Antioxidant Activity of Co-Products from Guava, Mango and Barbados Cherry Produced in the Brazilian Northeast
Abstract
:1. Introduction
2. Results and Discussion
2.1. Yield and Total Extractable Phenolic Content (TEP)
| Sample | Yield (%) | mg GAE/gof Extract | mg GAE/100 g of dc ** |
|---|---|---|---|
| Guava | 7.8 ± 0.29 b | 24.15 ± 1.59 b | 188.40 ± 12.38 c |
| Mango | 11.0 ± 0.31 a | 44.18 ± 1.73 a | 485.93 ± 19.08 a |
| Barbados cherry | 5.5 ± 0.21 c | 49.21 ± 3.70 a | 270.68 ± 20.37 b |
2.2. Radical-Scavenging Activity of the 2,2 '-Diphenyl-β-Picrylhydrazyl Radical (DPPH)
| Method | Guava | Mango | B. cherry | BHT |
|---|---|---|---|---|
| DPPH (EC 50 µg/mL) | 169 ± 6.00 a | 36 ± 0.58 c | 44 ± 4.36 c | 67 ± 3.06 b |
| DPPH (anti-radical efficiency) | 5.92 ± 0.21 d | 27.52 ± 0.43 a | 22.87 ± 2.15 b | 14.80 ± 0.66 c |
| FRAP (mmol Fe(II)/g extract) | 5.15 ± 0.57 c | 26.80 ± 0.63 a | 24.90 ± 0.17 b | – |
| FRAP (mmol Fe(II)/100 g dc **) | 40.15 ± 4.40 c | 273.86 ± 1.83 a | 147.44 ± 3.50 b | – |
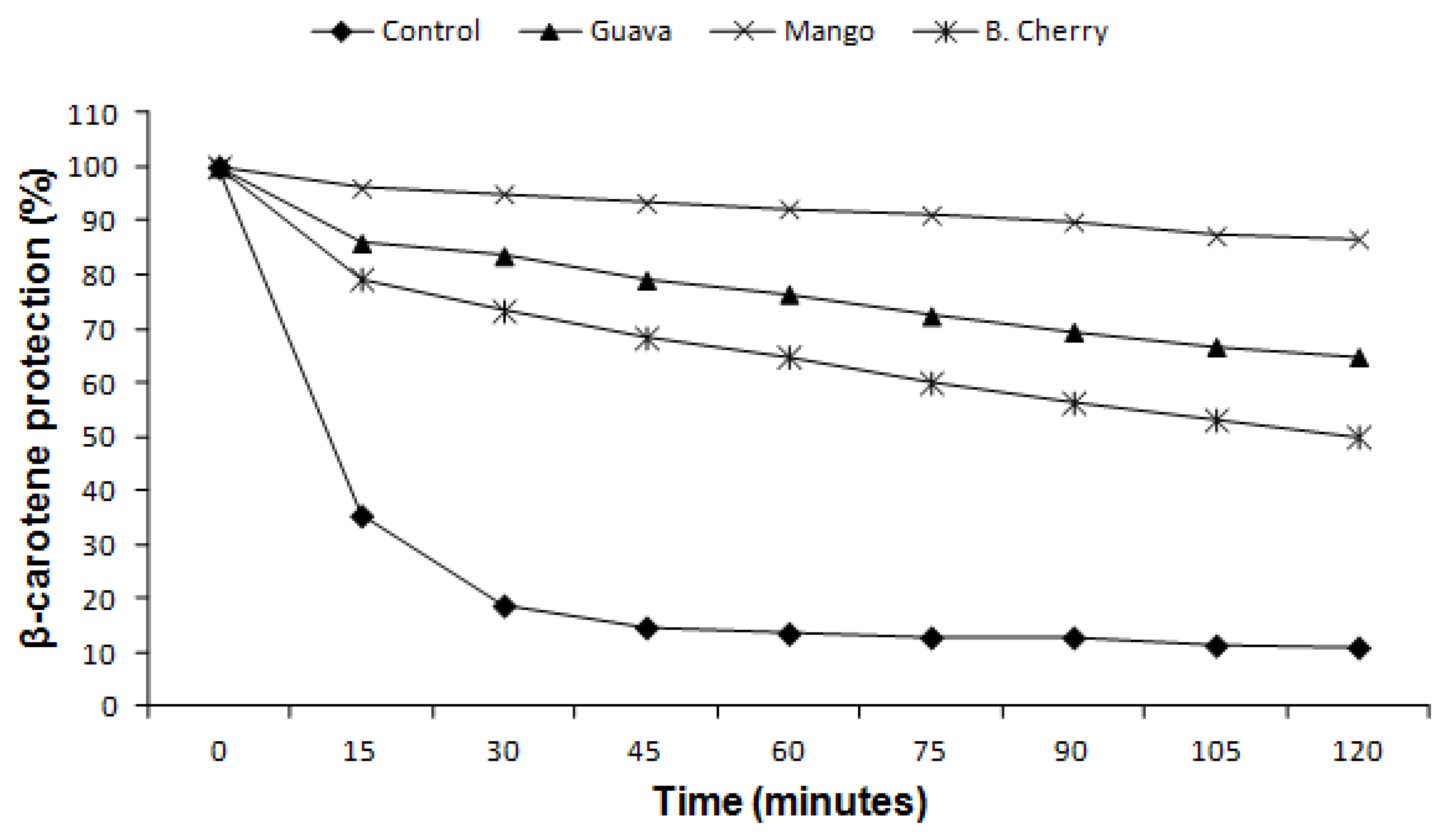
| β-Carotene bleaching (OI %) | 60.64 ± 0.24 b | 88.91 ± 0.32 a | 39.36 ± 0.24 c | – |
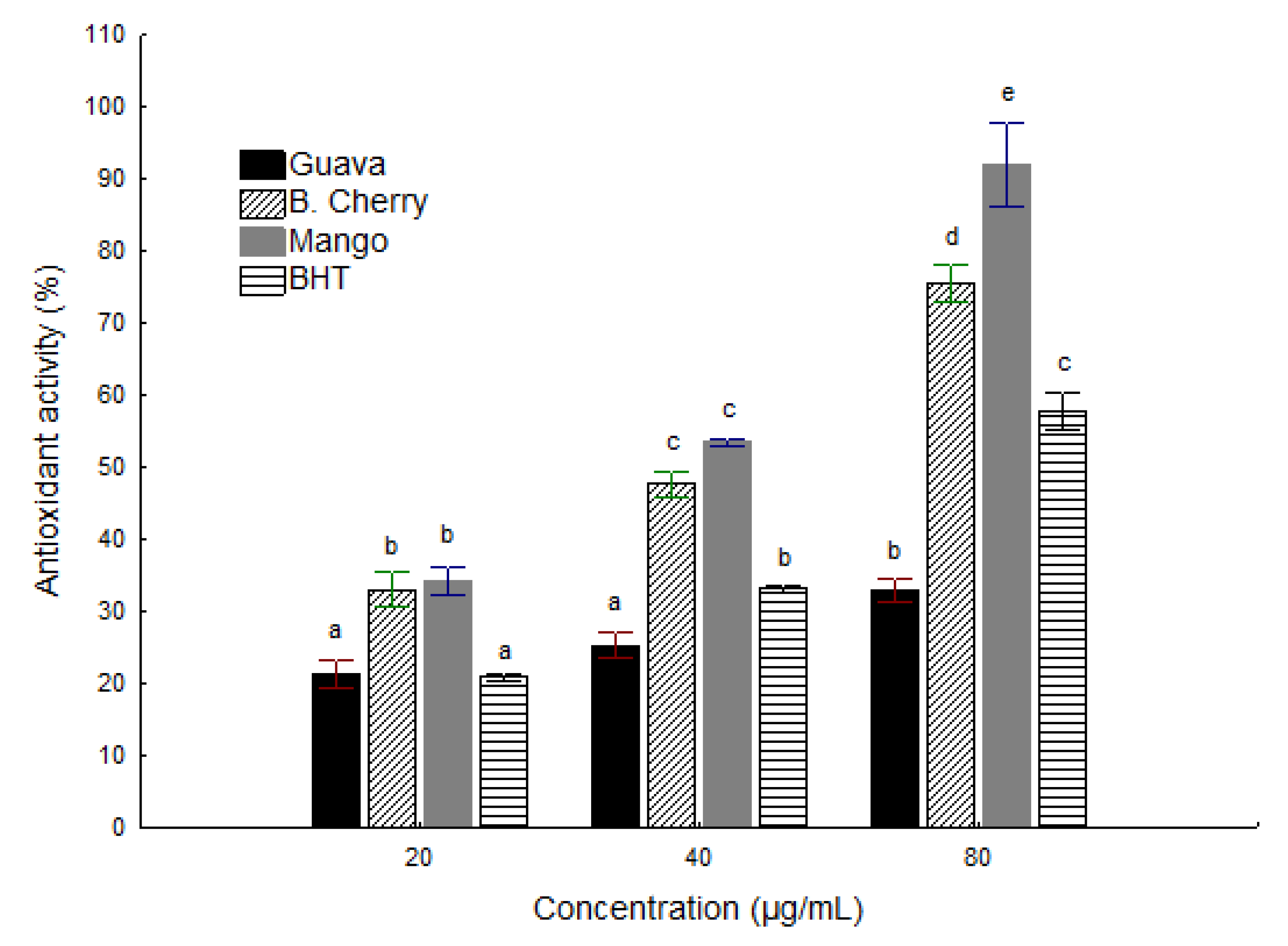
2.3. Ferric Reducing Antioxidant Power (FRAP)
2.4. β-Carotene Bleaching Method
2.5. Correlation between Total Phenolic Compounds and Antioxidant Activity using DPPH, FRAP and β-Carotene/Linoleic Acid Methods
3. Experimental
3.1. Reagents
3.2. Sample Preparation and Extraction
3.3. Determination of Total Extractable Phenolic Content
3.4. Radical-Scavenging Activity of the 2,2'-Diphenyl-β-Picrylhydrazyl Radical (DPPH) Assay
3.5. Ferric Reducing Antioxidant Power (FRAP) Assay
3.6. β-Carotene Bleaching Method
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- FAOSTAT. Available online: http://faostat.fao.org/site/339/defut.aspx (accessed on 1 March 2014).
