Agaricus Blazei Hot Water Extract Shows Anti Quorum Sensing Activity in the Nosocomial Human Pathogen Pseudomonas Aeruginosa
Abstract
:1. Introduction
2. Results and Discussion
| Concentration | 0.1 mg/mL * | 0.05 mg/mL | 0.025 mg/mL |
|---|---|---|---|
| A. blazei extract | 16.06 ± 0.47 | 42.58 ± 0.35 | 98.37 ± 0.97 |
| Ampicillin | 69.16 ± 0.65 | 56.46 ± 0.46 | 92.16 ± 0.37 |
| Streptomycin | 49.40 ± 0.46 | 70.97 ± 0.36 | 88.36 ± 0.42 |
| P. aeruginosa control | 98.90 ± 0.97 | 100.00 ± 0.00 | 99.30 ± 0.46 |
| Concentration mg/disc | 0.03 | 0.06 | 0.125 | 0.25 | 0.50 | |
|---|---|---|---|---|---|---|
| A. blazei extr. | MIC | - | - | - | - | - |
| AQ | 7.0 ± 1.0 | 7.3 ± 0.5 | 8.0 ± 1.0 | 9.3 ± 1.0 | 17.7 ± 1.0 | |
| Ampicillin | MIC | - | - | - | - | - |
| AQ | -- | -- | -- | 7.6 ± 0.6 | 9.0 ± 1.0 | |
| Streptomycin | MIC | - | - | 9.0 ± 1.0 15.0 ± 2.1 | 16.6 ± 1.5 22.6 ± 2.3 | 18.6 ± 1.2 26.7 ± 1.2 |
| AQ | ||||||
| P. aeruginosa Control | MIC AQ | ni | ni | ni | ni | ni |
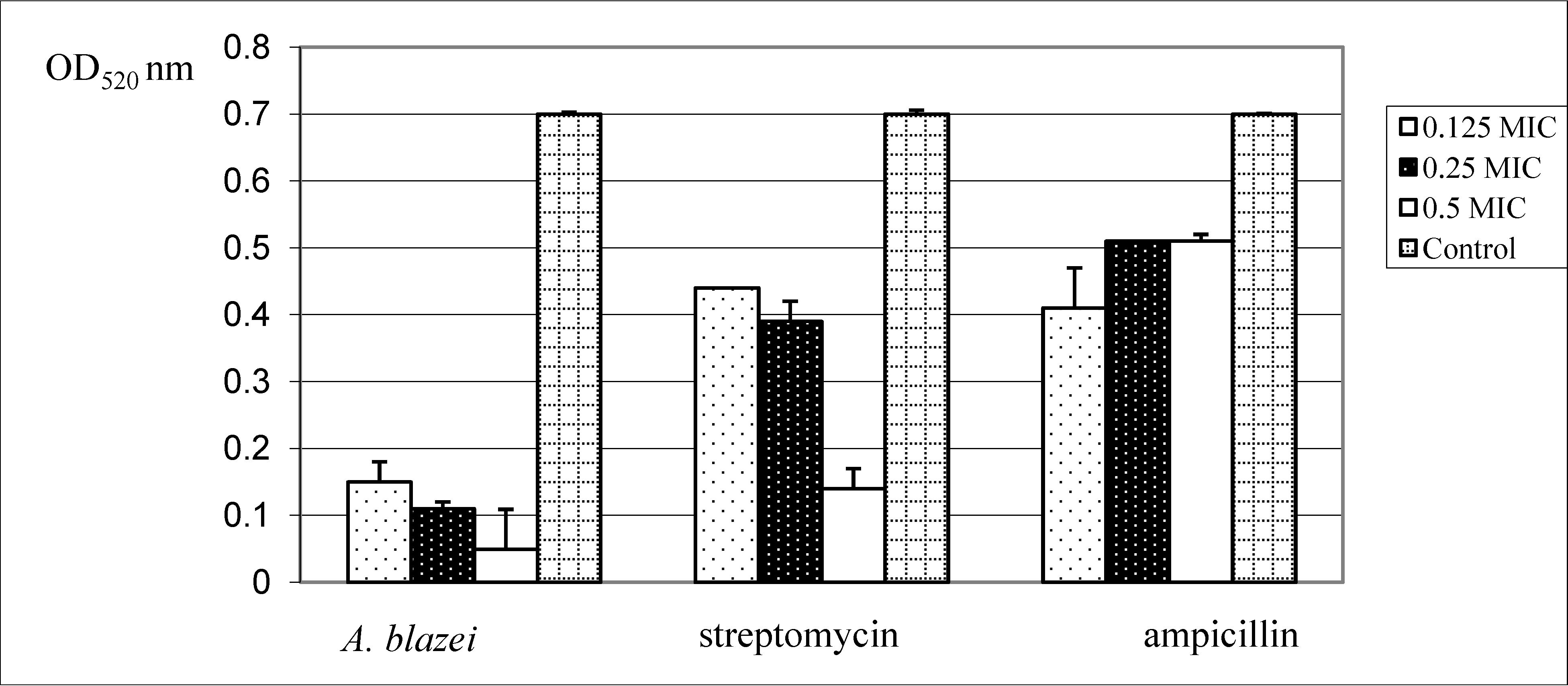
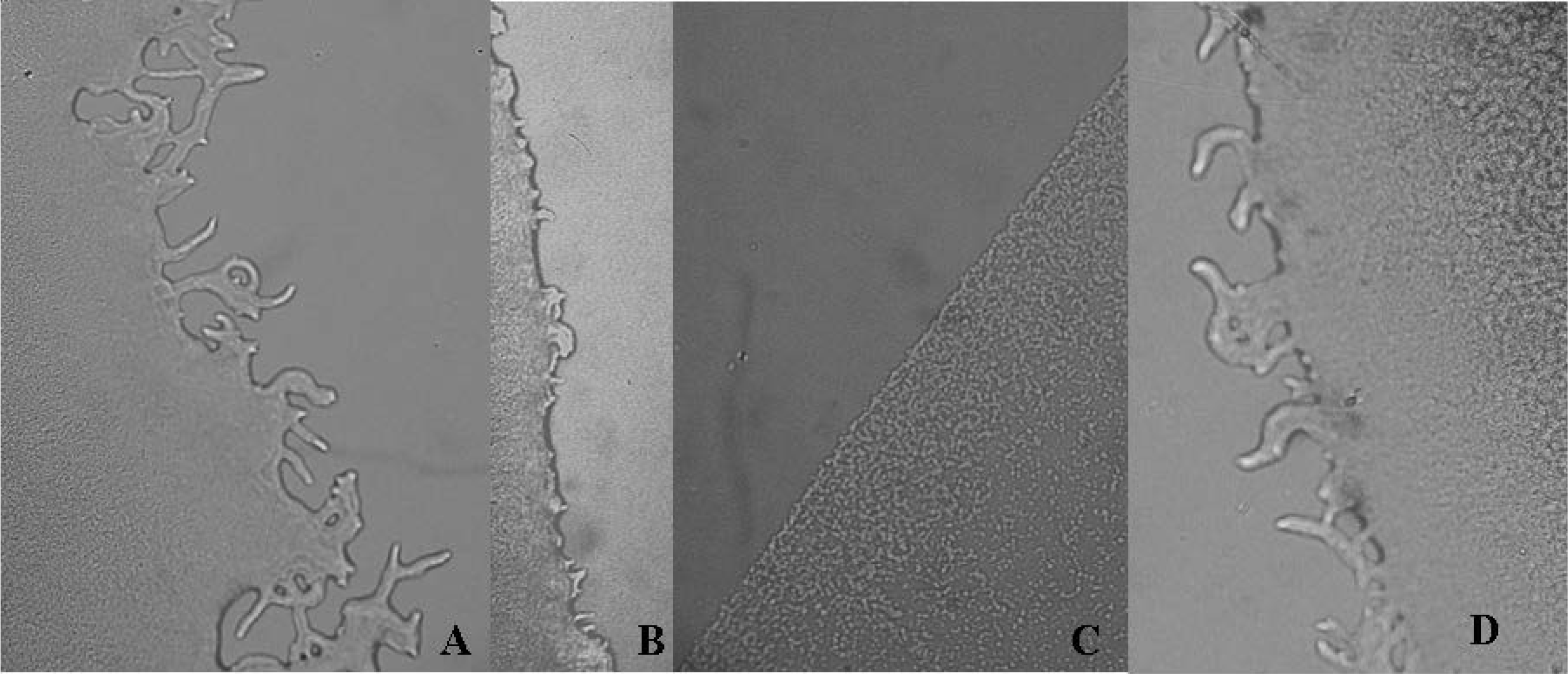
| Extracts | Colony diameter | Colony color | Colony edge |
|---|---|---|---|
| A. blazei H2O | 21.67 ± 0.51 | white | reduced flagella |
| Streptomycin | 5.00 ± 0.06 | white | flat |
| Ampicillin | 12.00 ± 1.00 | white | regular |
| Control P. aeruginosa 109 CFU | 14.00 ± 1.00 | green | regular |
3. Experimental
3.1. Extract Preparation
3.2. Bacterial Strains, Growth Media and Culture Conditions
3.3. Biofilm Formation
3.4. Inhibition of Biofilm Formation of P. aeruginosa
3.5. Inhibition of Twitching and Flagella Motility of P. aeruginosa
3.6. Inhibition of Synthesis of P. aeruginosa PA01 Pyocyanin
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heinemann, P. Essai d’une clé de détermination des genres Agaricus et Micropsalliota. Sydowia 1977, 30, 36–37. [Google Scholar]
- Wasser, S.P.; Didukh, M.Y.; Amazonas, M.A.L.; Nevo, E.; Stamets, P.; da Eira, A.F. Is a widely cultivated culinary-medicinal royal sun Agaricus. (the Himematsutake mushroom) indeed Agaricus. blazei Murill? Int. J. Med. Mushrooms 2002, 4, 274. [Google Scholar]
