Influence of Extractive Solvents on Lipid and Fatty Acids Content of Edible Freshwater Algal and Seaweed Products, the Green Microalga Chlorella kessleri and the Cyanobacterium Spirulina platensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Lipid Contents
| Sample † | Total lipid content [%] | |||
|---|---|---|---|---|
| Mean 1 | SD 1 | Mean 2 | SD 2 | |
| S | 10.23 a | 0.59 | 3.50 b | 0.37 |
| SP | 18.02 a | 1.15 | 13.41 b | 0.79 |
| C | 3.70 a | 0.31 | 3.41 a | 0.32 |
| CK | 18.01 a | 1.15 | 9.87 b | 0.89 |
| N | 1.61 a | 0.10 | 0.93 b | 0.13 |
| D | 1.32 a | 0.18 | 0.64 b | 0.10 |
| K | 3.11 a | 0.26 | 1.83 b | 0.19 |
| A | 2.02 a | 0.22 | 1.58 b | 0.12 |
| W | 1.66 a | 0.15 | 0.73 b | 0.17 |
| WI | 3.31 a | 0.34 | 1.11 b | 0.15 |
| H | 1.55 a | 0.16 | 1.17 b | 0.08 |
2.2. Influence of Extractive Solvents on the Number and Amount of Identified Fatty Acids
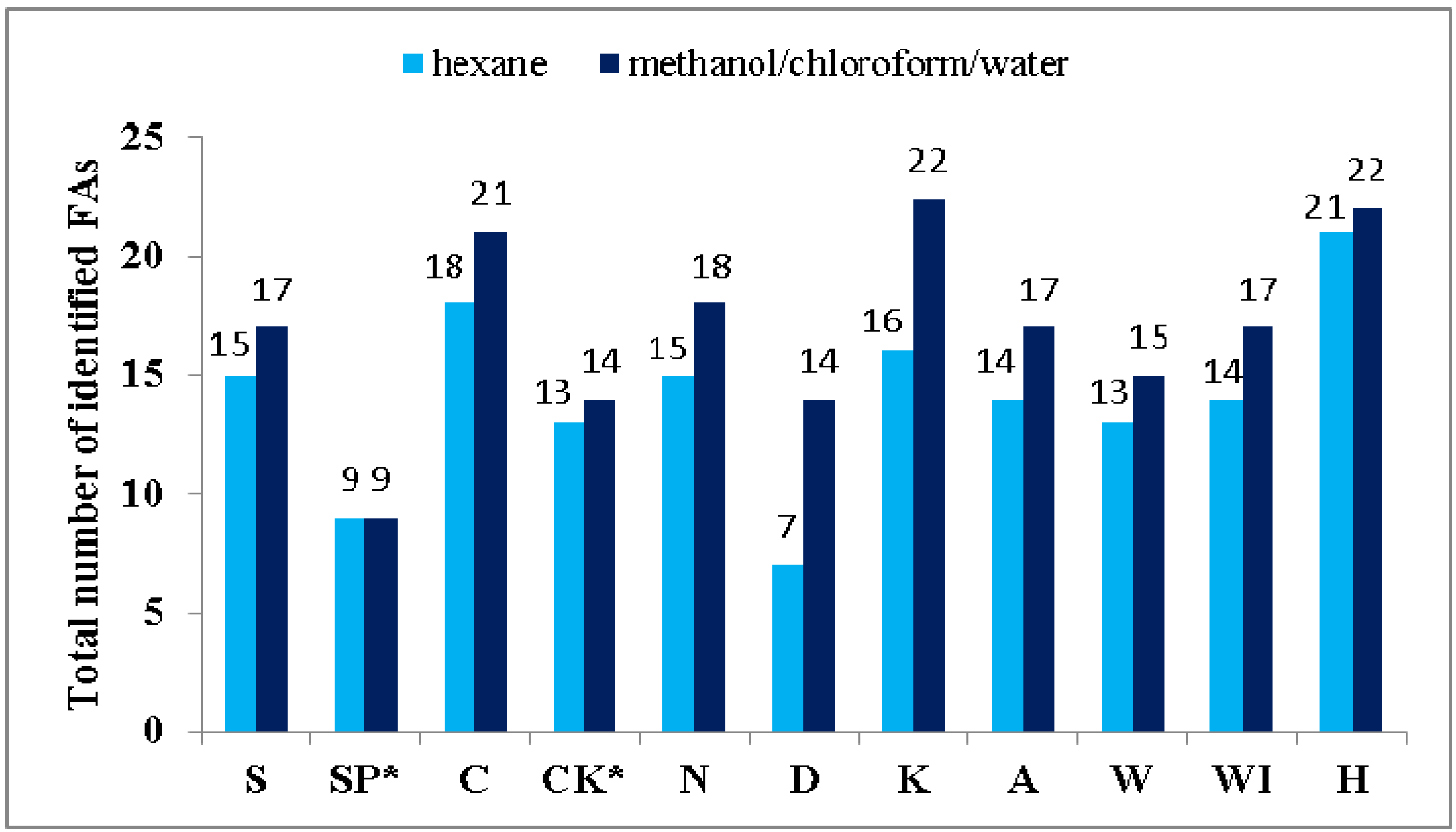
2.3. Fatty Acid Profiles
| Cyanobacteria | Green microalgae | Red seaweeds | Brown seaweeds | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| FAs | S | SP * | C | CK * | N | D | K | A | W | WI | H |
| C10:0 | 0.11 | nd | 0.20 | nd | 1.40 | 0.57 | 0.04 | 0.13 | 0.10 | 0.09 | 0.16 |
| C12:0 | 0.21 | nd | 0.08 | 0.11 | 0.29 | 0.58 | 0.05 | 0.08 | 0.10 | 0.11 | 0.13 |
| C14:0 | 0.56 | 0.46 | 0.44 | 1.14 | 1.99 | 12.32 | 5.82 | 8.18 | 5.88 | 5.16 | 5.09 |
| C15:0 | 0.10 | nd | 0.12 | 0.72 | 0.88 | 1.79 | 0.20 | 0.59 | 0.44 | 0.33 | 0.45 |
| C16:0 | 53.02 | 61.06 | 25.59 | 24.64 | 57.61 | 65.49 | 31.21 | 31.10 | 33.38 | 31.05 | 33.80 |
| C17:0 | 0.28 | 0.27 | 0.36 | 0.29 | 0.28 | 0.45 | 0.20 | 0.28 | 0.36 | 0.34 | 0.39 |
| C18:0 | 4.75 | 1.40 | 9.48 | 0.74 | 1.40 | 3.45 | 5.00 | 0.96 | 2.04 | 1.43 | 1.19 |
| C20:0 | 0.06 | nd | 0.09 | nd | nd | nd | 0.67 | 1.36 | 0.78 | 0.65 | 0.19 |
| C21:0 | nd | nd | nd | nd | 0.36 | nd | nd | nd | nd | nd | 0.92 |
| C22:0 | nd | nd | 0.16 | nd | 1.71 | nd | nd | nd | nd | nd | 0.49 |
| C24:0 | 0.38 | nd | 0.47 | 1.23 | nd | 1.95 | 4.06 | 3.42 | nd | nd | 4.68 |
| C16:1(n-7) | 3.36 | 2.59 | 1.49 | 1.61 | 2.20 | 5.01 | 0.79 | 10.04 | 1.80 | 1.04 | 5.25 |
