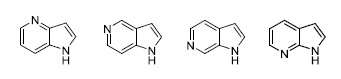
The Azaindole Framework in the Design of Kinase Inhibitors
Abstract
:1. Introduction
| Azaindole framework | 4-Azaindole | 5-Azaindole | 6-Azaindole | 7-Azaindole |
|---|---|---|---|---|
| on-line substructure research a |  |  |  |  |
| Scifinder | ||||
| chemical structures | 16,505 | 13,632 | 34,762 | 100,384 |
| commercially available | 2517 | 2006 | >2749 | 4273 |
| References (patents) | 2158 (1187) | 2923 (961) | 10,497 (1756) | 6576 (2863) |
| Reference Evolution from 2003 to 2013 |  | |||
| Reaxys | ||||
| chemical structures | 9701 | 8058 | 27,655 | 58,082 |
| commercially available | 707 | 639 | 1116 | 3503 |
| References (patents) | 583 (586) | 724 (477) | 3461 (890) | 3951 (2026) |
| E molecules | 128 | 150 | 138 | 2300 |
| Ambinter | 1105 | 1395 | 4132 | >to 10,000 |
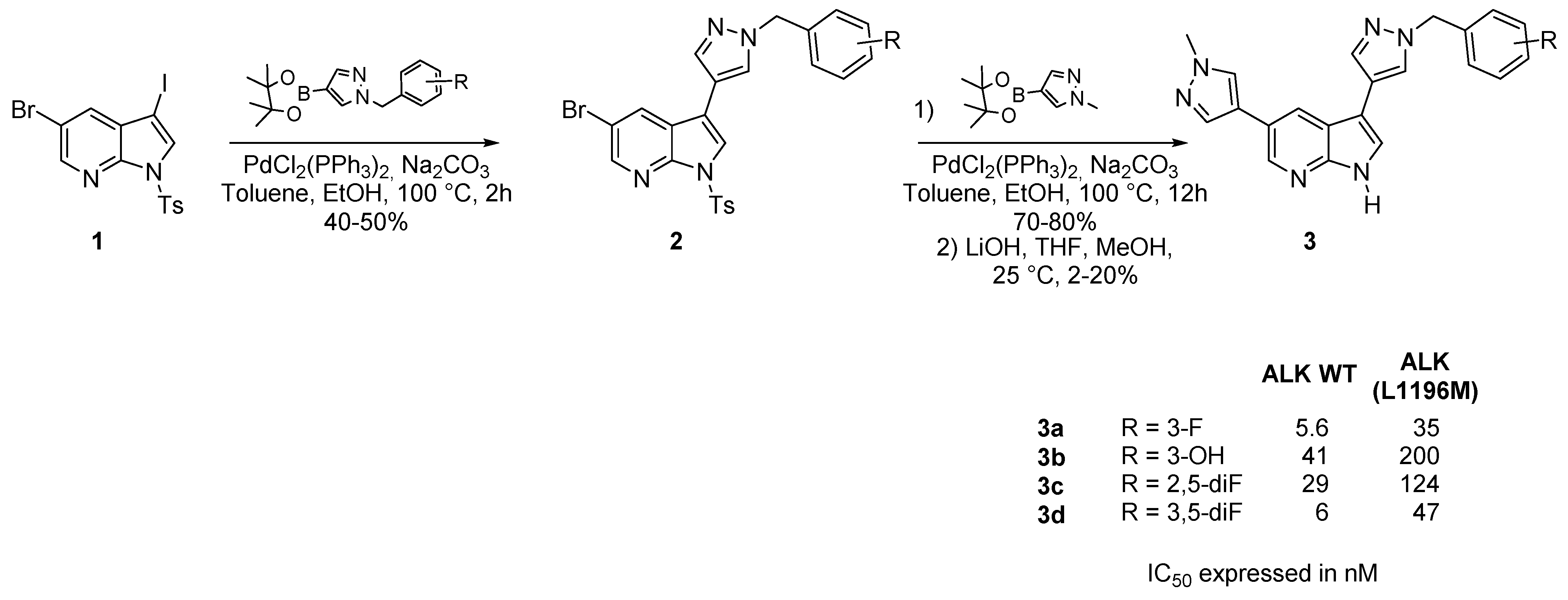
2. ALK Kinase Inhibitors
3. Aurora Kinase Inhibitors

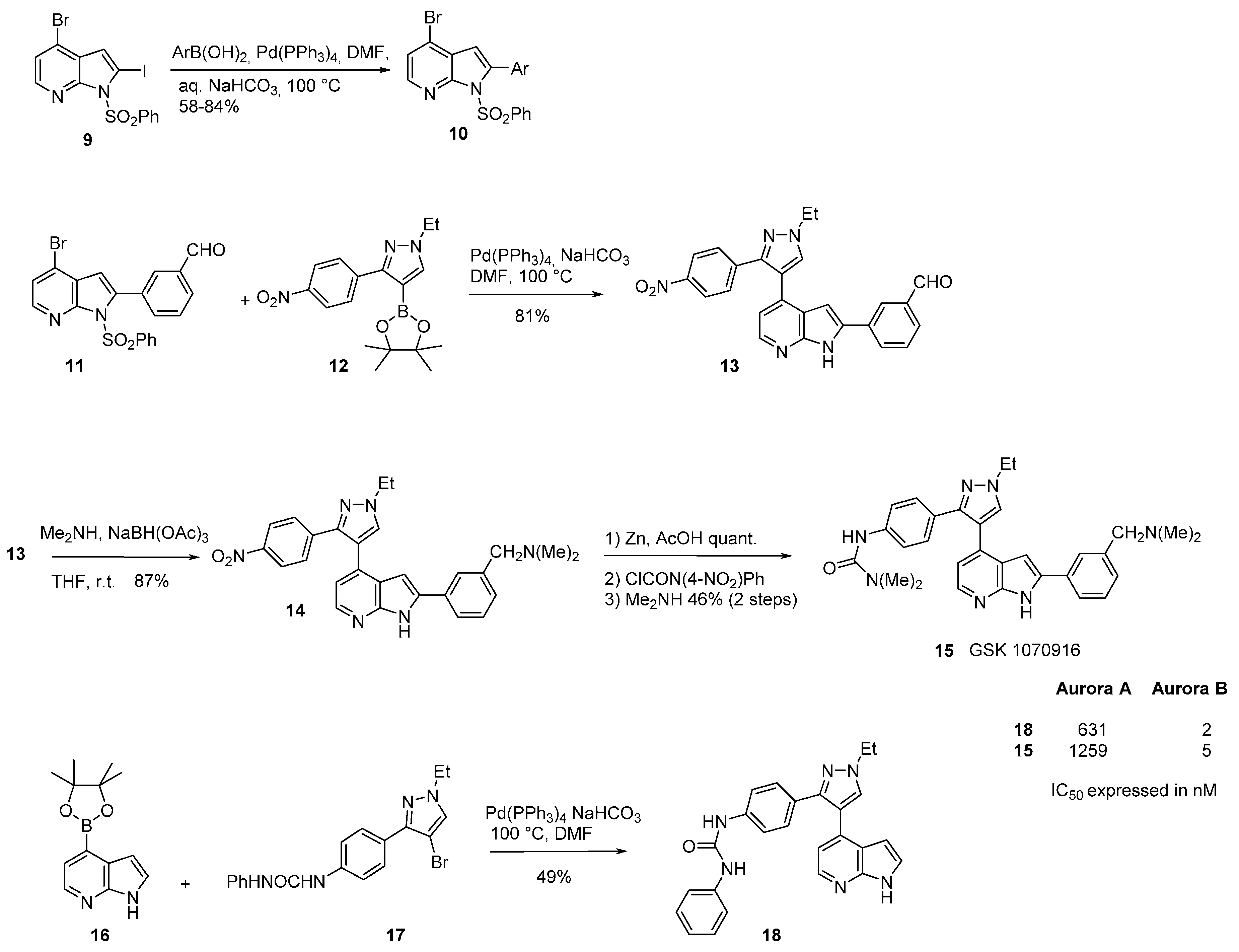
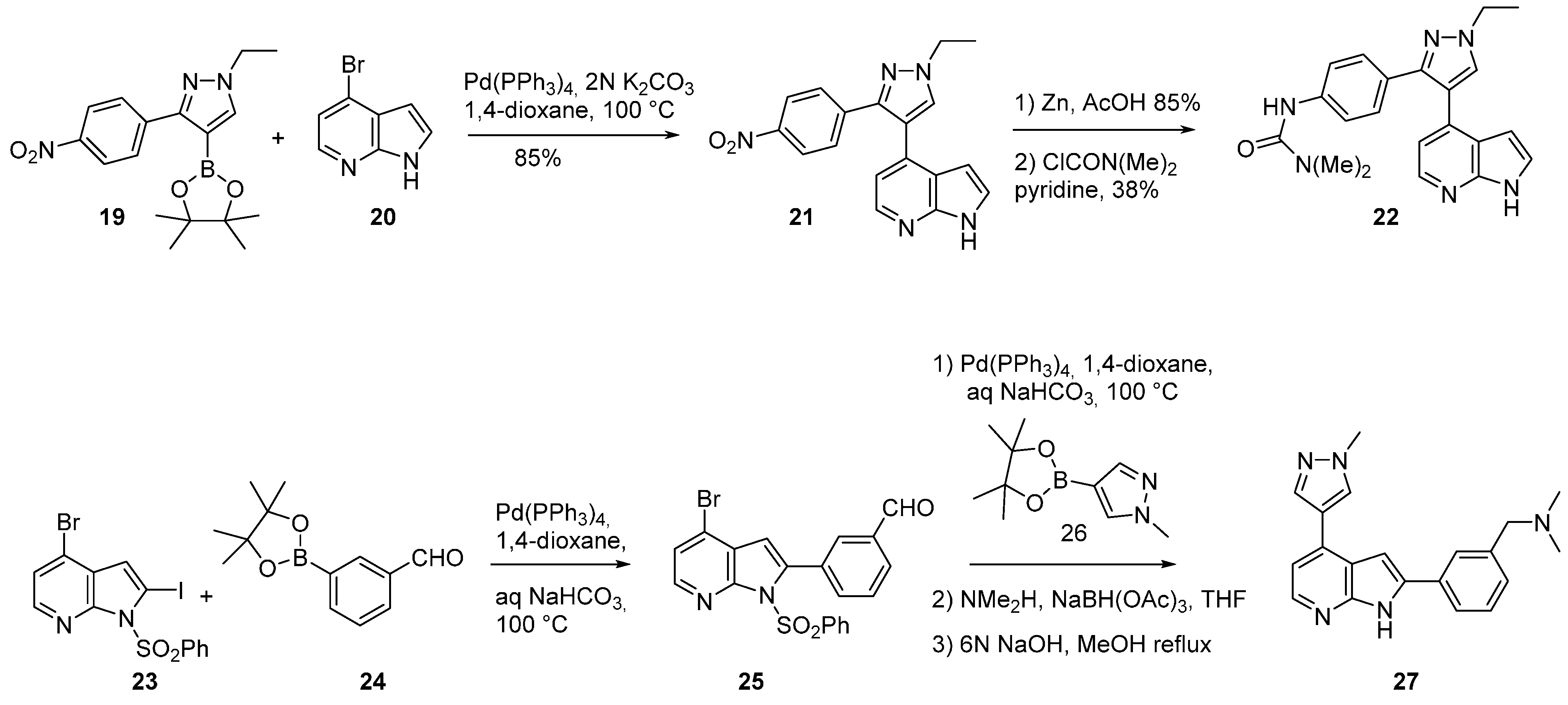
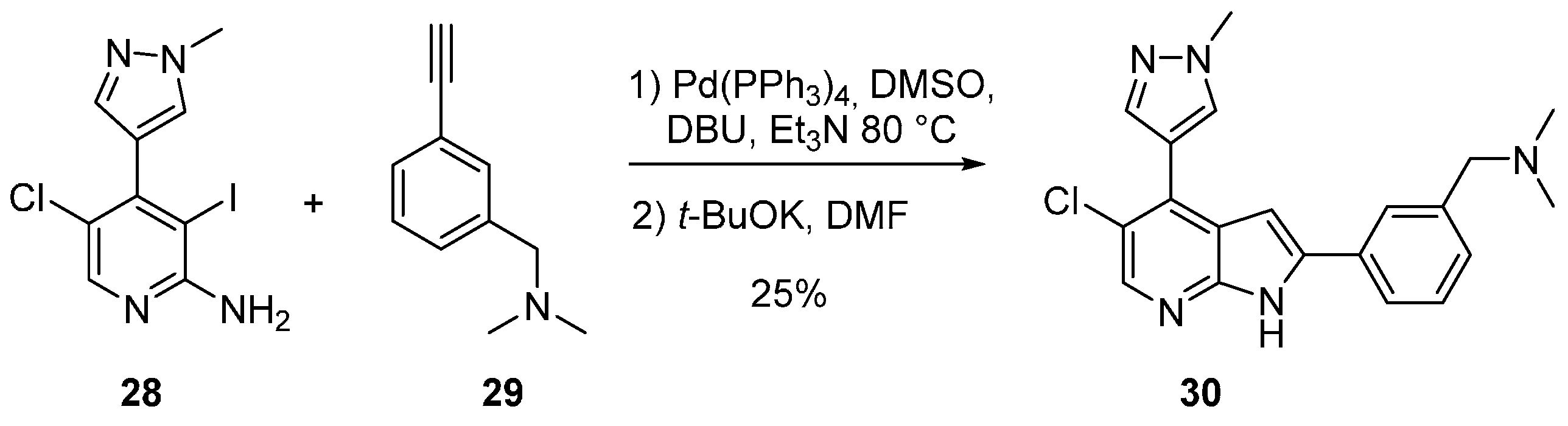
| IC50 μM | 22 | 27 | 30 | GSK1070916 |
|---|---|---|---|---|
| Aurora A | 10,000 | 128 | 6.2 | 1100 |
| Aurora B | 26 | 5.7 | 0.51 | 3.2 |
4. Cdc7 Inhibitors

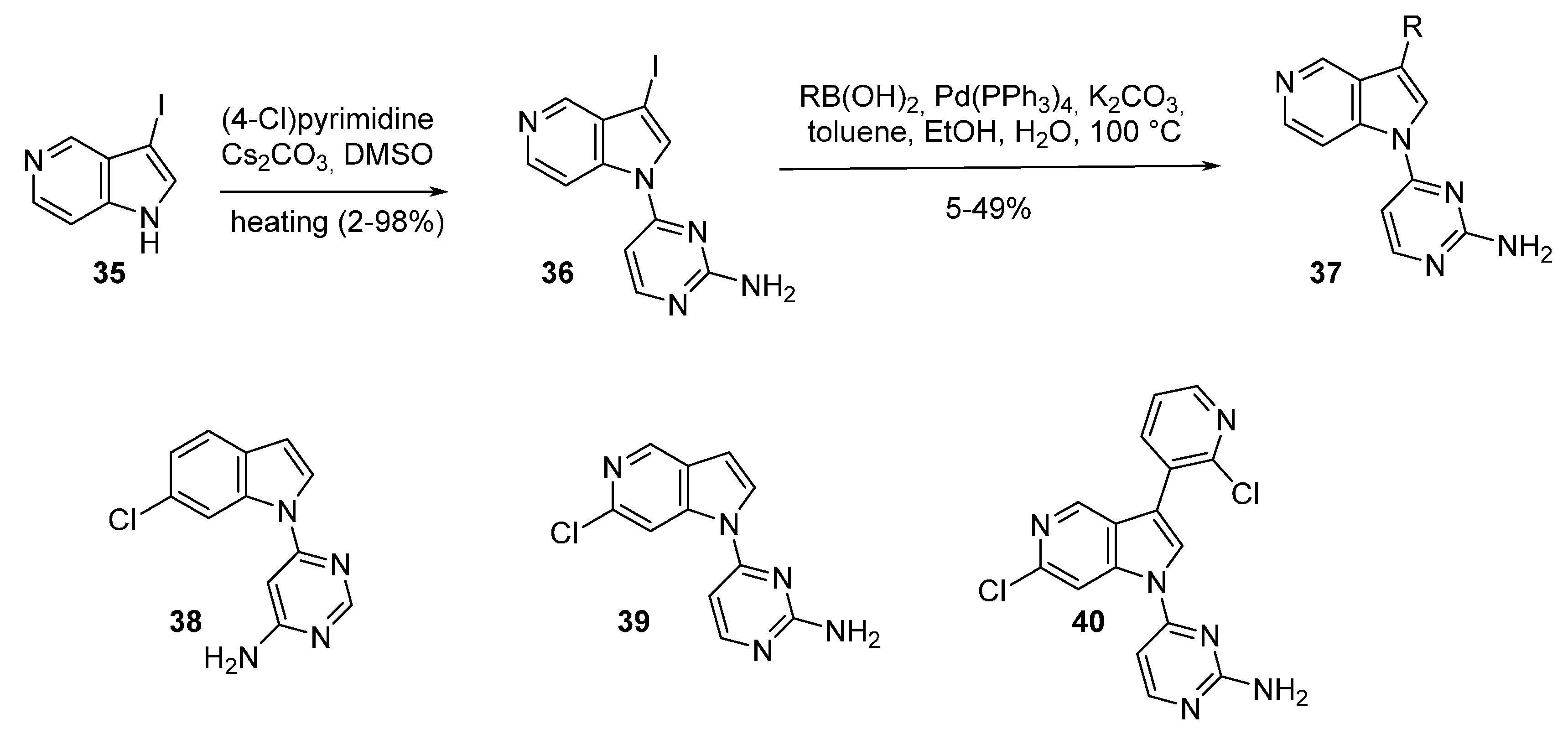
| Compound | R | IC50 Cdc7 (μM) | IC50 CDK2 (μM) | Compound | IC50 Cdc7 (μM) | IC50 CDK2 (μM) |
|---|---|---|---|---|---|---|
| 37a | H | 0.98 | 3.7 | 38 | 0.066 | 3.7 |
| 37b | C6H4 | 0.10 | 0.31 | 39 | 0.66 | 38 |
| 37c | 2-ClC6H4 | 0.011 | 0.11 | 40 | 0.03 | >80 |
| 37d | 3-ClC6H4 | 0.16 | >83 | |||
| 37e | 4-ClC6H4 | 0.33 | 0.06 | |||
| 37f | 3-pyridinyl | 0.16 | 1.1 | |||
| 37g | 3-(2-Clpyridinyl) | 0.007 | 0.31 | |||
| 37h | 4-pyridinyl | 0.25 | 0.16 | |||
| 37i | 3-furanyl | 0.12 | 0.10 | |||
| 37j | 3-pyrazole | 0.58 | 0.55 |


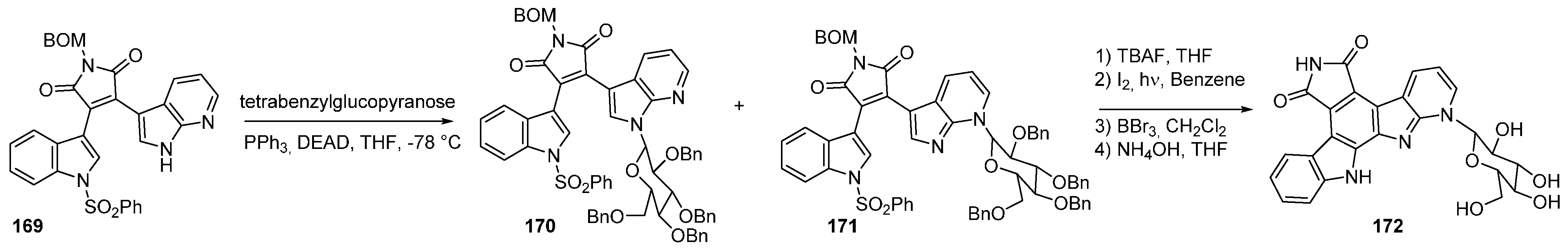
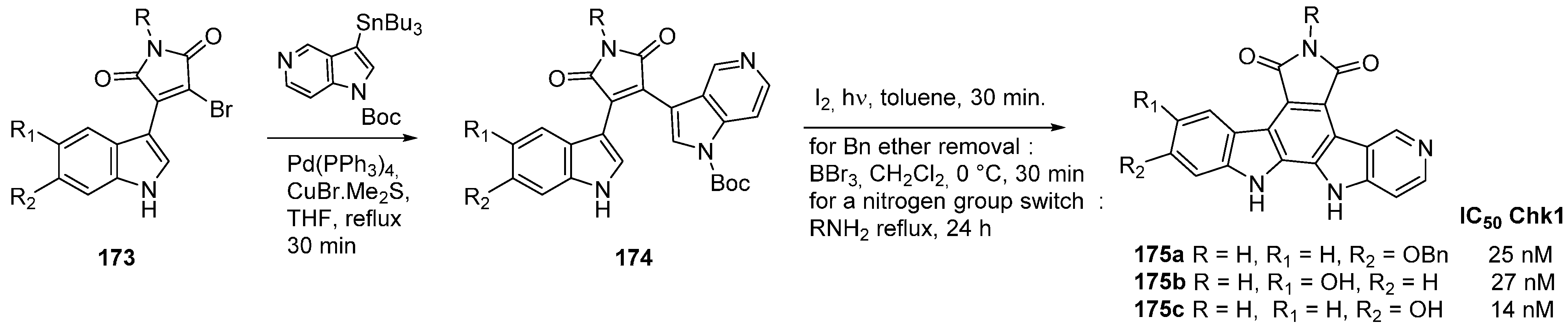
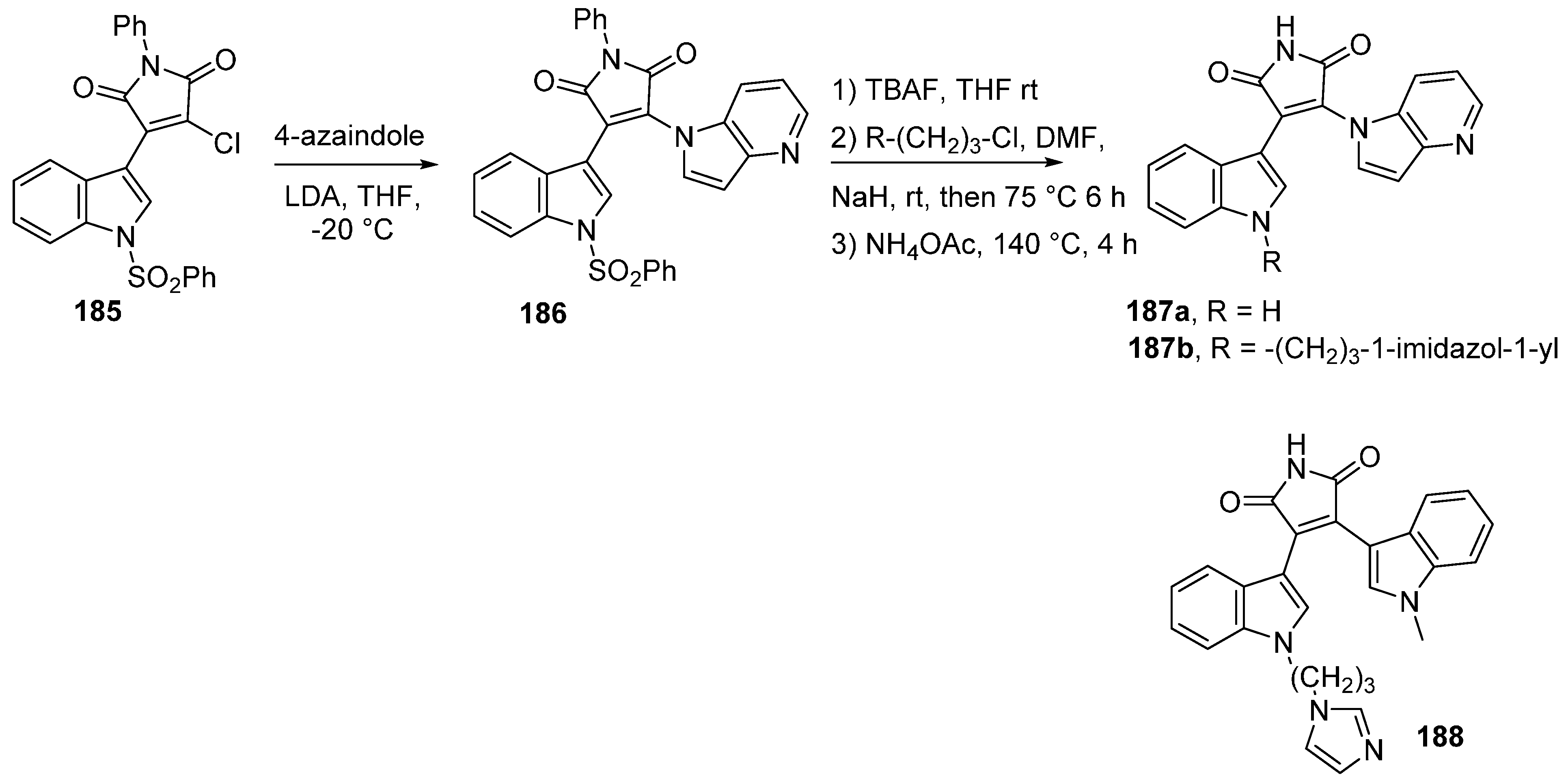
5. Check-Point Kinase (CHK1) Inhibitors
6. C-Met Kinase Inhibitors

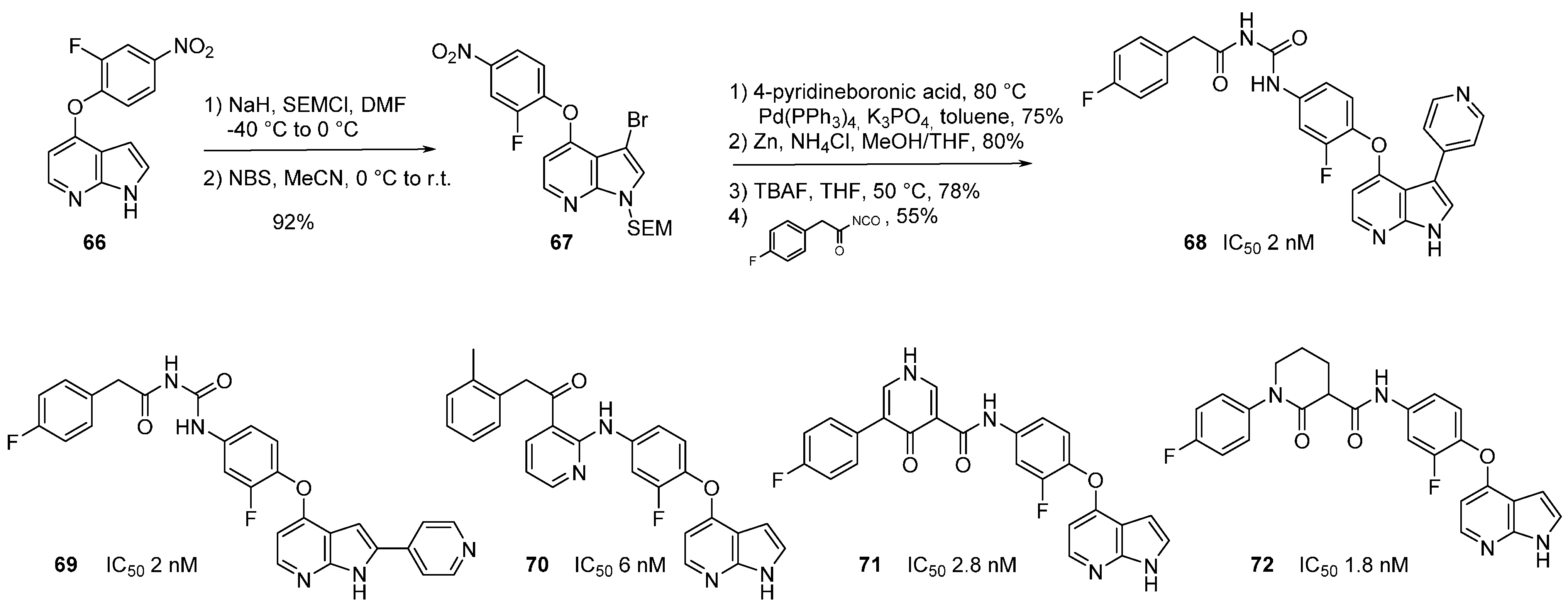
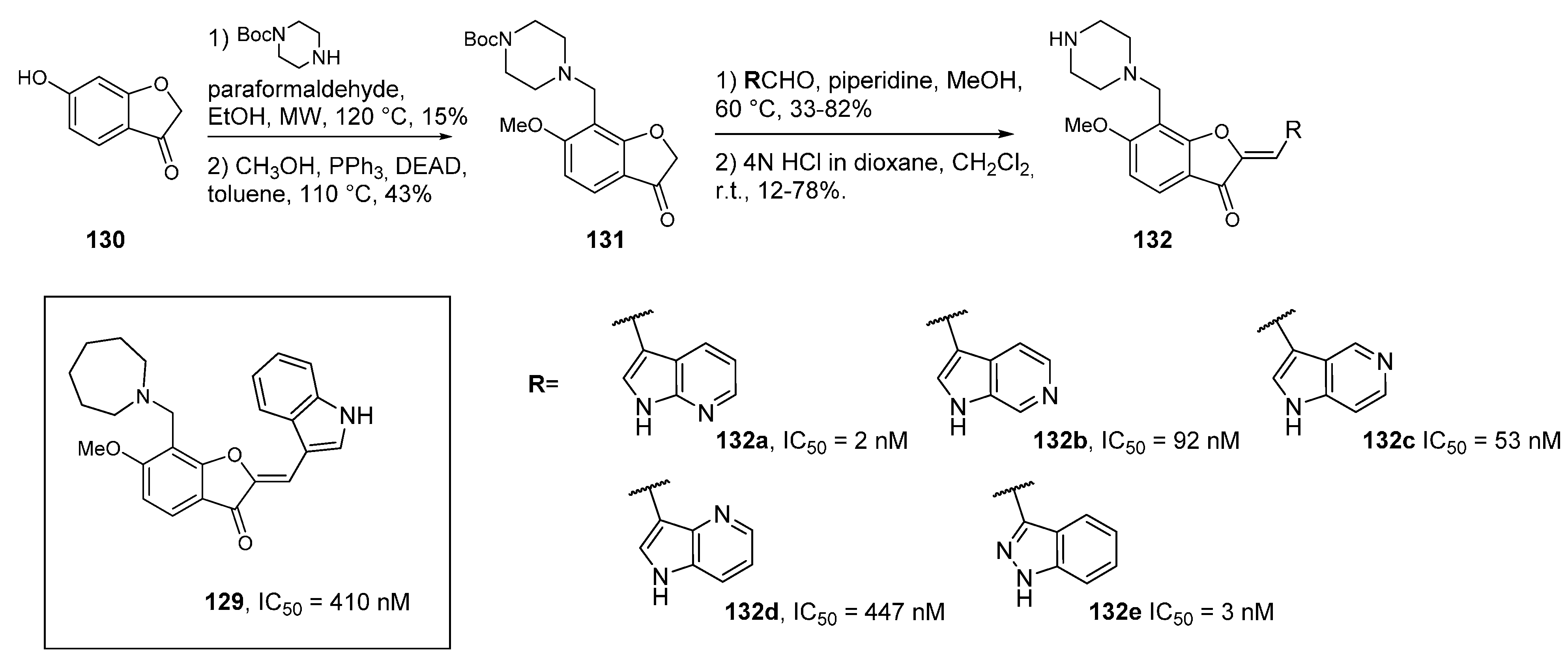
7. DYRK1A Kinase Inhibitors


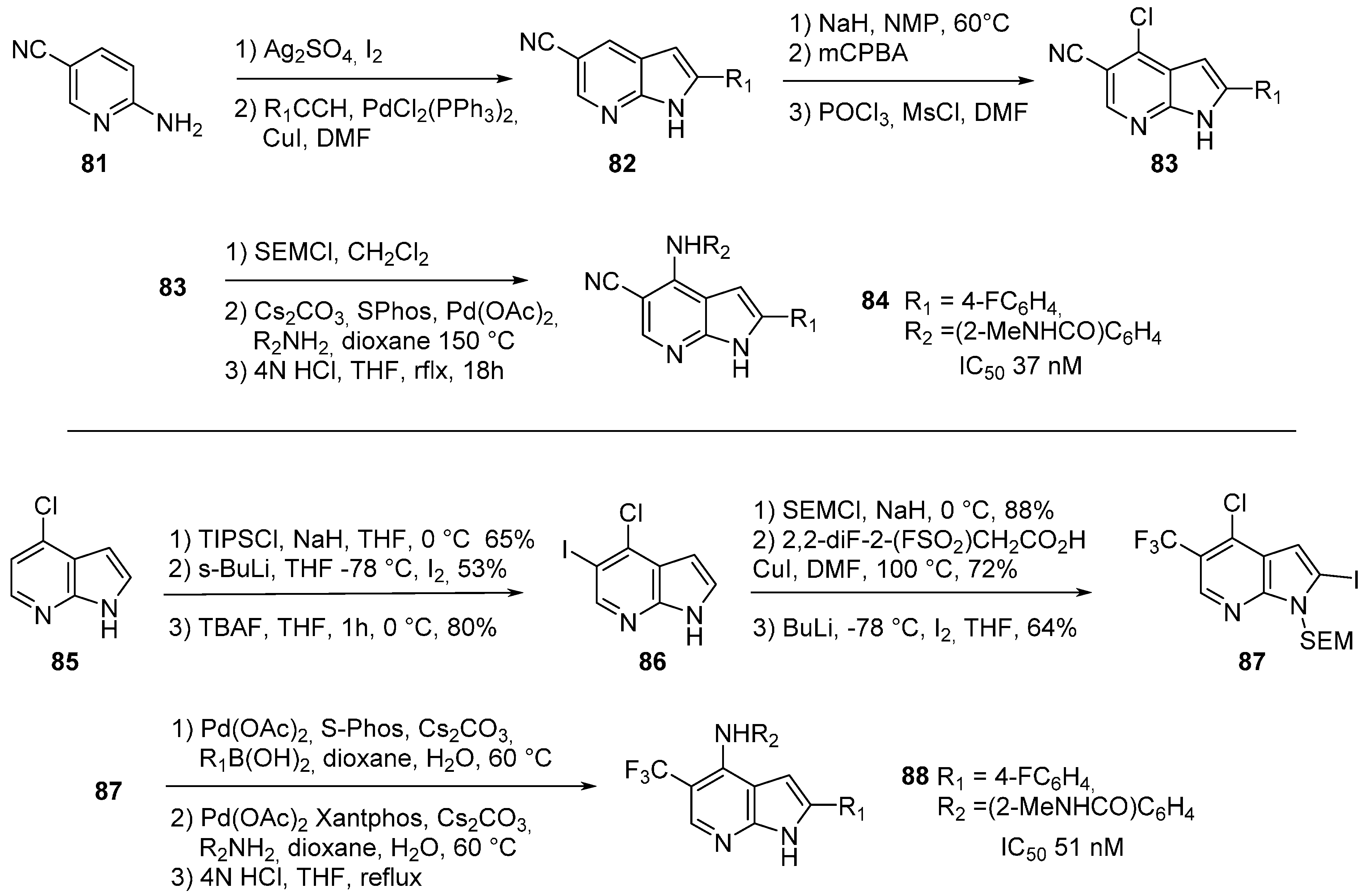
8. FAK Inhibitors
9. IKK2 Inhibitors
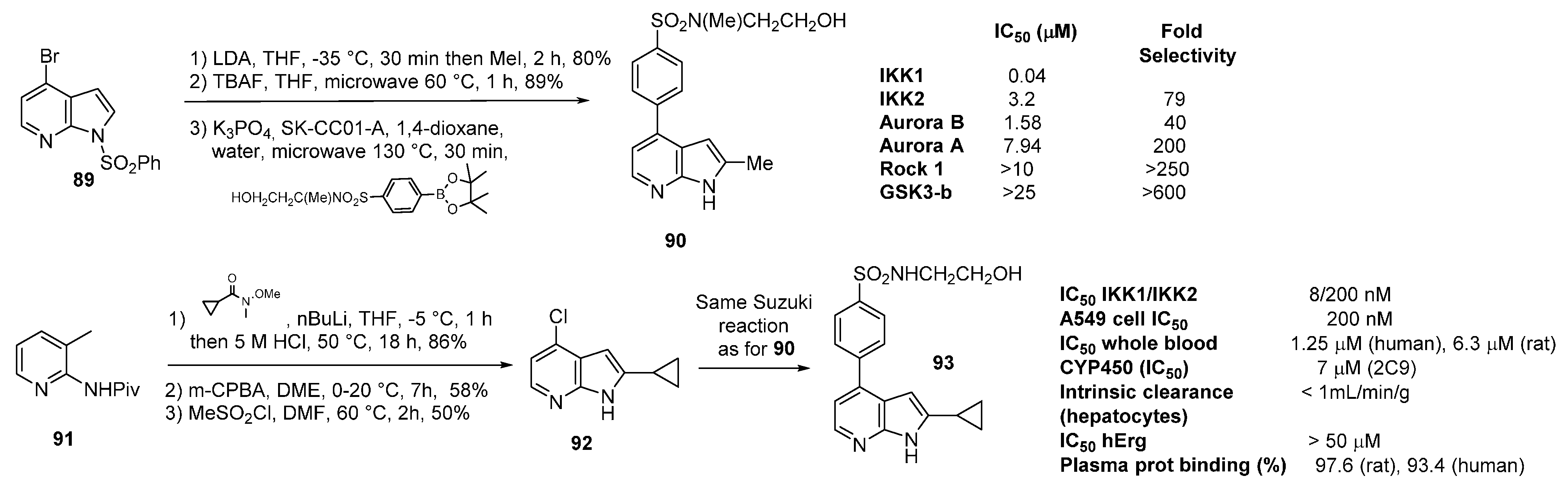
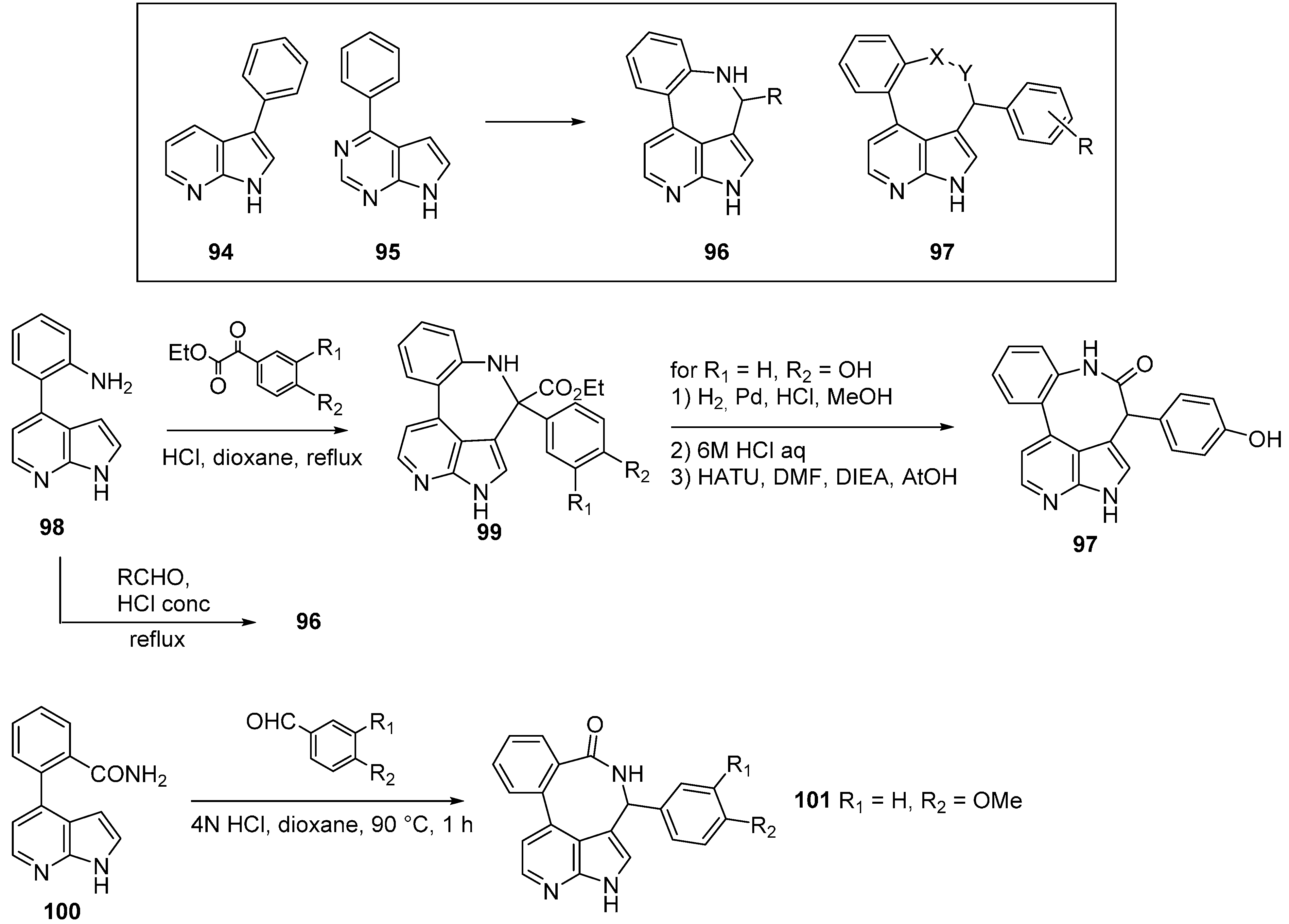
10. JAK2 Inhibitors

11. KIT/FMS Dual Kinase Inhibitors

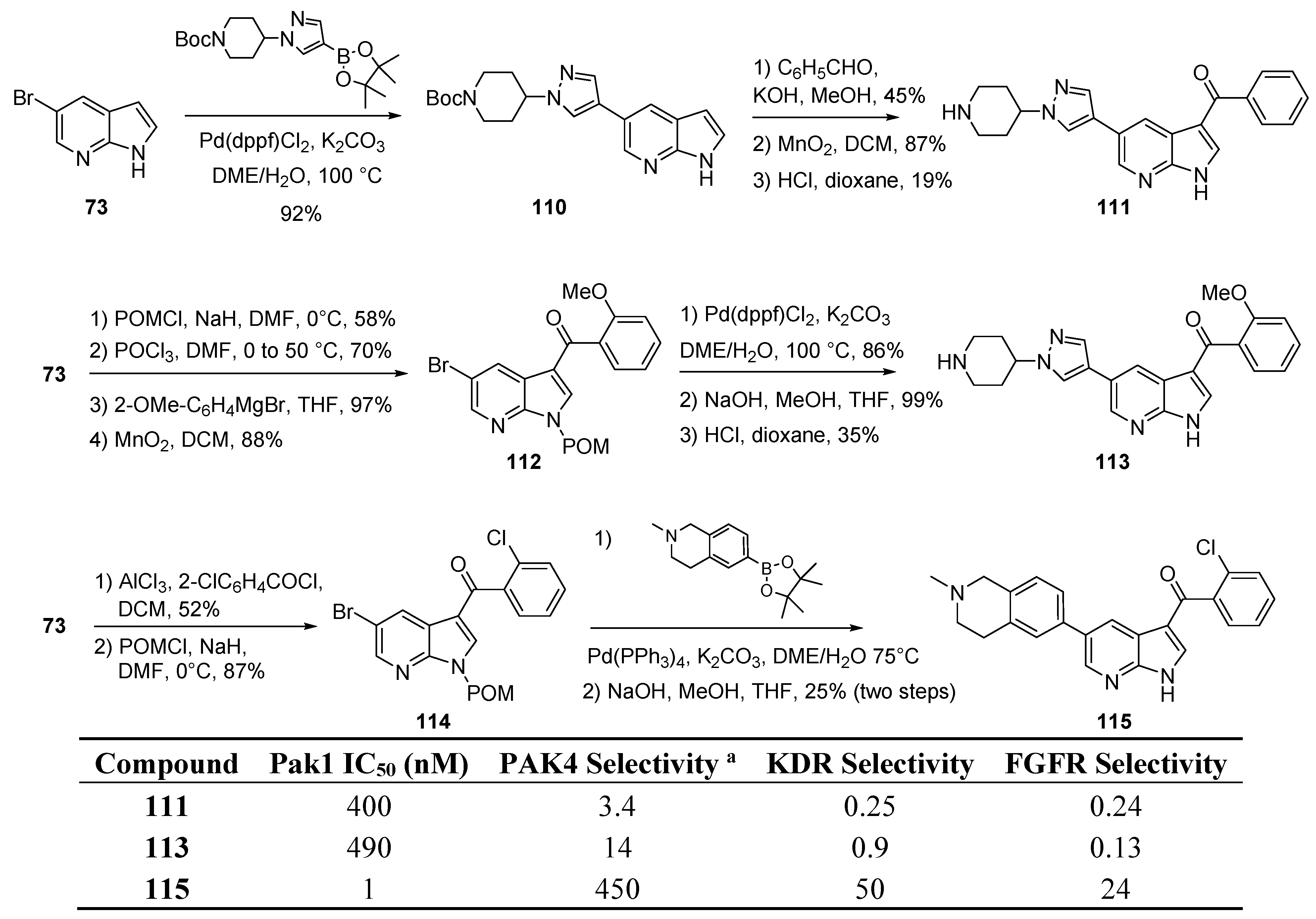
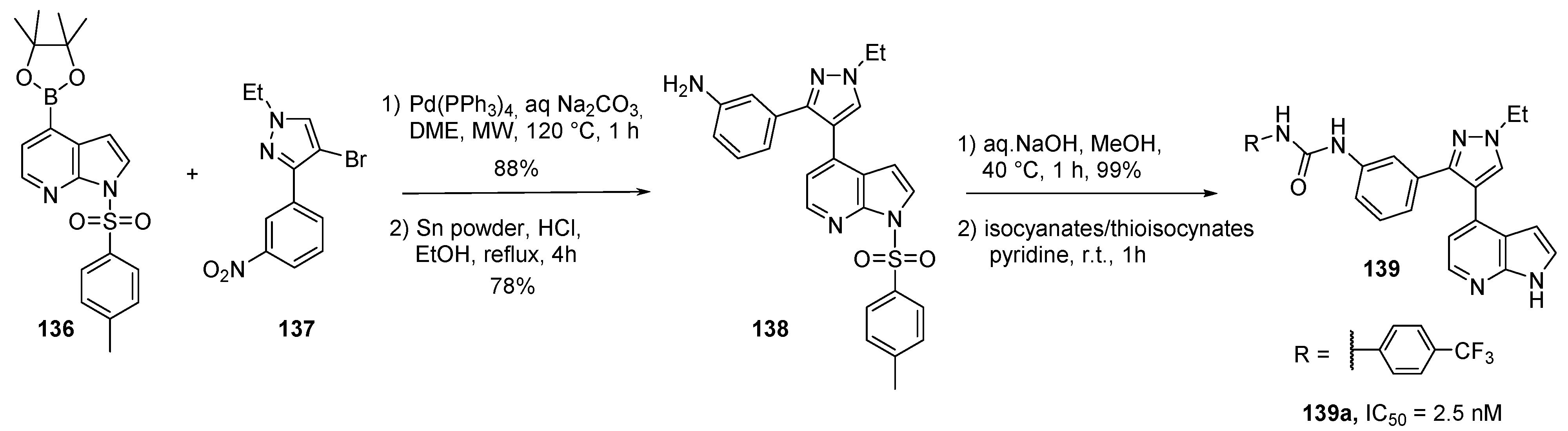
12. PAK1 Kinase Inhibitors
13. p38α MAP Kinase Inhibitors


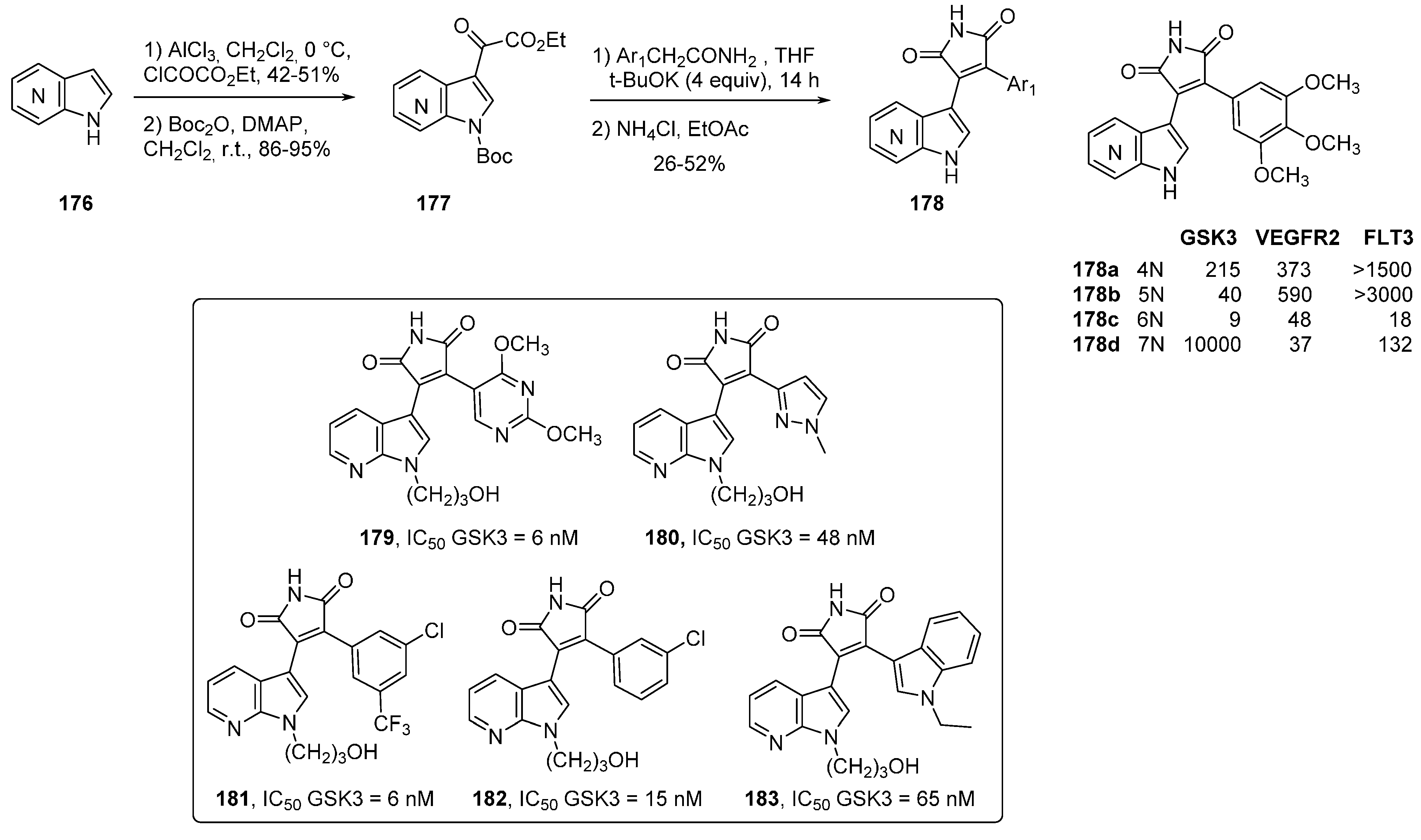
14. PIM Kinase Inhibitors
15. PI3 Kinase Inhibitors
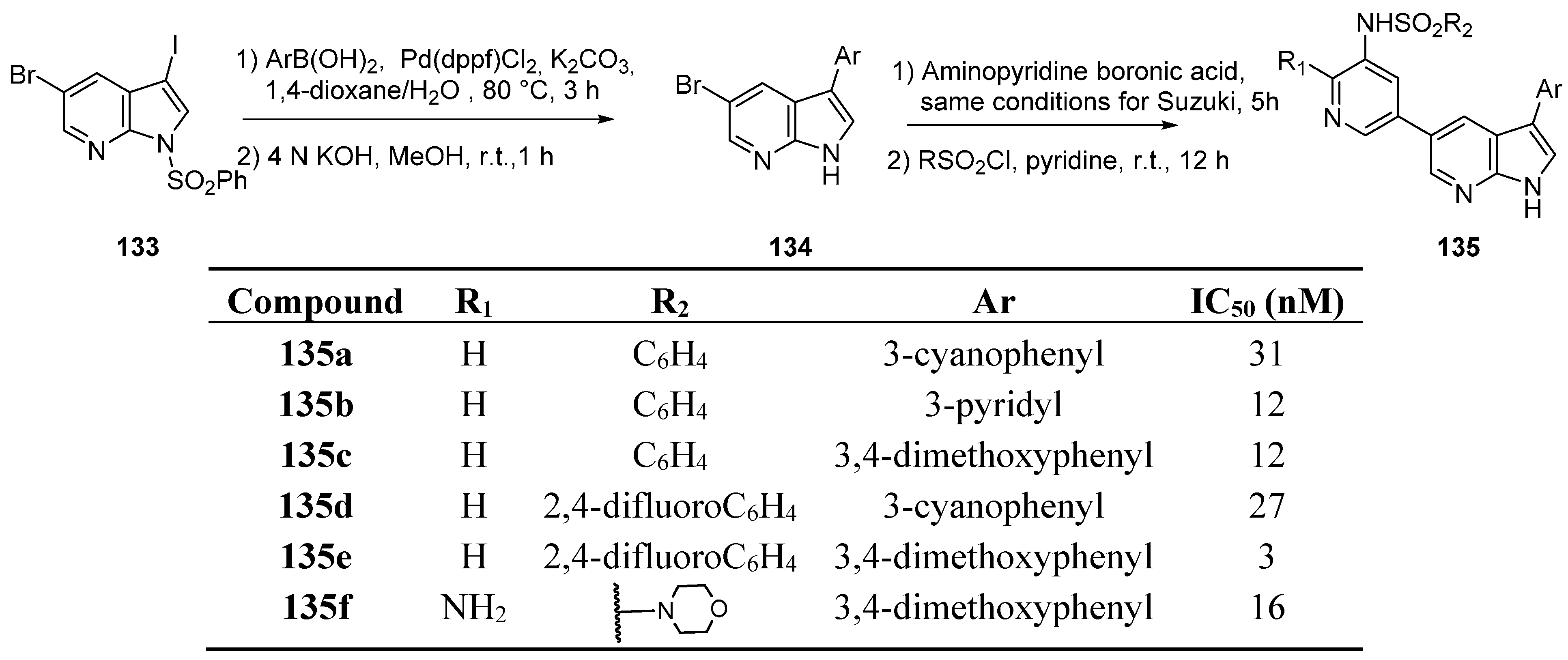
16. B-Raf Kinase Inhibitors
17. Rho Kinase (ROCK) Inhibitors
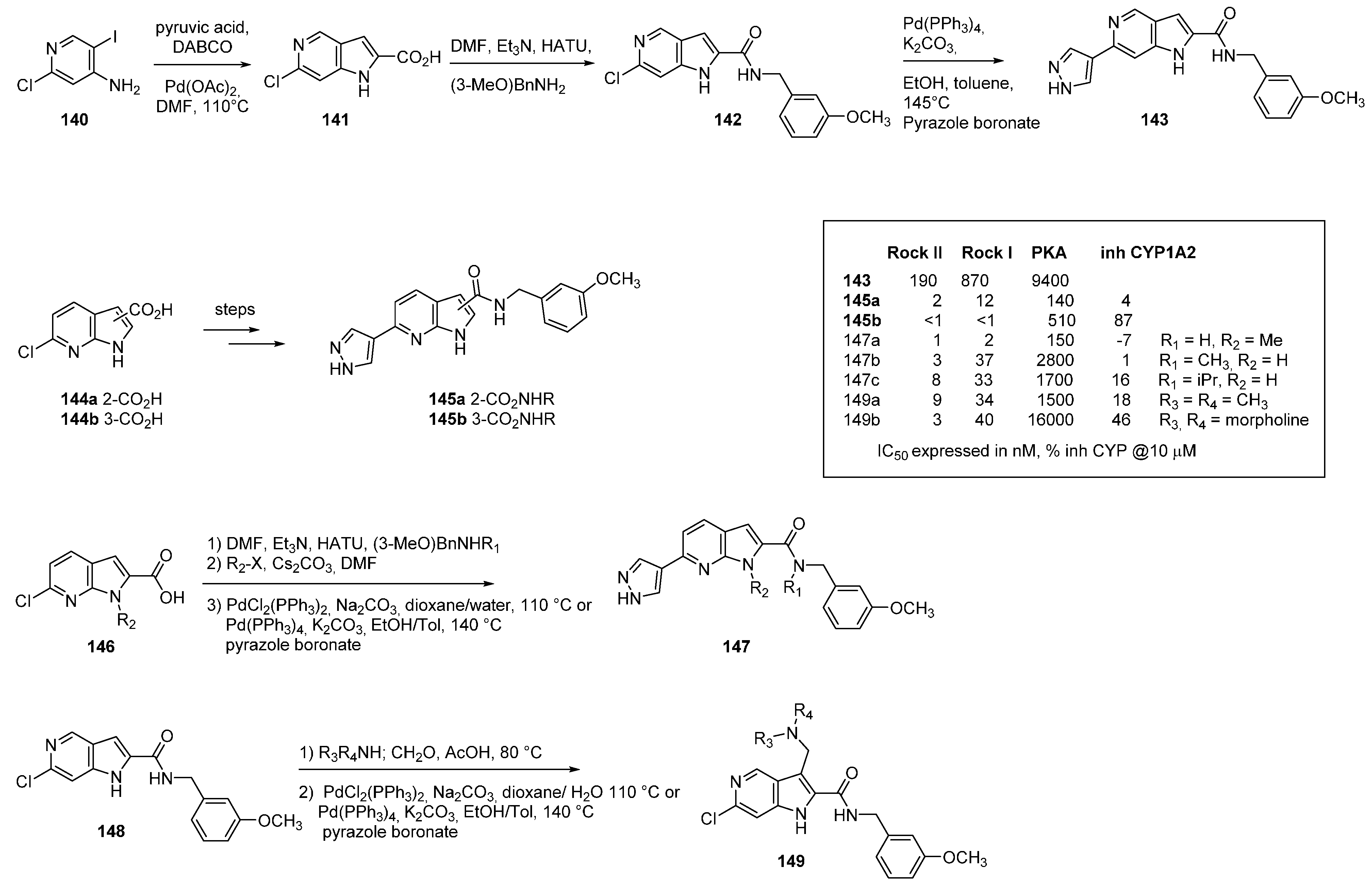
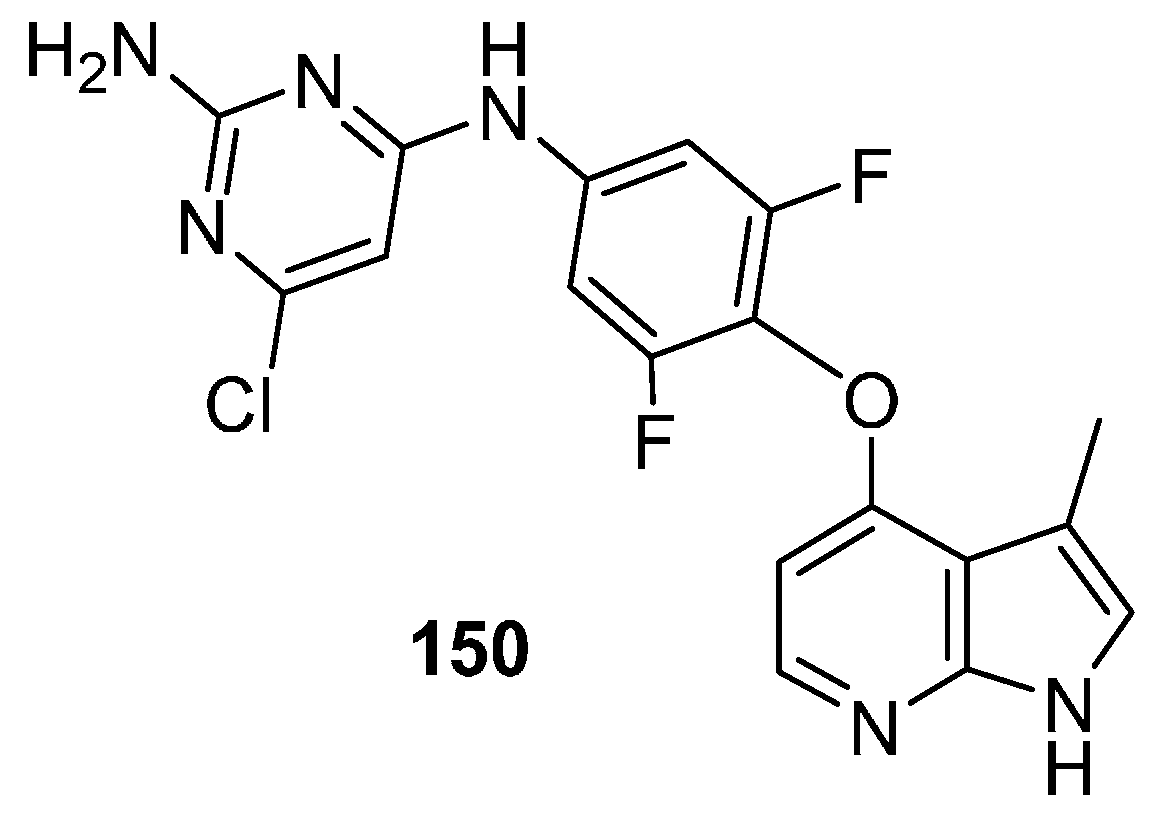
18. m-TOR Kinase Inhibitors

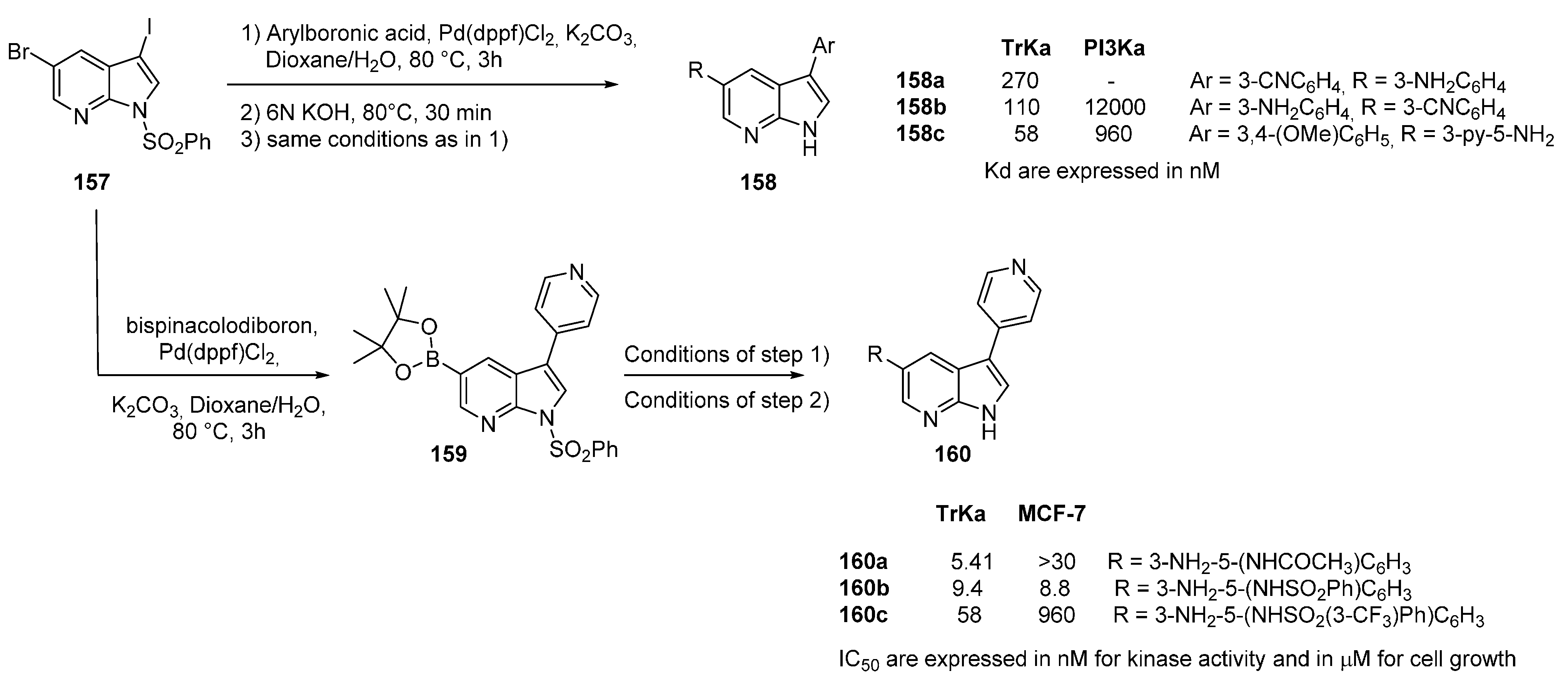
19. TrkA Kinase Inhibitors
20. Azaindole Binding Mode Analysis
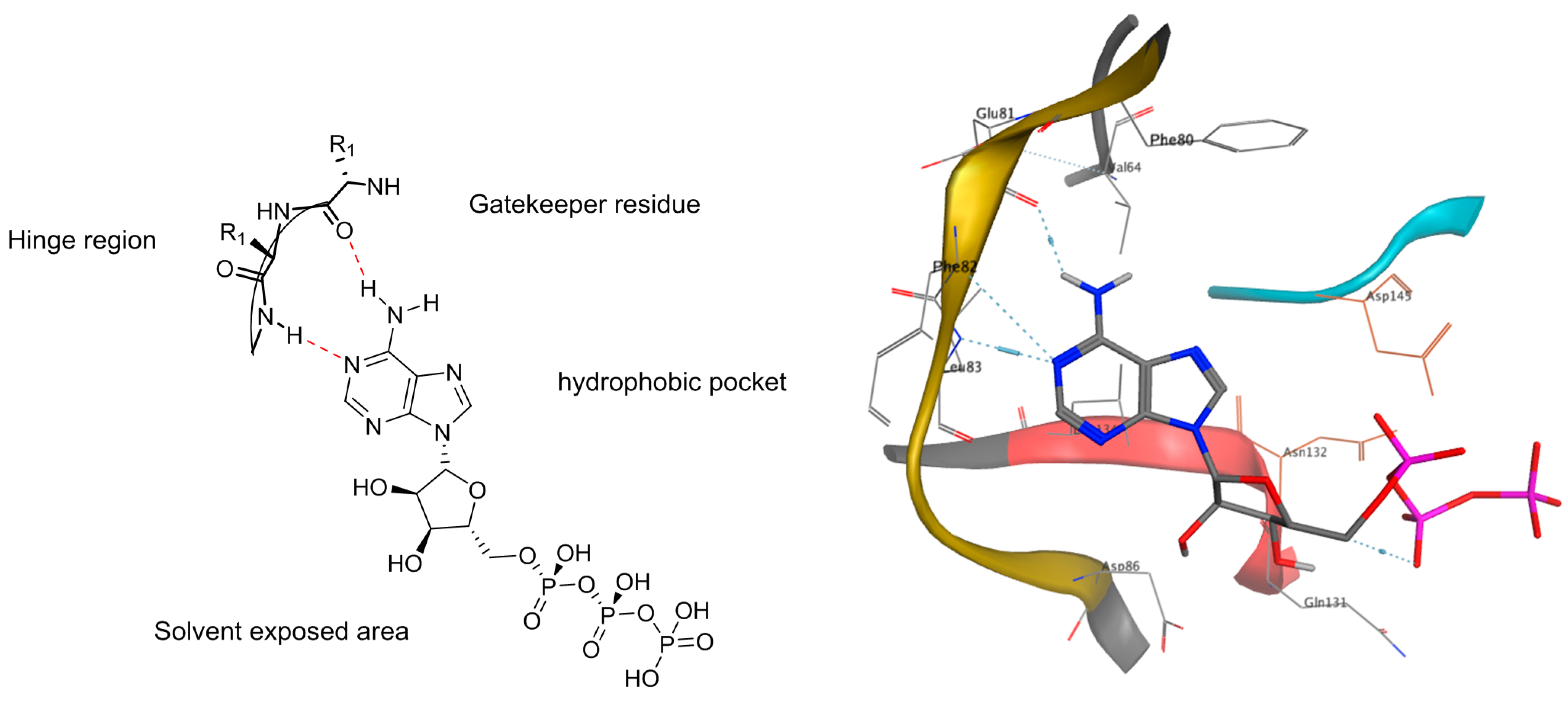
| Azaindole Binding to the Hinge Region and Mimicking the Adenine of ATP | 4-Azaindole | 5-Azaindole | 6-Azaindole | 7-Azaindole |
|---|---|---|---|---|
| Yes | 2WD1 | 4C4E 4C4F 4C4G 4C4H 4C4I 4C4J | None | 1ZYS 2QHM 2QOH 2UVX 2Z60 3BHT 3BHU 3C4C 3C4D 3C4E 3C4F 3CE3 3CTJ 3DJ6 3DK3 3DK6 3DK7 3E87 3ETA 3FQH 3GFW 3GQL 3HDM 3HDN 3JY9 3LJ3 3LVP 3OG7 3RCJ 3ZCL 3ZLS 4AOI 4AWI 4BIC 4BID 4BIE 4FK3 4FV9 4GU6 4HVS 4HW7 4IQ6 4JOA 4K1B 4O91 |
| No | 1OZ1 3LVP | 4PMS | None | 2QD9 3EN4 4JG7 |

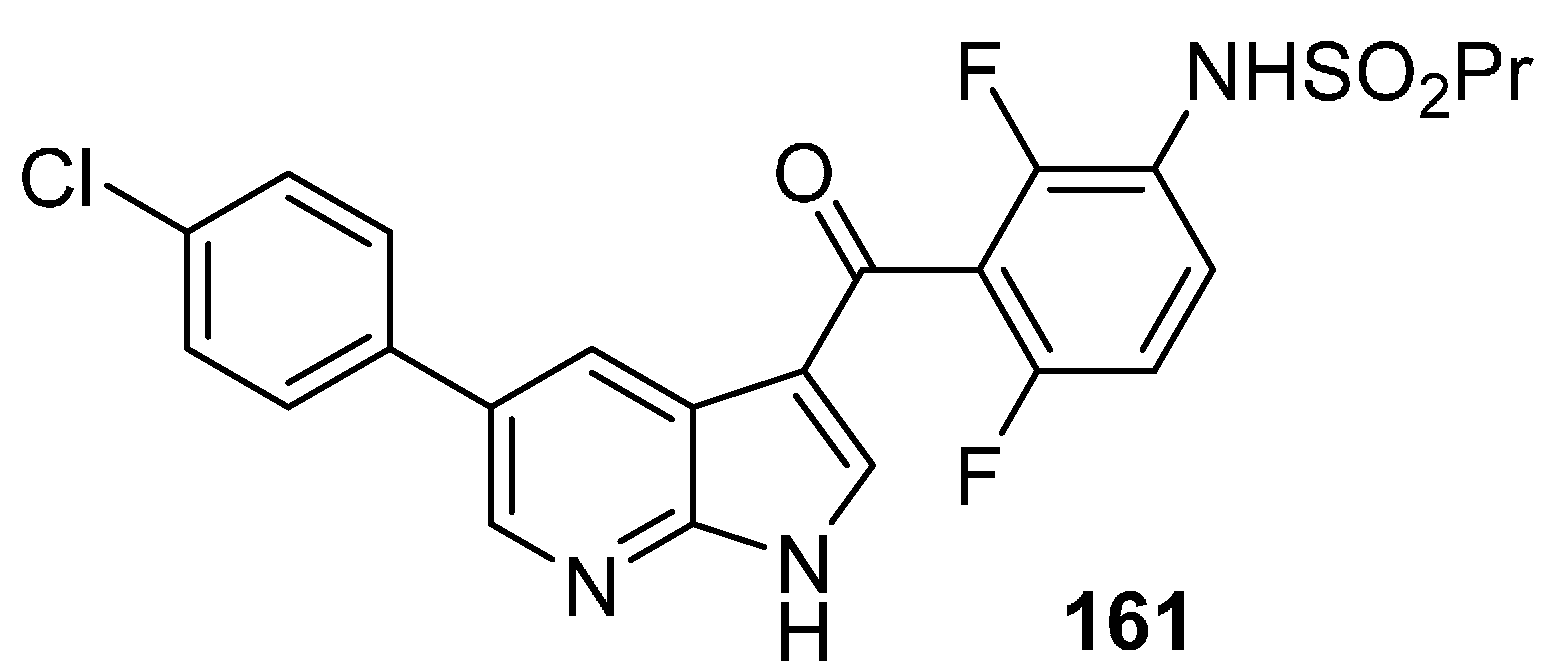
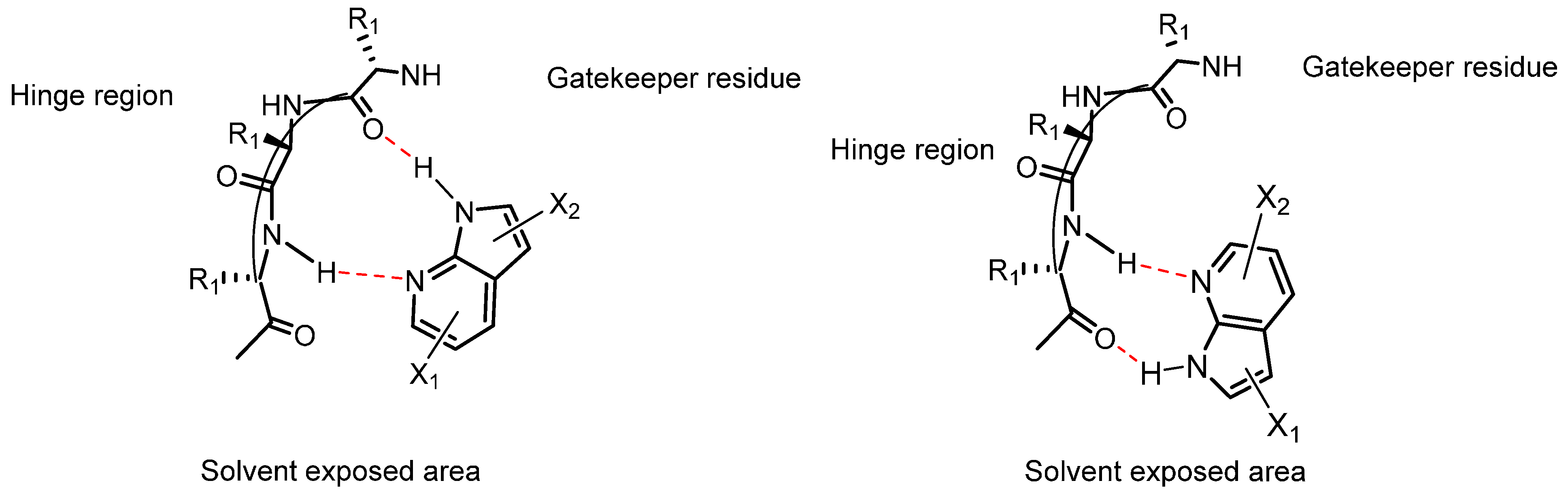
21. Natural Products as Kinase Inhibitors
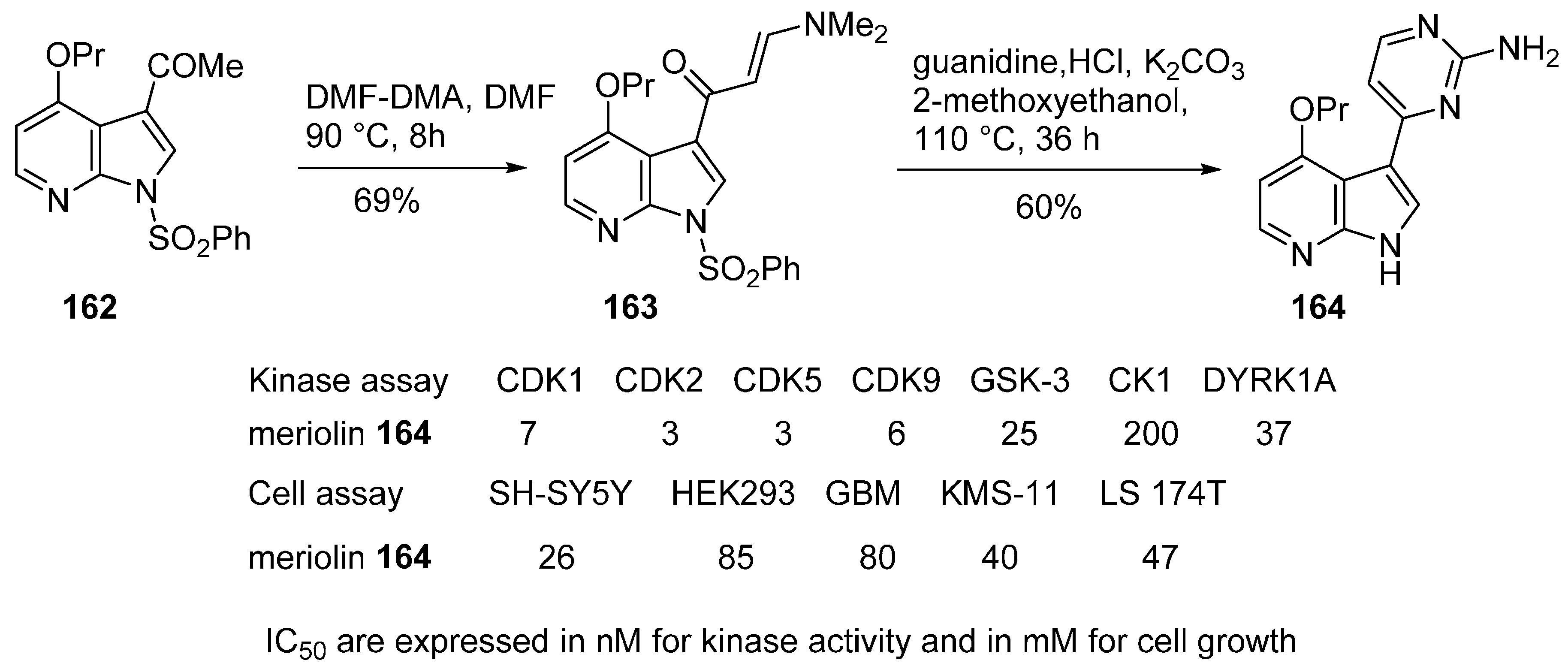
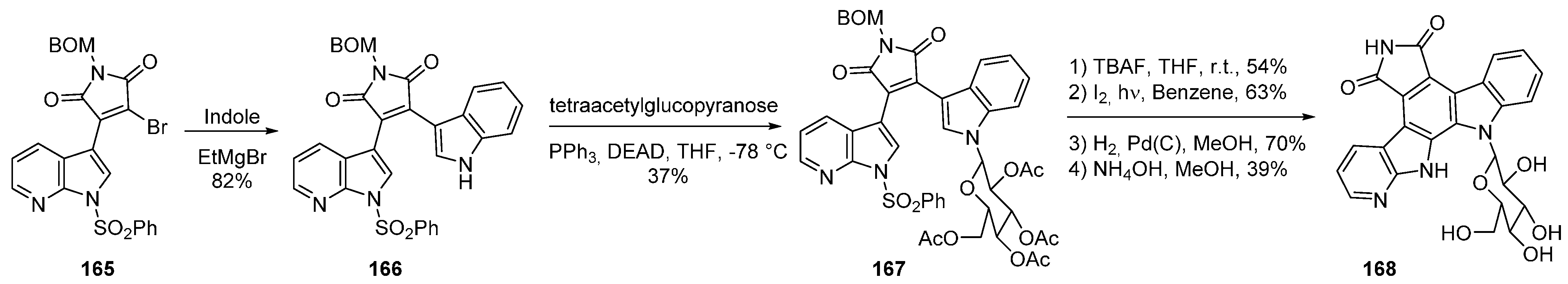


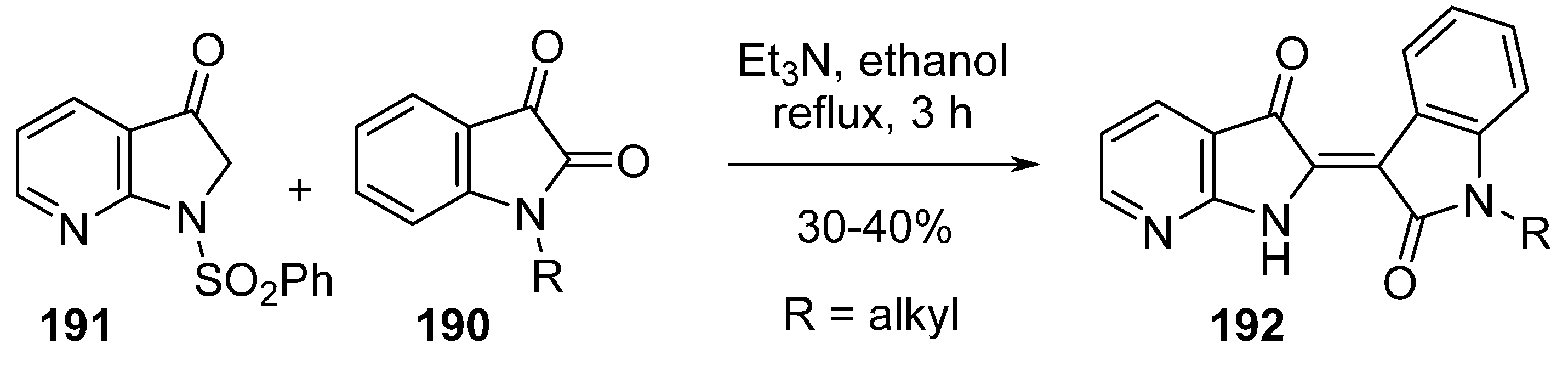
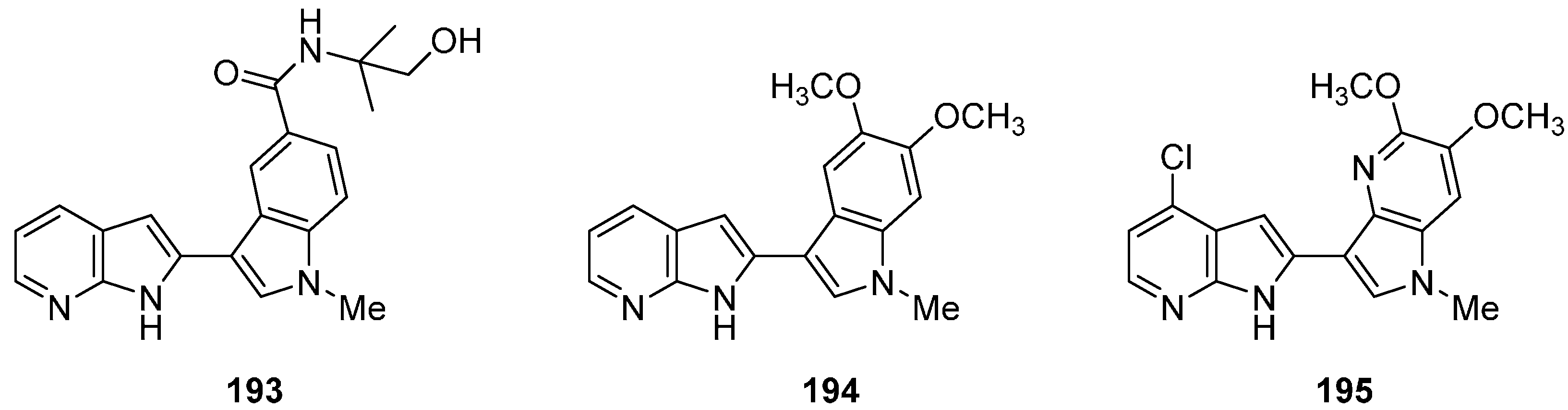

22. Conclusions
Acknowledgments
Conflicts of Interest
References
- Yang, S.-W.; Abdel-Kader, M.; Malone, S.; Werkhoven, M.C.M.; Wisse, J.H.; Bursuker, I.; Neddermann, K.; Fairchild, C.; Raventos-Suarez, C.; Menendez, A.T.; et al. Synthesis and Biological Evaluation of Analogues of Cryptolepine, an Alkaloid Isolated from the Suriname Rainforest1. J. Nat. Prod. 1999, 62, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Paulo, A.; Gomes, E.T.; Houghton, P.J. New Alkaloids from Cryptolepis sanguinolenta. J. Nat. Prod. 1995, 58, 1485–1491. [Google Scholar] [CrossRef]
- Ahaidar, A.; Fernández, D.; Danelón, G.; Cuevas, C.; Manzanares, I.; Albericio, F.; Joule, J.A.; Alvarez, M. Total syntheses of variolin B and deoxyvariolin B. J. Org. Chem. 2003, 68, 10020–10029. [Google Scholar] [CrossRef] [PubMed]
- Choshi, T.; Yamada, S.; Sugino, E.; Kuwada, T.; Hibino, S. Total synthesis of grossularines-1 and -2. J. Org. Chem. 1995, 60, 5899–5904. [Google Scholar] [CrossRef]
- Simone, M.; Erba, E.; Damia, G.; Vikhanskaya, F.; di Francesco, A.M.; Riccardi, R.; Bailly, C.; Cuevas, C.; Fernandez Sousa-Faro, J.M.; D’Incalci, M. Variolin B and its derivate deoxy-variolin B: New marine natural compounds with cyclin-dependent kinase inhibitor activity. Eur. J. Cancer Oxf. Engl. 1990 2005, 41, 2366–2377. [Google Scholar]
- Prudhomme, M. Rebeccamycin analogues as anti-cancer agents. Eur. J. Med. Chem. 2003, 38, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-C.; Ye, H.; Conway, B.R.; Derian, C.K.; Addo, M.F.; Kuo, G.-H.; Hecker, L.R.; Croll, D.R.; Li, J.; Westover, L.; et al. 3-(7-Azaindolyl)-4-arylmaleimides as potent, selective inhibitors of glycogen synthase kinase-3. Bioorg. Med. Chem. Lett. 2004, 14, 3245–3250. [Google Scholar] [CrossRef] [PubMed]
