Effects of Croton rhamnifolioides Essential Oil on Aedes aegypti Oviposition, Larval Toxicity and Trypsin Activity
Abstract
:1. Introduction
2. Results and Discussion
| Component a | Fresh Oil [%] | LRI calc. b | Stored Oil [%] | LRI calc. b | LRI Lit. c | |
|---|---|---|---|---|---|---|
| Tricyclene | 0.16 | 921 | 0.52 | 920 | 920 | |
| α-Thujene | 1.49 | 924 | 2.17 | 926 | 926 | |
| α-Pinene | 4.74 | 932 | 9.52 | 932 | 932 | |
| Camphene | 0.65 | 946 | 2.4 | 946 | 946 | |
| Sabinene | 3.04 | 969 | 7.18 | 972 | 972 | |
| β-Pinene | 0.55 | 974 | 0.69 | 974 | 974 | |
| Myrcene | 0.36 | 988 | 0.68 | 991 | 991 | |
| α-Phellandrene | 12.83 | 1002 | 8.37 | 1003 | 1003 | |
| α-Terpinene | 0.31 | 1014 | - | - | 1016 | |
| o-Cymene | 4.60 | 1022 | 14.64 | 1023 | 1023 | |
| Sylvestrene | 3.62 | 1025 | 3.99 | 1028 | 1027 | |
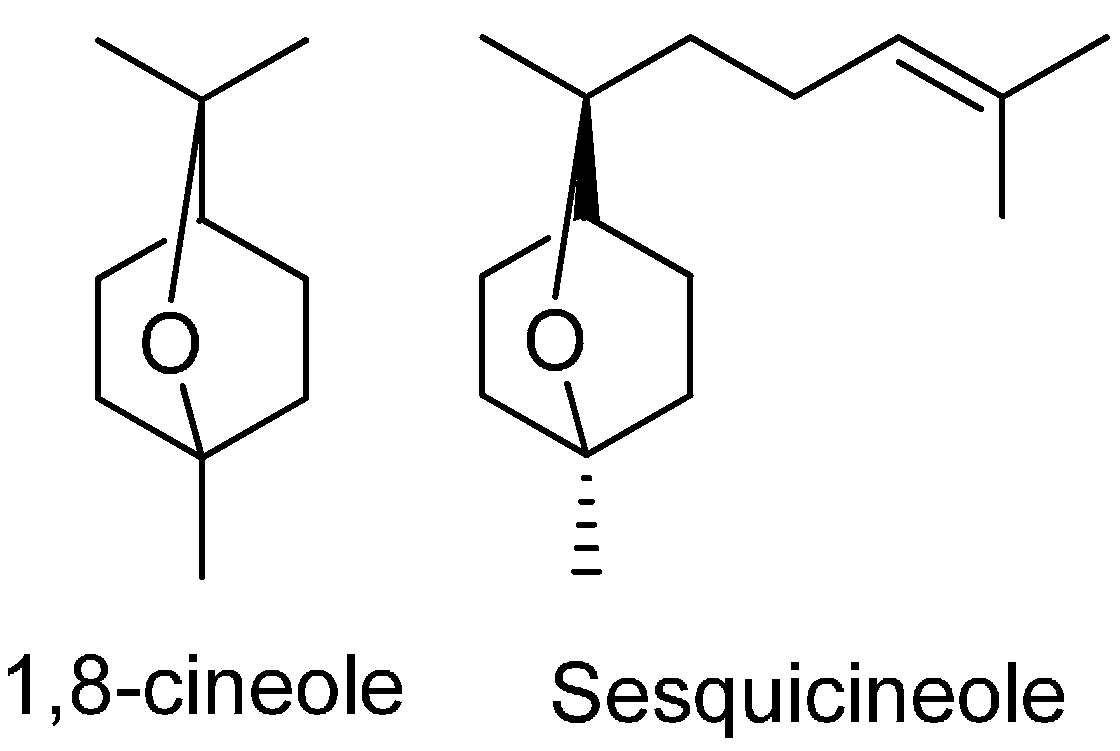
| 1,8-Cineole | 7.24 | 1026 | 18.61 | 1030 | 1030 | |
| Benzyl alcohol | 0.20 | 1026 | - | - | 1033 | |
| (E)-β Ocimene | 0.75 | 1044 | 0.34 | 1049 | 1048 | |
| γ-Terpinene | 1.09 | 1054 | - | - | 1058 | |
| Terpinolene | 0.24 | 1086 | - | - | 1088 | |
| Linalool | 1.32 | 1095 | 1.27 | 1100 | 1100 | |
| Dehydro-sabina ketone | 0.16 | 1117 | - | - | 1120 | |
| (Z)-β-Terpineol | 0.08 | 1140 | - | - | 1139 | |
| Camphor | 0.06 | 1141 | 0.45 | 1144 | 1144 | |
| Pinocarvone | 0.02 | 1160 | - | - | 1162 | |
| Borneol | 0.83 | 1165 | - | - | 1165 | |
| Terpinen-4-ol | 1.90 | 1174 | 0.48 | 1177 | 1177 | |
| α-Terpineol | 1.91 | 1186 | 0.7 | 1190 | 1190 | |
| Myrtenol | 0.31 | 1194 | - | - | 1196 | |
| Methyl chavicol | 0.06 | 1195 | - | - | 1198 | |
