Effects of Honey and Its Mechanisms of Action on the Development and Progression of Cancer
Abstract
:1. Introduction
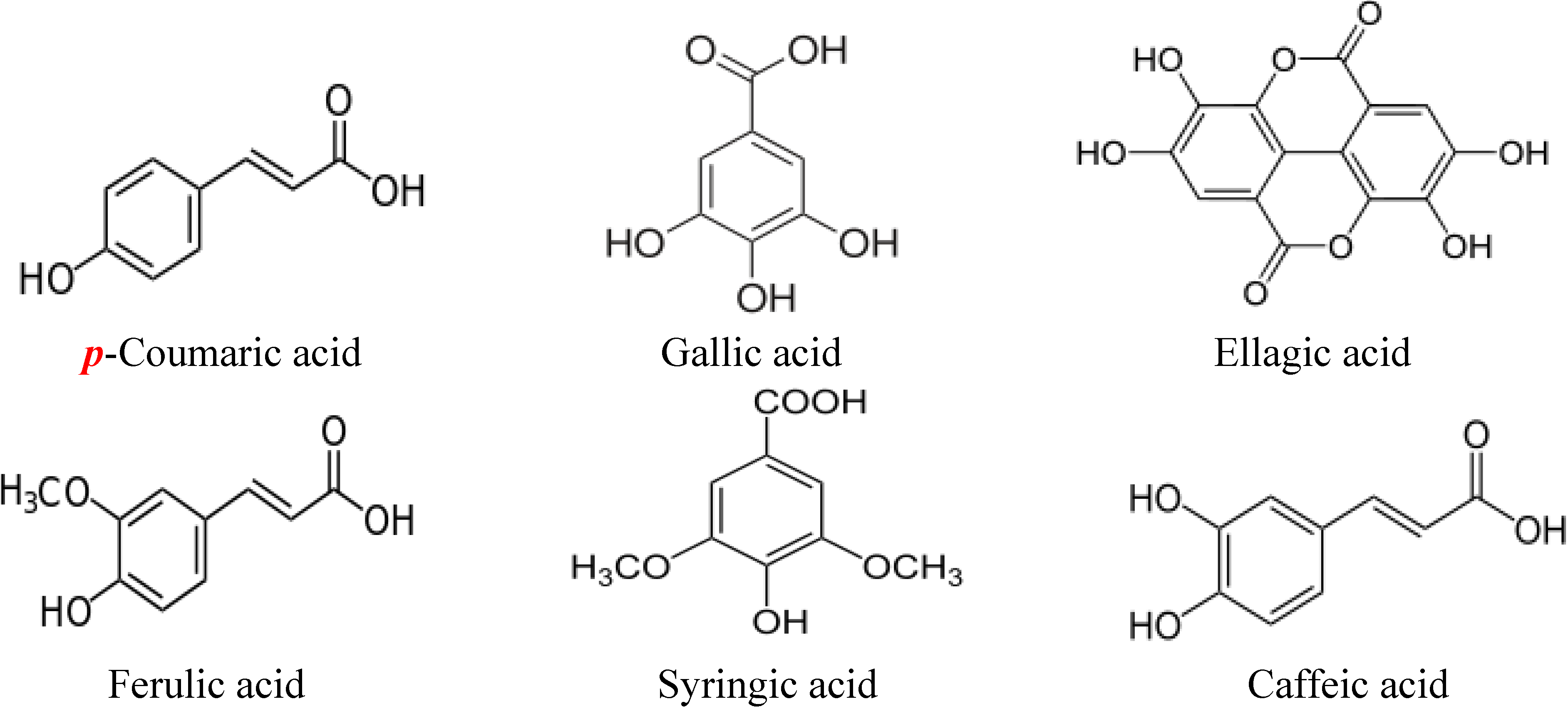
2. Structures of Major Flavonoids and Phenolic Acids in Honey


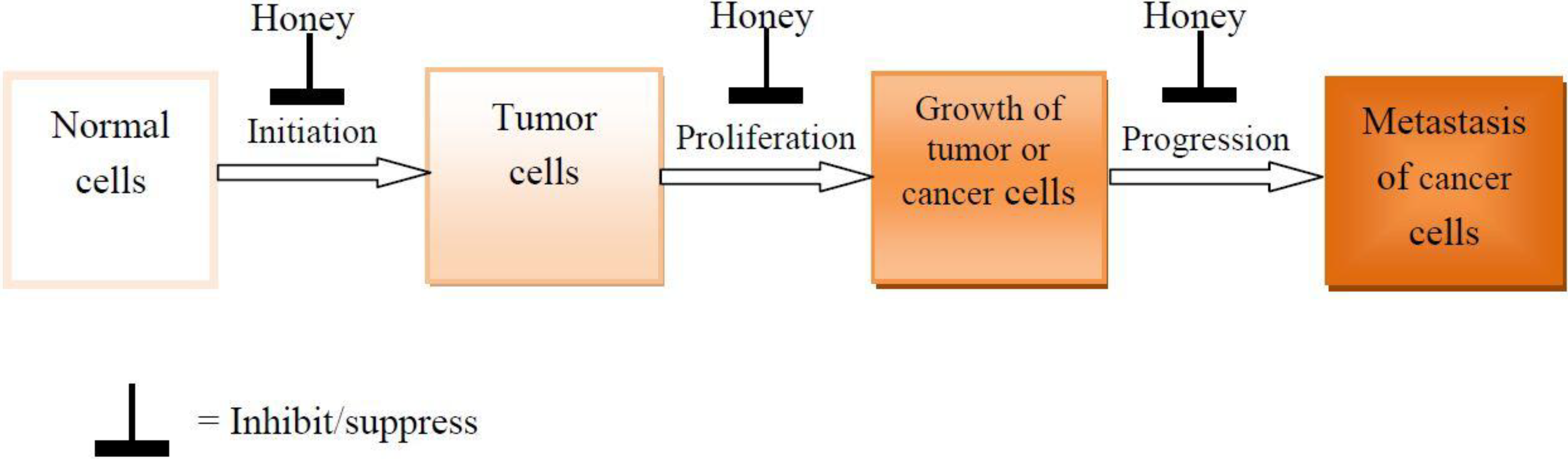
3. Effects of Honey on the Development and Progression of Tumor and Cancer
3.1. Breast Cancer
3.2. Liver Cancer
3.3. Colorectal Cancer
3.4. Prostate Cancer
3.5. Other Forms of Cancer
| Type of tumor/cancer and cancer cell type | Effects of honey (key findings) | References |
|---|---|---|
| In vitro studies | ||
| Human breast cancer (MCF-7 & MDA-MB-231) | Antagonizes estrogen activity, inhibits cell proliferation, induces apoptosis, reduces mitochondrial membrane potential | [31,36,37,38] |
| Human liver cancer (HepG2) | Inhibits cell proliferation, suppresses angiogenesis, induces apoptosis, protects against mutagen-induced DNA damage | [47,50,52,53,55,56] |
| Human colorectal cancer (HT 29, HCT 15 & CT 26) | Inhibits cell proliferation, induces apoptosis, arrests cell cycle, reduces mitochondrial membrane potential, increases generation of ROS, depletes intracellular non-protein thiols, induces DNA damage, suppresses inflammation | [62,63,64,75] |
| Human prostate cancer (PC-3) | Inhibits cell proliferation, induces apoptosis | [26,31] |
| Human bladder cancer (T24, 253J, RT4 & MBT-2) | Inhibits cell proliferation | [68] |
| Human kidney cancer (Renal cancer cell line) | Induces apoptosis | [69] |
| Human oral cancer (Oral carcinoma) | Inhibits cell proliferation | [72] |
| Human bone cancer (Osteosarcoma) | Inhibits cell proliferation | [72] |
| Human skin cancer (Melanoma cells) | Inhibits cell proliferation, arrests cell cycle | [70] |
| Human leukemia | Induces apoptosis | [73] |
| Human endometrial cancer | Inhibits cell proliferation | [31] |
| Human lung cancer NCI-H460 | Inhibits cell proliferation | [71] |
| Human cervical cancer | Induces apoptosis, disrupts mitochondrial membrane potential | [36] |
| In vivo studies | ||
| Walker 256 carcinoma | Inhibits cell proliferation, arrests cell cycle, induces apoptosis | [43] |
| DMBA-induced breast cancer in rats | Delays the development of tumors, reduces the number and size of tumors, prevents the development of high grade cancer | [44] |
| Rats with DEN-induced hepatic cancer | Protects against transformation of normal liver cells to neoplastic hepatic cells, restores the PCNA and P53 expression | [57] |
| Mice/rats with colon carcinoma or adenocarcinoma | Inhibits formation of metastases and tumor growth | [40,65,66,67] |
| Mice implanted with bladder cancer cells | Inhibits tumor growth | [68] |
| Mice with melanoma | Inhibits tumor growth, induces apoptosis | [75] |
4. Mechanisms of the Antiproliferative, Antimetastatic and Anticancer Effects of Honey
4.1. Cell Cycle Arrest
4.2. Activation of the Mitochondrial Pathway
4.3. Induction of Mitochondrial Outer Membrane Permeabilization
4.4. Induction of Apoptosis
4.5. Modulation of Oxidative Stress
4.6. Amelioration of Inflammation
4.7. Modulation of Insulin Signaling
4.8. Inhibition of Angiogenesis
5. Conclusions
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA: Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Bray, F.; Ren, J.S.; Masuyer, E.; Ferlay, J. Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int. J. Cancer 2013, 132, 1133–1145. [Google Scholar] [CrossRef]
- Kanavos, P. The rising burden of cancer in the developing world. Ann. Oncol. 2006, 17, vii15–vii23. [Google Scholar] [CrossRef]
- Othman, N.H. Honey and cancer: Sustainable inverse relationship particularly for developing nations—A review. Evid. Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Pitot, H.C. The molecular biology of carcinogenesis. Cancer 1993, 72, 962–970. [Google Scholar] [CrossRef]
- Tubiana, M. Carcinogenesis: From epidemiology to molecular biology. Bull. Acad. Natl. Med. 1998, 182, 19–29. [Google Scholar]
- Chidambaram, M.; Manavalan, R.; Kathiresan, K. Nanotherapeutics to overcome conventional cancer chemotherapy limitations. J. Pharm. Pharm. Sci. 2011, 14, 67–77. [Google Scholar]
- Wedding, U. Chances and limitations of chemotherapy in elderly patients. Internist 2010, 51. [Google Scholar] [CrossRef]
- Mendel, J. Evidenced based medicine. Benefits, limitations and issues for complementary and alternative medicine. Aust. J. Holist. Nurs. 2004, 11, 21–29. [Google Scholar]
- Heimall, J.; Bielory, L. Defining complementary and alternative medicine in allergies and asthma: Benefits and risks. Clin. Rev. Allergy Immunol. 2004, 27, 93–103. [Google Scholar] [CrossRef]
