Biomolecule-Mediated Synthesis of Selenium Nanoparticles using Dried Vitis vinifera (Raisin) Extract
Abstract
:1. Introduction
2. Results and Discussion
2.1. Atomic Absorption Spectroscopy Analysis
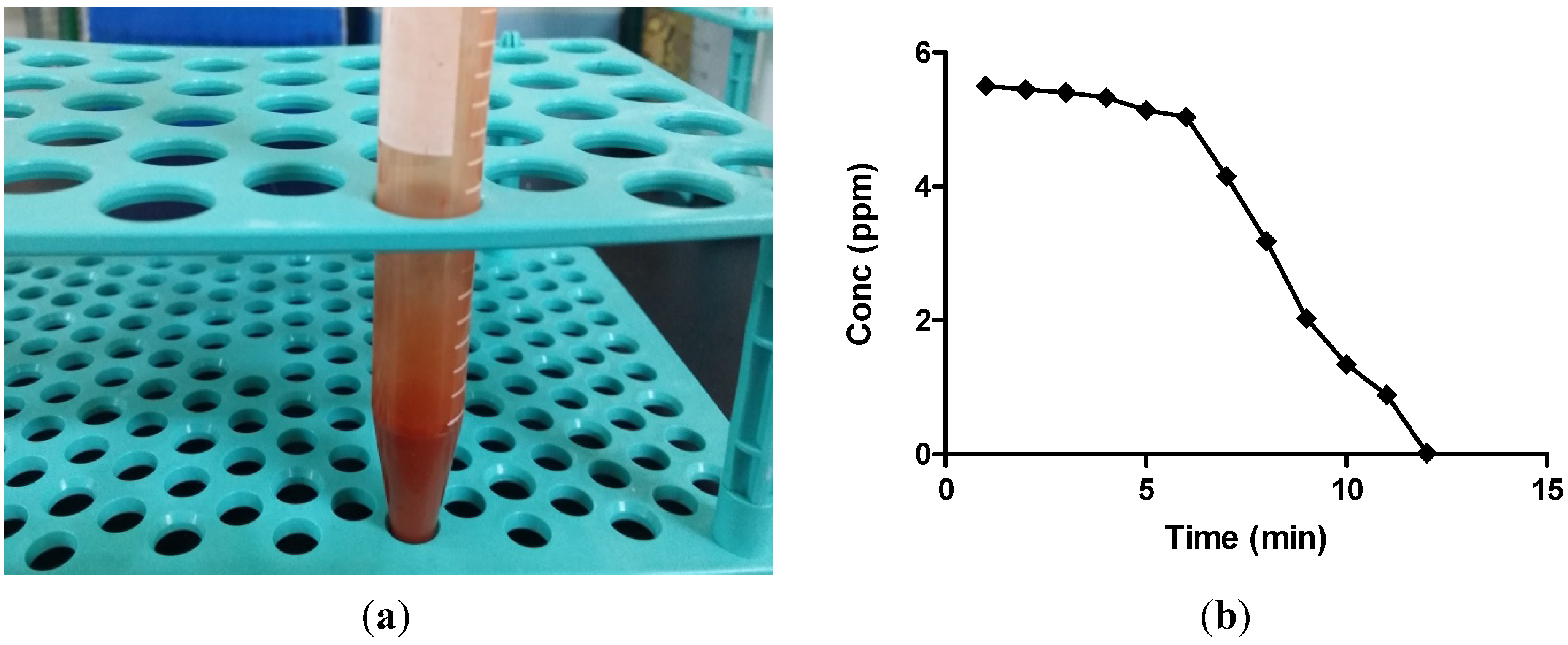
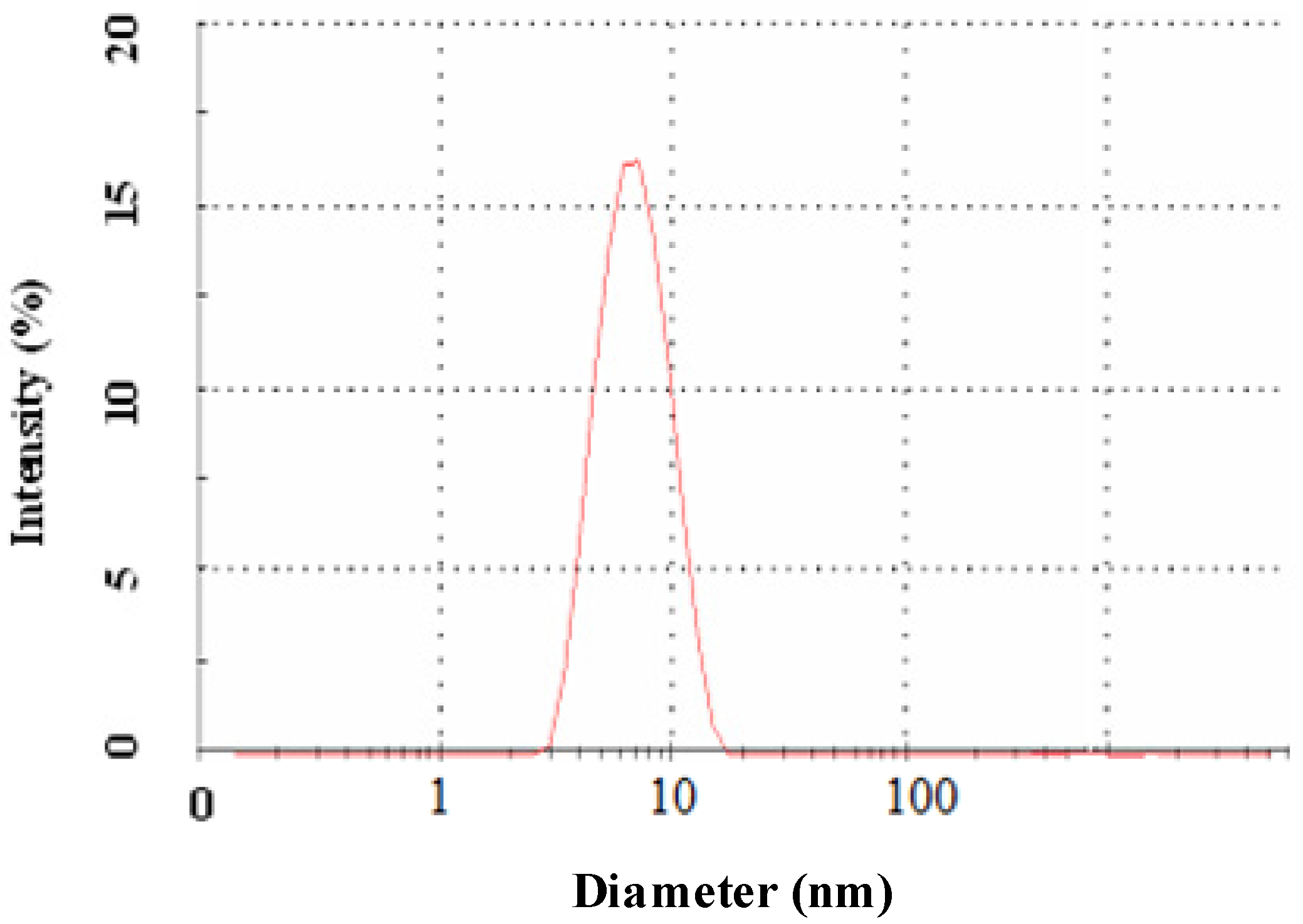
2.2. Morphology and Size Analysis

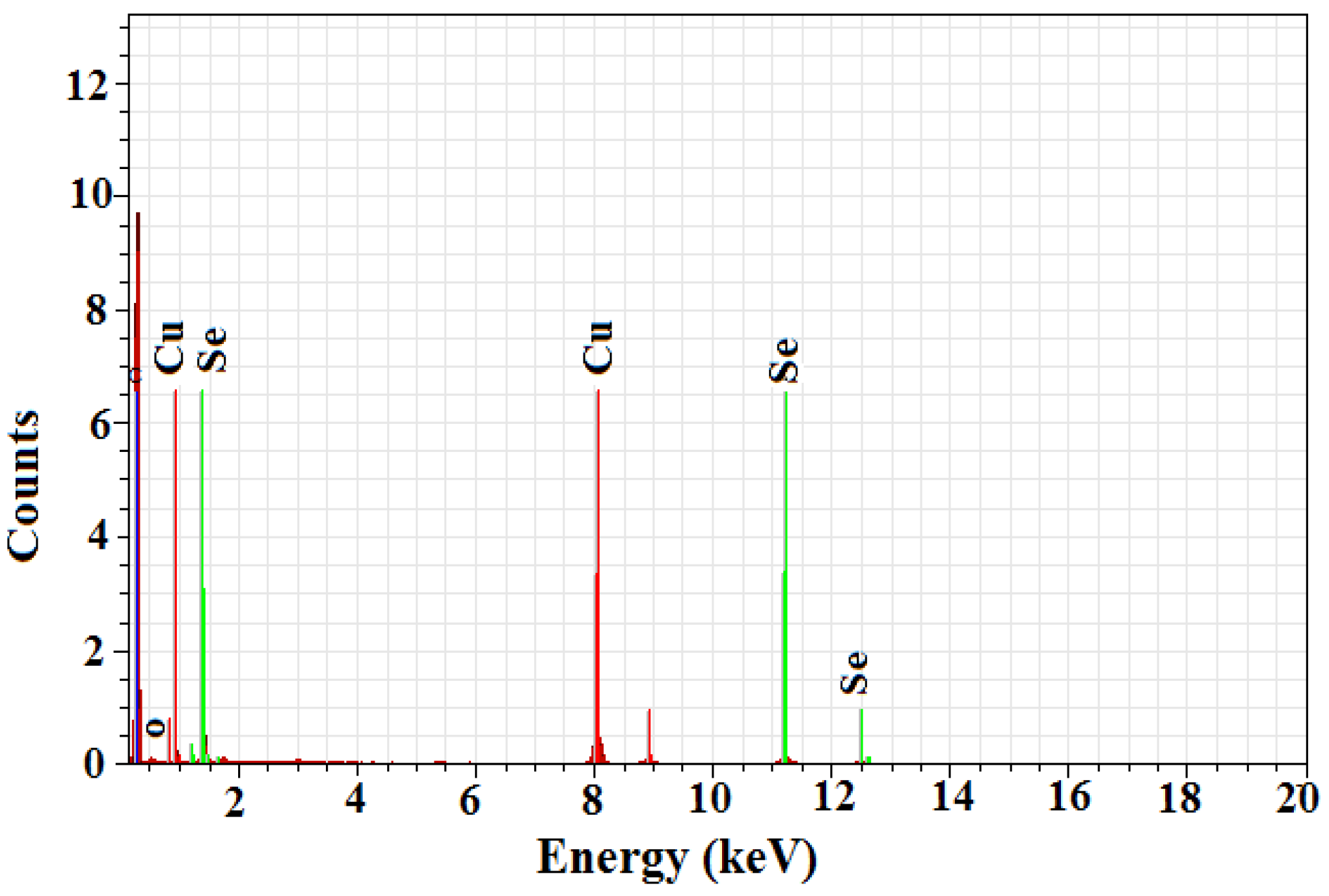
2.3. Energy Dispersive X-ray Analysis
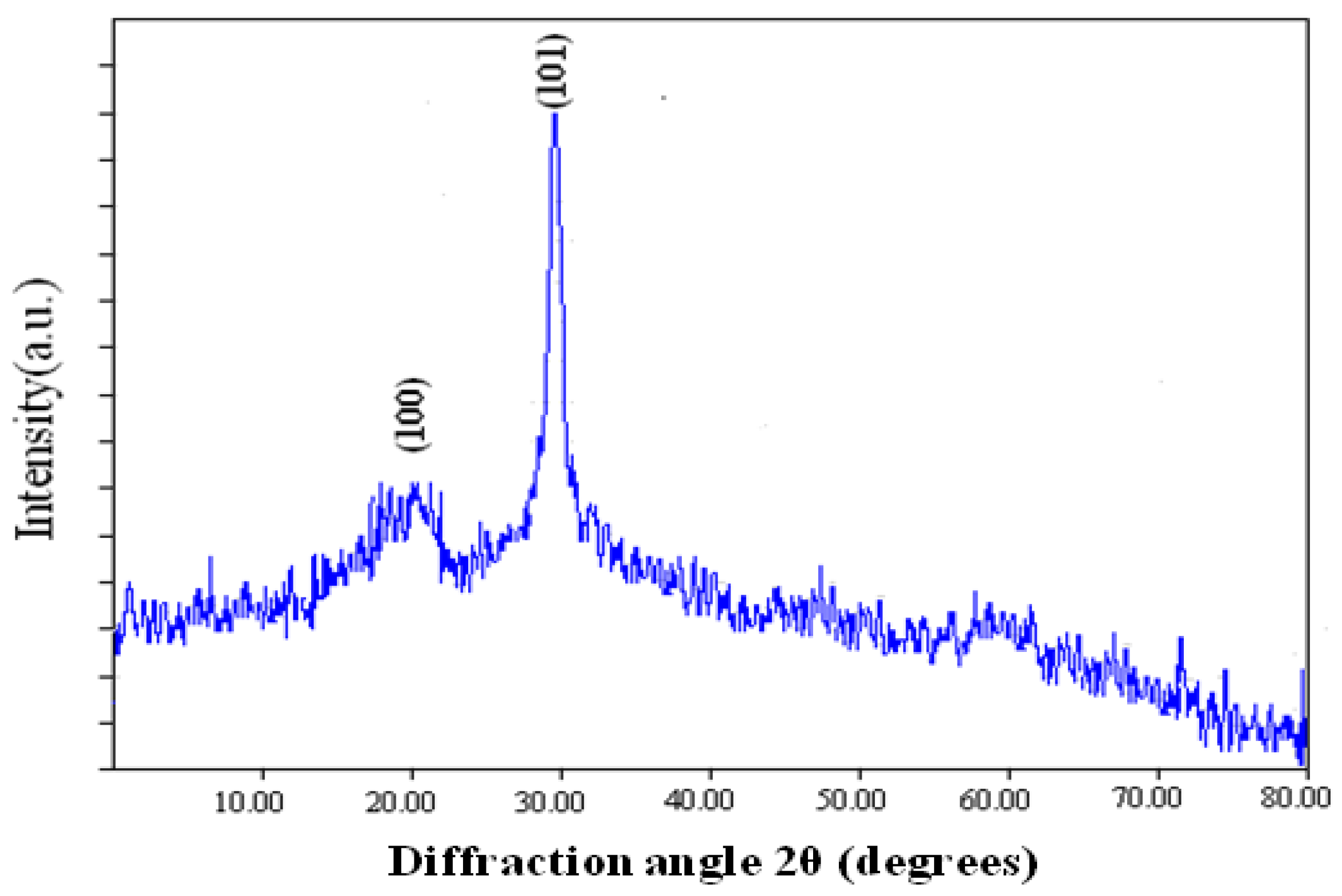
2.4. X-ray Diffraction Analysis
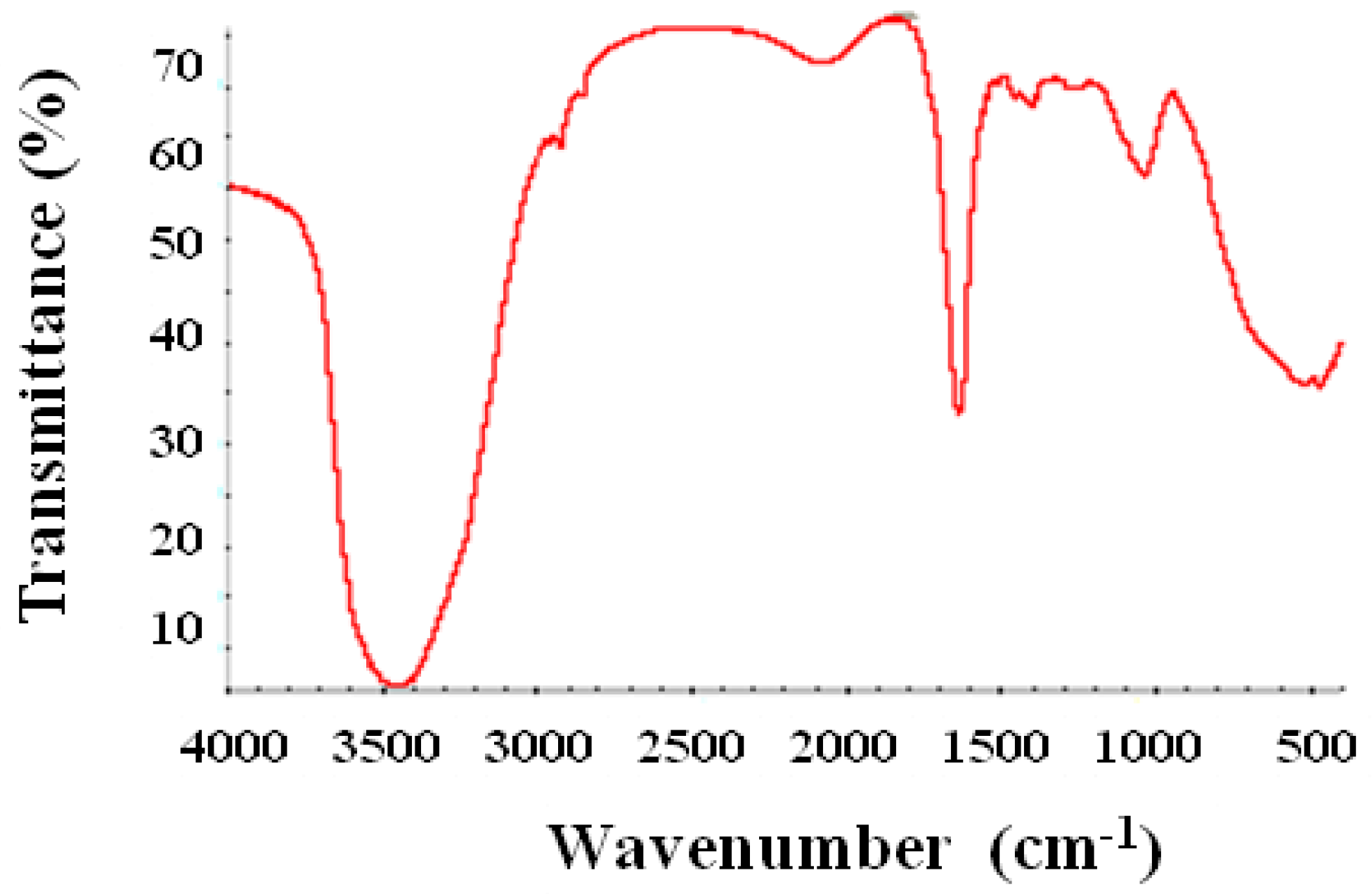
2.5. Fourier Transform Infrared Spectroscopy
3. Experimental
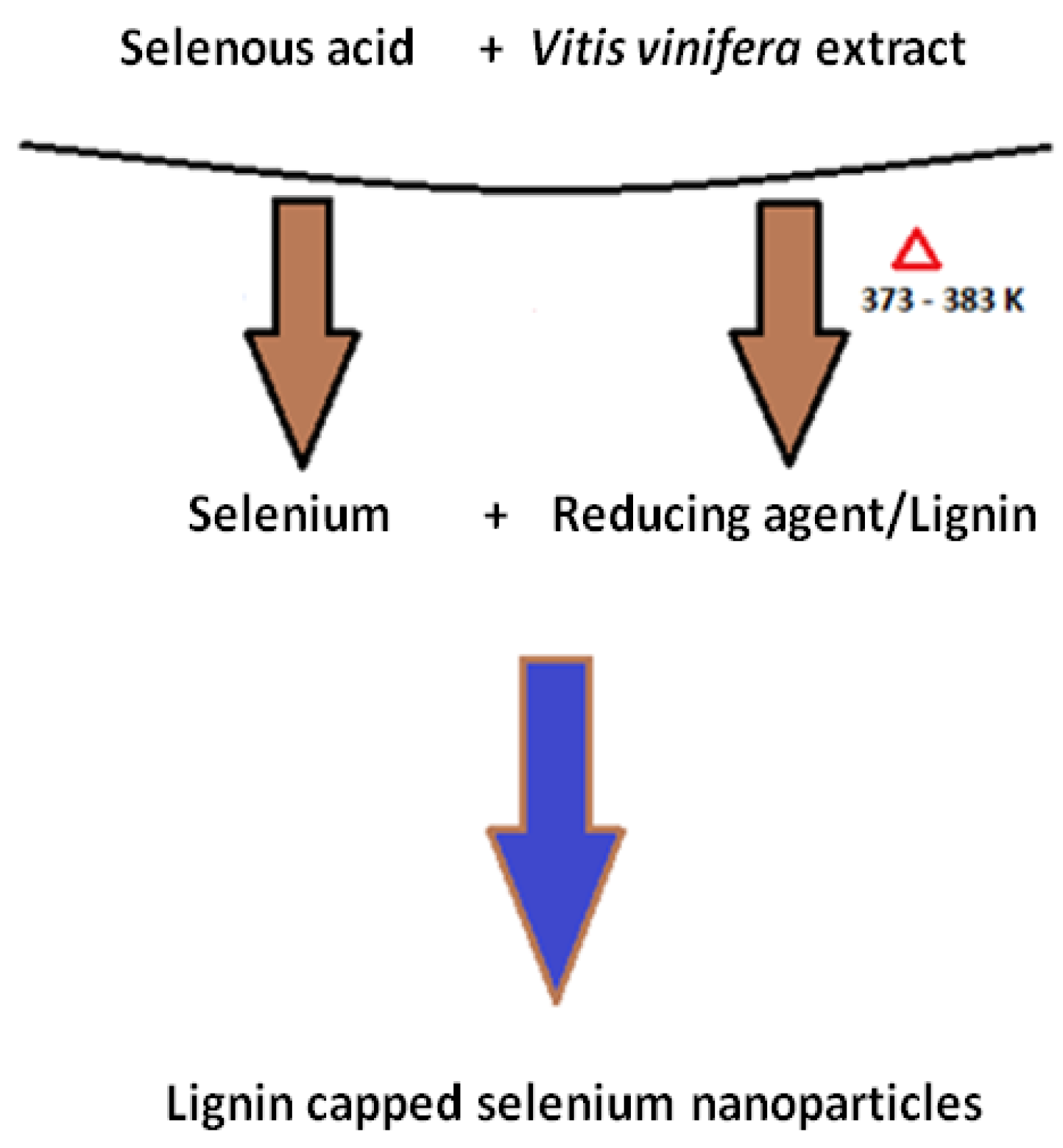
3.1. Synthesis of Selenium Nanoparticles
3.2. Characterization of Selenium Nanoparticles
3.2.1. Atomic Absorption Spectroscopy
3.2.2. Transmission Electron Microscopy
3.2.3. Dynamic Light Scattering Spectroscopy
3.2.4. Energy Dispersive X-ray Study
3.2.5. X-ray Diffraction Study
3.2.6. Fourier Transform Infrared Spectrpscopy