| Thymol methyl ether | 0.03 | 1232 | - | - | 1235 | |
| Bornyl acetate | 0.33 | 1284 | 0.72 | 1287 | 1286 | |
| Thymol | 0.05 | 1289 | - | - | 1292 | |
| Carvacrol | 0.20 | 1298 | - | - | 1301 | |
| Myrtenyl acetate | 0.02 | 1324 | - | - | 1326 | |
| δ-Elemene | 0.03 | 1335 | - | - | 1338 | |
| α-Copaene | 0.19 | 1374 | 0.43 | 1378 | 1377 | |
| β-Bourbonene | 0.07 | 1387 | - | - | 1386 | |
| β-Elemene | 0.35 | 1389 | 1.36 | 1394 | 1393 | |
| (Z)-α-Bergamotene | 0.05 | 1411 | - | - | 1417 | |
| (E)-Caryophyllene | 6.33 | 1417 | 4.37 | 1422 | 1422 | |
| (E)α-Bergamotene | 0.14 | 1432 | - | - | 1438 | |
| (Z)-β-Farnesene | 0.04 | 1440 | - | - | 1445 | |
| α-Humulene | 1.17 | 1452 | 0.96 | 1456 | 1457 | |
| 9-epi-(E)-Caryophyllene | 0.80 | 1464 | 0.95 | 1464 | 1465 | |
| γ-Muurolene | 0.04 | 1478 | - | - | 1481 | |
| Germacrene D | 0.99 | 1484 | - | - | 1485 | |
| Viridiflorene | 0.14 | 1496 | 0.71 | 1489 | 1490 | |
| Bicyclogermacrene | 4.59 | 1500 | 2.44 | 1499 | 1501 | |
| (Z)-β-Guaiene | 0.10 | 1502 | - | - | 1504 | |
| α-Bulnesene | 0.05 | 1509 | - | - | 1506 | |
| Germacrene A | 0.21 | 1508 | 0.84 | 1508 | 1510 | |
| Sesquicineole | 16.79 | 1515 | 1.77 | 1516 | 1518 | |
| δ-Cadinene | 0.45 | 1522 | 0.44 | 1526 | 1527 | |
| Germacrene B | 0.06 | 1559 | - | - | 1556 | |
| Spathulenol | 4.14 | 1577 | 4.98 | 1580 | 1580 | |
| Caryophyllene oxide | 3.22 | 1582 | 2.89 | 1586 | 1586 | |
| Viridiflorol | 0.09 | 1592 | - | - | 1596 | |
| epi-α-Cadinol | 2.33 | 1638 | 3.16 | 1644 | 1643 | |
| α-Bisabolol | 0.72 | 1685 | - | - | 1685 | |
| Eudesma-4(15),7-dien-1β-ol | 0.23 | 1687 | - | - | 1688 | |
| Monoterpene hydrocarbons | 32.94 | 48.33 | ||||