- Sousa, M.S.; Vieira, L.; Lima, A.D. Total phenolics and in vitro antioxidant capacity of tropical fruit pulp wastes. Braz. J. Food Techol. 2011, 14, 202–210. [Google Scholar] [CrossRef]
- Oliveira, A.C.; Valetin, I.B.; Silva, C.A.; Bechara, E.J.; Barros, M.P.; Mano, C.M.; Goulart, M.O. Total phenolic content and free radical scavenging activities of methanolic. Food Chem. 2009, 115, 469–475. [Google Scholar] [CrossRef]
- Babbar, N.; Oberai, S.H.; Uppal, D.S.; Patil, R.T. Total phenolic content and antioxidant capacity of extracts obtained from six important fruit residues. Food Res. Int. 2011, 44, 391–396. [Google Scholar] [CrossRef]
- Contreras-Calderón, J.; Calderón-Jaimes, L.; Guerra-Hermández, E.; García-Villanova, B. Antioxidant capacity, phenolic content and vitamin C in pulp, peel and seed from 24 exotic fruits from Colombia. Food Res. Int. 2011, 44, 2047–2053. [Google Scholar] [CrossRef]
- Makris, D.P.; Boskou, G.; Andrikopoulos, N.K. Recoveredy of antioxidant phenolics from white vinification solid by-products employing water/ethanol mixtures. Bioresour. Technol. 2007, 1, 2963–2967. [Google Scholar]
- Ajila, C.M.; Bhat, S.G.; Prasasa Rao, U.J. Valuable components of raw and ripe peels from two Indian mango varieties. Food Chem. 2007, 102, 1006–1011. [Google Scholar] [CrossRef]
- Kedare, S.B.; Sing, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef]
- Niki, E. Assessment of Antioxidant Capacity in vitro and in vivi. Free Rad. Biol. Med. 2010, 49, 503–515. [Google Scholar] [CrossRef]
- Abidille, M.H.; Singh, R.P.; Jayaprakasha, G.K.; Jena, B.S. Antioxidant activity of the extracts from Dillenia indica fruits. Food Chem. 2005, 90, 891–896. [Google Scholar] [CrossRef]
- Shahidi, F.; Wanasundara, P.K. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr. 1992, 81, 215–217. [Google Scholar]
- Fresco, P.; Borges, F.; Diniz, C.; Marques, M.P. New insights on the anticancer properties of dietary polyphenols. Med. Res. Rev. 2006, 22, 747–766. [Google Scholar]
