Potential Mechanism of Action of meso-Dihydroguaiaretic Acid on Mycobacterium tuberculosis H37Rv
Abstract
:1. Introduction
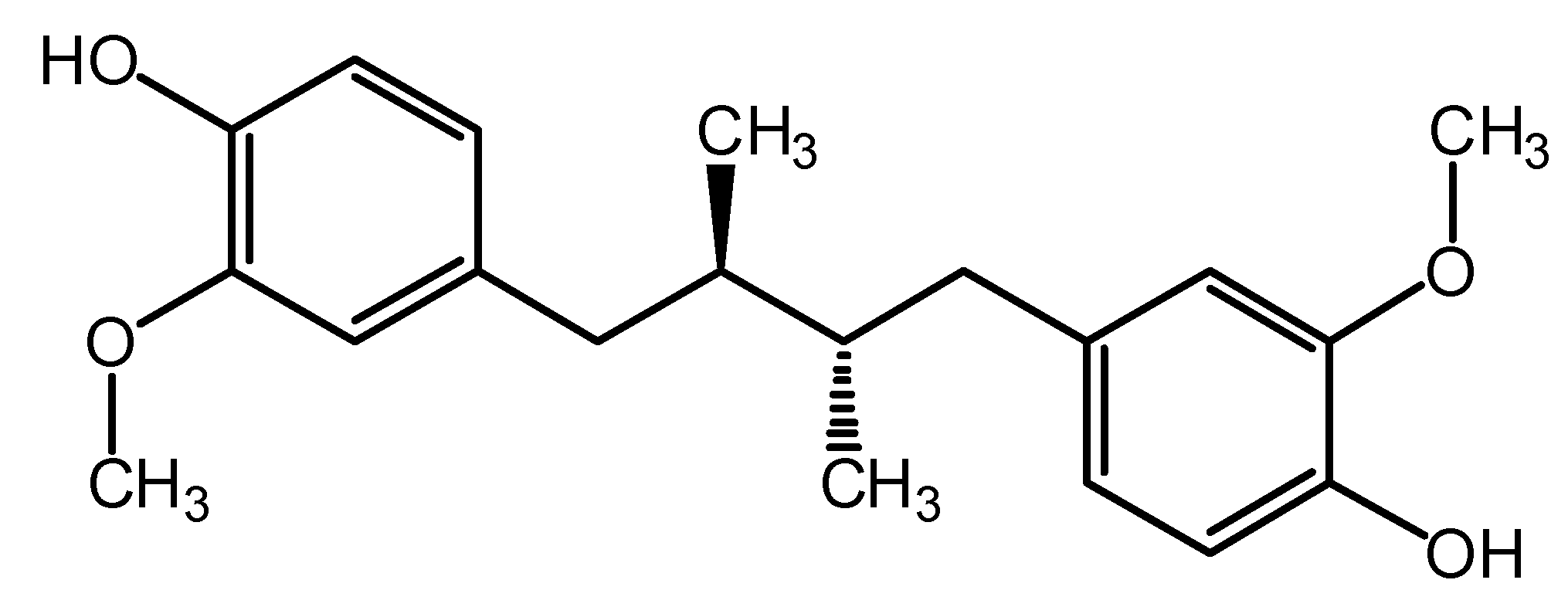
2. Results and Discussion
2.1. Growth Curve of M. tuberculosis H37Rv with Different Concentrations of MDGA
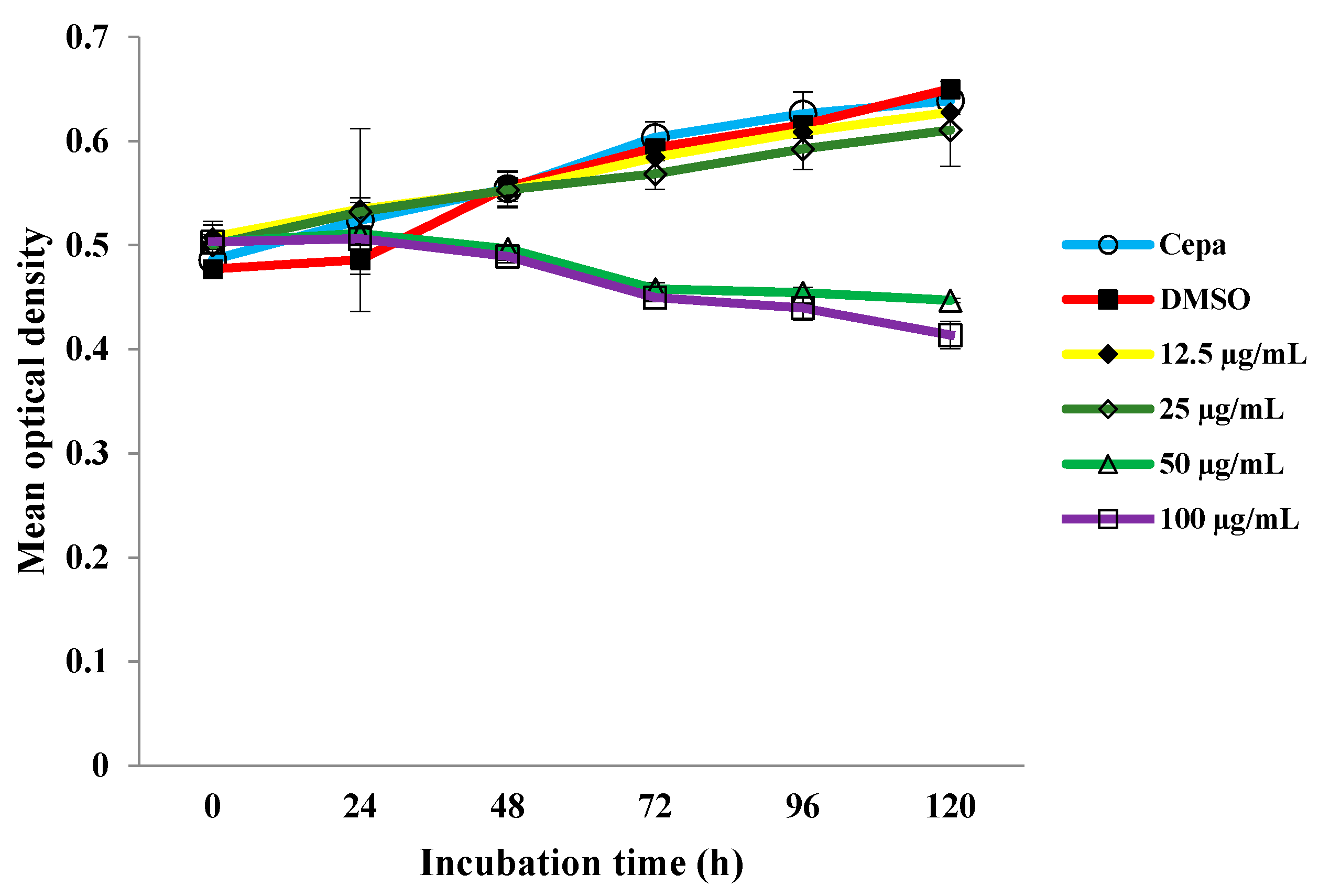
2.2. Microarray Assay and RT-PCR
| Functional Category | Up-Regulated Genes | Down-Regulated Genes |
|---|---|---|
| PE/PPE | 11.23% | --- |
| Conserved hypotheticals | 25.84% | 17.64% |
| Cell wall and cell processes | 22.47% | 29.41% |
| Intermediary metabolism and respiration | 17.99% | 35.29% |
| Regulatory proteins | 3.37% | 11.76% |
| Virulence, detoxification y adaptation | 6.74% | 5.88% |
| Information pathways | 6.74% | --- |
| Lipids metabolism | 4.49% | --- |
| Insertion sequences and phages | 1.12% | --- |
| Gen ID | Forward 5'→3' Reverse 5'→3' | Zscore | RQ |
|---|---|---|---|
| Rv3551 | CTTGAATCTCGGTGACAGCC ACCGATTTGACCAGTTCCTC | 2.473 | 2.780 ± 0.587 |
| Rv3903c | AGGAGATGCTGACTGGGAT CTAGCGCCTCATTAGGGTT | 3.549 | 18.580 ± 4.221 |
| Rv0584 | CACCCACTACGCCAATTTCT AGCGGTACCACACTGTCTC | 2.824 | 11.890 ± 0.572 |
| Rv0849 | GAGTCCTCGTCGGAAATCTG GAACCCGAGGTGAATGTGTC | 3.461 | 1.949 ± 0.261 |
| Rv0176 | GTTTGTGGGTTGGCTGTG CGGTTTGCCATTCATTGACG | 2.967 | 2.250 ± 0.3 |
| Rv1066 | GCTAGTGATCGAGCGCAA CAACCCCAAGTCCAGCA | 2.979 | 6.557 ± 0.958 |
| Rv2780 | TCCACACTCGCTACTCATCG TATCCACCAGTACCGCACCT | −3.285 | −1.950 ± 0.296 |
| Rv2895c | CTTTGAGGTTGTCGCTACCC CGACGAACACCAGCTTGAT | −4.931 | −2.740 ± 0.180 |
| Rv0954 | ACTCGGCGTATTTCTGATGG CTGTCCATACGGGTCGAACT | −3.311 | −2.207 ± 0.729 |
| Rv3692 | GAGCGTCAAGTCAGTGTGGA CAACTGATAGGTGCCCTCGT | −4.234 | −3.556 ± 1.176 |
| RvDB_6066 | CAAGGCTAAAACTCAAAGGA GGACTTAACCCAACATCTCA | -- | -- |
2.3. Molecular Docking Studies
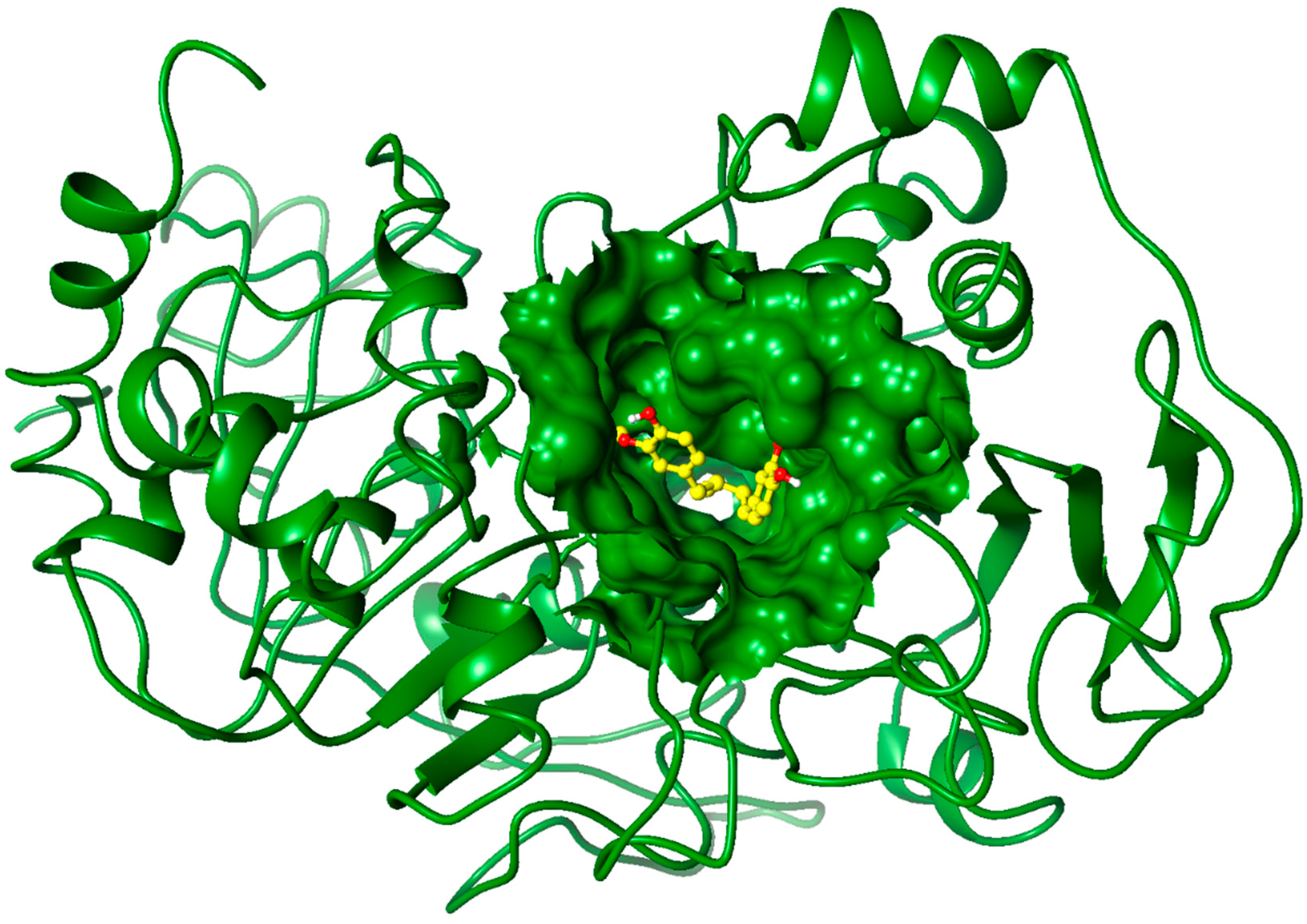
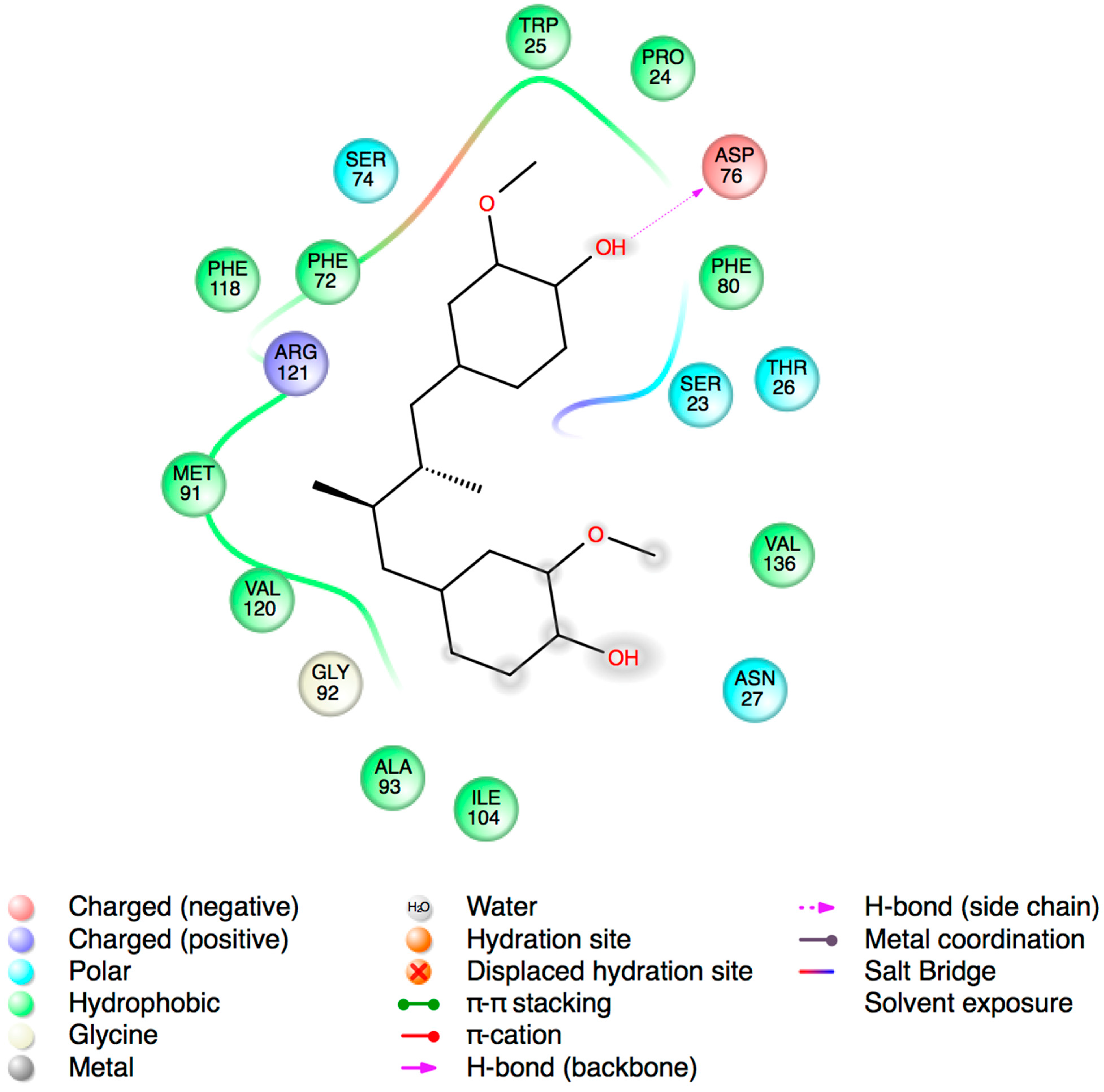
3. Experimental
3.1. Growth Curve of M. tuberculosis H37Rv with Different Concentrations of MDGA
3.2. Isolation of RNA
3.3. Synthesis and Labeling of Modified cDNA
3.4. Microarray Assay
3.5. Real-Time Reverse Transcription Polymerase Chain Reaction (RT-PCR)
3.6. Sequence Retrieving, Analysis, and Homology Modeling
3.7. Molecular Docking
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2013. Available online: http://www.who.int/iris/bitstream/10665/91355/1/9789241564656_eng.pdf (accessed on 15 September 2014).
- Akbar, A.; Reza, M.; Farnia, P.; Tabarsi, P.; Ghanavi, J.; Hassan, A. Emergence of New Forms of Totally Drug-Resistant Tuberculosis Bacilli. Chest 2009, 136, 420–425. [Google Scholar]
- Dover, L.G.; Coxon, G.D. Current Status and Research Strategies in Tuberculosis Drug Development. J. Med. Chem. 2011, 54, 6157–6165. [Google Scholar]
- Newman, D.; Cragg, G. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar]
- García, A.; Bocanegra, V.; Palma, J.; Rivera, G. Recent advances in antitubercular natural products. Eur. J. Med. Chem. 2012, 49, 1–23. [Google Scholar]
- Salinas-Salazar, C.; Camacho-Corona, M.R.; Bernes, S.; Waksman, N. 2,2'-Dimethoxy-4,4'-[rel-(2R,3S)-2,3-dimethylbutane-1,4-diyl]diphenol. Acta Cryst. 2009, E65, o1279. [Google Scholar]
- Favela-Hernández, J.M.J.; García, A.; Garza-González, E.; Rivas-Galindo, V.M.; Camacho-Corona, M.R. Antibacterial and antimycobacterial lignans and flavonoids from Larrea tridentata. Phytother. Res. 2012, 26, 1957–1960. [Google Scholar]
- Coy, E.D.; Cuca, L.E.; Sefkow, M. Macrophyllin-type bicycle[3.2.1]octanoid neolignans from the leaves of Pleurothyrium cinereum. J. Nat. Prod. 2009, 72, 1245–1248. [Google Scholar]
- Baquero, E.; Quiñones, W.; Ribbons, W.; Caldas, M.; Sarmiento, L.; Echeverria, F. Effect of an oxadiazoline and a lignin on mycolic acid biosynthesis and ultrastructural changes of Mycobacterium tuberculosis. Tuberc. Res. Treat. 2011, 1, 1–6. [Google Scholar]
- Alanís, B.; Salazar, R.; Ramírez, R.; Garza, E. A new antimycobacterial furanolignan from Leucophyllum frutescens. Nat. Prod. Commun. 2012, 7, 597–598. [Google Scholar]
- Villenueve, D.; Parissenti, A. The Use of DNA Microarrays to Investigate the Pharmacogenomics of Drug Response in Living Systems. Curr. Top. Med. Chem. 2004, 13, 1327–1343. [Google Scholar]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene list. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar]