| Oxygenated monoterpenes | 15.85 | 24.4 | ||||
| Sesquiterpene hydrocarbons | 15.75 | 12.5 | ||||
| Oxygenated sesquiterpenes | 27.52 | 12.8 | ||||
| Total | 92.47 | 98.03 | ||||
| Test Sample | LC50 ± SE [µg·mL−1] | Confidence Interval [µg·mL−1] | Χ2 Test | p Level |
|---|---|---|---|---|
| C. rhamnifolioides (fresh essential oil) | 122.3 ± 3.7 | 115.1–129.6 | 0.2 | 1.00 |
| C. rhamnifolioides (stored essential oil) | 89.0 ± 1.9 | 85.2–92.8 | 0.6 | 0.90 |
| α-Phellandrene | 39.3 ± 1.0 | 37.3-41.3 | 1.4 | 0.77 |
| 1,8-Cineole | >100 | - | - | - |
3. Experimental Section
3.1. Plant Material
3.2. Extraction of Essential Oil
3.3. GC-MS Analyses
3.4. Mosquito Population
3.5. Larvicidal Bioassays
3.6. Gut Extracts from A. aegypti 4th Instar Larvae
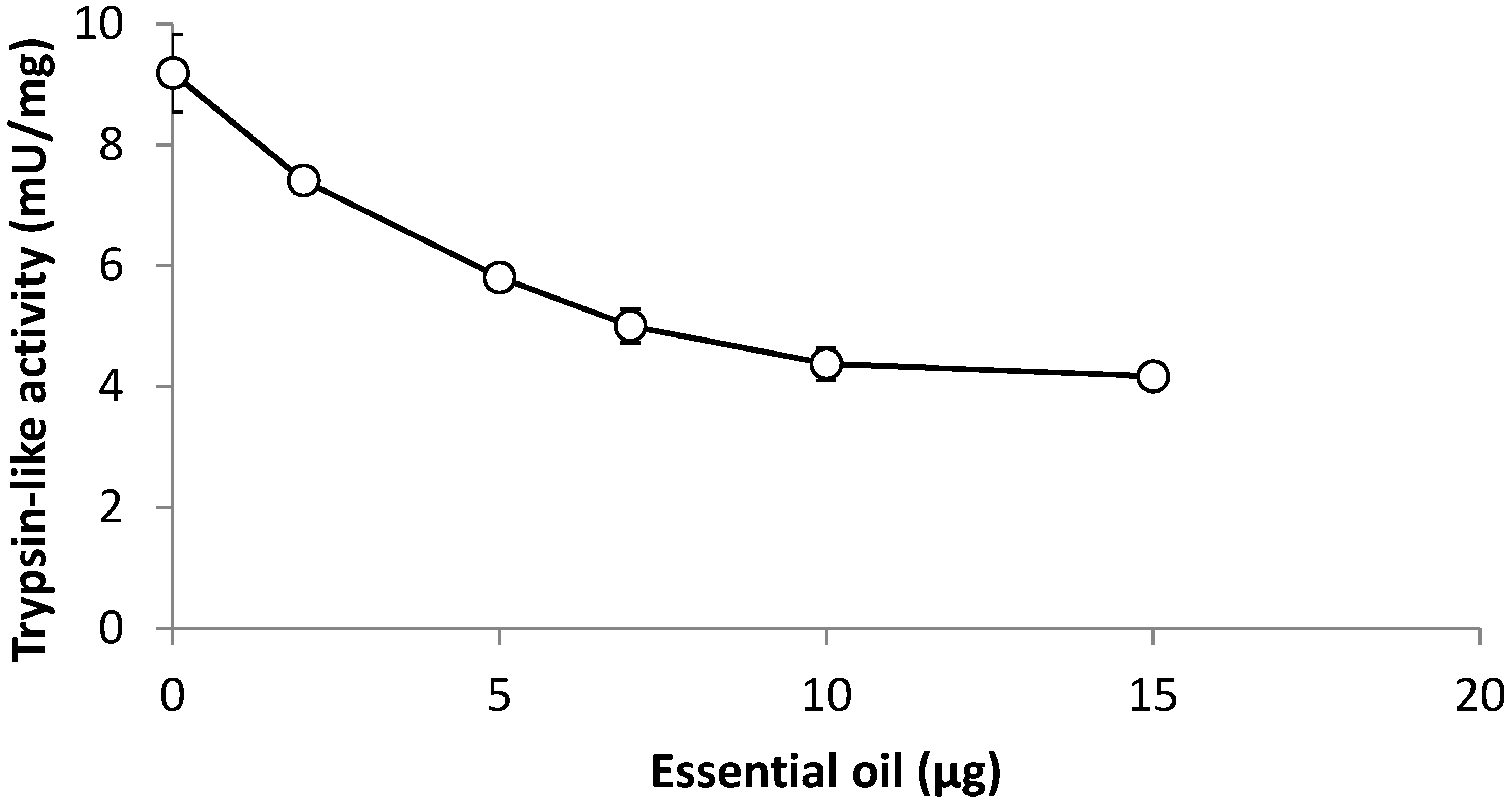
3.7. Effect of Leaf Oil on Trypsin-Like Activity from Larvae
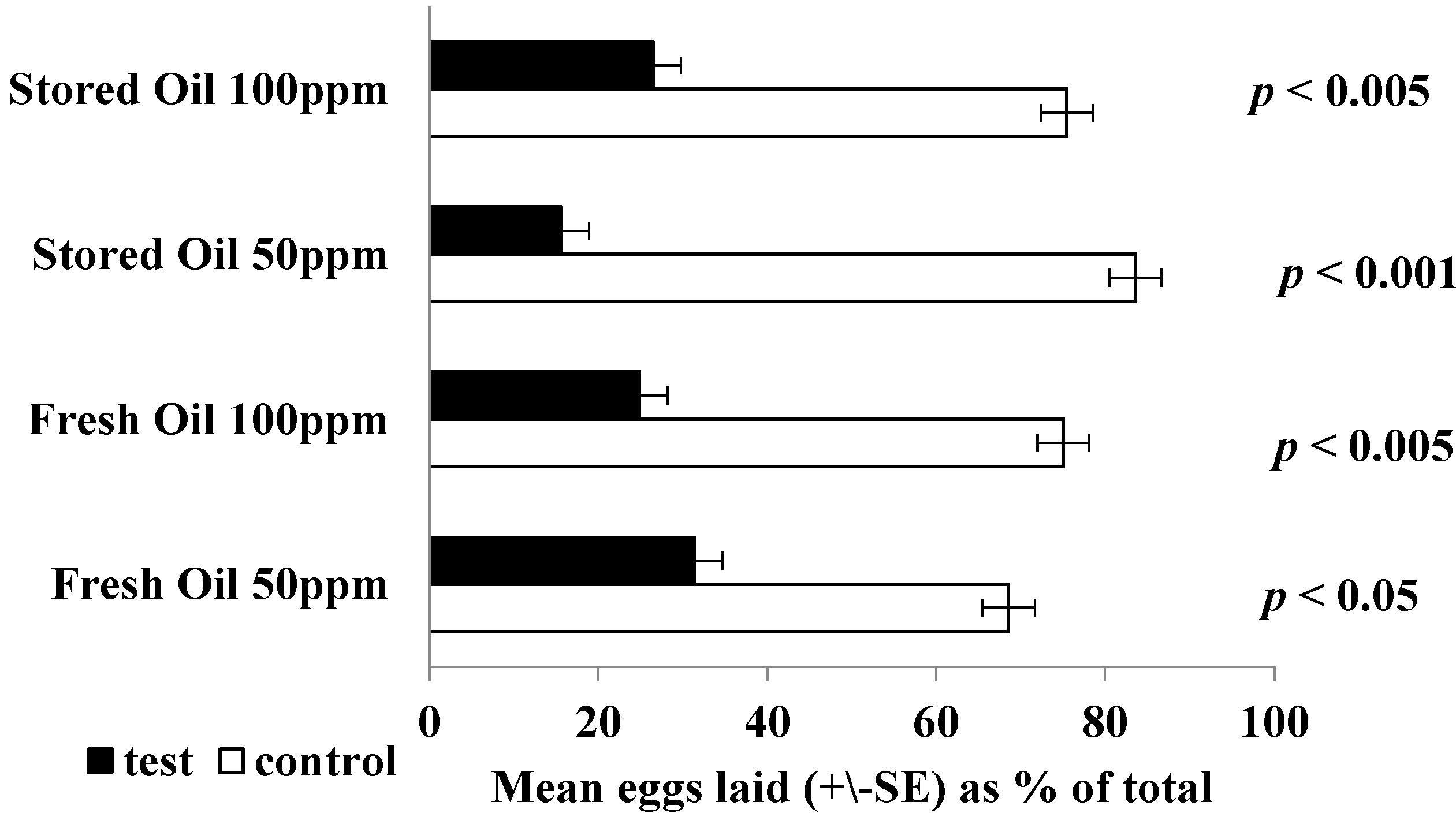
3.8. Oviposition Bioassays
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lima, L.R.; Pirani, J.R. Taxonomic revision of Croton sect. Lamprocroton (Müll. Arg.) Pax (Euphorbiaceae s.s.). Biota Neotrop. 2008, 8, 177–231. [Google Scholar]
- Salatino, A.; Salatino, M.L.F.; Negri, G. Traditional uses, chemistry and pharmacology of Croton species (Euphorbiaceae). J. Braz. Chem. Soc. 2007, 18, 11–33. [Google Scholar]
- Hiruma-Lima, C.A.; Gracioso, J.S.; Rodríguez, J.A.; Haun, M.; Nunes, D.S.; Souza Brito, A.R.M. Gastroprotective effect of essential oil from Croton cajucara Benth. (Euphorbiaceae). J. Ethnopharmacol. 2000, 69, 229–234. [Google Scholar]