- Oh, B.; Butow, P.; Mullan, B.; Beale, P.; Pavlakis, N.; Rosenthal, D.; Clarke, S. The use and perceived benefits resulting from the use of complementary and alternative medicine by cancer patients in Australia. Asia Pac. J. Clin. Oncol. 2010, 6, 342–349. [Google Scholar] [CrossRef]
- Tan, H.T.; Rahman, R.A.; Gan, S.H.; Halim, A.S.; Hassan, S.A.; Sulaiman, S.A.; Kirnpal-Kaur, B. The antibacterial properties of Malaysian tualang honey against wound and enteric microorganisms in comparison to manuka honey. BMC Complement. Altern. Med. 2009, 9, 34. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Gurtu, S.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.; Salleh, M.S. Hypoglycemic and antioxidant effects of honey supplementation in streptozotocin-induced diabetic rats. Int. J. Vitam. Nutr. Res. 2010, 80, 74–82. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.; Salleh, S.; Gurtu, S. Honey supplementation in spontaneously hypertensive rats elicits antihypertensive effect via amelioration of renal oxidative stress. Oxid. Med. Cell. Longev. 2012, 2012, 374037. [Google Scholar]
- Kassim, M.; Achoui, M.; Mustafa, M.R.; Mohd, M.A.; Yusoff, K.M. Ellagic acid, phenolic acids, and flavonoids in Malaysian honey extracts demonstrate in vitro anti-inflammatory activit. Nutr. Res. 2010, 30, 650–659. [Google Scholar] [CrossRef]
- Erejuwa, O.; Sulaiman, S.; Wahab, M.; Sirajudeen, K.; Salleh, M.; Gurtu, S. Hepatoprotective effect of tualang honey supplementation in streptozotocin-induced diabetic rats. Int. J. Appl. Res. Nat. Prod. 2012, 4, 37–41. [Google Scholar]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Honey—A Novel Antidiabetic Agent. Int. J. Biol. Sci. 2012, 8, 913–934. [Google Scholar]
- Alvarez-Suarez, J.M.; Giampieri, F.; Battino, M. Honey as a source of dietary antioxidants: Structures, bioavailability and evidence of protective effects against human chronic diseases. Curr. Med. Chem. 2013, 20, 621–638. [Google Scholar] [CrossRef]
- White, J.W. Composition of honey. Honey: Compr. Surv. 1975, 157–206. [Google Scholar]
- Hermosín, I.; Chicón, R.M.; Dolores Cabezudo, M. Free amino acid composition and botanical origin of honey. Food Chem. 2003, 83, 263–268. [Google Scholar] [CrossRef]
- Sergiel, I.; Pohl, P.; Biesaga, M. Characterisation of honeys according to their content of phenolic compounds using high performance liquid chromatography/tandem mass spectrometry. Food Chem. 2014, 145, 404–408. [Google Scholar] [CrossRef]
- Chan, C.W.; Deadman, B.J.; Manley-Harris, M.; Wilkins, A.L.; Alber, D.G.; Harry, E. Analysis of the flavonoid component of bioactive New Zealand manuka (Leptospermum scoparium) honey and the isolation, characterisation and synthesis of an unusual pyrrole. Food Chem. 2013, 141, 1772–1781. [Google Scholar] [CrossRef]
- Candiracci, M.; Piatti, E.; Dominguez-Barragan, M.; Garcia-Antras, D.; Morgado, B.; Ruano, D.; Gutierrez, J.F.; Parrado, J.; Castano, A. Anti-inflammatory activity of a honey flavonoid extract on lipopolysaccharide-activated N13 microglial cells. J. Agric. Food Chem. 2012, 60, 12304–12311. [Google Scholar] [CrossRef]
- Xie, F.; Su, M.; Qiu, W.; Zhang, M.; Guo, Z.; Su, B.; Liu, J.; Li, X.; Zhou, L. Kaempferol promotes apoptosis in human bladder cancer cells by inducing the tumor suppressor, PTEN. Int. J. Mol. Sci. 2013, 14, 21215–21226. [Google Scholar]
- Jaganathan, S.K. Growth inhibition by caffeic acid, one of the phenolic constituents of honey, in HCT 15 colon cancer cells. Sci. World J. 2012, 2012. [Google Scholar] [CrossRef]
- Samarghandian, S.; Afshari, J.T.; Davoodi, S. Chrysin reduces proliferation and induces apoptosis in the human prostate cancer cell line pc-3. Clinics 2011, 66, 1073–1079. [Google Scholar] [CrossRef]
- Zand, R.S.; Jenkins, D.J.; Diamandis, E.P. Steroid hormone activity of flavonoids and related compounds. Breast Cancer Res. Treat. 2000, 62, 35–49. [Google Scholar]
- Kyselova, Z. Toxicological aspects of the use of phenolic compounds in disease prevention. Interdiscip. Toxicol. 2011, 4, 173–183. [Google Scholar] [CrossRef]
- Han, D.H.; Denison, M.S.; Tachibana, H.; Yamada, K. Relationship between estrogen receptor-binding and estrogenic activities of environmental estrogens and suppression by flavonoids. Biosci. Biotechnol. Biochem. 2002, 66, 1479–1487. [Google Scholar] [CrossRef]
- Germain, D. Estrogen carcinogenesis in breast cancer. Endocrinol. Metab Clin North. Am. 2011, 40, 473–484. [Google Scholar] [CrossRef]