- Almdahl, S.M.; Seljelid, R. Semisoluble aminated glucan: Long-term efficacy against an intraperitoneal E. coli challenge and its effect on formation of abdominal adhesions. Res. Exp. Med. 1987, 187, 369–377. [Google Scholar]
- Brown, G.D.; Gordon, S. Fungal β-glucans and mammalian immunity. Immunology 2003, 19, 311–315. [Google Scholar]
- Hetland, G.; Johnson, E.; Eide, D.M.; Samuelsen, A.B.C.; Wiker, H.G. Antimicrobial effects of β-glucans and pectin and of the Agaricus. blazei-based mushroom extract, Andosan™ Examples of mouse models for pneumococcal-, fecal bacterial-, and mycobacterial infections. In Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Mendez-Vilas, A., Ed.; Formatex: Badajoz, Spain, 2013; pp. 889–898. [Google Scholar]
- Smiderle, F.S.; Alquini, G.; Tadra-Sfeir, M.Z.; Iacomini, M.; Wichers, H.J.; van Griensven, L.J.L.D. Agaricus. bisporus and Agaricus. brasiliensis (1–6)→β-d-glucans show immunostimulatory activity on human THP-1 derived macrophages. In Carbohydr. Polym.; 2013; Volume 94, pp. 91–99. [Google Scholar]
- Bernardshaw, S.; Johnson, E.; Hetland, G. An extract of the mushroom Agaricus. blazei Murill administered orally against systemic Streptococcus pneumoniae infection in mice. Scand. J. Immunol. 2005, 62, 393–398. [Google Scholar] [CrossRef]
- Fantuzzi, E.; Rezende Anastacio, L.; Nicoli, J.R.; Oliveira de Paula, S.; Esteves Arantes, R.M.; Dantas Vanetti, M.C. Evaluation of Royal Sun Agaricus. brasiliensis S. Wasser et al. aqeous extract in mice challenged with Salmonella enterica serovar typhimurium. Int. J. Med. Mushrooms 2011, 13, 281–288. [Google Scholar] [CrossRef]
- Lewis, S.K.; Camper, A.; Ehrlich, G.; Costerton, J.; Davies, D. Pseudomonas aeruginosa displays multiple phenotypes during development as a biofilm. J. Bacteriol. 2002, 184, 1140–1154. [Google Scholar] [CrossRef]
- Li, Y.-H.; Tian, X. Quorum sensing and bacterial social interactions in biofilms. Sensors 2012, 12, 2519–2538. [Google Scholar] [CrossRef]
- Rumbaugh, K.P.; Diggle, S.P.; Watters, S.M.; Ross-Gillespie, A.; Griffin, A.S.; West, S.A. Quorum sensing and the social evolution of bacterial virulence. Curr. Biol. 2009, 19, 341–345. [Google Scholar]