- Gurgel, L.A.; Silva, R.M.; Santos, F.A.; Martins, D.T.; Mattos, P.O.; Rao, V.S. Studies on the antidiarrhoeal effect of dragon’s blood from Croton urucurana. Phytother. Res. 2001, 15, 319–322. [Google Scholar]
- Appendino, G.; Borrelli, F.; Capasso, R.; Campagnuolo, C.; Fattorusso, E.; Petrucci, F.; Taglialatela-Scafati, O. Minor diterpenoids from cascarilla (Croton eluteriaBennet) and evaluation of the cascarilla extract and cascarillin effects on gastric acid secretion. J. Agric. Food Chem. 2003, 51, 6970–6974. [Google Scholar]
- Nardi, G.M.; Felippi, R.; DalBó, S.; Siqueira-Junior, J.M.; Arruda, D.C.; Delle Monache, F.; Timbola, A.K.; Pizzolatti, M.G.; Ckless, K.; Ribeiro-do-Valle, R.M. Anti-inflammatory and antioxidant effects of Croton celtidifolius bark. Phytomedicine 2003, 10, 176–184. [Google Scholar]
- Suárez, A.I.; Compagnone, R.S.; Salazar-Bookaman, M.M.; Tillett, S.; Delle Monache, F.; di Giulio, C.; Bruges, G. Antinociceptive and anti-inflammatory effects of Croton malambo bark aqueous extract. J. Ethnopharmacol. 2003, 88, 11–14. [Google Scholar]
- Aguilar-Guadarrama, A.B.; Rios, M.Y. Three new sesquiterpenes from Croton arboreous. J. Nat. Prod. 2004, 67, 914–917. [Google Scholar]
- Cavalcanti, J.M.; Leal-Cardoso, J.H.; Diniz, L.R.L.; Portella, V.G.; Costa, C.O.; Linard, C.F.B.M.; Alves, K.; Rocha, M.V.A.P.; Lima, C.C.; Cecatto, V.M.; et al. The essential oil of Croton zehntneri and trans-anethole improves cutaneous wound healing. J. Ethnopharmacol. 2012, 144, 240–247. [Google Scholar]
- Ximenes, R.M.; Morais Nogueira, L.; Cassundé, N.M.R.; Jorge, R.J.B.; dos Santos, S.M.; Magalhães, L.P.M.; Silva, M.R.; Barros Viana, G.S.; Araújo, R.M.; de Sena, K.X.F.R.; et al. Antinociceptive and wound healing activities of Croton adamantinus Müll. Arg. essential oil. J. Nat. Med. 2013, 67, 758–764. [Google Scholar]