- Amineni, U.; Pradhan, D.; Marisetty, H. In silico identification of common putative drug targets in Leptospira interrogans. J. Chem. Biol. 2010, 3, 165–173. [Google Scholar]
- Rohmer, M. The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat. Prod. Rep. 1999, 16, 565–574. [Google Scholar]
- Wolken, W.; Tramper, J.; van der Werf, M. Toxicity of terpenes to spores and mycelium of Penicillium digitatum. Biotechnol. Bioeng. 2002, 80, 685–690. [Google Scholar]
- Bard, M.; Albrecht, M.; Gupta, N.; Guynn, C.; Stillwell, W. Geraniol interferes with membrane functions in strains of Candida and Saccharomyces. Lipids 1988, 23, 534–538. [Google Scholar]
- Prashar, A.; Hili, P.; Veness, R.G.; Evans, Ch.S. Antimicrobial action of palmarosa oil (Cymbopogon martinii) on Saccharomyces cerevisiae. Phytochemitry 2003, 63, 569–575. [Google Scholar]
- Trombetta, D.; Castelli, F.; Sarprieto, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of Antibacterial Action of Three Monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar]
- Inoue, Y.; Shiraishi, A.; Hada, T.; Hirose, K.; Hamashima, H.; Shimada, J. The antibacterial effects of terpene alcohols on Staphylococcus aureus and their mode of action. FEMS Microbiol. Lett. 2004, 237, 325–331. [Google Scholar]
- Togashi, N.; Inoue, Y.; Hamashima, H.; Takano, A. Effects of two terpene alcohols on the antibacterial activity and the mode of action of farnesol against Staphylococcus aureus. Molecules 2008, 13, 3069–3076. [Google Scholar]