- Grant, E.R.; Errico, M.A.; Emanuel, S.L.; Benjamin, D.; McMillian, M.K.; Wadsworth, S.A.; Zivin, R.A.; Zhong, Z. Protection against glutamate toxicity through inhibition of the p44/42 mitogen-activated protein kinase pathway in neuronally differentiated P19 cells. Biochem. Pharmacol. 2001, 62, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Dugar, S. Azaindole Derivatives as Inhibitors of p38 Kinase. Patent WO2004032874, 9 October 2003. [Google Scholar]
- Showell, G.A.; Emms, F.; Marwood, R.; O’Connor, D.; Patel, S.; Leeson, P.D. Binding of 2,4-disubstituted morpholines at human D4 dopamine receptors. Bioorg. Med. Chem. 1998, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kulagowski, J.J.; Broughton, H.B.; Curtis, N.R.; Mawer, I.M.; Ridgill, M.P.; Baker, R.; Emms, F.; Freedman, S.B.; Marwood, R.; Patel, S.; et al. 3-((4-(4-Chlorophenyl)piperazin-1-yl)-methyl)-1H-pyrrolo-2,3-b-pyridine: An antagonist with high affinity and selectivity for the human dopamine D4 receptor. J. Med. Chem. 1996, 39, 1941–1942. [Google Scholar] [CrossRef] [PubMed]
- Fonquerna, S.; Miralpeix, M.; Pagès, L.; Puig, C.; Cardús, A.; Antón, F.; Vilella, D.; Aparici, M.; Prieto, J.; Warrellow, G.; et al. Synthesis and structure-activity relationships of piperidinylpyrrolopyridine derivatives as potent and selective H1 antagonists. Bioorg. Med. Chem. Lett. 2005, 15, 1165–1167. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Becker, M.; Choi-Sledeski, Y.M.; Davis, R.S.; Salvino, J.M.; Chu, V.; Brown, K.D.; Pauls, H.W. Solid-phase parallel synthesis of azarene pyrrolidinones as factor Xa inhibitors. Bioorg. Med. Chem. Lett. 2000, 10, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, P.E.J.; Stanton, M.G.; Dorsey, B.D.; Lyle, T.A.; McDonough, C.; Sanders, W.M.; Savage, K.L.; Naylor-Olsen, A.M.; Krueger, J.A.; Lewis, S.D.; et al. Azaindoles: Moderately basic P1 groups for enhancing the selectivity of thrombin inhibitors. Bioorg. Med. Chem. Lett. 2003, 13, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Horton, D.A.; Bourne, G.T.; Smythe, M.L. The Combinatorial Synthesis of Bicyclic Privileged Structures or Privileged Substructures. Chem. Rev. 2003, 103, 893–930. [Google Scholar] [CrossRef] [PubMed]
- De Sá Alves, F.R.; Barreiro, E.J.; Fraga, C.A.M. From nature to drug discovery: The indole scaffold as a “privileged structure”. Mini Rev. Med. Chem. 2009, 9, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Kumar, P.; Pathak, D. Biological importance of the indole nucleus in recent years: A comprehensive review. J. Heterocycl. Chem. 2010, 47, 491–502. [Google Scholar]
- Perry, N.B.; Ettouati, L.; Litaudon, M.; Blunt, J.W.; Munro, M.H.G.; Parkin, S.; Hope, H. Alkaloids from the antarctic sponge Kirkpatrickia varialosa: Part 1: Variolin b, a new antitumour and antiviral compound. Tetrahedron 1994, 50, 3987–3992. [Google Scholar] [CrossRef]
- Welsch, M.E.; Snyder, S.A.; Stockwell, B.R. Privileged scaffolds for library design and drug discovery. Curr. Opin. Chem. Biol. 2010, 14, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.R.; Carter, E.J.; Huff, B.C.; Morris, J.C. Variolins and related alkaloids. Chem. Rev. 2009, 109, 3080–3098. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J. Indoles, oxindoles, and azaindoles. In Heterocyclic Chemistry in Drug Discovery; John wiley and Sons Inc.: Hoboken, NJ, USA, 2013; pp. 54–118. [Google Scholar]
- Ila, H.; Markiewicz, J.; Malakhov, V.; Knochel, P. Metalated Indoles, Indazoles, Benzimidazoles, and Azaindoles and Their Synthetic Applications. Synthesis 2013, 45, 2343–2371. [Google Scholar] [CrossRef]
- Mérour, J.-Y.; Routier, S.; Suzenet, F.; Joseph, B. Recent advances in the synthesis and properties of 4-, 5-, 6- or 7-azaindoles. Tetrahedron 2013, 69, 4767–4834. [Google Scholar] [CrossRef]
- Ölgen, S. Recent development of new substituted indole and azaindole derivatives as anti-HIV agents. Mini Rev. Med. Chem. 2013, 13, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Boulahjar, R.; Ouach, A.; Matteo, C.; Bourg, S.; Ravache, M.; le Guével, R.; Marionneau, S.; Oullier, T.; Lozach, O.; Meijer, L.; et al. Novel tetrahydropyrido[1,2-a]isoindolone derivatives (valmerins): Potent cyclin-dependent kinase/glycogen synthase kinase 3 inhibitors with antiproliferative activities and antitumor effects in human tumor xenografts. J. Med. Chem. 2012, 55, 9589–9606. [Google Scholar] [CrossRef] [PubMed]
- Dehbi, O.; Tikad, A.; Bourg, S.; Bonnet, P.; Lozach, O.; Meijer, L.; Aadil, M.; Akssira, M.; Guillaumet, G.; Routier, S. Synthesis and optimization of an original V-shaped collection of 4–7-disubstituted Pyrido[3,2-d]pyrimidines as CDK5 and DYRK1A inhibitors. Eur. J. Med. Chem. 2014, 80, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Kassis, P.; Brzeszcz, J.; Bénéteau, V.; Lozach, O.; Meijer, L.; le Guével, R.; Guillouzo, C.; Lewiński, K.; Bourg, S.; Colliandre, L.; et al. Synthesis and biological evaluation of new 3-(6-hydroxyindol-2-yl)-5-(Phenyl) pyridine or pyrazine V-Shaped molecules as kinase inhibitors and cytotoxic agents. Eur. J. Med. Chem. 2011, 46, 5416–5434. [Google Scholar] [CrossRef] [PubMed]