- Amaral, A.C.F.; Barnes, R.A. A tetrahydroprotoberberine alkaloid from Croton hemiargyreus. Phytochemistry 1998, 47, 1445–1447. [Google Scholar]
- Milanowski, D.J.; Winter, R.E.K.; Elvin-Lewis, M.P.F.; Lewis, W.H. Geographic distribution of three alkaloid chemotypes of Croton lechleri. J. Nat. Prod. 2002, 65, 814–819. [Google Scholar]
- Block, S.; Baccelli, C.; Tinant, B.; van Meervelt, L.; Rozenberg, R.; Habib Jiwan, J.-L.; Llabrès, G.; de Pauw-Gillet, M.-C.; Quetin-Leclercq, J. Diterpenes from the leaves of Croton zambesicus. Phytochemistry 2004, 65, 1165–1171. [Google Scholar]
- Aguiar, L.A.; Porto, R.S.; Lahlou, S.; Ceccatto, V.M.; Barbosa, R.; Lemos, T.L.G.; dos Santos, H.S.; Coelho-de-Souza, A.N.; Magalhães, P.J.C.; Zin, W.A.; et al. Antispasmodic effects of a new kaurene diterpene isolated from Croton argyrophylloides on rat airway smooth muscle. J. Pharm. Pharmacol. 2012, 64, 1155–1164. [Google Scholar]
- Fontenelle, R.O.S.; Morais, S.M.; Brito, E.H.S.; Brilhante, R.S.N.; Cordeiro, R.A.; Nascimento, N.R.F.; Kerntopf, M.R.; Sidrim, J.J.C.; Rocha, M.F.G. Antifungal activity of essential oils of Croton species from the Brazilian Caatinga biome. J. Appl. Microbiol. 2008, 104, 1383–1390. [Google Scholar]
- Brasil, D.S.B.; Muller, A.H.; Guilhon, G.M.S.P.; Alves, C.N.; Andrade, E.H.A.; da Silva, J.K.R.; Maia, J.G.S. Essential oil composition of Croton palanostigma Klotzsch from north Brazil. J. Braz. Chem. Soc. 2009, 20, 1188–1192. [Google Scholar]