- Tsiapara, A.V.; Jaakkola, M.; Chinou, I.; Graikou, K.; Tolonen, T.; Virtanen, V.; Moutsatsou, P. Bioactivity of Greek honey extracts on breast cancer (MCF-7), prostate cancer (PC-3) and endometrial cancer (Ishikawa) cells: Profile analysis of extracts. Food Chem. 2009, 116, 702–708. [Google Scholar] [CrossRef]
- Patisaul, H.B.; Jefferson, W. The pros and cons of phytoestrogens. Front. Neuroendocrinol. 2010, 31, 400–419. [Google Scholar] [CrossRef]
- Ziegler, R.G. Phytoestrogens and breast cancer. Am. J. Clin. Nutr. 2004, 79, 183–184. [Google Scholar]
- Bulzomi, P.; Galluzzo, P.; Bolli, A.; Leone, S.; Acconcia, F.; Marino, M. The pro-apoptotic effect of quercetin in cancer cell lines requires ERbeta-dependent signals. J. Cell. Physiol. 2012, 227, 1891–1898. [Google Scholar] [CrossRef]
- Galluzzo, P.; Martini, C.; Bulzomi, P.; Leone, S.; Bolli, A.; Pallottini, V.; Marino, M. Quercetin-induced apoptotic cascade in cancer cells: Antioxidant versus estrogen receptor alpha-dependent mechanisms. Mol. Nutr. Food Res. 2009, 53, 699–708. [Google Scholar] [CrossRef]
- Fauzi, A.N.; Norazmi, M.N.; Yaacob, N.S. Tualang honey induces apoptosis and disrupts the mitochondrial membrane potential of human breast and cervical cancer cell lines. Food Chem. Toxicol. 2011, 49, 871–878. [Google Scholar] [CrossRef]
- Yaacob, N.S.; Nengsih, A.; Norazmi, M.N. Tualang honey promotes apoptotic cell death induced by tamoxifen in breast cancer cell lines. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mandal, S.M.; Jana, S.K.; Das, S.; Mandal, M. Studies on the phenolic profiling, anti-oxidant and cytotoxic activity of Indian honey: In vitro evaluation. Nat. Prod. Res. 2010, 24, 1295–1306. [Google Scholar] [CrossRef]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar]
- Orsolic, N.; Knezevic, A.; Sver, L.; Terzic, S.; Hackenberger, B.K.; Basic, I. Influence of honey bee products on transplantable murine tumours. Vet. Comp. Oncol. 2003, 1, 216–226. [Google Scholar] [CrossRef]
- Yang, B.; Huang, J.; Xiang, T.; Yin, X.; Luo, X.; Luo, F.; Li, H.; Ren, G. Chrysin inhibits metastatic potential of human triple-negative breast cancer cells by modulating matrix metalloproteinase-10, epithelial to mesenchymal transition, and PI3K/Akt signaling pathway. J. Appl. Toxicol. 2013, 2013. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mondhe, D.; Wani, Z.A.; Pal, H.C.; Mandal, M. Effect of honey and eugenol on Ehrlich ascites and solid carcinoma. J. Biomed. Biotechnol. 2010, 2010. [Google Scholar] [CrossRef]
- Tomasin, R.; Gomes-Marcondes, M.C. Oral administration of aloe vera and honey reduces walker tumour growth by decreasing cell proliferation and increasing apoptosis in tumour tissue. Phytother. Res. 2011, 25, 619–623. [Google Scholar] [CrossRef]
- Kadir, E.A.; Sulaiman, S.A.; Yahya, N.K.; Othman, N.H. Inhibitory effects of tualang honey on experimental breast cancer in rats: A preliminary study. Asian Pac. J. Cancer Prev. 2013, 14, 2249–2254. [Google Scholar] [CrossRef]
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics, 2002. Cancer J. Clin. 2005, 55, 74–108. [Google Scholar] [CrossRef]
- Venook, A.P.; Papandreou, C.; Furuse, J.; de Guevara, L.L. The incidence and epidemiology of hepatocellular carcinoma: A global and regional perspective. Oncologist 2010, 15, 5–13. [Google Scholar]
- Hassan, M.I.; Mabrouk, G.M.; Shehata, H.H.; Aboelhussein, M.M. Antineoplastic effects of bee honey and Nigella sativa on hepatocellular carcinoma cells. Integr. Cancer Ther. 2012, 11, 354–363. [Google Scholar] [CrossRef]
- Wartenberg, M.; Diedershagen, H.; Hescheler, J.; Sauer, H. Growth stimulation versus induction of cell quiescence by hydrogen peroxide in prostate tumor spheroids is encoded by the duration of the Ca2+ response. J. Biol. Chem. 1999, 274, 27759–27767. [Google Scholar] [CrossRef]
- Rao, G.N.; Berk, B.C. Active oxygen species stimulate vascular smooth muscle cell growth and proto-oncogene expression. Circ. Res. 1992, 70, 593–599. [Google Scholar] [CrossRef]
- Jubri, Z.; Narayanan, N.N.N.; Karim, N.A.; Ngah, W.Z.W. Antiproliferative activity and apoptosis induction by gelam honey on liver cancer cell line. Int. J. Appl. Sci. Technol. 2012, 2, 135–141. [Google Scholar]
- Ismail, W.I.W.; Abu, M.N.; Salleh, M.A.M.; Radzman, N.H.M.; Yusof, R.M.; Hassan, H.F. Insulin sensitivity enhancement of the mixture of Tinospora crispa and gelam (Melaleuca cajuputi) honey and its antiproliferative activity on hepatocellular carcinoma, HepG2: A preliminary study. J. Med. Res. Dev. 2013, 2, 48–54. [Google Scholar]
- Chanchao, C. Bioactivity of honey and propolis of Tetragonula Laeviceps in Thailand. In Bioactivity of honey and propolis of Tetragonula Laeviceps in Thailand.; In Pot-Honey; Springer: New York, NY, USA, 2013; pp. 495–505. [Google Scholar]
- Abdel Aziz, A.; Rady, H.; Amer, M.; Kiwan, H. Effect of some honey bee extracts on the proliferation, proteolytic and gelatinolytic activities of the hepatocellular carcinoma Hepg2 cell line. Aust. J. Basic Appl. Sci. 2009, 3, 2754–2769. [Google Scholar]
- Henriques, A.; Jackson, S.; Cooper, R.; Burton, N. Free radical production and quenching in honeys with wound healing potential. J. Antimicrob. Chemother. 2006, 58, 773–777. [Google Scholar] [CrossRef]
- Hanaa, M.; Shaymaa, M. Enhancement of the antitumor effect of honey and some of its extracts using adiponectin hormone. Aust. J. Basic Appl. Sci. 2011, 5, 100–108. [Google Scholar]
- Haza, A.I.; Morales, P. Spanish honeys protect against food mutagen-induced DNA damage. J. Sci. Food Agric. 2013, 93, 2995–3000. [Google Scholar] [CrossRef]
- El-kott, A.F.; Kandeel, A.A.; El-Aziz, S.F.A.; Ribea, H.M. Anti-tumor effects of bee honey on PCNA and P53 expression in the rat hepatocarcinogenesis. Int. J. Cancer Res. 2012, 8, 130–139. [Google Scholar] [CrossRef]
- Boyle, P.; Langman, J.S. ABC of colorectal cancer: Epidemiology. BMJ 2000, 321, 805–808. [Google Scholar] [CrossRef]
- Hlavata, I.; Mohelnikova-Duchonova, B.; Vaclavikova, R.; Liska, V.; Pitule, P.; Novak, P.; Bruha, J.; Vycital, O.; Holubec, L.; Treska, V.; et al. The role of ABC transporters in progression and clinical outcome of colorectal cancer. Mutagenesis 2012, 27, 187–196. [Google Scholar] [CrossRef]
- Boyle, P.; Leon, M.E. Epidemiology of colorectal cancer. Br. Med. Bull. 2002, 64, 1–25. [Google Scholar] [CrossRef]
- Midgley, R.S.; Kerr, D.J. ABC of colorectal cancer: Adjuvant therapy. BMJ 2000, 321, 1208–1211. [Google Scholar] [CrossRef]
- Wen, C.T.; Hussein, S.Z.; Abdullah, S.; Karim, N.A.; Makpol, S.; Mohd, Y.A. Gelam and Nenas honeys inhibit proliferation of HT 29 colon cancer cells by inducing DNA damage and apoptosis while suppressing inflammation. Asian Pac. J. Cancer Prev. 2012, 13, 1605–1610. [Google Scholar] [CrossRef]
- Jaganathan, S.; Mandal, M. Honey constituents and its apoptotic effect in colon cancer cells. J. Apiprod. Apimed. Sci. 2009, 1, 29–36. [Google Scholar] [CrossRef]
- Jaganathan, S.K.; Mandal, M. Involvement of non-protein thiols, mitochondrial dysfunction, reactive oxygen species and p53 in honey-induced apoptosis. Investig. N. Drugs 2010, 28, 624–633. [Google Scholar] [CrossRef]
- Oršolić, N.; Terzić, S.; Šver, L.; Bašić, I. Honey-bee products in prevention and/or therapy of murine transplantable tumours. J. Sci. Food Agric. 2005, 85, 363–370. [Google Scholar] [CrossRef]
- Orsolic, N.; Basic, I. Honey as a cancer-preventive agent. Period. Biol. 2004, 106, 397–401. [Google Scholar]
- Mabrouk, G.M.; Moselhy, S.S.; Zohny, S.F.; Ali, E.M.; Helal, T.E.; Amin, A.A.; Khalifa, A.A. Inhibition of methylnitrosourea (MNU) induced oxidative stress and carcinogenesis by orally administered bee honey and Nigella grains in Sprague Dawely rats. J. Exp. Clin. Cancer Res. 2002, 21, 341–346. [Google Scholar]
- Swellam, T.; Miyanaga, N.; Onozawa, M.; Hattori, K.; Kawai, K.; Shimazui, T.; Akaza, H. Antineoplastic activity of honey in an experimental bladder cancer implantation model: In vivo and in vitro studies. Int. J. Urol. 2003, 10, 213–219. [Google Scholar] [CrossRef]
- Samarghandian, S.; Afshari, J.T.; Davoodi, S. Honey induces apoptosis in renal cell carcinoma. Pharmacogn. Mag. 2011, 7, 46–52. [Google Scholar] [CrossRef]
- Pichichero, E.; Cicconi, R.; Mattei, M.; Muzi, M.G.; Canini, A. Acacia honey and chrysin reduce proliferation of melanoma cells through alterations in cell cycle progression. Int. J. Oncol. 2010, 37, 973–981. [Google Scholar]
- Aliyu, M.; Odunola, O.A.; Farooq, A.D.; Rasheed, H.; Mesaik, A.M.; Choudhary, M.I.; Channa, I.S.; Khan, S.A.; Erukainure, O.L. Molecular mechanism of antiproliferation potential of Acacia honey on NCI-H460 cell line. Nutr. Cancer 2013, 65, 296–304. [Google Scholar] [CrossRef]
- Ghashm, A.A.; Othman, N.H.; Khattak, M.N.; Ismail, N.M.; Saini, R. Antiproliferative effect of tualang honey on oral squamous cell carcinoma and osteosarcoma cell lines. BMC Complement. Altern. Med. 2010, 10, 49. [Google Scholar]
- Morales, P.; Haza, A.I. Antiproliferative and apoptotic effects of spanish honeys. Pharmacogn. Mag. 2013, 9, 231–237. [Google Scholar] [CrossRef]
- Ghaffari, A.; Somi, M.H.; Safaiyan, A.; Modaresi, J.; Ostadrahimi, A. Honey and apoptosis in human gastric mucosa. Health Promot. Perspect. 2012, 2, 53–59. [Google Scholar]
- Fernandez-Cabezudo, M.J.; El-Kharrag, R.; Torab, F.; Bashir, G.; George, J.A.; El-Taji, H.; Al-Ramadi, B.K. Intravenous administration of manuka honey inhibits tumor growth and improves host survival when used in combination with chemotherapy in a melanoma mouse model. PLoS One 2013, 8, e55993. [Google Scholar]
- Williams, G.H.; Stoeber, K. The cell cycle and cancer. J. Pathol. 2012, 226, 352–364. [Google Scholar] [CrossRef]
- Sherr, C.J. G1 phase progression: Cycling on cue. Cell 1994, 79, 551–555. [Google Scholar] [CrossRef]
- Collins, K.; Jacks, T.; Pavletich, N.P. The cell cycle and cancer. Proc. Natl. Acad. Sci. USA 1997, 94, 2776–2778. [Google Scholar] [CrossRef]
- Aliyu, M.; Odunola, O.A.; Farooq, A.D.; Mesaik, A.M.; Choudhary, M.I.; Fatima, B.; Qureshi, T.A.; Erukainure, O.L. Acacia honey modulates cell cycle progression, pro-inflammatory cytokines and calcium ions secretion in PC-3 cell line. J. Cancer Sci. Ther. 2012, 4, 401–407. [Google Scholar]
- Vidya Priyadarsini, R.; Senthil Murugan, R.; Maitreyi, S.; Ramalingam, K.; Karunagaran, D.; Nagini, S. The flavonoid quercetin induces cell cycle arrest and mitochondria-mediated apoptosis in human cervical cancer (HeLa) cells through p53 induction and NF-kappaB inhibition. Eur. J. Pharmacol. 2010, 649, 84–91. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, X.H.; Wang, Z.J. Flavones and flavonols exert cytotoxic effects on a human oesophageal adenocarcinoma cell line (OE33) by causing G2/M arrest and inducing apoptosis. Food Chem. Toxicol. 2008, 46, 2042–2053. [Google Scholar] [CrossRef]
- Mu, C.; Jia, P.; Yan, Z.; Liu, X.; Li, X.; Liu, H. Quercetin induces cell cycle G1 arrest through elevating Cdk inhibitors p21 and p27 in human hepatoma cell line (HepG2). Methods Find. Exp. Clin. Pharmacol. 2007, 29, 179–183. [Google Scholar] [CrossRef]
- Song, W.; Dang, Q.; Xu, D.; Chen, Y.; Zhu, G.; Wu, K.; Zeng, J.; Long, Q.; Wang, X.; He, D.; et al. Kaempferol induces cell cycle arrest and apoptosis in renal cell carcinoma through EGFR/p38 signaling. Oncol. Rep. 2014, 31, 1350–1356. [Google Scholar]
- Green, D.R.; Kroemer, G. The pathophysiology of mitochondrial cell death. Science 2004, 305, 626–629. [Google Scholar] [CrossRef]
- Crow, M.T.; Mani, K.; Nam, Y.J.; Kitsis, R.N. The mitochondrial death pathway and cardiac myocyte apoptosis. Circ. Res. 2004, 95, 957–970. [Google Scholar] [CrossRef]
- Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Multiple pathways of cytochrome c release from mitochondria in apoptosis. Biochim. Biophys. Acta 2006, 1757, 639–647. [Google Scholar] [CrossRef]
- Mouria, M.; Gukovskaya, A.S.; Jung, Y.; Buechler, P.; Hines, O.J.; Reber, H.A.; Pandol, S.J. Food-derived polyphenols inhibit pancreatic cancer growth through mitochondrial cytochrome C release and apoptosis. Int. J. Cancer 2002, 98, 761–769. [Google Scholar] [CrossRef]
- Ren, J.; Cheng, H.; Xin, W.Q.; Chen, X.; Hu, K. Induction of apoptosis by 7-piperazinethylchrysin in HCT-116 human colon cancer cells. Oncol. Rep. 2012, 28, 1719–1726. [Google Scholar]
- Chipuk, J.E.; Bouchier-Hayes, L.; Green, D.R. Mitochondrial outer membrane permeabilization during apoptosis: The innocent bystander scenario. Cell Death Differ. 2006, 13, 1396–1402. [Google Scholar] [CrossRef]
- Chien, S.Y.; Wu, Y.C.; Chung, J.G.; Yang, J.S.; Lu, H.F.; Tsou, M.F.; Wood, W.G.; Kuo, S.J.; Chen, D.R. Quercetin-induced apoptosis acts through mitochondrial- and caspase-3-dependent pathways in human breast cancer MDA-MB-231 cells. Hum. Exp. Toxicol. 2009, 28, 493–503. [Google Scholar] [CrossRef]
- Fadeel, B.; Orrenius, S. Apoptosis: A basic biological phenomenon with wide-ranging implications in human disease. J. Intern. Med. 2005, 258, 479–517. [Google Scholar] [CrossRef]
- O’Connor, O. Apoptosis: From biology to therapeutic targeting. Ann. Oncol. 2011, 22, iv76–iv79. [Google Scholar]
- Er, E.; Oliver, L.; Cartron, P.F.; Juin, P.; Manon, S.; Vallette, F.M. Mitochondria as the target of the pro-apoptotic protein Bax. Biochim. Biophys. Acta 2006, 1757, 1301–1311. [Google Scholar] [CrossRef]
- Tait, S.W.; Green, D.R. Mitochondria and cell death: Outer membrane permeabilization and beyond. Nat. Rev. Mol. Cell Biol. 2010, 11, 621–632. [Google Scholar] [CrossRef]
- Wang, Y.; Kim, N.S.; Haince, J.F.; Kang, H.C.; David, K.K.; Andrabi, S.A.; Poirier, G.G.; Dawson, V.L.; Dawson, T.M. Poly(ADP-ribose) (PAR) binding to apoptosis-inducing factor is critical for PAR polymerase-1-dependent cell death (parthanatos). Sci. Signal. 2011, 4, ra20. [Google Scholar]
- Saxena, S.; Gautam, S.; Maru, G.; Kawle, D.; Sharma, A. Suppression of error prone pathway is responsible for antimutagenic activity of honey. Food Chem. Toxicol. 2012, 50, 625–633. [Google Scholar] [CrossRef]
- Vogelstein, B.; Sur, S.; Prives, C. p53: The most frequently altered gene in human cancers. Nat. Educ. 2010, 3, 6. [Google Scholar]
- Rivlin, N.; Brosh, R.; Oren, M.; Rotter, V. Mutations in the p53 tumor suppressor gene: Important milestones at the various steps of tumorigenesis. Genes Cancer 2011, 2, 466–474. [Google Scholar] [CrossRef]
- Beckerman, R.; Prives, C. Transcriptional regulation by p53. Cold Spring Harb. Perspect. Biol. 2010, 2, a000935. [Google Scholar]
- Angst, E.; Park, J.L.; Moro, A.; Lu, Q.Y.; Lu, X.; Li, G.; King, J.; Chen, M.; Reber, H.A.; Go, V.L.; et al. The flavonoid quercetin inhibits pancreatic cancer growth in vitro and in vivo. Pancreas 2013, 42, 223–229. [Google Scholar] [CrossRef]
- Duo, J.; Ying, G.G.; Wang, G.W.; Zhang, L. Quercetin inhibits human breast cancer cell proliferation and induces apoptosis via Bcl-2 and Bax regulation. Mol. Med. Rep. 2012, 5, 1453–1456. [Google Scholar]
- Pichichero, E.; Cicconi, R.; Mattei, M.; Canini, A. Chrysin-induced apoptosis is mediated through p38 and Bax activation in B16-F1 and A375 melanoma cells. Int. J. Oncol. 2011, 38, 473–483. [Google Scholar]
- Placzek, W.J.; Wei, J.; Kitada, S.; Zhai, D.; Reed, J.C.; Pellecchia, M. A survey of the anti-apoptotic Bcl-2 subfamily expression in cancer types provides a platform to predict the efficacy of Bcl-2 antagonists in cancer therapy. Cell Death Dis. 2010, 1, e40. [Google Scholar] [CrossRef]
- Reed, J.C. Dysregulation of apoptosis in cancer. J. Clin. Oncol. 1999, 17, 2941–2953. [Google Scholar]
- Sauer, H.; Wartenberg, M.; Hescheler, J. Reactive oxygen species as intracellular messengers during cell growth and differentiation. Cell. Physiol. Biochem. 2001, 11, 173–186. [Google Scholar] [CrossRef]
- Bhagat, S.S.; Ghone, R.A.; Suryakar, A.N.; Hundekar, P.S. Lipid peroxidation and antioxidant vitamin status in colorectal cancer patients. Indian J. Physiol. Pharmacol. 2011, 55, 72–76. [Google Scholar]
- Balestrieri, M.L.; Dicitore, A.; Benevento, R.; di Maio, M.; Santoriello, A.; Canonico, S.; Giordano, A.; Stiuso, P. Interplay between membrane lipid peroxidation, transglutaminase activity, and cyclooxygenase 2 expression in the tissue adjoining to breast cancer. J. Cell. Physiol. 2012, 227, 1577–1582. [Google Scholar] [CrossRef]
- Peddireddy, V.; Siva Prasad, B.; Gundimeda, S.D.; Penagaluru, P.R.; Mundluru, H.P. Assessment of 8-oxo-7, 8-dihydro-2'-deoxyguanosine and malondialdehyde levels as oxidative stress markers and antioxidant status in non-small cell lung cancer. Biomarkers 2012, 17, 261–268. [Google Scholar] [CrossRef]
- Epplein, M.; Franke, A.A.; Cooney, R.V.; Morris, J.S.; Wilkens, L.R.; Goodman, M.T.; Murphy, S.P.; Henderson, B.E.; Kolonel, L.N.; Marchand, L. Association of plasma micronutrient levels and urinary isoprostane with risk of lung cancer: The multiethnic cohort study. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1962–1970. [Google Scholar] [CrossRef]
- Asombang, A.W.; Kayamba, V.; Mwanza-Lisulo, M.; Colditz, G.; Mudenda, V.; Yarasheski, K.; Chott, R.; Rubin, D.C.; Gyawali, C.P.; Sinkala, E.; et al. Gastric cancer in Zambian adults: A prospective case-control study that assessed dietary intake and antioxidant status by using urinary isoprostane excretion. Am. J. Clin. Nutr. 2013, 97, 1029–1035. [Google Scholar] [CrossRef]
- Yang, Y.; Karakhanova, S.; Werner, J.; Bazhin, A.V. Reactive oxygen species in cancer biology and anticancer therapy. Curr. Med. Chem. 2013, 20, 3677–3692. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S. Evidence in support of potential applications of lipid peroxidation products in cancer treatment. Oxid. Med. Cell. Longev. 2013, 2013, 931251. [Google Scholar]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Honey: A novel antioxidant. Molecules 2012, 17, 4400–4423. [Google Scholar] [CrossRef]
- Alzahrani, H.A.; Boukraa, L.; Bellik, Y.; Abdellah, F.; Bakhotmah, B.A.; Kolayli, S.; Sahin, H. Evaluation of the antioxidant activity of three varieties of honey from different botanical and geographical origins. Glob. J. Health Sci. 2012, 4, 191–196. [Google Scholar]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.; Salam, S.K.; Salleh, M.S.; Gurtu, S. Antioxidant protective effect of glibenclamide and metformin in combination with honey in pancreas of streptozotocin-induced diabetic rats. Int. J. Mol. Sci. 2010, 11, 2056–2066. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.; Sirajudeen, K.N.; Salleh, M.S.; Gurtu, S. Antioxidant protection of Malaysian tualang honey in pancreas of normal and streptozotocin-induced diabetic rats. Ann. Endocrinol. (Paris) 2010, 71, 291–296. [Google Scholar] [CrossRef]