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Daniel, M.C.; Astruc, D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-size Related Properties, and Applications towards Biology, Catalysis and Nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Mandal, D.; Bolander, M.E.; Mukhopadhyay, D.; Sarkar, G.; Mukherjee, P. The use of microorganisms for the formation of metal nanoparticles and their application. Appl. Microbiol. Biotechnol. 2006, 69, 485–492. [Google Scholar] [CrossRef]
- Pearce, C.I.; Coker, V.S.; Charnock, J.M.; Pattrick, R.A.D.; Mosselmans, J.F.W.; Law, N.; Beveridge, T.J.; Lloyd, J.R. Microbial manufacture of chalcogenide-based nanoparticles via the reduction of selenite using Veillonella atypica: An in situ EXAFS study. Nanotechnology 2008, 19, 155603. [Google Scholar] [CrossRef]
- Mahapatro, A.; Singh, D.K. Biodegradable nanoparticles are excellent vehicle for site directed in vivo delivery of drugs and vaccines. J. Nanobiotechnol. 2011, 9, 1–11. [Google Scholar] [CrossRef]
- Wu, C.D. Grape products and oral health. J. Nutr. 2009, 139, 1818S–1823S. [Google Scholar] [CrossRef]
- Poborchii, V.V.; Kolobov, A.V.; Tanaka, K. An in situ Raman study of polarization-dependent photocrystallization in amorphous selenium films. Appl. Phys. Lett. 1998, 72, 1167–1169. [Google Scholar] [CrossRef]
- Zhang, J.; Wanh, H.; Yan, X.; Zhang, L. Comparison of short-term toxicity between Nano-Se and selenite in mice. Life Sci. 2005, 76, 1099–1109. [Google Scholar] [CrossRef]
- Tran, P.A.; Webster, T.J. Selenium nanoparticles inhibit Staphylococcus aureus growth. Int. J. Nanomed. 2011, 6, 1553–1558. [Google Scholar]
- Chen, T.; Wonga, Y.S.; Zhengb, W.; Baib, Y.; Huang, L. Selenium nanoparticles fabricated in Undaria pinnatifida polysaccharide solutions induce mitochondria-mediated apoptosis in A375 human melanoma cells. Colloids Surf. B Biointer. 2008, 67, 26–31. [Google Scholar] [CrossRef]
- Ramamurthy, C.H.; Sampath, K.S.; Arunkumar, P.; Suresh Kumar, M.; Sujatha, V.; Premkumar, K.; Thirunavukkarasu, C. Green synthesis and characterization of selenium nanoparticles and its Augmented cytotoxicity with doxorubicin on cancer cells. Bioprocess Biosyst. Eng. 2012, 36, 1131–1139. [Google Scholar]
- Wang, H.; Zhang, J.; Yu, H. Elemental selenium at nano size possesses lower toxicity without compromising the fundamental effect on selenoenzymes: Comparison with selenomethionine in mice. Free Radic. Biol. Med. 2007, 42, 1524–1533. [Google Scholar] [CrossRef]
- Ingole, A.R.; Thakare, S.R.; Khati, N.T.; Wankhade, A.V.; Burghate, D.K. Green synthesis of selenium nanoparticles under ambient condition. Chalcogenide Lett. 2010, 7, 485–489. [Google Scholar]
- Fesharaki, P.J.; Nazari, P.; Shakibaie, M.; Rezaie, S.; Banoee, M.; Abdollahi, M.; Shahverdi, A.R. Biosynthesis of selenium nanoparticles using Klebsiella pneumoniae and their recovery by a simple sterilization process. Braz. J. Microbiol. 2010, 41, 461–466. [Google Scholar] [CrossRef]