| C17:1(n-7) | 0.50 | nd | 0.15 | nd | 0.42 | nd | 0.09 | 0.57 | nd | 0.13 | 0.44 |
| C18:1trans (n-9) | 0.08 | nd | 5.05 | 2.50 | 1.49 | nd | 0.20 | nd | nd | nd | nd |
| C18:1cis (n-9) | 2.79 | 5.29 | 8.06 | 5.50 | 3.79 | 5.29 | 26.45 | 13.39 | 13.32 | 11.91 | 10.00 |
| C20:1(n-9) | nd | nd | nd | nd | 3.98 | nd | 2.83 | 3.54 | nd | nd | 9.86 |
| C22:1(n-9) | nd | nd | nd | nd | nd | nd | nd | nd | nd | 0.22 | 3.51 |
| C18:2cis (n-6) | 16.17 | 18.42 | 18.79 | 36.38 | 8.90 | 0.71 | 8.76 | 9.55 | 10.12 | 9.85 | 5.07 |
| C18:3(n-3) | nd | nd | 21.29 | 19.76 | 3.52 | 0.38 | 1.22 | nd | 10.48 | 17.17 | 1.46 |
| C18:3(n-6) | 17.36 | 10.44 | 7.64 | 4.74 | 8.66 | 1.96 | 1.94 | 3.02 | 5.74 | 7.52 | 2.43 |
| C20:2(n-6) | 0.11 | nd | 0.26 | 0.64 | nd | nd | 0.93 | nd | nd | nd | nd |
| C20:3(n-6) | 0.15 | nd | 0.14 | nd | nd | nd | 0.94 | 0.34 | 0.79 | 0.61 | 0.35 |
| C20:4(n-6) | nd | nd | nd | nd | 1.48 | nd | 8.57 | 13.54 | 15.37 | 12.38 | 13.97 |
| Other a | 0.00 | 0.07 | 0.13 | 0.00 | 0.00 | 0.08 | 0.05 | 0.00 | 0.00 | 0.00 | 0.16 |
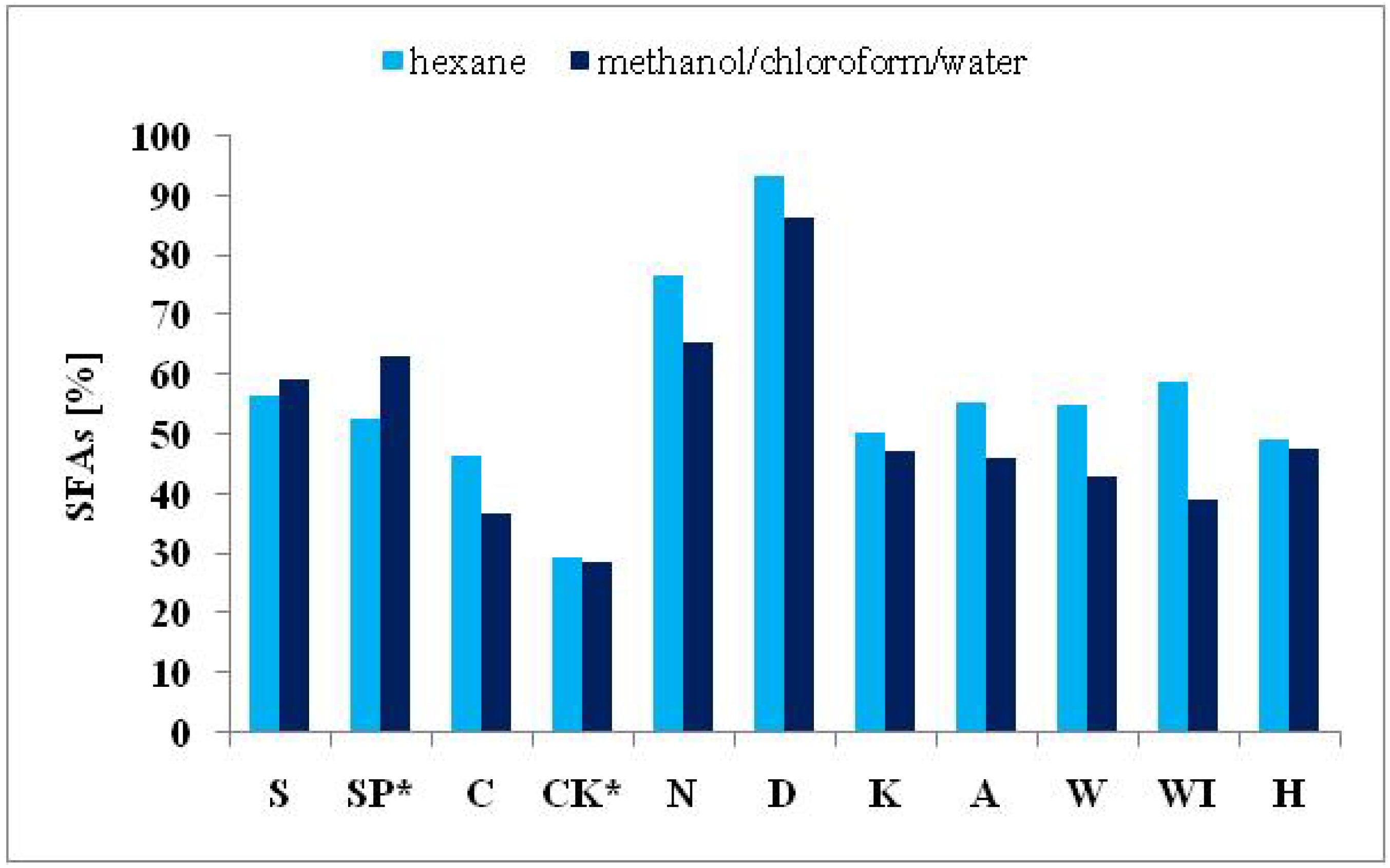
| ΣSFAs | 59.47 | 63.18 | 36.99 | 28.87 | 65.56 | 86.58 | 47.24 | 46.01 | 43.09 | 39.17 | 47.49 |
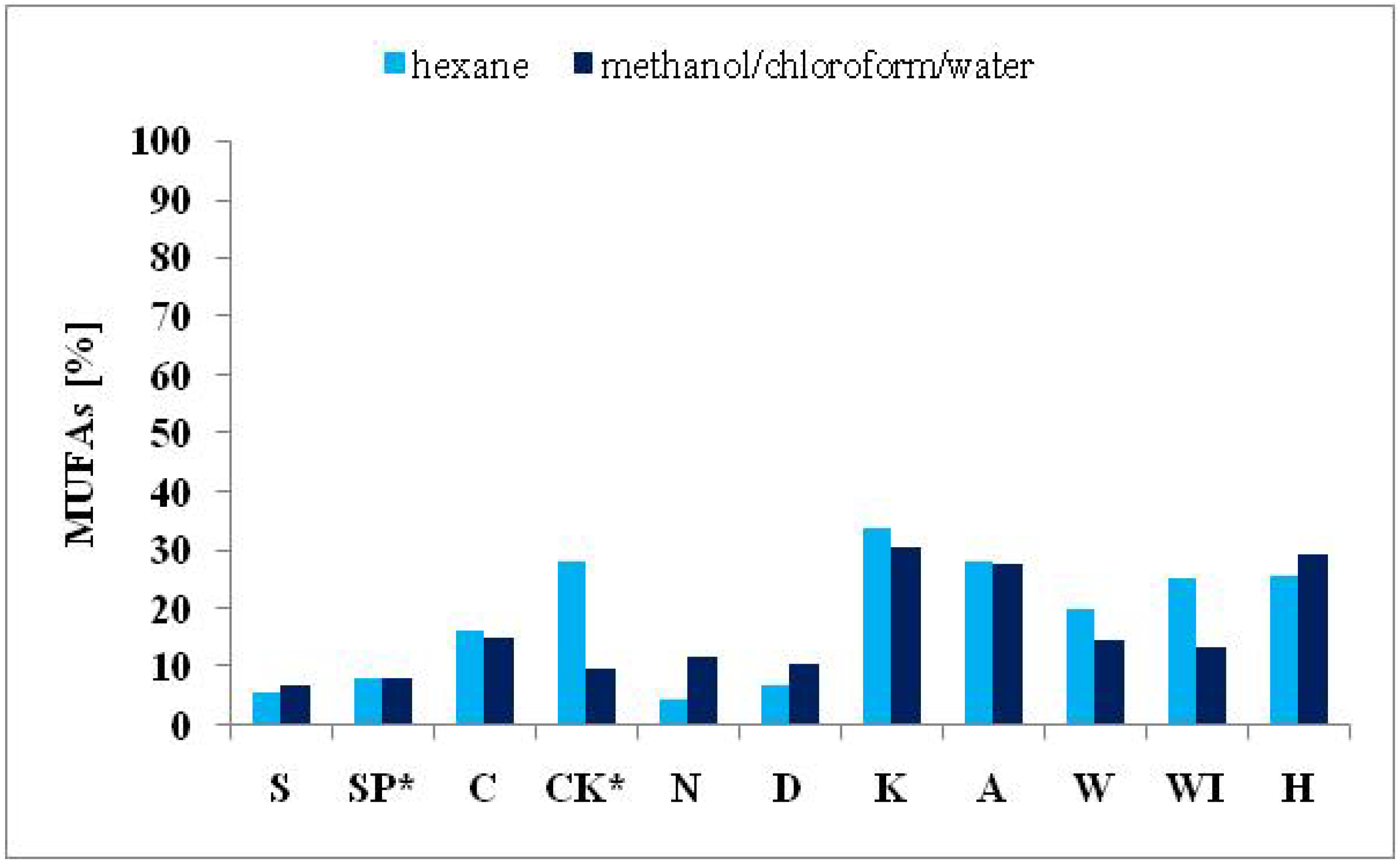
| ΣMUFAs | 6.73 | 7.96 | 14.78 | 9.61 | 11.88 | 10.38 | 30.38 | 27.54 | 14.40 | 13.30 | 29.07 |
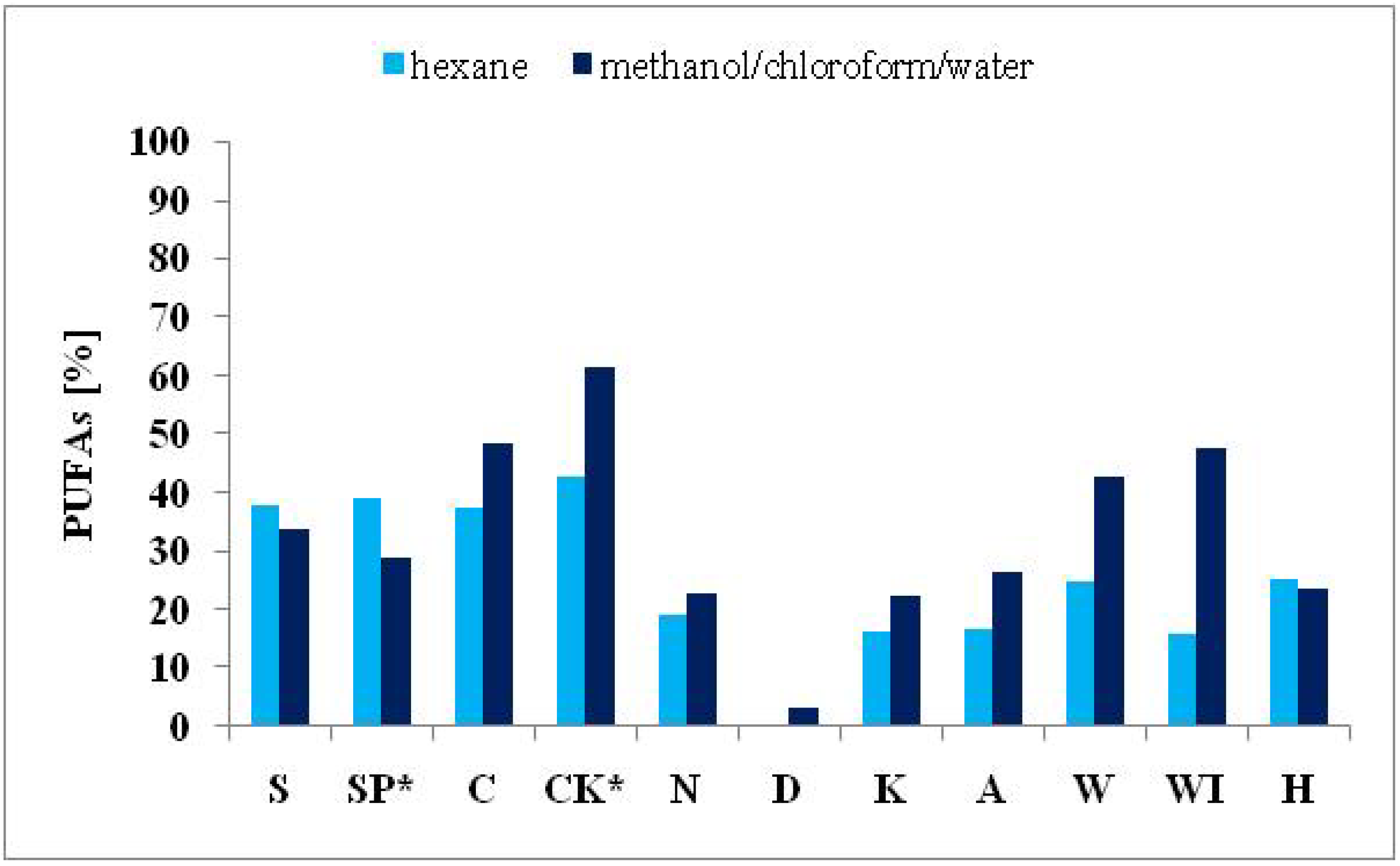
| ΣPUFAs | 33.79 | 28.86 | 48.23 | 61.52 | 22.56 | 3.04 | 22.39 | 26.45 | 42.51 | 47.53 | 23.44 |
| Σn3-FAs | 0.00 | 0.00 | 21.29 | 19.76 | 3.52 | 0.38 | 1.22 | 0.00 | 10.48 | 17.17 | 1.46 |
| Σn6-FAs | 33.79 | 28.86 | 26.93 | 41.42 | 19.04 | 2.67 | 21.17 | 26.45 | 32.02 | 30.36 | 21.98 |
| Cyanobacteria | Green microalgae | Red seaweeds | Brown seaweeds | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| FAs | S | SP * | C | CK * | N | D | K | A | W | WI | H |
| C10:0 | 0.19 | nd | 0.09 | nd | 0.58 | 3.73 | 0.16 | 0.42 | 0.71 | 0.84 | 0.30 |
| C12:0 | 0.36 | 0.10 | 0.17 | 0.47 | 0.96 | 2.45 | nd | nd | 0.29 | 0.60 | 0.17 |
| C14:0 | 0.71 | 0.48 | 0.57 | 2.25 | 1.97 | 13.16 | 6.67 | 9.56 | 6.64 | 7.47 | 5.49 |
| C15:0 | 0.12 | nd | 0.13 | 1.08 | 3.24 | nd | 0.13 | 0.71 | nd | 0.78 | 0.50 |