- Bjarnshoit, T.; Jensen, P.Q.; Jakobsen, T.H.; Phipps, R.; Nielsen, A.K.; Rybtke, M.T.; Tolker-Nielsen, T.; Givskov, M.; Haiby, N.; Ciofu, O. Quorum sensing and virulence of Pseudomonas aeruginosa during lung infection of cystic fibrosis patients. PLoS One 2010, 5, e10115. [Google Scholar] [CrossRef]
- Amadi, E.; Uzoaru, P.; Nwaziri, A.; Iroha, I. Antibiotic resistance in clinical isolates of Pseudomonas aeruginosa in Enugu and Abakaliki, Nigeria. Int. J. Infect. Dis. 2013, 7, 9474. [Google Scholar]
- Aloush, V.; Navon-Venezia, S.; Seigman-Igra, Y.; Cabili, S.; Carmeli, Y. Multidrug resistant Pseudomonas aeruginosa: Risk factors and clinical impact. Antimicrob. Agents Chemother. 2006, 50, 43–48. [Google Scholar] [CrossRef]
- Hirsch, E.B.; Tam, V.H. Impact of multi drug resistant Pseudomonas aeruginosa infection on patient outcomes. Exp. Rev. Pharmacoecon. Outcomes Res. 2010, 10, 441–451. [Google Scholar] [CrossRef]
- Harbottle, H.; Thakur, S.; Zhao, S.; White, D.G. Genetics of antimicrobial resistance. Anim. Biotechnol. 2006, 17, 111–124. [Google Scholar] [CrossRef]
- Coates, A.; Hu, Y.; Bax, R.; Page, C. The future challenges facing the development of new antimicrobial drugs. Nat. Rev. Drug Discov. 2002, 1, 895–910. [Google Scholar] [CrossRef]
- Al-Hussaini, R.; Mahasneh, A.M. Antimicrobial and antiquorum sensing activity of different parts of Laurus. nobilis L. extracts. Jordan Med. J. 2009, 43, 286–298. [Google Scholar]
- Adonizio, A.; Kong, K.; Mathee, K. Inhibition of quorum sensing-controlled virulence factor production in Pseudomonas aeruginosa by south Florida plant extracts. Antimicrob. Agents Chemother. 2008, 52, 198–203. [Google Scholar] [CrossRef]
- Hentzer, M.; Givskov, M. Pharmacological inhibition of quorum sensing for the treatment of chronic bacterial infections. J. Clin. Invest. 2003, 112, 1300–1307. [Google Scholar] [CrossRef]
- Stojković, D.; Reis, F.S.; Glamočlija, J.; Ćirić, A.; Barros, L.; van Griensven, L.J.L.D.; Ferreira, I.C.F.R.; Soković, M. Cultivated strains of Agaricus. bisporus and A. brasiliensis: Chemical characterization and evaluation of antioxidant and antimicrobial properties for final healthy product—Natural preservatives in yoghurt. Food Funct. 2014. Submitted. [Google Scholar]
- Koh, C.K.; Sam, C.K.; Yin, W.F.; Tan, L.Y.; Krishnan, T.; Chong, Y.M.; Chan, K.G. Plant-Derived Natural Products as Sources of Anti-Quorum Sensing Compounds. Sensors 2013, 13, 6217–6228. [Google Scholar] [CrossRef]
- Uroz, S.; Heinonsalo, J. Degradation of N-acyl homoserine lactone quorum sensing signal molecules by forest root-associated fungi. FEMS Microbiol. Ecol. 2008, 65, 271–278. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, W.; Wang, S.; Tian, B.; Zhang, S. Evaluation of quorum-sensing activity of fermentation metabolites from different strains of a medicinal mushroom, Phellinus igniarius. Chemotherapy 2012, 58, 195–199. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, W.; Tian, B.; Liu, H.; Ning, S. Inhibition of quorum sensing in the opportunistic pathogenic bacterium Chromobacterium viuolaceum by an extract from fruiting bodies of linghzhi or reishi medicinal mushroom Ganoderma lucidum W.Curt.:Fr.) P.Karst (higher basidiomycetes). Int. J. Med. MushroomsT 2011, 13, 559–564. [Google Scholar] [CrossRef]
- Dietrich, L.E.P.; Price-Whelan, A.; Petersen, A.; Whitely, M.; Newman, D.K. The phenazine pyocyanin is a terminal signalling factor in the quorum sensing network of Pseudomonas aeruginosa. Mol. Microbiol. 2006, 61, 1308–1321. [Google Scholar] [CrossRef]
- Rasmussen, T.B.; Givskov, M. Quorum sensing inhibitors: A bargain of effects. Microbiology 2006, 152, 895–904. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: A genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Kolter, R. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol. Microbiol. 1998, 30, 295–304. [Google Scholar]
- Henrichsen, J. Bacterial surface translocation: A survey and a classification. Bacteriol. Rev. 1972, 36, 478–503. [Google Scholar]
- Venturini, M.E.; Reyes, J.E.; Rivera, C.S.; Oria, R.; Blanco, D. Microbiological quality and safety of fresh cultivated and wild mushrooms commercialized in Spain. Food Microbiol. 2011, 28, 1492–1498. [Google Scholar] [CrossRef]