- Li, S.; Shen, Y.; Xie, A.; Yu, X.; Zhang, X.; Yang, L.; Li, C. Rapid, room-temperature synthesis of amorphous selenium/protein composites using Capsicum annuum L. extract. Nanotechnology 2007, 18, 405101–405109. [Google Scholar]
- Huang, N.M.; Lim, H.N.; Radiman, S.; Khiew, P.S.; Chiu, W.S.; Hashin, R.; Chia, C.H. Sucrose ester micellar-mediated synthesis of Ag nanoparticles and their antibacterial properties. Colloids Surf. 2010, 353, 69–76. [Google Scholar] [CrossRef]
- Kasture, M.B.; Patel, P.; Prabhune, A.A.; Ramana, C.V.; Kulkarni, K.K.; Prasad, B.L.V. Synthesis of silver nanoparticles by sophorolipids: Effect of temperature and sophorolipid structure on the size of particles. J. Chem. Sci. 2008, 120, 515–520. [Google Scholar] [CrossRef]
- Dhanjal, S.; Cameotra, S.S. Aerobic biogenesis of selenium nanospheres by Bacillus cereus isolated from coalmine soil. Microb. Cell Fact. 2010, 9, 52. [Google Scholar] [CrossRef]
- Klug, H.P.; Alexander, L.E. X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials; Wiley: New York, NY, USA, 1967. [Google Scholar]
- Arias, C.M. Binderless Fiberboard Production from Cynara cardunculuc and Vitis vinifera. Ph.D. Dissertation, Rovira i Virgili University, Tarragona, Spain, 2008. [Google Scholar]
- Faix, O.; Lin, S.Y.; Dence, C.W. Fourier transform infrared spectroscopy. In Methods in Lignin Chemistry; Lin, S.Y., Dence, C.W., Eds.; Springer-Verlag: Berlin, Germany, 1992; pp. 83–109. [Google Scholar]
- Ibarra, D.; delRío, J.C.; Gutiérrez, A.; Rodríguez, I.M.; Romero, J.; Martínez, M.J.; Martínez, A.T. Isolation of high-purity residual lignins from eucalypt paper pulps by cellulase and proteinase treatments followed by solvent extraction. Enz. Microb. Tech. 2004, 35, 173–181. [Google Scholar] [CrossRef]
- Vázquez, G.; Antorrena, G.; González, J.; Freire, S. FTIR, H-1 and C-13NMR characterization of acetosolv solubilized pine and eucalyptuslignins. Holzforschung 1997, 51, 158–166. [Google Scholar] [CrossRef]
- Bunzel, M.; Ralph, J. NMR characterization of lignins isolated from fruit and vegetable insoluble dietary fiber. J. Agric. Food Chem. 2006, 54, 8352–8361. [Google Scholar] [CrossRef]