- Aoshima, H.; Ayabe, S. Prevention of the deterioration of polyphenol-rich beverages. Food Chem. 2007, 100, 350–355. [Google Scholar] [CrossRef]
- Zubair, H.; Khan, H.Y.; Sohail, A.; Azim, S.; Ullah, M.F.; Ahmad, A.; Sarkar, F.H.; Hadi, S.M. Redox cycling of endogenous copper by thymoquinone leads to ROS-mediated DNA breakage and consequent cell death: Putative anticancer mechanism of antioxidants. Cell Death Dis. 2013, 4, e660. [Google Scholar] [CrossRef]
- Azmi, A.S.; Bhat, S.H.; Hanif, S.; Hadi, S.M. Plant polyphenols mobilize endogenous copper in human peripheral lymphocytes leading to oxidative DNA breakage: A putative mechanism for anticancer properties. FEBS Lett. 2006, 580, 533–538. [Google Scholar]
- Bishayee, K.; Ghosh, S.; Mukherjee, A.; Sadhukhan, R.; Mondal, J.; Khuda-Bukhsh, A.R. Quercetin induces cytochrome-c release and ROS accumulation to promote apoptosis and arrest the cell cycle in G2/M, in cervical carcinoma: Signal cascade and drug-DNA interaction. Cell Prolif. 2013, 46, 153–163. [Google Scholar] [CrossRef]
- Triantafillidis, J.K.; Nasioulas, G.; Kosmidis, P.A. Colorectal cancer and inflammatory bowel disease: Epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategie. Anticancer Res. 2009, 29, 2727–2737. [Google Scholar]
- Zaghloul, M.S. Bladder cancer and schistosomiasis. J. Egypt. Natl. Cancer Inst. 2012, 24, 151–159. [Google Scholar] [CrossRef]
- Shaw, R.; Robinson, M. The increasing clinical relevance of human papillomavirus type 16 (HPV-16) infection in oropharyngeal cancer. Br. J. Oral Maxillofac. Surg. 2011, 49, 423–429. [Google Scholar] [CrossRef]
- Farrow, B.; Evers, B.M. Inflammation and the development of pancreatic cancer. Surg. Oncol. 2002, 10, 153–169. [Google Scholar] [CrossRef]
- Jiang, X.; Shapiro, D.J. The immune system and inflammation in breast cancer. Mol. Cell. Endocrinol. 2014, 382, 673–682. [Google Scholar] [CrossRef]
- Wang, H.; Cho, C.H. Effect of NF-kappaB signaling on apoptosis in chronic inflammation-associated carcinogenesis. Curr. Cancer Drug Targets 2010, 10, 593–599. [Google Scholar] [CrossRef]
- Naldini, A.; Carraro, F. Role of inflammatory mediators in angiogenesis. Curr. Drug Targets Inflamm. Allergy 2005, 4, 3–8. [Google Scholar] [CrossRef]
- Goldberg, J.E.; Schwertfeger, K.L. Proinflammatory cytokines in breast cancer: Mechanisms of action and potential targets for therapeutics. Curr. Drug Targets 2010, 11, 1133–1146. [Google Scholar] [CrossRef]
- Steele, C.W.; Jamieson, N.B.; Evans, T.R.; McKay, C.J.; Sansom, O.J.; Morton, J.P.; Carter, C.R. Exploiting inflammation for therapeutic gain in pancreatic cancer. Br. J. Cancer 2013, 108, 997–1003. [Google Scholar]
- Batumalaie, K.; Zaman Safi, S.; Mohd Yusof, K.; Shah Ismail, I.; Devi Sekaran, S.; Qvist, R. Effect of gelam honey on the oxidative stress-induced signaling pathways in pancreatic hamster cells. Int. J. Endocrinol. 2013, 2013. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.N.; Huang, J.M.; Xiong, X.K.; Chen, M.F.; Ong, C.N.; Shen, H.M.; Yang, X.F. Chrysin promotes tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) induced apoptosis in human cancer cell lines. Toxicol. In Vitro 2011, 25, 630–635. [Google Scholar] [CrossRef]
- Leong, A.G.; Herst, P.M.; Harper, J.L. Indigenous New Zealand honeys exhibit multiple anti-inflammatory activities. Innate Immun. 2012, 18, 459–466. [Google Scholar] [CrossRef]
- Hussein, S.Z.; Mohd Yusoff, K.; Makpol, S.; Mohd Yusof, Y.A. Gelam honey inhibits the production of proinflammatory, mediators NO, PGE(2), TNF-alpha, and IL-6 in carrageenan-induced acute paw edema in rats. Evid.-Based Complement. Altern. Med. 2012, 2012, 109636. [Google Scholar]
- Hussein, S.Z.; Mohd Yusoff, K.; Makpol, S.; Mohd Yusof, Y.A. Gelam honey attenuates carrageenan-induced rat paw inflammation via NF-kappaB pathway. PLoS One 2013, 8, e72365. [Google Scholar]
- Abubakar, M.B.; Abdullah, W.Z.; Sulaiman, S.A.; Suen, A.B. A review of molecular mechanisms of the anti-leukemic effects of phenolic compounds in honey. Int. J. Mol. Sci. 2012, 13, 15054–15073. [Google Scholar] [CrossRef]
- Rao, C.V.; Desai, D.; Simi, B.; Kulkarni, N.; Amin, S.; Reddy, B.S. Inhibitory effect of caffeic acid esters on azoxymethane-induced biochemical changes and aberrant crypt foci formation in rat colon. Cancer Res. 1993, 53, 4182–4188. [Google Scholar]
- Coughlin, S.S.; Calle, E.E.; Teras, L.R.; Petrelli, J.; Thun, M.J. Diabetes mellitus as a predictor of cancer mortality in a large cohort of US adults. Am. J. Epidemiol. 2004, 159, 1160–1167. [Google Scholar]