- Uribe, S.; Ramirez, J.; Peña, A. Effects of beta-pinene on yeast membrane functions. Effects of P-Pinene on Yeast Membrane Functions. J. Bacteriol. 1985, 161, 1195–1200. [Google Scholar]
- Sikkema, J.; Poolman, B.; Konings, W.; de Bont, J.A. Effects of the Membrane Action of Tetralin on the Functional and Structural Properties of Artificial and Bacterial Membranes. J. Bacteriol. 1992, 174, 2986–2992. [Google Scholar]
- Sikkema, J.; de Bont, J.A.; Poolman, B. Interactions of cyclic hydrocarbons with biological membranes. J. Biol. Chem. 1994, 269, 8022–8028. [Google Scholar]
- Cerniglia, C.E.; Freeman, J.P.; Althaus, J.R.; Baalen, C.V. Metabolism and toxicity of 1- and 2-methylnaphthalene and their derivatives in cyanobacteria. Arch. Microbiol. 1983, 136, 177–183. [Google Scholar]
- Prenafeta-Boldú, F.X.; Kuhn, A.; Luykx, D.M.A.M.; Anke, H.; van Groenestijn, J.W.; de Bont, J.A.M. Isolation and characterization of fungi growing on volatile aromatic hydrocarbons as their sole carbon and energy source. Mycol. Res. 2001, 105, 477–484. [Google Scholar]
- Park, E.Y.; Shin, S.M.; Ma, C.J.; Kim, Y.C.; Kim, S.G. meso-Dihydroguaiaretic Acid from Machilus thunbergii Down-RegulatesTGF-β1 Gene Expression in Activated Hepatic Stellate Cells via Inhibition of AP-1 Activity. Planta Med. 2005, 71, 393–398. [Google Scholar]