- Neagoie, C.; Vedrenne, E.; Buron, F.; Mérour, J.-Y.; Rosca, S.; Bourg, S.; Lozach, O.; Meijer, L.; Baldeyrou, B.; Lansiaux, A.; et al. Synthesis of chromeno[3,4-b]indoles as Lamellarin D analogues: A novel DYRK1A inhibitor class. Eur. J. Med. Chem. 2012, 49, 379–396. [Google Scholar] [CrossRef] [PubMed]
- Lefoix, M.; Coudert, G.; Routier, S.; Pfeiffer, B.; Caignard, D.-H.; Hickman, J.; Pierré, A.; Golsteyn, R.M.; Léonce, S.; Bossard, C.; et al. Novel 5-azaindolocarbazoles as cytotoxic agents and Chk1 inhibitors. Bioorg. Med. Chem. 2008, 16, 5303–5321. [Google Scholar] [CrossRef] [PubMed]
- Saurat, T.; Buron, F.; Rodrigues, N.; de Tauzia, M.-L.; Colliandre, L.; Bourg, S.; Bonnet, P.; Guillaumet, G.; Akssira, M.; Corlu, A.; et al. Design, synthesis, and biological activity of pyridopyrimidine scaffolds as novel PI3K/mTOR dual inhibitors. J. Med. Chem. 2014, 57, 613–631. [Google Scholar] [CrossRef] [PubMed]
- Zuccotto, F.; Ardini, E.; Casale, E.; Angiolini, M. Through the “gatekeeper door”: Exploiting the active kinase conformation. J. Med. Chem. 2010, 53, 2681–2694. [Google Scholar] [CrossRef] [PubMed]
- Youl, E.; Bardy, G.; Magous, R.; Cros, G.; Sejalon, F.; Virsolvy, A.; Richard, S.; Quignard, J.F.; Gross, R.; Petit, P.; et al. Quercetin potentiates insulin secretion and protects INS-1 pancreatic β-cells against oxidative damage via the ERK1/2 pathway. Br. J. Pharmacol. 2010, 161, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Gruson, D.; Ginion., A.; Decroly, N.; Lause, P.; Vanoverschelde, J.-L.; Ketelslegers, J.-M.; Bertrand, L.; Thissen, J.-P. Urocortin-induced cardiomyocytes hypertrophy is associated with regulation of the GSK-3β pathway. Heart Vessels 2012, 27, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.D.; Flanagan, M.E.; Telliez, J.-B. Discovery and development of Janus kinase (JAK) inhibitors for inflammatory diseases. J. Med. Chem. 2014, 57, 5023–5038. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.-F.; Meng, J.; Li, Y.-H.; Yu, J.-Z.; Liu, C.-Y.; Feng, L.; Yang, W.-F.; Li, J.-L.; Feng, Q.-J.; Xiao, B.-G.; et al. The inhibition of Rho kinase blocks cell migration and accumulation possibly by challenging inflammatory cytokines and chemokines on astrocytes. J. Neurol. Sci. 2014, 343, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Castanares-Zapatero, D.; Bouleti, C.; Sommereyns, C.; Gerber, B.; Lecut, C.; Mathivet, T.; Horckmans, M.; Communi, D.; Foretz, M.; Vanoverschelde, J.-L.; et al. Connection between cardiac vascular permeability, myocardial edema, and inflammation during sepsis: Role of the α1AMP-activated protein kinase isoform. Crit. Care Med. 2013, 41, e411–e422. [Google Scholar] [CrossRef] [PubMed]
- Demeulder, B.; Zarrinpashneh, E.; Ginion, A.; Viollet, B.; Hue, L.; Rider, M.H.; Vanoverschelde, J.-L.; Beauloye, C.; Horman, S.; Bertrand, L. Differential regulation of eEF2 and p70S6K by AMPKalpha2 in heart. Biochim. Biophys. Acta 2013, 1832, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Erickson, J.R. Mechanisms of CaMKII Activation in the Heart. Front. Pharmacol. 2014, 5, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Huynh, T.N.; Santini, E.; Klann, E. Requirement of Mammalian target of rapamycin complex 1 downstream effectors in cued fear memory reconsolidation and its persistence. J. Neurosci. 2014, 34, 9034–9039. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Liu, N.-K.; Xu, X.-M. PTEN/PI3K and MAPK signaling in protection and pathology following CNS injuries. Front. Biol. 2013, 8, 421–433. [Google Scholar] [CrossRef]
- De Cárcer, G.; Perez de Castro, I.; Malumbres, M. Inhibiting Cell Kinase in Cancer Therapy. Front. Med Chem. 2012, 6, 154–188. [Google Scholar]
- Zámečníkova, A. Novel approaches to the development of tyrosine kinase inhibitors and their role in the fight against cancer. Expert Opin. Drug Discov. 2014, 9, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Hebert-Chatelain, E. Src kinases are important regulators of mitochondrial functions. Int. J. Biochem. Cell Biol. 2013, 45, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Pitts, T.M.; Davis, S.L.; Eckhardt, S.G.; Bradshaw-Pierce, E.L. Targeting nuclear kinases in cancer: Development of cell cycle kinase inhibitors. Pharmacol. Ther. 2014, 142, 258–269. [Google Scholar] [CrossRef]
- Roskoski, R. The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol. Res. 2014, 79, 34–74. [Google Scholar] [CrossRef] [PubMed]
- Rupani, A.; Modi, C.; Desai, S.; Rege, J. Primary anaplastic large cell lymphoma of central nervous system—A case report. J. Postgrad. Med. 2005, 51, 326–327. [Google Scholar] [PubMed]
- Gummadi, V.R.; Rajagopalan, S.; Looi, C.-Y.; Paydar, M.; Renukappa, G.A.; Ainan, B.R.; Krishnamurthy, N.R.; Panigrahi, S.K.; Mahasweta, K.; Raghuramachandran, S.; et al. Discovery of 7-azaindole based anaplastic lymphoma kinase (ALK) inhibitors: Wild type and mutant (L1196M) active compounds with unique binding mode. Bioorg. Med. Chem. Lett. 2013, 23, 4911–4918. [Google Scholar] [CrossRef] [PubMed]
- Pollard, J.R.; Mortimore, M. Discovery and development of aurora kinase inhibitors as anticancer agents. J. Med. Chem. 2009, 52, 2629–2651. [Google Scholar] [CrossRef] [PubMed]
- Bouloc, N.; Large, J.M.; Kosmopoulou, M.; Sun, C.; Faisal, A.; Matteucci, M.; Reynisson, J.; Brown, N.; Atrash, B.; Blagg, J.; et al. Structure-based design of imidazo[1,2-a]pyrazine derivatives as selective inhibitors of Aurora-A kinase in cells. Bioorg. Med. Chem. Lett. 2010, 20, 5988–5993. [Google Scholar] [CrossRef] [PubMed]
- Bavetsias, V.; Faisal, A.; Crumpler, S.; Brown, N.; Kosmopoulou, M.; Joshi, A.; Atrash, B.; Pérez-Fuertes, Y.; Schmitt, J.A.; Boxall, K.J.; et al. Aurora isoform selectivity: Design and synthesis of imidazo[4,5-b]pyridine derivatives as highly selective inhibitors of Aurora-A kinase in cells. J. Med. Chem. 2013, 56, 9122–9135. [Google Scholar] [CrossRef]
- Adams, N.D.; Adams, J.L.; Burgess, J.L.; Chaudhari, A.M.; Copeland, R.A.; Donatelli, C.A.; Drewry, D.H.; Fisher, K.E.; Hamajima, T.; Hardwicke, M.A.; et al. Discovery of GSK1070916, a potent and selective inhibitor of Aurora B/C kinase. J. Med. Chem. 2010, 53, 3973–4001. [Google Scholar] [CrossRef] [PubMed]
- Medina, J.R.; Grant, S.W.; Axten, J.M.; Miller, W.H.; Donatelli, C.A.; Hardwicke, M.A.; Oleykowski, C.A.; Liao, Q.; Plant, R.; Xiang, H. Discovery of a new series of Aurora inhibitors through truncation of GSK1070916. Bioorg. Med. Chem. Lett. 2010, 20, 2552–2555. [Google Scholar] [CrossRef] [PubMed]
- Seefeld, M.A.; Hamajima, T.; Jung, D.K.; Nakamura, H.; Reid, P.R.; Reno, M.J.; Rouse, M.B.; Heerding, D.A.; Tang, J.; Wang, J. Preparation of Pyrrolo[2,3-b]pyridines as Inhibitors of Akt Activity. Patent WO 2007076423, 5 July 2007. [Google Scholar]
- Hardwicke, M.A.; Oleykowski, C.A.; Plant, R.; Wang, J.; Liao, Q.; Moss, K.; Newlander, K.; Adams, J.L.; Dhanak, D.; Yang, J.; et al. GSK1070916, a potent Aurora B/C kinase inhibitor with broad antitumor activity in tissue culture cells and human tumor xenograft models. Mol. Cancer Ther. 2009, 8, 1808–1817. [Google Scholar] [CrossRef] [PubMed]
- Lan, P.; Chen, W.-N.; Sun, P.-H.; Chen, W.-M. 3D-QSAR and molecular docking studies of azaindole derivatives as Aurora B kinase inhibitors. J. Mol. Model. 2011, 17, 1191–1205. [Google Scholar] [CrossRef] [PubMed]
- Harrington, P.E.; Bourbeau, M.P.; Fotsch, C.; Frohn, M.; Pickrell, A.J.; Reichelt, A.; Sham, K.; Siegmund, A.C.; Bailis, J.M.; Bush, T.; et al. The optimization of aminooxadiazoles as orally active inhibitors of Cdc7. Bioorg. Med. Chem. Lett. 2013, 23, 6396–6400. [Google Scholar] [CrossRef] [PubMed]
- Bryan, M.C.; Falsey, J.R.; Frohn, M.; Reichelt, A.; Yao, G.; Bartberger, M.D.; Bailis, J.M.; Zalameda, L.; Miguel, T.S.; Doherty, E.M.; et al. N-substituted azaindoles as potent inhibitors of Cdc7 kinase. Bioorg. Med. Chem. Lett. 2013, 23, 2056–2060. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Stewart, K.D.; Florjancic, A.S.; Harlan, J.E.; Merta, P.J.; Przytulinska, M.; Soni, N.; Swinger, K.K.; Zhu, H.; Johnson, E.F.; et al. Azaindole-Based Inhibitors of Cdc7 Kinase: Impact of the Pre-DFG Residue, Val 195. ACS Med. Chem. Lett. 2013, 4, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Woods, K.W.; Lai, C.; Miyashiro, J.M.; Tong, Y.; Florjancic, A.S.; Han, E.K.; Soni, N.; Shi, Y.; Lasko, L.; Leverson, J.D.; et al. Aminopyrimidinone Cdc7 Kinase Inhibitors. Bioorg. Med. Chem. Lett. 2012, 22, 1940–1943. [Google Scholar] [CrossRef] [PubMed]
- Ermoli, A.; Bargiotti, A.; Brasca, M.G.; Ciavolella, A.; Colombo, N.; Fachin, G.; Isacchi, A.; Menichincheri, M.; Molinari, A.; Montagnoli, A.; et al. Cell division cycle 7 kinase inhibitors: 1H-pyrrolo[2,3-b]pyridines, synthesis and structure-activity relationships. J. Med. Chem. 2009, 52, 4380–4390. [Google Scholar] [CrossRef] [PubMed]
- Koolman, H.; Heinrich, T.; Böttcher, H.; Rautenberg, W.; Reggelin, M. Syntheses of novel 2,3-diaryl-substituted 5-cyano-4-azaindoles exhibiting c-Met inhibition activity. Bioorg. Med. Chem. Lett. 2009, 19, 1879–1882. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.; Lumb, S.; Franklin, R.J.; Gascon-Simorte, J.M.; Calmiano, M.; Riche, K.L.; Lallemand, B.; Keyaerts, J.; Edwards, H.; Maloney, A.; et al. Discovery of 4-azaindoles as novel inhibitors of c-Met kinase. Bioorg. Med. Chem. Lett. 2009, 19, 2780–2784. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-W.; Wei, D.; Schroeder, G.M.; Cornelius, L.A.M.; Kim, K.; Chen, X.-T.; Schmidt, R.J.; Williams, D.K.; Tokarski, J.S.; An, Y.; et al. Discovery of orally active pyrrolopyridine- and aminopyridine-based Met kinase inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 3224–3229. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.K.; Chen, X.-T.; Tarby, C.; Kaltenbach, R.; Cai, Z.-W.; Tokarski, J.S.; An, Y.; Sack, J.S.; Wautlet, B.; Gullo-Brown, J.; et al. Design, synthesis and structure-Activity relationships of novel biarylamine-based Met kinase inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 2998–3002. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Zhang, L.; Schmidt, R.; Cai, Z.-W.; Wei, D.; Williams, D.K.; Lombardo, L.J.; Trainor, G.L.; Xie, D.; Zhang, Y.; et al. Discovery of pyrrolopyridine-pyridone based inhibitors of Met kinase: Synthesis, X-ray crystallographic analysis, and biological activities. J. Med. Chem. 2008, 51, 5330–5341. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.; Joost, H.G. Structural and functional characteristics of Dyrk, a novel subfamily of protein kinases with dual specificity. Prog. Nucleic Acid Res. Mol. Biol. 1999, 62, 1–17. [Google Scholar] [PubMed]
- Becker, W.; Sippl, W. Activation, regulation, and inhibition of DYRK1A. FEBS J. 2011, 278, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Aranda, S.; Laguna, A.; de la Luna, S. DYRK family of protein kinases: Evolutionary relationships, biochemical properties, and functional roles. FASEB J. 2011, 25, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Hämmerle, B.; Elizalde, C.; Galceran, J.; Becker, W.; Tejedor, F.J. The MNB/DYRK1A protein kinase: Neurobiological functions and Down syndrome implications. J. Neural Transm. 2003, 67, 129–137. [Google Scholar]
- Park, J.; Song, W.-J.; Chung, K.C. Function and regulation of Dyrk1A: Towards understanding Down syndrome. Cell. Mol. Life Sci. CMLS 2009, 66, 3235–3240. [Google Scholar] [CrossRef]
- Tejedor, F.J.; Hämmerle, B. MNB/DYRK1A as a multiple regulator of neuronal development. FEBS J. 2011, 278, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Martí, E.; Altafaj, X.; Dierssen, M.; de la Luna, S.; Fotaki, V.; Alvarez, M.; Pérez-Riba, M.; Ferrer, I.; Estivill, X. Dyrk1A expression pattern supports specific roles of this kinase in the adult central nervous system. Brain Res. 2003, 964, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, I.; Barrachina, M.; Puig, B.; Martínez de Lagrán, M.; Martí, E.; Avila, J.; Dierssen, M. Constitutive Dyrk1A is abnormally expressed in Alzheimer disease, Down syndrome, Pick disease, and related transgenic models. Neurobiol. Dis. 2005, 20, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Wegiel, J.; Gong, C.-X.; Hwang, Y.-W. The role of DYRK1A in neurodegenerative diseases. FEBS J. 2011, 278, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Ionescu, A.; Dufrasne, F.; Gelbcke, M.; Jabin, I.; Kiss, R.; Lamoral-Theys, D. DYRK1A kinase inhibitors with emphasis on cancer. Mini Rev. Med. Chem. 2012, 12, 1315–1329. [Google Scholar] [PubMed]
- Gourdain, S.; Dairou, J.; Denhez, C.; Bui, L.C.; Rodrigues-Lima, F.; Janel, N.; Delabar, J.M.; Cariou, K.; Dodd, R.H. Development of DANDYs, new 3,5-diaryl-7-azaindoles demonstrating potent DYRK1A kinase inhibitory activity. J. Med. Chem. 2013, 56, 9569–9585. [Google Scholar] [CrossRef] [PubMed]
- Pin, F.; Buron, F.; Saab, F.; Colliandre, L.; Bourg, S.; Schoentgen, F.; Guevel, R.L.; Guillouzo, C.; Routier, S. Synthesis and biological evaluation of 2,3-bis(het)aryl-4-azaindole derivatives as protein kinase inhibitors. MedChemComm 2011, 2, 899–903. [Google Scholar] [CrossRef]
- Heinrich, T.; Seenisamy, J.; Emmanuvel, L.; Kulkarni, S.S.; Bomke, J.; Rohdich, F.; Greiner, H.; Esdar, C.; Krier, M.; Grädler, U.; et al. Fragment-based discovery of new highly substituted 1H-pyrrolo[2,3-b]- and 3H-imidazolo[4,5-b]-pyridines as focal adhesion kinase inhibitors. J. Med. Chem. 2013, 56, 1160–1170. [Google Scholar] [CrossRef] [PubMed]
- Liddle, J.; Bamborough, P.; Barker, M.D.; Campos, S.; Cousins, R.P.C.; Cutler, G.J.; Hobbs, H.; Holmes, D.S.; Ioannou, C.; Mellor, G.W.; et al. 4-Phenyl-7-azaindoles as potent and selective IKK2 inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 2504–2508. [Google Scholar] [CrossRef] [PubMed]
- Liddle, J.; Bamborough, P.; Barker, M.D.; Campos, S.; Chung, C.-W.; Cousins, R.P.C.; Faulder, P.; Heathcote, M.L.; Hobbs, H.; Holmes, D.S.; et al. 4-Phenyl-7-azaindoles as potent, selective and bioavailable IKK2 inhibitors demonstrating good in vivo efficacy. Bioorg. Med. Chem. Lett. 2012, 22, 5222–5226. [Google Scholar] [CrossRef] [PubMed]
- James, C.; Ugo, V.; le Couédic, J.-P.; Staerk, J.; Delhommeau, F.; Lacout, C.; Garçon, L.; Raslova, H.; Berger, R.; Bennaceur-Griscelli, A.; et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005, 434, 1144–1148. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.L.; Wadleigh, M.; Cools, J.; Ebert, B.L.; Wernig, G.; Huntly, B.J.P.; Boggon, T.J.; Wlodarska, I.; Clark, J.J.; Moore, S.; et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005, 7, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Kralovics, R.; Passamonti, F.; Buser, A.S.; Teo, S.-S.; Tiedt, R.; Passweg, J.R.; Tichelli, A.; Cazzola, M.; Skoda, R.C. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N. Engl. J. Med. 2005, 352, 1779–1790. [Google Scholar] [CrossRef] [PubMed]
- Baxter, E.J.; Scott, L.M.; Campbell, P.J.; East, C.; Fourouclas, N.; Swanton, S.; Vassiliou, G.S.; Bench, A.J.; Boyd, E.M.; Curtin, N.; et al. Cancer Genome Project Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005, 365, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Xing, S.; Li, Z.; Fu, X.; Li, Q.; Krantz, S.B.; Zhao, Z.J. Identification of an acquired JAK2 mutation in polycythemia vera. J. Biol. Chem. 2005, 280, 22788–22792. [Google Scholar] [CrossRef] [PubMed]
- Tono, C.; Xu, G.; Toki, T.; Takahashi, Y.; Sasaki, S.; Terui, K.; Ito, E. JAK2 Val617Phe activating tyrosine kinase mutation in juvenile myelomonocytic leukemia. Leukemia 2005, 19, 1843–1844. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, T.; Ledeboer, M.W.; Duffy, J.P.; Salituro, F.G.; Pierce, A.C.; Zuccola, H.J.; Block, E.; Shlyakter, D.; Hogan, J.K.; Bennani, Y.L. A novel chemotype of kinase inhibitors: Discovery of 3,4-ring fused 7-azaindoles and deazapurines as potent JAK2 inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Duffy, J.P.; Wang, J.; Halas, S.; Salituro, F.G.; Pierce, A.C.; Zuccola, H.J.; Black, J.R.; Hogan, J.K.; Jepson, S.; et al. Janus kinase 2 inhibitors. Synthesis and characterization of a novel polycyclic azaindole. J. Med. Chem. 2009, 52, 7938–7941. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R. Novel small-molecular therapeutics for rheumatoid arthritis. Curr. Opin. Rheumatol. 2012, 24, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Farmer, L.; Martinez-Botella, G.; Pierce, A.; Salituro, F.; Wang, J.; Wannamker, M.; Wang, T. Azaindoles Useful as Inhibitors of Janus Kinases. Patent WO2007084557, 26 July 2007. [Google Scholar]
- Zhang, C.; Ibrahim, P.N.; Zhang, J.; Burton, E.A.; Habets, G.; Zhang, Y.; Powell, B.; West, B.L.; Matusow, B.; Tsang, G.; et al. Design and pharmacology of a highly specific dual FMS and KIT kinase inhibitor. Proc. Natl. Acad. Sci. USA 2013, 110, 5689–5694. [Google Scholar] [CrossRef] [PubMed]
- Tunduguru, R.; Chiu, T.T.; Ramalingam, L.; Elmendorf, J.S.; Klip, A.; Thurmond, D.C. Signaling of the p21-activated kinase (PAK1) coordinates insulin-stimulated actin remodeling and glucose uptake in skeletal muscle cells. Biochem. Pharmacol. 2014. [Google Scholar] [CrossRef]
- Ji, X.; Zhang, W.; Zhang, L.; Zhang, L.; Zhang, Y.; Tang, P. Inhibition of p21-activated kinase 1 by IPA-3 attenuates secondary injury after traumatic brain injury in mice. Brain Res. 2014, 1585, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Prudnikova, T.Y.; Rawat, S.J.; Chernoff, J. Molecular Pathways: Targeting the Kinase Effectors of RHO-Family GTPases. Clin. Cancer Res. 2014. [Google Scholar] [CrossRef]
- McCoull, W.; Hennessy, E.J.; Blades, K.; Box, M.R.; Chuaqui, C.; Dowling, J.E.; Davies, C.D.; Ferguson, A.D.; Goldberg, F.W.; Howe, N.J.; et al. Identification and optimisation of 7-azaindole PAK1 inhibitors with improved potency and kinase selectivity. MedChemComm 2014, 5, 1533–1539. [Google Scholar] [CrossRef]
- Young, P.R.; McLaughlin, M.M.; Kumar, S.; Kassis, S.; Doyle, M.L.; McNulty, D.; Gallagher, T.F.; Fisher, S.; McDonnell, P.C.; Carr, S.A.; et al. Pyridinyl imidazole inhibitors of p38 mitogen-activated protein kinase bind in the ATP site. J. Biol. Chem. 1997, 272, 12116–12121. [Google Scholar] [CrossRef] [PubMed]
- Trejo, A.; Arzeno, H.; Browner, M.; Chanda, S.; Cheng, S.; Comer, D.D.; Dalrymple, S.A.; Dunten, P.; Lafargue, J.; Lovejoy, B.; et al. Design and synthesis of 4-azaindoles as inhibitors of p38 MAP kinase. J. Med. Chem. 2003, 46, 4702–4713. [Google Scholar] [CrossRef] [PubMed]
- Mavunkel, B.J.; Perumattam, J.J.; Tan, X.; Luedtke, G.R.; Lu, Q.; Lim, D.; Kizer, D.; Dugar, S.; Chakravarty, S.; Xu, Y.; et al. Piperidine-based heterocyclic oxalyl amides as potent p38α MAP kinase inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Aparicio, C.; Carnero, A. Pim kinases in cancer: Diagnostic, prognostic and treatment opportunities. Biochem. Pharmacol. 2013, 85, 629–643. [Google Scholar] [CrossRef] [PubMed]
- Nakano, H.; Saito, N.; Parker, L.; Tada, Y.; Abe, M.; Tsuganezawa, K.; Yokoyama, S.; Tanaka, A.; Kojima, H.; Okabe, T.; et al. Rational evolution of a novel type of potent and selective proviral integration site in Moloney murine leukemia virus kinase 1 (PIM1) inhibitor from a screening-hit compound. J. Med. Chem. 2012, 55, 5151–5164. [Google Scholar] [CrossRef] [PubMed]
- Brader, S.; Eccles, S.A. Phosphoinositide 3-kinase signalling pathways in tumor progression, invasion and angiogenesis. Tumori 2004, 90, 2–8. [Google Scholar] [PubMed]
- Samuels, Y.; Wang, Z.; Bardelli, A.; Silliman, N.; Ptak, J.; Szabo, S.; Yan, H.; Gazdar, A.; Powell, S.M.; Riggins, G.J.; et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 2004, 304, 554. [Google Scholar] [CrossRef] [PubMed]
- Wymann, M.P.; Marone, R. Phosphoinositide 3-kinase in disease: Timing, location, and scaffolding. Curr. Opin. Cell Biol. 2005, 17, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Parsons, D.W.; Wang, T.-L.; Samuels, Y.; Bardelli, A.; Cummins, J.M.; DeLong, L.; Silliman, N.; Ptak, J.; Szabo, S.; Willson, J.K.V.; et al. Colorectal cancer: Mutations in a signalling pathway. Nature 2005, 436, 792. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Bader, A.G.; Vogt, P.K. Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic. Proc. Natl. Acad. Sci. USA 2005, 102, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.-W.; Knight, Z.A.; Goldenberg, D.D.; Yu, W.; Mostov, K.E.; Stokoe, D.; Shokat, K.M.; Weiss, W.A. A dual PI3 kinase/mTOR inhibitor reveals emergent efficacy in glioma. Cancer Cell 2006, 9, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Choi, M.-J.; Hong, S.; Lee, H.; Hong, S.-W.; Zheng, H.-M.; Lee, H.-S.; Hong, S.; Hong, S.-S. HS-116, a novel phosphatidylinositol 3-kinase inhibitor induces apoptosis and suppresses angiogenesis of hepatocellular carcinoma through inhibition of the PI3K/AKT/mTOR pathway. Cancer Lett. 2012, 316, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Lee, S.; Kim, B.; Lee, H.; Hong, S.-S.; Hong, S. Discovery of new azaindole-based PI3Kα inhibitors: Apoptotic and antiangiogenic effect on cancer cells. Bioorg. Med. Chem. Lett. 2010, 20, 7212–7215. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Hamajima, T.; Nakano, M.; Sato, H.; Dickerson, S.H.; Lackey, K.E. Knowledge-based design of 7-azaindoles as selective B-Raf inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 4610–4614. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Sessions, E.H.; Pocas, J.R.; Grant, W.; Schröter, T.; Lin, L.; Ruiz, C.; Cameron, M.D.; Schürer, S.; LoGrasso, P.; et al. Discovery and optimization of indoles and 7-azaindoles as Rho kinase (ROCK) inhibitors (part-I). Bioorg. Med. Chem. Lett. 2011, 21, 7107–7112. [Google Scholar] [CrossRef] [PubMed]
- Sessions, E.H.; Chowdhury, S.; Yin, Y.; Pocas, J.R.; Grant, W.; Schröter, T.; Lin, L.; Ruiz, C.; Cameron, M.D.; LoGrasso, P.; et al. Discovery and optimization of indole and 7-azaindoles as Rho kinase (ROCK) inhibitors (part-II). Bioorg. Med. Chem. Lett. 2011, 21, 7113–7118. [Google Scholar] [CrossRef] [PubMed]
- Schirok, H.; Paulsen, H.; Kroh, W.; Chen, G.; Gao, P. Improved Synthesis of the Selective Rho-Kinase Inhibitor 6-Chloro-N4-{3,5-difluoro-4-[(3-methyl-1H-pyrrolo[2,3-b]pyridin-4-yl)oxy]phenyl}pyrimidin-2,4-diamine. Org. Process Res. Dev. 2010, 14, 168–173. [Google Scholar] [CrossRef]
- Schirok, H.; Kast, R.; Figueroa-Pérez, S.; Bennabi, S.; Gnoth, M.J.; Feurer, A.; Heckroth, H.; Thutewohl, M.; Paulsen, H.; Knorr, A.; et al. Design and synthesis of potent and selective azaindole-based Rho kinase (ROCK) inhibitors. ChemMedChem 2008, 3, 1893–1904. [Google Scholar] [CrossRef] [PubMed]
- Kast, R.; Schirok, H.; Figueroa-Pérez, S.; Mittendorf, J.; Gnoth, M.J.; Apeler, H.; Lenz, J.; Franz, J.K.; Knorr, A.; Hütter, J.; et al. Cardiovascular effects of a novel potent and highly selective azaindole-based inhibitor of Rho-kinase. Br. J. Pharmacol. 2007, 152, 1070–1080. [Google Scholar] [CrossRef] [PubMed]
- Dahal, B.K.; Kosanovic, D.; Pamarthi, P.K.; Sydykov, A.; Lai, Y.-J.; Kast, R.; Schirok, H.; Stasch, J.-P.; Ghofrani, H.A.; Weissmann, N.; et al. Therapeutic efficacy of azaindole-1 in experimental pulmonary hypertension. Eur. Respir. J. 2010, 36, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Zoncu, R.; Efeyan, A.; Sabatini, D.M. mTOR: From growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 2011, 12, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Tsou, H.-R.; MacEwan, G.; Birnberg, G.; Zhang, N.; Brooijmans, N.; Toral-Barza, L.; Hollander, I.; Ayral-Kaloustian, S.; Yu, K. 4-Substituted-7-azaindoles bearing a ureidobenzofuranone moiety as potent and selective, ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR). Bioorg. Med. Chem. Lett. 2010, 20, 2259–2263. [Google Scholar] [CrossRef] [PubMed]
- Tsou, H.-R.; MacEwan, G.; Birnberg, G.; Grosu, G.; Bursavich, M.G.; Bard, J.; Brooijmans, N.; Toral-Barza, L.; Hollander, I.; Mansour, T.S.; et al. Discovery and optimization of 2-(4-substituted-pyrrolo[2,3-b]pyridin-3-yl)methylene-4-hydroxybenzofuran-3(2H)-ones as potent and selective ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR). Bioorg. Med. Chem. Lett. 2010, 20, 2321–2325. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Kim, J.; Seo, J.H.; Jung, K.H.; Hong, S.-S.; Hong, S. Design, synthesis, and evaluation of 3,5-disubstituted 7-azaindoles as Trk inhibitors with anticancer and antiangiogenic activities. J. Med. Chem. 2012, 55, 5337–5349. [Google Scholar] [CrossRef] [PubMed]
- Molecular Operating Environment (MOE), 2013.08; Chemical Computing Group Inc.: Montreal, QC, Canada, 2014.
- RCSB Protein Data Bank. Available online: http://www.rcsb.org/ (accessed on 26 August 2014).
- Manning, G.; Whyte, D.B.; Martinez, R.; Hunter, T.; Sudarsanam, S. The protein kinase complement of the human genome. Science 2002, 298, 1912–1934. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, P.D.; Russo, A.A.; Polyak, K.; Gibbs, E.; Hurwitz, J.; Massagué, J.; Pavletich, N.P. Mechanism of CDK activation revealed by the structure of a cyclinA-CDK2 complex. Nature 1995, 376, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Southall, N.T.; Ajay. Kinase patent space visualization using chemical replacements. J. Med. Chem. 2006, 49, 2103–2109. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.A.; Goldberg, F.W. Kinase Drug Discovery; RSC: Cambridge, UK, 2011. [Google Scholar]
- Zhang, J.; Yang, P.L.; Gray, N.S. Targeting cancer with small molecule kinase inhibitors. Nat. Rev. Cancer 2009, 9, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Bollag, G.; Tsai, J.; Zhang, J.; Zhang, C.; Ibrahim, P.; Nolop, K.; Hirth, P. Vemurafenib: The first drug approved for BRAF-mutant cancer. Nat. Rev. Drug Discov. 2012, 11, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Donald, A.; McHardy, T.; Rowlands, M.G.; Hunter, L.-J.K.; Davies, T.G.; Berdini, V.; Boyle, R.G.; Aherne, G.W.; Garrett, M.D.; Collins, I. Rapid evolution of 6-phenylpurine inhibitors of protein kinase B through structure-based design. J. Med. Chem. 2007, 50, 2289–2292. [Google Scholar] [CrossRef] [PubMed]
- Echalier, A.; Bettayeb, K.; Ferandin, Y.; Lozach, O.; Clément, M.; Valette, A.; Liger, F.; Marquet, B.; Morris, J.C.; Endicott, J.A.; et al. Meriolins (3-(pyrimidin-4-yl)-7-azaindoles): Synthesis, kinase inhibitory activity, cellular effects, and structure of a CDK2/cyclin A/meriolin complex. J. Med. Chem. 2008, 51, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Routier, S.; Coudert, G.; Mérour, J.-Y.; Caignard, D.H. First synthesis of symmetrical and non-symmetrical aza indolocarbazoles derivatives. Tetrahedron Lett. 2002, 43, 2561–2564. [Google Scholar] [CrossRef]
- Routier, S.; Ayerbe, N.; Mérour, J.-Y.; Coudert, G.; Bailly, C.; Pierré, A.; Pfeiffer, B.; Caignard, D.-H.; Renard, P. Synthesis and biological evaluation of 7-azaindolocarbazoles. Tetrahedron 2002, 58, 6621–6630. [Google Scholar] [CrossRef]
- Messaoudi, S.; Anizon, F.; Peixoto, P.; David-Cordonnier, M.-H.; Golsteyn, R.M.; Léonce, S.; Pfeiffer, B.; Prudhomme, M. Synthesis and biological activities of 7-aza rebeccamycin analogues bearing the sugar moiety on the nitrogen of the pyridine ring. Bioorg. Med. Chem. 2006, 14, 7551–7562. [Google Scholar] [CrossRef] [PubMed]
- Ganser, C.; Lauermann, E.; Maderer, A.; Stauder, T.; Kramb, J.-P.; Plutizki, S.; Kindler, T.; Moehler, M.; Dannhardt, G. Novel 3-Azaindolyl-4-arylmaleimides exhibiting potent antiangiogenic efficacy, protein kinase inhibition, and antiproliferative activity. J. Med. Chem. 2012, 55, 9531–9540. [Google Scholar] [CrossRef] [PubMed]
- Faul, M.M.; Winneroski, L.L.; Krumrich, C.A. A new one step synthesis of maleimides by condensation of glyoxylate esters with acetamides. Tetrahedron Lett. 1999, 40, 1109–1112. [Google Scholar]
- O’Neill, D.J.; Shen, L.; Prouty, C.; Conway, B.R.; Westover, L.; Xu, J.Z.; Zhang, H.-C.; Maryanoff, B.E.; Murray, W.V.; Demarest, K.T.; et al. Design, synthesis, and biological evaluation of novel 7-azaindolyl-heteroaryl-maleimides as potent and selective glycogen synthase kinase-3β (GSK-3β) inhibitors. Bioorg. Med. Chem. 2004, 12, 3167–3185. [Google Scholar] [PubMed]
- Maderer, A.; Plutizki, S.; Kramb, J.-P.; Göpfert, K.; Linnig, M.; Khillimberger, K.; Ganser, C.; Lauermann, E.; Dannhardt, G.; Galle, P.R.; et al. Moguntinones—New Selective Inhibitors for the Treatment of Human Colorectal Cancer. Mol. Cancer Ther. 2014, 13, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Xu, G.; Lv, D.; Cheng, Z.; Li, J.; Hu, Y. Synthesis and biological evaluation of novel 4-azaindolyl-indolyl-maleimides as glycogen synthase kinase-3β (GSK-3β) inhibitors. Bioorg. Med. Chem. 2009, 17, 4302–4312. [Google Scholar] [CrossRef] [PubMed]
- Kuo, G.-H.; Prouty, C.; DeAngelis, A.; Shen, L.; O’Neill, D.J.; Shah, C.; Connolly, P.J.; Murray, W.V.; Conway, B.R.; Cheung, P.; et al. Synthesis and discovery of macrocyclic polyoxygenated bis-7-azaindolylmaleimides as a novel series of potent and highly selective glycogen synthase kinase-3β inhibitors. J. Med. Chem. 2003, 46, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.H.; Dong, Y.; Wang, T.; Shang, M.H.; Hua, W.Y.; Yao, Q.Z. Synthesis and CDK2 kinase inhibitory activity of 7/7'-azaindirubin derivatives. Chin. Chem. Lett. 2010, 21, 297–300. [Google Scholar] [CrossRef]
- Villaseñor, A.G.; Kondru, R.; Ho, H.; Wang, S.; Papp, E.; Shaw, D.; Barnett, J.W.; Browner, M.F.; Kuglstatter, A. Structural insights for design of potent spleen tyrosine kinase inhibitors from crystallographic analysis of three inhibitor complexes. Chem. Biol. Drug Des. 2009, 73, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.J.; Majid, T.N.; Lai, J.Y.Q.; Morley, A.D.; Amendola, S.; Deprets, S.; Edlin, C. Preparation of Azaindoles as Protein Kinase Inhibitors. Patent No. WO 2001047922, 5 July 2001. [Google Scholar]
- Nemecek, C.; Metz, W.A.; Wentzler, S.; Ding, F.-X.; Venot, C.; Souaille, C.; Dagallier, A.; Maignan, S.; Guilloteau, J.-P.; Bernard, F.; et al. Design of potent IGF1-R inhibitors related to bis-azaindoles. Chem. Biol. Drug Des. 2010, 76, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.H.; Wan, X.Z.; Jiang, B. Syntheses and biological activities of bis(3-indolyl)thiazoles, analogues of marine bis(indole)alkaloid nortopsentins. Bioorg. Med. Chem. Lett. 1999, 9, 569–572. [Google Scholar] [PubMed]
- Jiang, B.; Gu, X.H. Syntheses and cytotoxicity evaluation of bis(indolyl)thiazole, bis(indolyl)pyrazinone and bis(indolyl)pyrazine: Analogues of cytotoxic marine bis(indole) alkaloid. Bioorg. Med. Chem. 2000, 8, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Diana, P.; Carbone, A.; Barraja, P.; Montalbano, A.; Parrino, B.; Lopergolo, A.; Pennati, M.; Zaffaroni, N.; Cirrincione, G. Synthesis and antitumor activity of 3-(2-phenyl-1,3-thiazol-4-yl)-1H-indoles and 3-(2-phenyl-1,3-thiazol-4-yl)-1H-7-azaindoles. ChemMedChem 2011, 6, 1300–1309. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mérour, J.-Y.; Buron, F.; Plé, K.; Bonnet, P.; Routier, S. The Azaindole Framework in the Design of Kinase Inhibitors. Molecules 2014, 19, 19935-19979. https://doi.org/10.3390/molecules191219935
Mérour J-Y, Buron F, Plé K, Bonnet P, Routier S. The Azaindole Framework in the Design of Kinase Inhibitors. Molecules. 2014; 19(12):19935-19979. https://doi.org/10.3390/molecules191219935
Chicago/Turabian StyleMérour, Jean-Yves, Frédéric Buron, Karen Plé, Pascal Bonnet, and Sylvain Routier. 2014. "The Azaindole Framework in the Design of Kinase Inhibitors" Molecules 19, no. 12: 19935-19979. https://doi.org/10.3390/molecules191219935
APA StyleMérour, J.-Y., Buron, F., Plé, K., Bonnet, P., & Routier, S. (2014). The Azaindole Framework in the Design of Kinase Inhibitors. Molecules, 19(12), 19935-19979. https://doi.org/10.3390/molecules191219935