- Coccia, F.; Tonucci, L.; Bosco, D.; Bressand, M.; D’Alessandro, N. One pot synthesis of lignin-stabilized platinum and palladium nanoparticles and their catalytic behavior on oxidation and reduction reactions. Green Chem. 2012, 14, 1073–1078. [Google Scholar] [CrossRef]
- Chen, A.; Shin, D.; Nam, J.; Kwon, K.; Yoo, J. Selenium nanowires and nanutubes synthesized via a facile template-free solution method. Mater. Res. Bull. 2010, 45, 699–704. [Google Scholar] [CrossRef]
- Singhal, G.; Bhavesh, R.; Kasariya, K.; Sharma, A.R.; Singh, R.P. Biosynthesis of silver nanopartiucles using Ocimum sanctum (Tulsi) leaf extract and screening its antimicrobial activity. J. Nanopart. Res. 2011, 13, 2981–2988. [Google Scholar] [CrossRef]
- Sample Availability: Samples of dried vitis vinefera are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sharma, G.; Sharma, A.R.; Bhavesh, R.; Park, J.; Ganbold, B.; Nam, J.-S.; Lee, S.-S. Biomolecule-Mediated Synthesis of Selenium Nanoparticles using Dried Vitis vinifera (Raisin) Extract. Molecules 2014, 19, 2761-2770. https://doi.org/10.3390/molecules19032761
Sharma G, Sharma AR, Bhavesh R, Park J, Ganbold B, Nam J-S, Lee S-S. Biomolecule-Mediated Synthesis of Selenium Nanoparticles using Dried Vitis vinifera (Raisin) Extract. Molecules. 2014; 19(3):2761-2770. https://doi.org/10.3390/molecules19032761
Chicago/Turabian StyleSharma, Garima, Ashish Ranjan Sharma, Riju Bhavesh, Jongbong Park, Bilguun Ganbold, Ju-Suk Nam, and Sang-Soo Lee. 2014. "Biomolecule-Mediated Synthesis of Selenium Nanoparticles using Dried Vitis vinifera (Raisin) Extract" Molecules 19, no. 3: 2761-2770. https://doi.org/10.3390/molecules19032761
APA StyleSharma, G., Sharma, A. R., Bhavesh, R., Park, J., Ganbold, B., Nam, J.-S., & Lee, S.-S. (2014). Biomolecule-Mediated Synthesis of Selenium Nanoparticles using Dried Vitis vinifera (Raisin) Extract. Molecules, 19(3), 2761-2770. https://doi.org/10.3390/molecules19032761