- Vigneri, P.; Frasca, F.; Sciacca, L.; Frittitta, L.; Vigneri, R. Obesity and cancer. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 1–7. [Google Scholar] [CrossRef]
- Fair, A.M.; Dai, Q.; Shu, X.O.; Matthews, C.E.; Yu, H.; Jin, F.; Gao, Y.T.; Zheng, W. Energy balance, insulin resistance biomarkers, and breast cancer risk. Cancer Detect. Prev. 2007, 31, 214–219. [Google Scholar] [CrossRef] [Green Version]
- Belfiore, A.; Malaguarnera, R. Insulin receptor and cancer. Endocr. Relat. Cancer 2011, 18, R125–R147. [Google Scholar] [CrossRef]
- Vincent, E.E.; Elder, D.J.; Curwen, J.; Kilgour, E.; Hers, I.; Tavare, J.M. Targeting non-small cell lung cancer cells by dual inhibition of the insulin receptor and the insulin-like growth factor-1 receptor. PLoS One 2013, 8, e66963. [Google Scholar]
- Carnero, A.; Blanco-Aparicio, C.; Renner, O.; Link, W.; Leal, J.F. The PTEN/PI3K/AKT signalling pathway in cancer, therapeutic implications. Curr. Cancer Drug Targets 2008, 8, 187–198. [Google Scholar] [CrossRef]
- Mehlen, P.; Puisieux, A. Metastasis: A question of life or death. Nat. Rev. Cancer 2006, 6, 449–458. [Google Scholar] [CrossRef]
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef]
- Hoff, P.M.; Machado, K.K. Role of angiogenesis in the pathogenesis of cancer. Cancer Treat. Rev. 2012, 38, 825–833. [Google Scholar] [CrossRef]
- Somani, R.R.; Bhanushali, U.V. Targeting angiogenesis for treatment of human cancer. Indian J. Pharm. Sci. 2013, 75, 3–10. [Google Scholar] [CrossRef]
- Rossiter, K.; Cooper, A.J.; Voegeli, D.; Lwaleed, B.A. Honey promotes angiogeneic activity in the rat aortic ring assay. J. Wound Care 2010, 19, 442–446. [Google Scholar]
- Al-Waili, N.; Salom, K.; Al-Ghamdi, A.A. Honey for wound healing, ulcers, and burns; data supporting its use in clinical practice. Sci. World J. 2011, 11, 766–787. [Google Scholar] [CrossRef]
- Khiati, B.; Bacha, S.; Ahmed, M.; Aissat, S.; Meslem, A.; Djebli, N. Wound care with euphorbia honey after nucleation: A case report. Clin. Microbial. 2013, 2, 129. [Google Scholar]
- Yasuda, M.; Ohzeki, Y.; Shimizu, S.; Naito, S.; Ohtsuru, A.; Yamamoto, T.; Kuroiwa, Y. Stimulation of in vitro angiogenesis by hydrogen peroxide and the relation with ETS-1 in endothelial cells. Life Sci. 1999, 64, 249–258. [Google Scholar]
- Oak, M.H.; El Bedoui, J.; Schini-Kerth, V.B. Antiangiogenic properties of natural polyphenols from red wine and green tea. J. Nutr. Biochem. 2005, 16, 1–8. [Google Scholar] [CrossRef]
- Davalli, P.; Rizzi, F.; Caporali, A.; Pellacani, D.; Davoli, S.; Bettuzzi, S.; Brausi, M.; D’Arca, D. Anticancer activity of green tea polyphenols in prostate gland. Oxid. Med. Cell. Longev. 2012, 2012. [Google Scholar] [CrossRef]
- Spatafora, C.; Tringali, C. Natural-derived polyphenols as potential anticancer agents. Anticancer Agents Med. Chem. 2012, 12, 902–918. [Google Scholar] [CrossRef]
- Sawicka, D.; Car, H.; Borawska, M.H.; Niklinski, J. The anticancer activity of propolis. Folia Histochem. Cytobiol. 2012, 50, 25–37. [Google Scholar] [CrossRef]
- Ahn, M.R.; Kunimasa, K.; Ohta, T.; Kumazawa, S.; Kamihira, M.; Kaji, K.; Uto, Y.; Hori, H.; Nagasawa, H.; Nakayama, T. Suppression of tumor-induced angiogenesis by Brazilian propolis: Major component artepillin C inhibits in vitro tube formation and endothelial cell proliferation. Cancer Lett. 2007, 252, 235–243. [Google Scholar] [CrossRef]
- Hwang, H.J.; Park, H.J.; Chung, H.J.; Min, H.Y.; Park, E.J.; Hong, J.Y.; Lee, S.K. Inhibitory effects of caffeic acid phenethyl ester on cancer cell metastasis mediated by the down-regulation of matrix metalloproteinase expression in human HT1080 fibrosarcoma cells. J. Nutr. Biochem. 2006, 17, 356–362. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A. Effects of Honey and Its Mechanisms of Action on the Development and Progression of Cancer. Molecules 2014, 19, 2497-2522. https://doi.org/10.3390/molecules19022497
Erejuwa OO, Sulaiman SA, Wahab MSA. Effects of Honey and Its Mechanisms of Action on the Development and Progression of Cancer. Molecules. 2014; 19(2):2497-2522. https://doi.org/10.3390/molecules19022497
Chicago/Turabian StyleErejuwa, Omotayo O., Siti A. Sulaiman, and Mohd S. Ab Wahab. 2014. "Effects of Honey and Its Mechanisms of Action on the Development and Progression of Cancer" Molecules 19, no. 2: 2497-2522. https://doi.org/10.3390/molecules19022497
APA StyleErejuwa, O. O., Sulaiman, S. A., & Wahab, M. S. A. (2014). Effects of Honey and Its Mechanisms of Action on the Development and Progression of Cancer. Molecules, 19(2), 2497-2522. https://doi.org/10.3390/molecules19022497