- Santos, H.S.; Furtado, E.F.; Bertini, L.M.; Bandeira, P.N.; Albuquerque, M.R.J.R.; Menezes, J.E.S.A.; Trevisan, M.T.S.; Lemos, T.L.G. Chemical composition and cholinesterase inhibition of essential oils of three chemotypes from Croton zehntneri. Rev. Latinoam. Quim. 2010, 38, 45–51. [Google Scholar]
- De Almeida, T.S.; Rocha, J.B.T.; Rodrigues, F.F.G.; Campos, A.R.; da Costa, J.G.M. Chemical composition, antibacterial and antibiotic modulatory effect of Croton campestris essential oils. Ind. Crop. Prod. 2013, 44, 630–633. [Google Scholar]
- Secco, R.S.; Cordeiro, I.; Senna-Vale, L.; Sales, M.F.; Lima, L.R.; Medeiros, D.; sá Haiad, B.; de Oliveira, A.S.; Caruzo, M.B.R.; Carneiro-Torres, D.; et al. An overview of recent taxonomic studies on Euphorbiaceae s.l. in Brazil. Rodriguésia 2012, 63, 227–242. [Google Scholar]
- Silva, J.S.; Sales, M.F.; Gomes, A.P.S.; Carneiro-Torres, D.S. Synopsis of the species of Croton L. (Euphorbiaceae) in Pernambuco state, Brasil. Acta Bot. Bras. 2010, 24, 441–453. [Google Scholar]
- Randau, K.P. Estudo Farmacognóstico (Farmacobotânico e Farmacoquímico) e Atividade Biológica do Croton Rhamnifolius H.B.K. e Croton Rhamnifolioides Pax & Hoffm. (Euphorbiaceae). Master’s Thesis, Universidade Federal de Pernambuco, Recife, Brazil, February 2001. [Google Scholar]
- Randau, K.P.; Florêncio, D.C.; Ferreira, C.P.; Xavier, H.S. Estudo farmacognóstico de Croton rhamnifolius H.B.K. e Croton rhamnifolioides Pax & Hoffm. (Euphorbiaceae). Rev. Bras. Farmacogn. 2004, 14, 89–96. [Google Scholar]
- World Health Organization, Dengue: Guidelines for Diagnosis Treatment, Prevention and Control; WHO Press: Geneva, Switzerland, 2009; 3–87.