| C16:0 | 44.85 | 50.43 | 27.53 | 20.33 | 46.50 | 48.82 | 34.67 | 38.35 | 35.49 | 36.93 | 34.36 |
| C17:0 | 0.43 | 0.51 | 0.39 | 1.37 | 3.81 | 13.17 | 0.32 | 1.04 | 1.85 | 0.42 | 0.54 |
| C18:0 | 9.90 | 1.30 | 16.83 | 3.79 | 7.58 | 11.93 | 5.55 | 2.97 | 8.61 | 6.71 | 1.31 |
| C20:0 | 0.12 | nd | 0.16 | nd | 3.23 | nd | 0.71 | 1.50 | 1.42 | 0.57 | 0.40 |
| C21:0 | nd | nd | 0.03 | nd | nd | nd | nd | 0.91 | nd | nd | 0.99 |
| C22:0 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 1.36 |
| C24:0 | nd | nd | 0.65 | nd | 8.70 | nd | 2.20 | nd | nd | 4.53 | 3.91 |
| C16:1(n-7) | 2.56 | 3.10 | 1.52 | 1.36 | 1.28 | nd | 0.75 | 9.44 | 2.76 | 1.10 | 2.67 |
| C17:1(n-7) | 0.42 | nd | 0.10 | nd | 0.21 | nd | nd | nd | nd | nd | 0.33 |
| C18:1trans (n-9) | 0.08 | nd | 3.44 | 1.91 | 0.04 | nd | 0.20 | nd | nd | nd | nd |
| C18:1cis (n-9) | 2.46 | 5.13 | 11.00 | 7.86 | 2.87 | 6.74 | 30.49 | 16.28 | 9.35 | 15.30 | 11.28 |
| C20:1(n-9) | nd | nd | nd | nd | nd | nd | 2.11 | 2.11 | 7.95 | 8.85 | 8.46 |
| C22:1(n-9) | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 2.78 |
| C18:2cis (n-6) | 13.65 | 23.73 | 16.12 | 25.89 | 8.44 | nd | 8.30 | 7.94 | 4.92 | 9.88 | 5.60 |
| C18:3(n-3) | nd | nd | 13.41 | 10.86 | nd | nd | nd | nd | nd | nd | 1.97 |
| C18:3(n-6) | 23.98 | 15.22 | 7.78 | 5.95 | 10.60 | nd | 1.71 | 2.47 | 19.16 | nd | 2.28 |
| C20:2(n-6) | 0.16 | nd | 0.08 | nd | nd | nd | nd | nd | nd | nd | nd |
| C20:3(n-6) | nd | nd | nd | nd | nd | nd | 0.62 | nd | nd | nd | 0.91 |
| C20:4(n-6) | nd | nd | nd | nd | nd | nd | 5.41 | 6.29 | 0.85 | 6.03 | 14.39 |
| Other a | 0.00 | 0.00 | 0.00 | 16.88 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| ΣSFAs | 56.70 | 52.82 | 46.56 | 29.29 | 76.56 | 93.26 | 50.41 | 55.46 | 55.02 | 58.84 | 49.34 |
| ΣMUFAs | 5.51 | 8.23 | 16.06 | 28.01 | 4.41 | 6.74 | 33.55 | 27.84 | 20.05 | 25.24 | 25.52 |
| ΣPUFAs | 37.79 | 38.95 | 37.38 | 42.70 | 19.04 | 0.00 | 16.03 | 16.70 | 24.93 | 15.91 | 25.15 |
| Σn3-FAs | 0.00 | 0.00 | 13.41 | 10.86 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 1.97 |
| Σn6-FAs | 37.79 | 38.95 | 23.97 | 31.84 | 19.04 | 0.00 | 16.03 | 16.70 | 24.93 | 15.91 | 23.18 |
2.3.1. Saturated Fatty Acids
2.3.2. Monounsaturated Fatty Acids
2.3.3. Polyunsaturated Fatty Acids
2.4. Fatty Acid Profiles of Autotrophically Cultivated Cyanobacteria and Microalga
2.5. PUFAs/SFAs Fatty Acids Ratio
3. Experimental
3.1. Samples and Chemicals
| Algal group | Algal species | Sample | Product | Country | Form |
|---|---|---|---|---|---|
| Green | Chlorella pyrenoidosa | C | Chlorella Tabs | Taiwan | pills |
| Chlorella kessleri | CK * | - | Czech Republic | dried alga | |
| Cyanobacteria | Spirulina platensis | S | Spirulina Bio | India | pills |
| Spirulina platensis | SP * | - | Czech Republic | dried alga | |
| Red | Porphyra tenera | N | Nori flakes | Japan | dried seaweed |
| Palmaria palmata | D | Dulse flakes Bio | USA | dried seaweed | |
| Brown | Laminaria japonica | K | Kombu | Japan | dried seaweed |
| Eisenia bicyclis | A | Arame | Japan | dried seaweed | |
| Undaria pinnatifida | W | Wakame | Japan | dried seaweed | |
| Undaria pinnatifida | WI | Wakame-instant | Japan | dried seaweed | |
| Hizikia fusiformis | H | Hijiky | Japan | dried seaweed |
3.2. Total Lipids Determination
3.3. GC Analysis of FAMEs
3.4. Statistics
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Cardoso, K.H.M.; Guaratini, T.; Barros, M.P.; Falcão, V.R.; Tonon, A.P.; Lopes, N.P.; Campos, S.; Torres, M.A.; Souza, A.O.; Colepicolo, P.; et al. Metabolites from algae with economical impact. Comp. Biochem. Physiol. C 2007, 146, 60–78. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Mišurcová, L. Chemical Composition of Seaweeds. In Handbook of Marine Macroalgae: Biotechnology and Applied Phycology; John Wiley & Sons: Chichester, UK, 2011; pp. 173–192. [Google Scholar]
- Mišurcová, L.; Ambrožová, L.; Samek, D. Seaweed lipids as nutraceuticals. Adv. Food Nutr. Res. 2011, 64, 339–355. [Google Scholar] [CrossRef]
- Mišurcová, L.; Machů, L.; Orsavová, J. Seaweed minerals as nutraceuticals. Adv. Food Nutr. Res. 2011, 64, 371–390. [Google Scholar] [CrossRef]
- Mišurcová, L.; Škrovánková, S.; Samek, D.; Ambrožová, J.; Machů, L. Health benefits of algal polysaccharides in human nutrition. HAdv. Food Nutr. Res. 2012, 66, 75–146. [Google Scholar] [CrossRef]
- Juríková, T.; Balla, S.; Sochor, J.; Pohanka, M.; Mlcek, J.; Baron, M. Flavonoid profile of saskatoon berries (Amelanchier alnifolia Nutt.) and their health promoting effects. Molecules 2013, 18, 12571–12586. [Google Scholar] [CrossRef]
- Rop, O.; Balík, J.; Řezníček, V.; Juríková, T.; Škardová, P.; Salaš, P.; Sochor, J.; Mlček, J.; Kramářová, D. Chemical characteristics of fruits of some selected quince (Cydonia oblonga Mill.) cultivars. Czech. J. Food Sci. 2011, 29, 65–73. [Google Scholar]
- Van, T.T.T.; Hieu, V.M.N.; Vi, T.N.H.; Ly, B.M.; Thuy, T.T.T. Antioxidant activities and total phenolic content of macroalgae from central coast of Vietnam. Asian J. Chem. 2013, 25, 6639–6642. [Google Scholar]
- Gamal, A.A.E. Biological importance of marine algae. Saudi Pharm. J. 2010, 18, 1–25. [Google Scholar]
- Gupta, S.; Abu-Ghannam, N. Bioactive potential and possible health effects of edible brown seaweeds. Trends Food Sci. Technol. 2011, 22, 315–326. [Google Scholar] [CrossRef]
- Kucekova, Z.; Mlcek, J.; Humpolicek, P.; Rop, O. Edible flowers—Antioxidant activity and impact on cell viability. Cent. Eur. J. Biol. 2013, 8, 1023–1031. [Google Scholar] [CrossRef]
- Mlcek, J.; Rop, O. Fresh edible flowers of ornamental plants—A new source of nutraceutical foods. Trends Food Sci. Technol. 2011, 22, 561–569. [Google Scholar] [CrossRef]
- Grofová, Z. Fatty acids. (In Czech)Med. Praxi 2010, 7, 388–390. [Google Scholar]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Polat, S.; Ozogul, Y. Seasonal proximate and fatty acid variations of some seaweeds from the northeastern Mediterranean coast. Oceanologia 2013, 55, 375–391. [Google Scholar] [CrossRef]
- Badzhanov, A.S.; Abdusamatova, N.; Yusupova, F.M.; Faizullaeva, N.; Mezhlumyan, L.G.; Malikova, M.K. Chemical composition of Spirulina platensis cultivated in Uzbekistan. Chem. Nat. Compd. 2004, 40, 276–279. [Google Scholar] [CrossRef]
- D’Oca, M.G.M.; Viegas, C.V.; Lemoes, J.S.; Miyasaki, E.K.; Morón-Villarreyes, J.A.; Primel, E.G.; Abreu, P.C. Production of FAMEs from several microalgal lipidic extracts and direct transesterification of the Chlorella pyrenoidosa. Biomass Bioenergy 2011, 35, 1533–1538. [Google Scholar] [CrossRef]
- Ortega-Calvo, J.J.; Mazuelos, C.; Hermosin, B.; Saiz-Jimenez, C. Chemical composition of Spirulina and eukaryotic algae food products marked in Spain. J. Appl. Phycol. 1993, 5, 425–435. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; Lopéz-Cervantes, J.; Lopéz-Hernandéz, J.; Paseiro-Losada, P. Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Burtin, P. Nutritional value of seaweeds. Electron. J. Environ. Agric. Food Chem. 2003, 2, 498–503. [Google Scholar]
- Habib, M.A.B.; Parvin, M.; Huntington, T.C.; Hasan, M.R. A Review on Culture, Production and Use of Spirulina as Food for Humans and Feeds for Domestic Animals and Fish; FAO Fisheries and Aquaculture Circular: Rome, Italy, 2008; pp. 1–33. [Google Scholar]
- Simopoulos, A.P. The importance of the Omega-6/Omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Žák, A.; Tvrzická, E.; Zeman, M.; Vecka, M. Pathophysiology of and clinical significance of polyunsaturated fatty acids n-3 family. (In Czech)Čas. Lék. Česk. 2005, 144, 6–18. [Google Scholar]
- McHugh, D.J. A Guide to the Seaweed Industry; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003. [Google Scholar]
- Sanina, N.M.; Goncharova, S.N.; Kostetsky, E.Y. Seasonal changes of fatty acid composition and thermotropic behavior of polar lipids from marine macrophytes. Phytochemistry 2008, 69, 1517–1527. [Google Scholar] [CrossRef]
- Colla, L.M.; Bertolin, T.E.; Costa, J.A.V. Fatty acids profile of Spirulina platensis grown under different temperatures and nitrogen concentrations. Z. Naturforsch. C 2004, 59, 55–59. [Google Scholar]
- Mishra, V.K.; Temelli, F.; Ooraikul, B.; Shacklock, P.F.; Craigie, J.S. Lipids of the red alga, palmaria palmata. Bot. Mar. 1993, 36, 169–174. [Google Scholar]
- Khotimchenko, S.V.; Kulikova, I.V. Lipids of different parts of the lamina of Laminaria japonica Aresch. Bot. Mar. 2000, 43, 87–91. [Google Scholar]