- Kim, H.; Moon, J.Y.; Kim, H.; Lee, D.-S.; Cho, M.; Choi, H.-K. Antioxidant and antiproliferative activities of mango (Mangifera indica L.) flesh and peel. Food Chem. 2010, 121, 429–436. [Google Scholar] [CrossRef]
- Kuskoski, E.M.; Asuero, G.A.; Troncoso, A.M.; Mancini-Filho, J.; Fett, R. Aplicación de diverso métodos químicos para determinar actividad antioxidante em pulpa de frutos. Food Sci. Technol. 2005, 25, 726–732. [Google Scholar]
- Moure, A.; Cruz, J.M.; Franco, D.; Domíngues, J.M.; Sineiro, J.; Domíngues, H. Natural antioxidants from residual sources. Food Chem. 2001, 72, 145–171. [Google Scholar] [CrossRef]
- Muller, L.; Gnoyke, S.; Popken, A.M.; Bohm, V. Antioxidant capacity and related parameters of different fruit formulations. LWT-Food Sci. Technol. 2010, 42, 992–999. [Google Scholar]
- Rufino, M.D.; Alves, R.E.; Brito, E.S.; Pérez-Jiménez, J.; Saura-Calixto, F.; Mancini-Filho, J. Bioactive Compounds and antioxidant capacities of 18 non-tradicional tropical fruits from Brazil. Food Chem. 2010, 121, 996–1002. [Google Scholar] [CrossRef]
- Alves, R.E.; Brito, E.A.; Rufino, M.S.; Sampaio, C.G. Antioxidant activity measurement in tropical fruits: A case study with acerola. Acta Horticul. 2008, 773, 299–305. [Google Scholar]
- Hassimoto, N.M.; Genovese, I.S.; Lajolo, F.M. Antioxidant activity of dietary fruits, vegetables and commercial frozen fruit pulps. J. Agric. Food Chem. 2005, 46, 2928–2935. [Google Scholar] [CrossRef]
- Santos, N.A.; Cordeiro, A.M.T.M.; Damasceno, S.S.; Aguiar, R.T.; Rosenhaim, R.; Carvalho Filho, J.R.; Santos, I.M.G. Commercial antioxidants and thermal stability evaluations. Fuel 2012, 97, 638–643. [Google Scholar] [CrossRef]
- How, W.C.; Lin, R.D.; Chang, K.T.; Huang, Y.T.; Cho, C.H.; Chen, C.H. Free radical scavenging activity of Taiwanes native plants. Phytomedicine 2003, 10, 170–175. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vit. 1977, 28, 49–55. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 80, 25–30. [Google Scholar]
- Pulido, R.; Bravo, L.; Fulgencio, S.-C. Antioxidant activity of dietary polyphenols As determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef]
- Miller, H.E. A simplified method for the evaluation of antioxidants. J. Am. Oil Chem. Soc. 1971, 48, 91. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the co-products assayed are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Araújo, K.L.G.V.; Magnani, M.; Nascimento, J.A.; Souza, A.L.; Epaminondas, P.S.; Souza, A.L.; Queiroz, N.; Souza, A.G. Antioxidant Activity of Co-Products from Guava, Mango and Barbados Cherry Produced in the Brazilian Northeast. Molecules 2014, 19, 3110-3119. https://doi.org/10.3390/molecules19033110
Araújo KLGV, Magnani M, Nascimento JA, Souza AL, Epaminondas PS, Souza AL, Queiroz N, Souza AG. Antioxidant Activity of Co-Products from Guava, Mango and Barbados Cherry Produced in the Brazilian Northeast. Molecules. 2014; 19(3):3110-3119. https://doi.org/10.3390/molecules19033110
Chicago/Turabian StyleAraújo, Kassandra L. G. V., Marciane Magnani, Jaqueline A. Nascimento, Alline L. Souza, Poliana S. Epaminondas, Antônia L. Souza, Neide Queiroz, and Antonio G. Souza. 2014. "Antioxidant Activity of Co-Products from Guava, Mango and Barbados Cherry Produced in the Brazilian Northeast" Molecules 19, no. 3: 3110-3119. https://doi.org/10.3390/molecules19033110
APA StyleAraújo, K. L. G. V., Magnani, M., Nascimento, J. A., Souza, A. L., Epaminondas, P. S., Souza, A. L., Queiroz, N., & Souza, A. G. (2014). Antioxidant Activity of Co-Products from Guava, Mango and Barbados Cherry Produced in the Brazilian Northeast. Molecules, 19(3), 3110-3119. https://doi.org/10.3390/molecules19033110