- Rodríguez, M.M.; Bisset, J.; Ruiz, M.; Soca, A. Cross-resistance to pyrethroid and organophosphorus insecticides induced by selection with temephos in Aedes aegypti (Diptera: Culicidae) from Cuba. J. Med. Entomol. 2002, 39, 882–888. [Google Scholar]
- García, G.P.; Flores, A.E.; Fernández-Salas, I.; Saavedra-Rodríguez, K.; Reyes-Solis, G.; Lozano-Fuentes, S.; Guillermo Bond, J.; Casas-Martínez, M.; Ramsey, J.M.; García-Rejón, J.; et al. Recent rapid rise of a permethrin knock down resistance allele in Aedes aegypti in México. PLoS Negl. Trop. Dis. 2009, 3, e531. [Google Scholar]
- Melo-Santos, M.A.V.; Varjal-Melo, J.J.M.; Araújo, A.P.; Gomes, T.C.S.; Paiva, M.H.S.; Regis, L.N.; Furtado, A.F.; Magalhaes, T.; Macoris, M.L.G.; Andrighetti, M.T.M.; et al. Resistance to the organophosphate temephos: Mechanisms, evolution and reversion in an Aedes aegypti laboratory strain from Brazil. Acta Trop. 2010, 113, 180–189. [Google Scholar]
- Tantely, M.L.; Tortosa, P.; Alout, H.; Berticat, C.; Berthomieu, A.; Rutee, A.; Dehecq, J.-S.; Makoundou, P.; Labbé, P.; Pasteur, N.; et al. Insecticide resistance in Culex pipiens quinquefasciatus and Aedes albopictus mosquitoes from La Réunion Island. Insect Biochem. Mol. Biol. 2010, 40, 317–324. [Google Scholar]
- Ocampo, C.B.; Salazar-Terreros, M.J.; Mina, N.J.; McAllister, J.; Brogdon, W. Insecticide resistance status of Aedes aegypti in 10 localities in Colombia. Acta Trop. 2011, 118, 37–44. [Google Scholar]
- Vontas, J.; Kioulos, E.; Pavlidi, N.; Morou, E.; della Torre, A.; Ranson, H. Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic. Biochem. Physiol. 2012, 104, 126–131. [Google Scholar]
- Carvalho, A.F.U.; Melo, V.M.M.; Craveiro, A.A.; Machado, M.I.L.; Bantim, M.B.; Rabelo, E.F. Larvicidal activity of the essential oil from Lippia sidoides Cham. against Aedes aegypti Linn. Mem. Inst. Oswaldo Cruz 2003, 98, 569–571. [Google Scholar]
- Cavalcanti, E.S.B.; de Morais, S.M.; Lima, M.A.A.; Santana, E.W.P. Larvicidal activity of essential oils from Brazilian plants against Aedes aegypti L. Mem. Inst. Oswaldo Cruz 2004, 99, 541–544. [Google Scholar]
- Santos, R.P.; Nunes, E.P.; Nascimento, R.F.; Santiago, G.M.P.; Menezes, G.H.A.; Silveira, E.R.; Pessoa, O.D.L. Chemical composition and larvicidal activity of the essential oils of Cordia leucomalloides and Cordia curassavica from the northeast of Brazil. J. Braz. Chem. Soc. 2006, 17, 1027–1030. [Google Scholar]
- Kovendan, K.; Murugan, K.; Mahesh Kumar, P.; Thiyagarajan, P.; William, S.J. Ovicidal, repellent, adulticidal and field evaluations of plant extract against dengue, malaria and filarial vectors. Parasitol. Res. 2013, 112, 1205–1219. [Google Scholar]
- Wang, Z.; Kim, J.-R.; Wang, M.; Shu, S.; Ahn, Y.-J. Larvicidal activity of Cnidium monnieri fruit coumarins and structurally related compounds against insecticide-susceptible and insecticide-resistant Culex pipiens pallens and Aedes aegypti. Pest Manag. Sci. 2012, 68, 1041–1047. [Google Scholar]
- Senthilkumar, A.; Jayaraman, M.; Venkatesalu, V. Chemical constituents and larvicidal potential of Feronia limonia leaf essential oil against Anopheles stephensi, Aedesaegypti and Culex quinquefasciatus. Parasitol. Res. 2013, 112, 1337–1342. [Google Scholar]
- Navarro, D.M.A.F.; da Silva, P.C.B.; da Silva, M.F.R.; Napoleao, T.H.; Paiva, P.M.G. Larvicidal activity of plant and algae extracts, essential oils and isolated chemical constituents against Aedes aegypti. Nat. Prod. J. 2014, 3, 268–291. [Google Scholar]
- Dória, G.A.A.; Silva, W.J.; Carvalho, G.A.; Alves, P.B.; Cavalcanti, S.C.H. A study of the larvicidal activity of two Croton species from northeastern Brazil against Aedes aegypti. Pharm. Biol. 2010, 48, 615–620. [Google Scholar]
- Camurça-Vasconcelos, A.L.F.; Bevilaqua, C.M.L.; Morais, S.M.; Maciel, M.V.; Costa, C.T.C.; Macedo, I.T.F.; Oliveira, L.M.B.; Braga, R.R.; Silva, R.A.; Vieira, L.S. Anthelmintic activity of Croton zehntneri and Lippia sidoides essential oils. Vet. Parasitol. 2007, 148, 288–294. [Google Scholar]
- De Lima, G.P.G.; de Souza, T.M.; de Paula Freire, G.; Farias, D.F.; Cunha, A.P.; Ricardo, N.M.P.S.; de Morais, S.M.; Carvalho, A.F.U. Further insecticidal activities of essential oils from Lippia sidoides and Croton species against Aedes aegypti L. Parasitol. Res. 2013, 112, 1953–1958. [Google Scholar]
- Morais, S.M.; Cavalcanti, E.S.B.; Bertini, L.M.; Oliveira, C.L.L.; Rodrigues, J.R.B.; Cardoso, J.H.L. Larvicidal activity of essential oils from Brazilian Croton species against Aedes aegypti L. J. Am. Mosq. Control Assoc. 2006, 22, 161–164. [Google Scholar]
- Torres, M.C.M.; Assunção, J.C.; Santiago, G.M.P.; Andrade-Neto, M.; Silveira, E.R.; Costa-Lotufo, L.V.; Bezerra, D.P.; Marinho Filho, J.D.B.; Viana, F.A.; Pessoa, O.D.L. Larvicidal and nematicidal activities of the leaf essential oil of Croton regelianus. Chem. Biodivers. 2008, 5, 2724–2728. [Google Scholar]
- Misharina, T.A.; Polshkov, A.N.; Ruchkina, E.L.; Medvedeva, I.B. Changes in the composition of the essential oil of marjoram during storage. Appl. Biochem. Microbiol. 2003, 39, 311–316. [Google Scholar]
- El-Nikeety, M.M.A.; el-Akel, A.T.M.; el-Hady, M.M.I.A.; Badei, A.Z.M. Changes in physical properties and chemical constituents of parsley herb volatile oil during storage. Egypt. J. Food Sci. 2000, 26, 35–49. [Google Scholar]
- Turek, C.; Stintzing, F.C. Impact of different storage conditions on the quality of selected essential oils. Food Res. Int. 2012, 46, 341–353. [Google Scholar]
- Wise, M.L.; Urbansky, M.; Helms, G.L.; Coates, R.M.; Croteau, R. Syn stereochemistry of cyclic ether formation in 1,8-cineole biosynthesis catalyzed by recombinant synthase from Salvia officinalis. J. Am. Chem. Soc. 2002, 124, 8546–8547. [Google Scholar]
- Weyerstahl, P.; Krohn, K. Structure—Odor correlation—IX from 1,8-cineole to sesquicineol—Change of odor with structure. Tetrahedron 1990, 46, 3503–3514. [Google Scholar]
- Santos, G.K.N.; Dutra, K.A.; Barros, R.A.; da Câmara, C.A.G.; Lira, D.D.; Gusmão, N.B.; Navarro, D.M.A.F. Essential oils from Alpinia purpurata (Zingiberaceae): Chemical composition, oviposition deterrence, larvicidal and antibacterial activity. Ind. Crop. Prod. 2012, 40, 254–260. [Google Scholar]
- Perumalsamy, H.; