar] [CrossRef]
- Guo, Z.-G.; Liu, W.-M.; Su, B.-L. A stable lotus-leaf-like water-repellent copper. Appl. Phys. Lett. 2008, 92, 063104:1–063104:3. [Google Scholar]
- Guo, Z.; Zhou, F.; Hao, J.; Liu, W. Stable biomimetic super-hydrophobic engineering materials. J. Am. Chem. Soc. 2005, 127, 15670–15671. [Google Scholar] [CrossRef]
- Xi, W.; Qiao, Z.; Zhu, C.; Jia, A.; Li, M. The preparation of lotus-like super-hydrophobic copper surfaces by electroplating. Appl. Surf. Sci. 2009, 255, 4836–4839. [Google Scholar] [CrossRef]
- Marmur, A. Wetting on hydrophobic rough surfaces: To be heterogeneous or not to be? Langmuir 2003, 19, 8343–8348. [Google Scholar] [CrossRef]
- Wu, X.; Shi, G. Fabrication of a lotus-like micro–nanoscale binary structured surface and wettability modulation from superhydrophilic to superhydrophobic. Nanotechnology 2005, 16, 2056–2060. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, J.; Zhang, Y.; Zhao, F.; Zhang, Y. Fabrication of superhydrophobic surface by hierarchical growth of lotus-leaf-like boehmite on aluminum foil. J. Colloid Interf. Sci. 2011, 358, 277–283. [Google Scholar] [CrossRef]
- Zhao, N.; Xu, J.; Xie, Q.; Weng, L.; Guo, X.; Zhang, X.; Shi, L. Fabrication of biomimetic superhydrophobic coating with a micro-nano-binary structure. Macromol. Rapid Commun. 2005, 26, 1075–1080. [Google Scholar] [CrossRef]
- Wei, Z.J.; Liu, W.L.; Tian, D.; Xiao, C.L.; Wang, X.Q. Preparation of lotus-like superhydrophobic fluoropolymer films. Appl. Surf. Sci. 2010, 256, 3972–3976. [Google Scholar] [CrossRef]
- Li, Y.; Huang, X.J.; Heo, S.H.; Li, C.C.; Choi, Y.K.; Cai, W.P.; Cho, S.O. Superhydrophobic bionic surfaces with hierarchical microsphere/SWCNT composite arrays. Langmuir 2007, 23, 2169–2174. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, X.; Xin, J. Super-hydrophobic surfaces from a simple coating method: A bionic nanoengineering approach. Nanotechnology 2006, 17, 3259. [Google Scholar] [CrossRef]
- Kim, T.-Y.; Ingmar, B.; Bewilogua, K.; Oh, K.H.; Lee, K.-R. Wetting behaviours of aC: H: Si: O film coated nano-scale dual rough surface. Chem. Phys. Lett. 2007, 436, 199–203. [Google Scholar] [CrossRef]
- Shirtcliffe, N.J.; McHale, G.; Newton, M. The superhydrophobicity of polymer surfaces: Recent developments. J. Polym. Sci. Part B: Polym. Phys. 2011, 49, 1203–1217. [Google Scholar] [CrossRef]
- Latthe, S.S.; Terashima, C.; Nakata, K.; Sakai, M.; Fujishima, A. Development of sol–gel processed semi-transparent and self-cleaning superhydrophobic coatings. J. Mater. Chem. A 2014, 2, 5548–5553. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Latthe, S.S.; Terashima, C.; Nakata, K.; Fujishima, A. Superhydrophobic Surfaces Developed by Mimicking Hierarchical Surface Morphology of Lotus Leaf. Molecules 2014, 19, 4256-4283. https://doi.org/10.3390/molecules19044256
Latthe SS, Terashima C, Nakata K, Fujishima A. Superhydrophobic Surfaces Developed by Mimicking Hierarchical Surface Morphology of Lotus Leaf. Molecules. 2014; 19(4):4256-4283. https://doi.org/10.3390/molecules19044256
Chicago/Turabian StyleLatthe, Sanjay S., Chiaki Terashima, Kazuya Nakata, and Akira Fujishima. 2014. "Superhydrophobic Surfaces Developed by Mimicking Hierarchical Surface Morphology of Lotus Leaf" Molecules 19, no. 4: 4256-4283. https://doi.org/10.3390/molecules19044256
APA StyleLatthe, S. S., Terashima, C., Nakata, K., & Fujishima, A. (2014). Superhydrophobic Surfaces Developed by Mimicking Hierarchical Surface Morphology of Lotus Leaf. Molecules, 19(4), 4256-4283. https://doi.org/10.3390/molecules19044256