- Yu, Y.U.; Kang, S.Y.; Park, H.Y.; Sung, S.H.; Lee, E.J.; Kim, S.Y.; Kim, Y.C. Antioxidant lignans from Machilus thunbergii protect CCl4-injured primary cultures of rat hepatocytes. J. Pharm. Pharmacol. 2000, 52, 1163–1169. [Google Scholar]
- Filleur, F.; le Bail, J.C.; Duroux, J.L.; Simon, A.; Chulia, A.J. Antiproliferative, Anti-Aromatase, Anti-17β-HSD and Antioxidant Activities of Lignans Isolated from Myristica argentea. Planta Med. 2001, 67, 700–704. [Google Scholar]
- Jacob, U.; Mack, M.; Clausen, T.; Huber, R.; Buckel, W.; Messerschmidt, A. Glutaconate CoA-transferase from Acidaminococcus fermentans: The crystal structure reveals homology with other CoA-transferases. Structure 1997, 5, 415–426. [Google Scholar]
- Korolev, S.; Koroleva, O.; Petterson, K.; Gu, M.; Collart, F.; Dementieva, I.; Joachimiak, A. Autotracing of Escherichia coli acetate CoA-transferase alpha-subunit structure using 3.4 A MAD and 1.9 A native data. Acta Crystallogr. D Biol. Crystallogr. 2002, 58, 2116–2121. [Google Scholar]
- Hetényi, C.; van der Spoel, D. Efficient docking of peptides to proteins without prior knowledge of the binding site. Protein Sci. 2002, 11, 1729–1737. [Google Scholar]
- Hetényi, C.; van der Spoel, D. Blind docking of drug-sized compounds to proteins with up to a thousand residues. FEBS Lett. 2006, 580, 1447–1450. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera-a visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar]
- Schrödinger Release 2014-2: Maestro; Schrödinger, LLC: New York, NY, USA, 2014; version 9.8.
- Lew, J.M.; Kapopoulou, A.; Jones, L.M.; Cole, S.T. TubercuList—10 years after. Tuberculosis (Edinb) 2011, 91, 1–7. [Google Scholar]
- Kanehisa, M.; Goto, S. KEGG: Kyoto enciclopedia of genes and genomes. Nucleis Acid Res. 2000, 28, 27–30. [Google Scholar]
- Benson, D.A.; Cavanaugh, M.; Clark, K.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Sayers, E.W. GenBank. Nucleic Acids Res. 2013, 41, D36–D42. [Google Scholar]
- Reddy, T.B.K.; Riley, R.; Wymore, F.; Montgomery, P.; DeCaprio, D.; Engels, R.; Gellesch, M.; Hubble, J.; Jen, D.; Jin, H.; et al. TB database: An integrated platform for tuberculosis research. Nucleic Acid Res. 2009, 37, D499–D508. [Google Scholar]
- Sali, A.; Blundell, T.L. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 1993, 234, 779–815. [Google Scholar]
- Melo, F.; Feytmans, E. Assessing protein structures with a non-local atomic interaction energy. J. Mol. Biol. 1998, 277, 1141–1152. [Google Scholar]
- PubChem. Available online: http://pubchem.ncbi.nlm.nih.gov (accessed on 1 December 2014).
- Sanner, M.F. Python: A programming language for software integration and development. J. Mol. Graph. Model. 1999, 17, 57–61. [Google Scholar]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. Autodock4 and AutoDockTools4: Automated docking with selective receptor flexiblity. J. Comput. Chem. 2009, 16, 5–91. [Google Scholar]
- Sample Availability: Sample of the compound is available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clemente-Soto, A.F.; Balderas-Rentería, I.; Rivera, G.; Segura-Cabrera, A.; Garza-González, E.; Del Rayo Camacho-Corona, M. Potential Mechanism of Action of meso-Dihydroguaiaretic Acid on Mycobacterium tuberculosis H37Rv. Molecules 2014, 19, 20170-20182. https://doi.org/10.3390/molecules191220170
Clemente-Soto AF, Balderas-Rentería I, Rivera G, Segura-Cabrera A, Garza-González E, Del Rayo Camacho-Corona M. Potential Mechanism of Action of meso-Dihydroguaiaretic Acid on Mycobacterium tuberculosis H37Rv. Molecules. 2014; 19(12):20170-20182. https://doi.org/10.3390/molecules191220170
Chicago/Turabian StyleClemente-Soto, Aldo F., Isaías Balderas-Rentería, Gildardo Rivera, Aldo Segura-Cabrera, Elvira Garza-González, and María Del Rayo Camacho-Corona. 2014. "Potential Mechanism of Action of meso-Dihydroguaiaretic Acid on Mycobacterium tuberculosis H37Rv" Molecules 19, no. 12: 20170-20182. https://doi.org/10.3390/molecules191220170