- Andolfi, A.; Cimmino, A.; Cantore, P.L.; Iacobellis, N.S.; Evidente, A. Bioactive and structural metabolites of Pseudomonas and Burkholderia. species causal agents of cultivated mushrooms diseases. Perspect. Med. Chem. 2008, 2, 81–112. [Google Scholar]
- Drenkard, E.; Ausubel, F.M. Pseudomonas biofilm formation and antibiotic resistance are linked to phenotypic variation. Nature 2002, 416, 740–743. [Google Scholar] [CrossRef]
- Spoering, A.L.; Lewis, K. Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing of antimicrobials. J. Bacteriol. 2001, 183, 6746–6751. [Google Scholar] [CrossRef]
- O’May, V.; Tufenkji, N. The Swarming Motility of Pseudomonas aeruginosa Is Blocked by Cranberry Proanthocyanidins and Other Tannin-Containing Materials. Appl. Environ. Microbiol. 2011, 77, 3061–3067. [Google Scholar] [CrossRef]
- Sandy, S.M.; Foong-Yee, T. Anti-quorum sensing and antimicrobial activities of some traditional Chinese medicinal plants commonly used in South-East Asia. Malays. J. Microb. 2012, 8, 11–20. [Google Scholar]
- Essar, D.W.; Eberly, L.; Hadero, A.; Crawford, I. Identification and characterization of genes for a second anthranilate synthase in Pseudomonas aeruginosa: Interchangeability of the two anthranilate synthases and evolutionary implications. J. Bacteriol. 1990, 172, 884–900. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Soković, M.; Ćirić, A.; Glamočlija, J.; Nikolić, M.; Van Griensven, L.J.L.D. Agaricus Blazei Hot Water Extract Shows Anti Quorum Sensing Activity in the Nosocomial Human Pathogen Pseudomonas Aeruginosa. Molecules 2014, 19, 4189-4199. https://doi.org/10.3390/molecules19044189
Soković M, Ćirić A, Glamočlija J, Nikolić M, Van Griensven LJLD. Agaricus Blazei Hot Water Extract Shows Anti Quorum Sensing Activity in the Nosocomial Human Pathogen Pseudomonas Aeruginosa. Molecules. 2014; 19(4):4189-4199. https://doi.org/10.3390/molecules19044189
Chicago/Turabian StyleSoković, Marina, Ana Ćirić, Jasmina Glamočlija, Miloš Nikolić, and Leo J. L. D. Van Griensven. 2014. "Agaricus Blazei Hot Water Extract Shows Anti Quorum Sensing Activity in the Nosocomial Human Pathogen Pseudomonas Aeruginosa" Molecules 19, no. 4: 4189-4199. https://doi.org/10.3390/molecules19044189







