Determination of Pesticide Residues in Honeybees using Modified QUEChERS Sample Work-Up and Liquid Chromatography-Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction Procedure
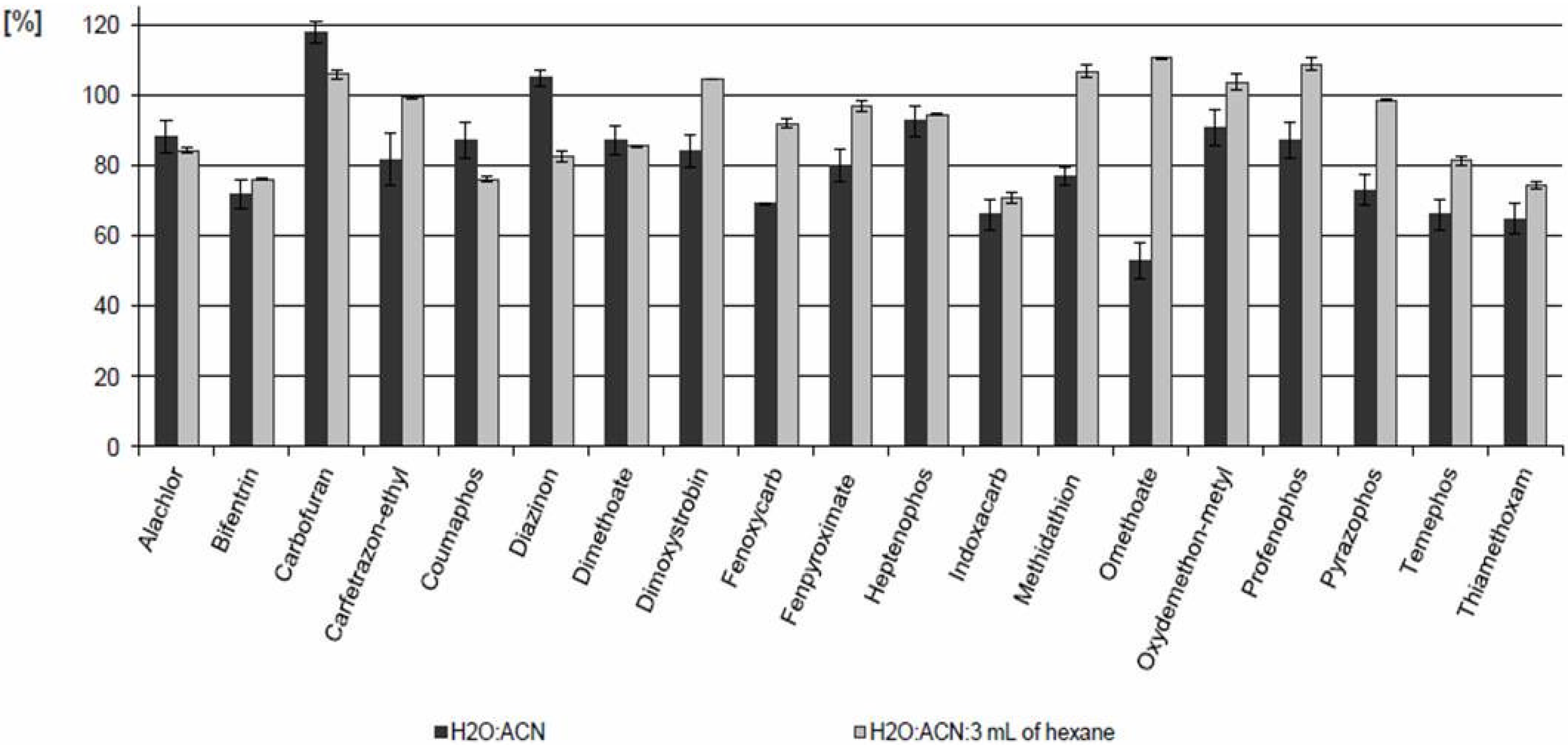
2.2. Method Performance
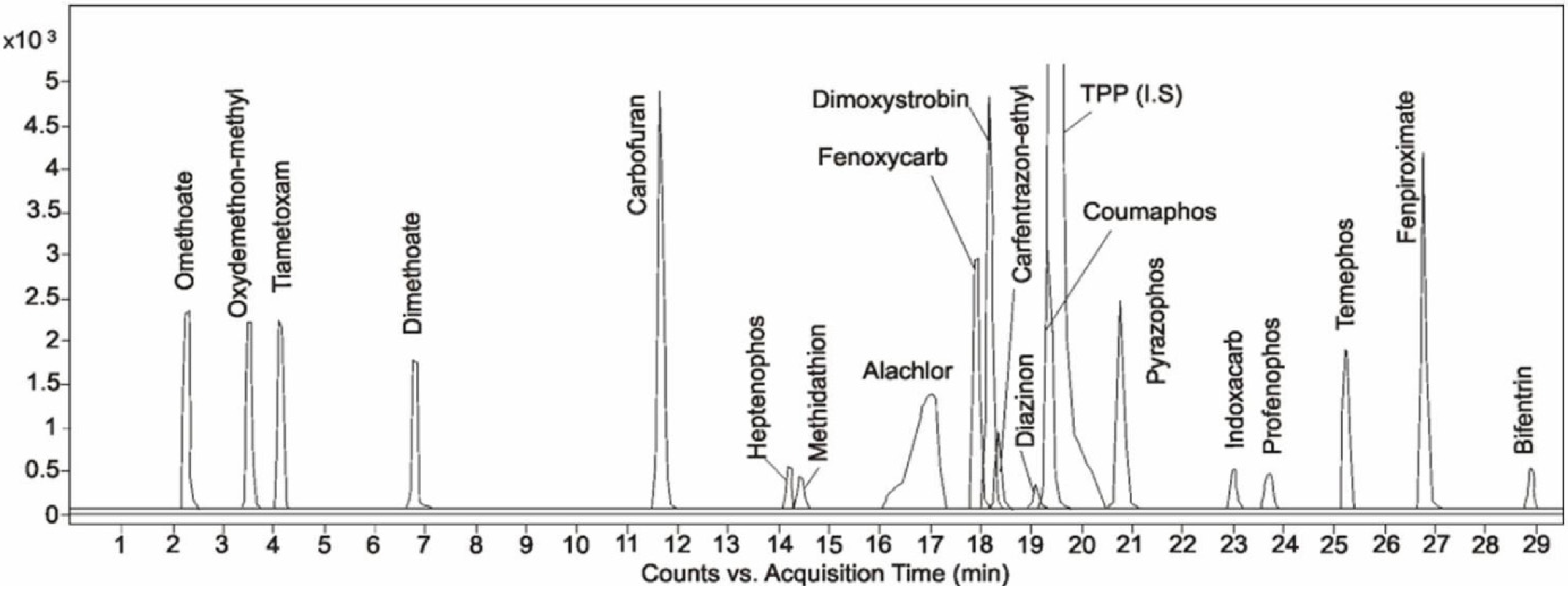
| Analyte | Range [ng/g] | Recovery [%] ± RSD [%] n = 5 | LOD [ng/g] | LOQ [ng/g] | |
|---|---|---|---|---|---|
| LOQ | 3LOQ | ||||
| Alachlor | 75/450 | 80.5 ± 5.4 | 84.34 ± 0.76 | 25 | 75 |
| Bifentrin | 4.05/24.40 | 85 ± 17 | 76.08 ± 0.22 | 1.3 | 4.1 |
| Carbofuran | 3.65/22.00 | 106 ± 11 | 106.1 ± 1.2 | 1.2 | 3.6 |
| Carfetrazon-ethyl | 3.78/22.60 | 79.0 ± 9.0 | 99.48 ± 0.45 | 1.3 | 3.8 |
| Coumaphos | 4.95/29.80 | 80.4 ± 7.7 | 76.18 ± 0.95 | 1.6 | 4.9 |
| Diazinon | 4.02/24.20 | 83 ± 14 | 82.7 ± 1.6 | 1.3 | 4.0 |
| Dimethoate | 3.51/21.00 | 85.3 ± 7.2 | 85.63 ± 0.28 | 1.2 | 3.5 |
| Dimoxystrobin | 3.87/23.20 | 76.8 ± 5.7 | 104.45 ± 0.40 | 1.3 | 3.9 |
| Fenoxycarb | 3.69/22.20 | 70.8 ± 6.3 | 91.9 ± 1.3 | 1.2 | 3.7 |
| Fenpiroxymate | 3.69/22.20 | 83 ± 10 | 97.0 ± 1.5 | 1.2 | 3.7 |
| Heptenophos | 2.97/17.80 | 93.0 ± 8.0 | 94.63 ± 0.10 | 1.0 | 3.0 |
| Indoxacarb | 3.66/22.00 | 70.8 ± 5.1 | 70.8 ± 1.6 | 1.2 | 3.7 |
| Methidathion | 4.53/27.20 | 74.6 ± 6.7 | 106.8 ± 1.8 | 1.5 | 4.5 |
| Omethoate | 3.63/21.80 | 71.6 ± 2.1 | 110.58 ± 0.14 | 1.2 | 3.6 |
| Oxydemeton-methyl | 3.63/21.80 | 87.3 ± 9.3 | 103.9 ± 2.2 | 1.2 | 3.6 |
| Profenophos | 3.42/20.60 | 80.1 ± 5.7 | 109.1 ± 1.7 | 1.1 | 3.4 |
| Pyrazophos | 3.87/23.20 | 70.4 ± 1.8 | 98.69 ± 0.32 | 1.3 | 3.9 |
| Temephos | 3.69/22.20 | 72.70 ± 0.36 | 81.5 ± 1.2 | 1.2 | 3.7 |
| Thiamethoxam | 3.42/20.60 | 70.1 ± 2.0 | 74.38 ± 0.92 | 1.1 | 3.4 |
2.3. Application to Real Samples
| Pesticides | Number of samples | The districts in Pomerania (Poland) | No. of samples above LOD (%) | Min level [ng/g] (RSD) | Max level [ng/g] (RSD) |
|---|---|---|---|---|---|
| Alachlor | 11–15 | Kartuzy, Tczew | 5 (26.3%) | >LOD | 95.0 (8.4) |
| Bifenthrin | 1, 5, 6, 7, 10–14, 19 | Gdansk, Kartuzy, Tczew | 10 (52.6%) | nd | <LOQ |
| Carbofuran | 1, 11 | Gdańsk, Kartuzy | 2 (10.5%) | nd | <LOQ |
| Carfentrazon-ethyl | 1, 11, 14, 15 | Gdańsk, Kartuzy, Tczew | 4 (21%) | >LOD | 18.1 (1.3) |
| Coumaphos | 11 | Kartuzy | 1 (5.3%) | nd | <LOQ |
| Diazinon | 1, 5, 11–13, 15 | Gdańsk, Kartuzy, Tczew | 6 (31.6%) | >LOD | 13.3 (1.4) |