- Erickson, M.C. Lipid extraction from channel catfish muscle: Comparison of solvent systems. J. Food Sci. 1993, 58, 84–89. [Google Scholar] [CrossRef]
- Hernández-Carmona, G.; Carrillo-Domínguez, S.; Arvizu-Higuera, D.L.; Rodríguez-Montesinos, Y.E.; Murillo-Álvarez, J.I.; Muñoz-Ochoa, M.; Castillo-Domínguez, R.M. Monthly variation in the chemical composition of Eisenia arborea J.E. Areschoug. J. Appl. Phycol. 2009, 21, 607–616. [Google Scholar] [CrossRef]
- Nelson, M.M.; Phleger, C.F.; Nichols, P.D. Seasonal lipid composition in macroalgae of the northeastern Pacific Ocean. Bot. Mar. 2002, 45, 58–65. [Google Scholar]
- Gressler, V.; Yokoya, N.S.; Fujii, M.T.; Colepico, P.; Filho, J.H.; Torres, R.P.; Pinto, E. Lipid, fatty acid, protein, amino acid and ash contents in four Brazilian red algae species. Food Chem. 2010, 120, 585–590. [Google Scholar] [CrossRef]
- Petkov, G.; Garcia, G. Which are fatty acids of the green alga Chlorella? Biochem. Syst. Ecol. 2007, 35, 281–285. [Google Scholar] [CrossRef]
- Fleurence, J.; Gutbier, G.; Mabeau, S.; Leray, C. Fatty acids from 11 marine macroalgae of the French Brittany coast. J. Appl. Phycol. 1994, 6, 527–532. [Google Scholar] [CrossRef]
- Khotimchenko, S.V. Fatty acids of brown algae from the Russian Far East. Phytochemistry 1998, 49, 2363–2369. [Google Scholar] [CrossRef]
- Li, X.; Fan, X.; Han, L.; Lou, Q. Fatty acids of some algae from Bohai Sea. Phytochemistry 2002, 59, 157–161. [Google Scholar] [CrossRef]
- Tvrzická, E.; Staňková, B.; Vecka, M.; Žák, A. Fatty acids 1. Occurrence and biological significance. (In Czech)Čas. Lék Čes. 2009, 148, 16–24. [Google Scholar]
- Flachs, P.; Horakova, O.; Brauner, P.; Rossmeisl, M.; Pecina, P.; Franssen-van Hal, N.; Ruzickova, J.; Sponarova, J.; Drahota, Z.; Vlcek, C.; et al. Polyunsaturated fatty acids of marine origin upregulate mitochondrial biogenesis and induce β-oxidation in white fat. Diabetologia 2005, 48, 2365–2375. [Google Scholar] [CrossRef]
- Kinsella, J.E.; Lokesh, B.; Stone, R.A. Dietary n-3 polyunsaturated fatty acids and amelioration of cardiovascular disease: Possible mechanisms. Am. J. Clin. Nutr. 1990, 52, 1–28. [Google Scholar]
- Weiss, L.A.; Barrett-Connor, E.; von Mühlen, D. Ratio of n-6 to n-3 fatty acids and bone mineral density in older adults: The Rancho Bernardo Study. Am. J. Clin. Nutr. 2005, 81, 934–938. [Google Scholar]
- Hu, F.B.; Manson, J.A.E.; Willett, W.C. Types of dietary fat and risk of coronary heart disease: A critical review. J. Am. Coll. Nutr. 2001, 20, 5–19. [Google Scholar] [CrossRef]
- Masojídek, J.; Sergejevová, M.; Rottnerová, K.; Jirka, V.; Korečko, J.; Kopecký, J.; Zaťková, I.; Torzillo, G.; Štys, D. A two-stage solar photobioreactor for cultivation of microalgae based on solar concentrators. J. Appl. Phycol. 2009, 21, 55–63. [Google Scholar] [CrossRef]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Baziere, G. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Sample Availability: All samples are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an openaccess article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ambrozova, J.V.; Misurcova, L.; Vicha, R.; Machu, L.; Samek, D.; Baron, M.; Mlcek, J.; Sochor, J.; Jurikova, T. Influence of Extractive Solvents on Lipid and Fatty Acids Content of Edible Freshwater Algal and Seaweed Products, the Green Microalga Chlorella kessleri and the Cyanobacterium Spirulina platensis. Molecules 2014, 19, 2344-2360. https://doi.org/10.3390/molecules19022344
Ambrozova JV, Misurcova L, Vicha R, Machu L, Samek D, Baron M, Mlcek J, Sochor J, Jurikova T. Influence of Extractive Solvents on Lipid and Fatty Acids Content of Edible Freshwater Algal and Seaweed Products, the Green Microalga Chlorella kessleri and the Cyanobacterium Spirulina platensis. Molecules. 2014; 19(2):2344-2360. https://doi.org/10.3390/molecules19022344
Chicago/Turabian StyleAmbrozova, Jarmila Vavra, Ladislava Misurcova, Robert Vicha, Ludmila Machu, Dusan Samek, Mojmir Baron, Jiri Mlcek, Jiri Sochor, and Tunde Jurikova. 2014. "Influence of Extractive Solvents on Lipid and Fatty Acids Content of Edible Freshwater Algal and Seaweed Products, the Green Microalga Chlorella kessleri and the Cyanobacterium Spirulina platensis" Molecules 19, no. 2: 2344-2360. https://doi.org/10.3390/molecules19022344