| Dimethoate | 1, 8, 13, 14, 16 | Gdansk, Kartuzy, Tczew | 5 (26.3%) | >LOD | 20.5 (0.4) |
| Dimoxystrobin | 11 | Kartuzy | 1 (5.3%) | nd | <LOQ |
| Fenoxycarb | 11 | Kartuzy | 1 (5.3%) | nd | 15.0 (0.2) |
| Fenpyroximate | 1, 11 | Gdansk, Kartuzy | 2 (10.5%) | nd | <LOQ |
| Heptenophos | 1, 2, 3, 6, 8–16 | Gdańsk, Kartuzy, Tczew | 13 (68.4%) | >LOD | 18.5 (0.6) |
| Indoxacarb | 11, 12 | Kartuzy | 2 (10.5%) | >LOD | 11.8 (2.3) |
| Methidathion | 1, 5, 6, 10, 11, 13–16 | Gdańsk, Kartuzy, Tczew | 9 (47.4%) | >LOD | 22.4 (1.9) |
| Omethoate | 11, 14, 16 | Kartuzy, Tczew | 3 (15.8%) | >LOD | 15.8 (2.9) |
| Oxydemeton-methyl | 11 | Kartuzy | 1 (5.3%) | nd | 18.4 (1.5) |
| Profenophos | 1, 11, 12 | Gdańsk, Kartuzy | 3 (15.8%) | >LOD | 7.6 (2.7) |
| Pyrazophos | 2, 5, 6, 10, 11, 15 | Gdańsk, Kartuzy, Tczew | 6 (31.6%) | >LOD | 14.3 (0.6) |
| Temephos | 1, 2, 13 | Gdańsk, Kartuzy | 3 (15.8%) | nd | <LOQ |
| Thiamethoxam | 11, 14 | Kartuzy, Tczew | 2 (10.5%) | >LOD | 4.1 (0.7) |
3. Experimental
3.1. Materials
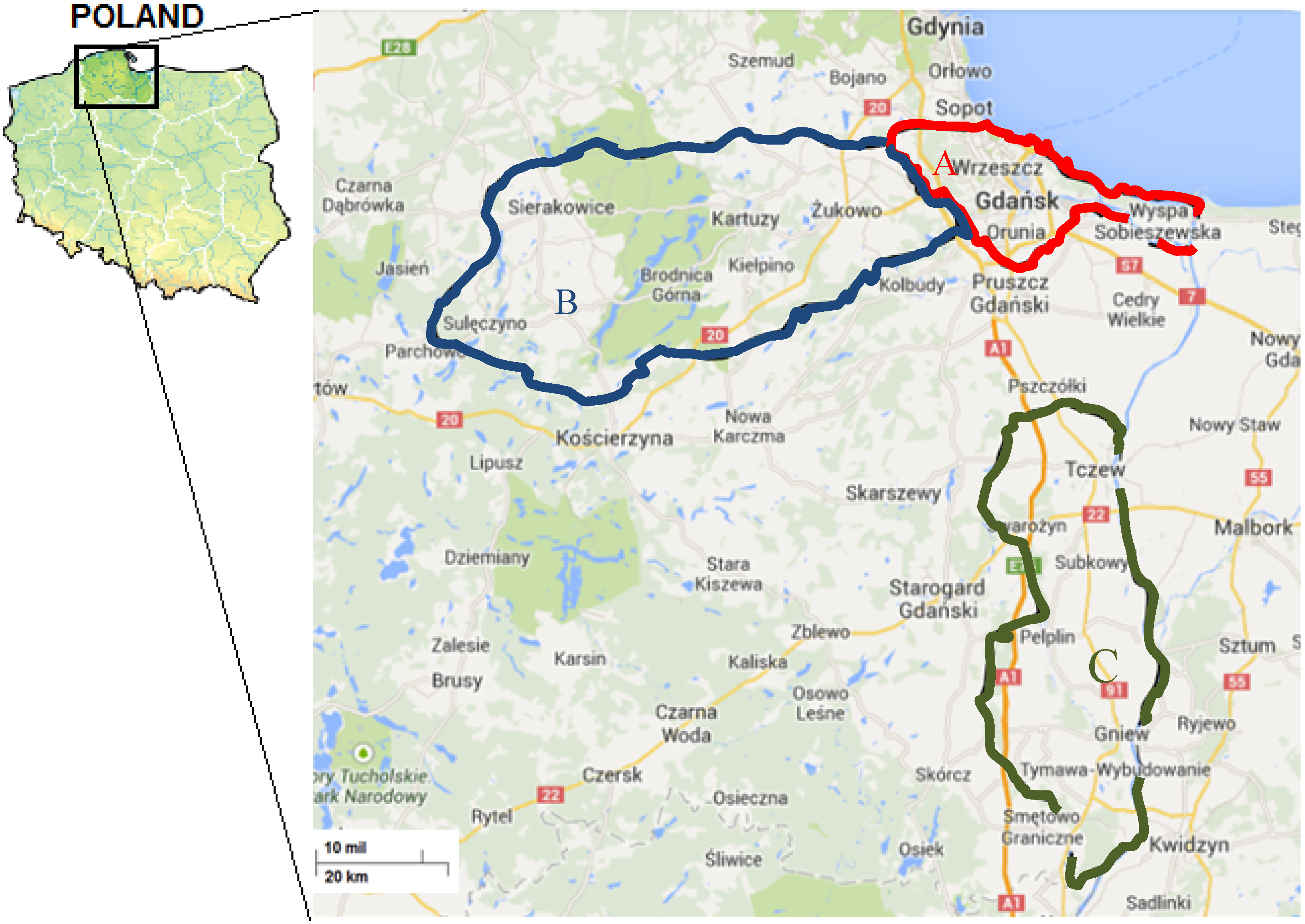
3.2. Sample Collection
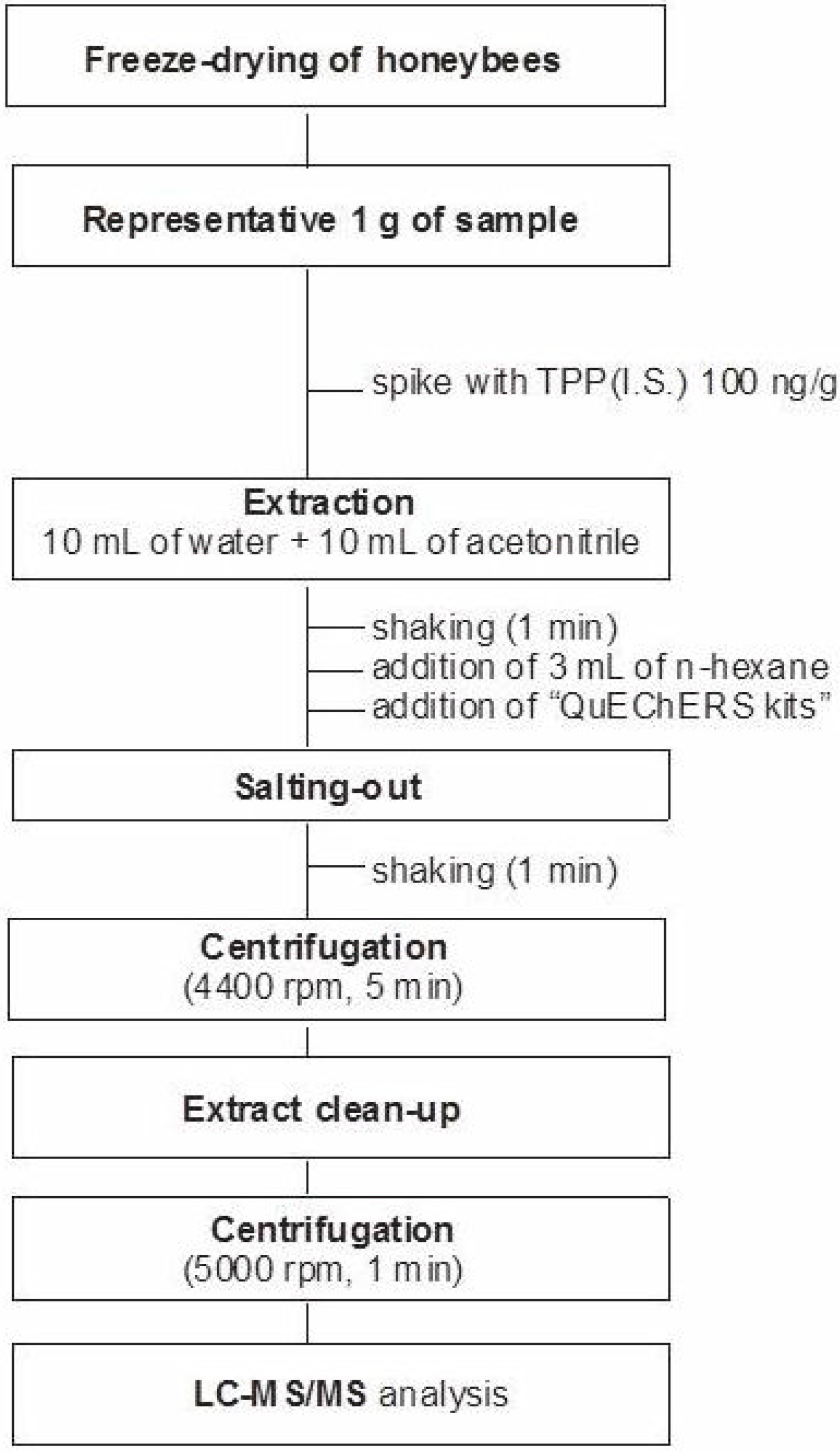
3.3. Sample Preparation
3.4. LC–MS/MS Analysis
| Compound | MW [g/mol] | Precursor ions (m/z) | Product ions (m/z) | tr [min] | Fragmentor potential [V] | Collision energy (CE) [V] | |
|---|---|---|---|---|---|---|---|
| Quantifier ion | Qualifier ion | ||||||
| Alachlor | 269.1 | 270.1 | 238.0 | 162.1 | 17.01 | 103 | 13 |
| 117.0 | 61 | ||||||
| Bifentrin | 422.1 | 440.2 | 181.0 | 165.0 | 28.94 | 103 | 77 |
| 115.0 | 141 | ||||||
| Carbofuran | 221.1 | 222.1 | 123.0 | 165.0 | 11.63 | 103 | 9 |
| 51.0 | 69 | ||||||
| Carfetrazon-ethyl | 411.1 | 412.1 | 345.9 | 383.9 | 18.42 | 152 | 9 |
| 365.9 | 13 | ||||||
| Coumaphos | 362.0 | 363.0 | 226.9 | 334.9 | 19.43 | 152 | 9 |
| 306.9 | 13 | ||||||
| Diazinon | 304.1 | 305.1 | 169.1 | 153.1 | 19.19 | 103 | 17 |
| 96.9 | 33 | ||||||
| Dimethoate | 229.0 | 230.0 | 124.9 | 198.9 | 6.70 | 54 | 5 |
| 170.9 | 9 | ||||||
| Dimoxystrobin | 326.2 | 327.2 | 205.0 | 116.0 | 18.24 | 103 | 17 |
| 58.1 | 29 | ||||||
| Fenoxycarb | 301.1 | 302.1 | 88.0 | 256.0 | 17.98 | 103 | 9 |
| 116.0 | 9 | ||||||
| Fenpyroximate | 421.2 | 422.2 | 366.0 | 107.0 | 26.84 | 152 | 61 |
| 77.0 | 101 | ||||||
| Heptenophos | 250.0 | 251.0 | 127.0 | 125.0 | 14.17 | 103 | 9 |
| 89.0 | 33 | ||||||
| Indoxacarb | 527.1 | 528.1 | 149.9 | 292.9 | 23.13 | 152 | 9 |
| 248.9 | 9 | ||||||
| Methidathion | 302.0 | 303.0 | 145 | 85.1 | 14.42 | 54 | 13 |
| 58.1 | 29 | ||||||
| Omethoate | 213.0 | 214.0 | 124.9 | 182.9 | 2.19 | 103 | 9 |
| 154.9 | 9 | ||||||
| Oxydemethon-methyl | 246.0 | 247.0 | 168.9 | 124.9 | 3.43 | 103 | 17 |
| 109.0 | 25 | ||||||
| Profenophos | 372.0 | 373.0 | 302.8 | 344.8 | 23.79 | 103 | 9 |
| 128.0 | 49 | ||||||
| Pyrazophos | 373.1 | 374.1 | 222.0 | 238.0 | 20.90 | 152 | 17 |
| 148.0 | 53 | ||||||
| Temephos | 466.0 | 467.0 | 124.9 | 418.9 | 25.31 | 152 | 13 |
| 404.9 | 9 | ||||||
| Thiamethoxam | 291.0 | 292.0 | 211.0 | 181.0 | 4.05 | 103 | 17 |
| 131.9 | 17 | ||||||
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Rissato, S.R.; Galhiane, M.S.; de Almeida, M.V.; Gerenutti, M.; Apon, B.M. Multiresidue determination of pesticides in honey samples by gas chromatography–mass spectrometry and application in environmental contamination. Food Chem. 2007, 101, 1719–1726. [Google Scholar] [CrossRef]
- Rial-Otero, R.; Gaspar, E.M.; Moura, I.; Capelo, J.L. Chromatographic-based methods for pesticide determination in honey: An overview. Talanta 2007, 71, 503–514. [Google Scholar] [CrossRef]
- Kujawski, M.; Namieśnik, J. Challenges in preparing honey samples for chromatographic determination of contaminants and trace residues. TrAC Trends Anal. Chem. 2008, 27, 785–793. [Google Scholar] [CrossRef]
- Thompson, H.M. Risk assessment for honey bees and pesticides—Recent developments and ‘new issues’. Pest Manag. Sci. 2010, 66, 1157–1162. [Google Scholar] [CrossRef]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; van Engelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS One 2010, 5, e9754. [Google Scholar] [CrossRef]
- Bargańska, Ż.; Namieśnik, J. Pesticide analysis of bee and bee product samples. Crit. Rev. Anal. Chem. 2010, 40, 159–171. [Google Scholar] [CrossRef]
- Ghini, S.; Fernández, M.; Picó, Y.; Marín, R.; Fini, F.; Mañes, J.; Girotti, S. Occurrence and distribution of pesticides in the province of Bologna, Italy, using Honeybees as bioindicators. Arch. Environ. Contam. Toxicol. 2004, 47, 479–488. [Google Scholar] [CrossRef]
- Amorena, M.; Visciano, P.; Giacomelli, A.; Marinelli, E.; Sabatini, A.G.; Medrzycki, P.; Persano Oddo, L.; de Pace, F.M.; Belligoli, P.; di Serafino, G.; et al. Monitoring of levels of polycyclic aromatic hydrocarbons in bees caught from beekeeping: Remark 1. Vet. Res. Commun. 2009, 33, S165–S167. [Google Scholar] [CrossRef]
- Kamel, A. Refined Methodology for the Determination of Neonicotinoid Pesticides and Their Metabolites in Honey Bees and Bee Products by Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS). J. Agric. Food Chem. 2010, 58, 5926–5931. [Google Scholar] [CrossRef]
- Lambert, O.; Veyrand, B.; Durand, S.; Marchand, P.; le Bizec, B.; Piroux, M.; Puyo, S.; Thorin, C.; Delbac, F.; Pouliquen, H. Polycyclic aromatic hydrocarbons: Bees, honey and pollen as sentinels for environmental chemical contaminants. Chemosphere 2012, 86, 98–104. [Google Scholar] [CrossRef]
- Van Engelsdorp, D.; Evans, J.D.; Donovall, L.; Mullin, C.; Frazier, M.; Frazier, J.; Tarpy, D.R.; Hayes, J., Jr.; Pettis, J.S. Entombed Pollen: A new condition in honey bee colonies associated with increased risk of colony mortality. J. Invertebr. Pathol. 2009, 101, 147–149. [Google Scholar] [CrossRef]
- Topolska, G.; Gajda, A.; Pohorecka, K.; Bober, A.; Kasprzak, S.; Skubida, M.; Semkiw, P. Winter colony losses in Poland. J. Apic. Res. 2010, 49, 126–128. [Google Scholar] [CrossRef]
- Osborne, J.L. Bumblebees and pesticides. Nature 2012, 491, 43–45. [Google Scholar] [CrossRef]
- Stokstad, E. Field research on bees raises concern about low-dose. Science 2012, 335, 1555. [Google Scholar] [CrossRef]
- Higes, M.; Martín-Hernández, R.; Martínez-Salvador, A.; Garrido-Bailón, A.; González-Porto, A.V.; Meana, A.; Bernal, J.L.; del Nozal, M.J.; Bernal, J. A preliminary study of the epidemiological factors related to honey bee colony loss in Spain. Environ. Microbiol. Rep. 2010, 2, 243–250. [Google Scholar] [CrossRef]
- Dainat, B.; van Engelsdrop, D.; Neumann, P. Colony collapse disorder in Europe. Environ. Toxicol. Rep. 2012, 4, 123–125. [Google Scholar]
- Core, A.; Runckel, C.; Ivers, J.; Quock, C.; Siapno, T.; DeNault, S.; Brown, B.; DeRisi, J.; Smith, C.D.; Hafernik, J. A new threat to honey bees, the parasitic phorid flyapocephalus borealis. PLoS One 2012, 7, e29639. [Google Scholar] [CrossRef]
- Martin, S.J. The role of Varroa and viral pathogens in the collapse of honeybee colonies: A modelling approach. J. Appl. Ecol. 2001, 38, 1082–1093. [Google Scholar] [CrossRef]
- Nazzi, F.; Brown, S.P.; Annoscia, D.; del Piccolo, F.; di Prisco, G.; Varricchio, P.; Vedova, G.D.; Cattonaro, F.; Caprio, E.; Pennacchio, F. Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honeybee colonies. PLoS One 2012, 8, e1002735. [Google Scholar]
- Sharpe, R.J.; Heyden, L.C. Honey bee colony collapse disorder is possibly caused by a dietary pyrethrum deficiency. Biosci. Hypothesis 2009, 2, 439–440. [Google Scholar] [CrossRef]
- Naug, D. Nutritional stress due to habitat loss may explain recent honeybee colony collapses. Biol. Conserv. 2009, 142, 2369–2372. [Google Scholar] [CrossRef]
- Gill, R.J.; Ramos-Rodriguez, O.; Raine, N.E. Combined pesticide exposure severely affects individual- and colony—Level traits in bees. Nature 2012, 491, 105–108. [Google Scholar] [CrossRef]
- Morzycka, B. Simple method for the determination of trace levels of pesticides in honeybees using matrix solid-phase dispersion and gas chromatography. J. Chromatogr. A 2002, 982, 267–273. [Google Scholar] [CrossRef]
- Fernández, M.; Picó, Y.; Mañes, J. Rapid screening of organophosphorus pesticides in honey and bees by liquid chromatography—Mass spectrometry. Chromatographia 2002, 56, 577–583. [Google Scholar] [CrossRef]
- Kristenson, E.M.; Shahmiri, S.; Slooten, C.J.; Vreuls, R.J.J.; Brinkman, U.A.T. Matrix solid-phase dispersion micro-extraction of pesticides from single insects with subsequent GC–MS analysis. Chromatographia 2004, 59, 315–320. [Google Scholar]
- Rossi, S.; Dalpero, A.P.; Ghini, S.; Colombo, R.; Sabatini, A.G.; Girotti, S. Multiresidual method for the gas chromatographic analysis of pesticides in honeybees cleaned by gel permeation chromatography. J. Chromatogr. A 2001, 905, 223–232. [Google Scholar]
- Charlton, A.J.A.; Jones, A. Determination of imidazole and triazole fungicide residues in honeybees using gas chromatography–mass spectrometry. J. Chromatogr. A 2007, 1141, 117–122. [Google Scholar]
- Anastassiades, M.; Lehotay, S.J.; Štajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “Dispersive Solid-Phase Extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar]
- Garcia-Reyes, J.F.; Ferrer, C.; Gómez-Ramos, J.M.; Molina-Diaz, A.; Fernández-Alba, A.R. Comprehensive screening of target, non-target and unknown pesticides in food by LC-TOF-MS. TrAC Trends Anal. Chem. 2007, 26, 828–841. [Google Scholar] [CrossRef]
- Lehotay, S.J.; de Kok, A.; Hiemstra, M.; van Bodegraven, P. Validation of a fast and easy method for the determination of residues from 229 pesticides in fruits and vegetables using gas and liquid chromatography and mass spectrometric detection. J. AOAC Int. 2005, 88, 595–614. [Google Scholar]
- Kmellár, B.; Fodor, P.; Pareja, L.; Ferrer, C.; Martínez, M.A.; Valverde, A.; Fernandez-Alba, A.R. Validation and uncertainty study of a comprehensive list of 160 pesticide residues in multi-class vegetables by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2008, 1215, 37–50. [Google Scholar]
- Lesueur, C.; Knittl, P.; Gartner, M.; Mentler, A.; Fuerhacker, M. Analysis of 140 pesticides from conventional farming foodstuff samples after extraction with the modified QuECheRS method. Food Control 2008, 19, 906–914. [Google Scholar] [CrossRef]
- Koesukwiwat, U.; Lehotay, S.J.; Miao, S.; Leepipatpiboon, N. High throughput analysis of 150 pesticides in fruits and vegetables using QuEChERS and low-pressure gas chromatography-Time-of-flight mass spectrometry. J. Chromatogr. A 2010, 1217, 6692–6703. [Google Scholar]
- Nocentini, M.; Focardi, C.; Fortini, M. Single laboratory validation and uncertainty estimation of multiresidues method for determination of pesticides in vegetables and fruits by using QuEChERS sample extraction and GC–MS (Abstract: P308-041). Toxicol. Lett. 2010, 196, S325. [Google Scholar] [CrossRef]
- Li, J.; Dong, F.; Xu, J.; Liu, X.; Li, Y.; Shan, W.; Zheng, Y. Enantioselective determination of triazole fungicide simeconazole in vegetables, fruits, and cereals using modified QuEChERS (quick, easy, cheap, effective, rugged and safe) coupled to gas chromatography/tandem mass spectrometry. Anal. Chim. Acta 2011, 702, 127–135. [Google Scholar] [CrossRef]
- Romero-González, R.; Garrido Frenich, A.; Martínez Vidal, J.L. Multiresidue method for fast determination of pesticides in fruit juices by ultra performance liquid chromatography coupled to tandem mass spectrometry. Talanta 2008, 76, 211–225. [Google Scholar] [CrossRef]
- Kolberg, D.I.; Prestes, O.D.; Adaime, M.B.; Zanella, R. Development of a fast multiresidue method for the determination of pesticides in dry samples (wheat grains, flour and bran) using QuEChERS based method and GC–MS. Food Chem. 2011, 125, 1436–1442. [Google Scholar] [CrossRef]
- Norli, H.R.; Christiansen, A.; Deribe, E. Application of QuEChERS method for extraction of selected persistent organic pollutants in fish tissue and analysis by gas chromatography mass spectrometry. J. Chromatogr. A 2011, 1218, 7234–7241. [Google Scholar]
- Nguyen, T.D.; Han, E.M.; Seo, M.S.; Kim, S.R.; Yun, M.Y.; Lee, D.M.; Lee, G.-H. A multi-residue method for the determination of 203 pesticides in rice paddies using gas chromatography/mass spectrometry. Anal. Chim. Acta 2008, 619, 67–74. [Google Scholar] [CrossRef]
- Pinto, C.G.; Laespada, M.E.F.; Martín, S.H.; Ferreira, A.M.C.; Pavón, J.L.P; Cordero, B.M. Simplified QuEChERS approach for the extraction of chlorinated compounds from soil samples. Talanta 2010, 81, 385–391. [Google Scholar] [CrossRef]
- García-Reyes, J.F.; Ferrer, C.; Gómez-Ramos, M.J.; Fernández-Alba, A.R.; García-Reyes, J.F.; Molina-Díaz, A. Determination of pesticide residues in olive oil and olives. TrAC Trends Anal. Chem. 2007, 26, 239–251. [Google Scholar]
- Wilkowska, A.; Biziuk, M. Determination of pesticide residues in food matrices using the QuEChERS methodology. Food Chem. 2009, 125, 803–812. [Google Scholar] [CrossRef]
- Blasco, C.; Vazquez-Roig, P.; Onghena, M.; Masia, A.; Picó, Y. Analysis of insecticides in honey by liquid chromatography–ion trap-mass spectrometry: Comparison of different extraction procedures. J. Chromatogr. A 2011, 1218, 4892–4901. [Google Scholar] [CrossRef]
- Wiest, L.; Buleté, A.; Giroud, B.; Fratta, C.; Amic, S.; Lambert, O.; Pouliquen, H.; Arnaudguilhem, C. Multi-residue analysis of 80 environmental contaminants in honeys, honeybees and pollens by one extraction procedure followed by liquid and gas chromatography coupled with mass spectrometric detection. J. Chromatogr. A 2011, 1218, 5743–5786. [Google Scholar] [CrossRef]
- Walorczyk, S.; Gnusowski, B. Development and validation of a multi-residue method for the determination of pesticides in honeybees using acetonitrile-based extraction and gas chromatography-tandem quadrupole mass spectrometry. J. Chromatogr. A 2009, 1216, 6522–6531. [Google Scholar] [CrossRef]
- Tapparo, A.; Giorio, C.; Soldà, L.; Bogialli, S.; Marton, D.; Marzaro, M.; Girolami, V. UHPLC-DAD method for the determination of neonicotinoid insecticides in single bees and its relevance in honeybee colony loss investigations. Anal.Bioanal. Chem. 2013, 405, 1007–1014. [Google Scholar] [CrossRef]
- Bargańska, Ż.; Ślebioda, M.; Namieśnik, J. Pesticide residues levels in honey from apiaries located of Northern Poland. Food Control 2013, 31, 196–201. [Google Scholar] [CrossRef]
- SANCO/12495/2011. Method Validation and Quality Control Procedures for Pesticide Residues Analysis in Food and Feed. Available online: http://ec.europa.eu/food/plant/plant_protection_products/guidance_documents/docs/qualcontrol_en.pdf (accessed on 11 November 2013).
- Johnson, R.M.; Ellis, M.D.; Mullin, C.A.; Frazier, M. Pesticides and honey bee toxicity—USA. Apidologie 2010, 41, 312–331. [Google Scholar] [CrossRef]
- Tomlin, C.D.S. The Pesticide Manual, A World Compendium, 12th ed.; British Crop Protection Council: Surry, UK, 2000; pp. 502–504. [Google Scholar]
- Qualls, W.A.; Xue, R.D.; Zhong, H. Impact of bifenthrin on honeybees and Culex quinquefasciatus. J. Am. Mosq. Control Assoc. 2010, 26, 223–225. [Google Scholar] [CrossRef]
- Washburn, A.D. The Environmental Fate of Methidathion. Available online: http://www.cdpr.ca.gov/docs/emon/pubs/fatememo/methidathion.pdf (accessed on 8 August 2013).
- Snedeker, S. Pesticides and Breast Cancer Risk, an Evaluation of Alachlor. Cornell University Program on Breast Cancer and Environmental Risk Factors in New York State (BCERF). Fact Sheet 2000, 32, 1–6. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bargańska, Ż.; Ślebioda, M.; Namieśnik, J. Determination of Pesticide Residues in Honeybees using Modified QUEChERS Sample Work-Up and Liquid Chromatography-Tandem Mass Spectrometry. Molecules 2014, 19, 2911-2924. https://doi.org/10.3390/molecules19032911
Bargańska Ż, Ślebioda M, Namieśnik J. Determination of Pesticide Residues in Honeybees using Modified QUEChERS Sample Work-Up and Liquid Chromatography-Tandem Mass Spectrometry. Molecules. 2014; 19(3):2911-2924. https://doi.org/10.3390/molecules19032911
Chicago/Turabian StyleBargańska, Żaneta, Marek Ślebioda, and Jacek Namieśnik. 2014. "Determination of Pesticide Residues in Honeybees using Modified QUEChERS Sample Work-Up and Liquid Chromatography-Tandem Mass Spectrometry" Molecules 19, no. 3: 2911-2924. https://doi.org/10.3390/molecules19032911
APA StyleBargańska, Ż., Ślebioda, M., & Namieśnik, J. (2014). Determination of Pesticide Residues in Honeybees using Modified QUEChERS Sample Work-Up and Liquid Chromatography-Tandem Mass Spectrometry. Molecules, 19(3), 2911-2924. https://doi.org/10.3390/molecules19032911




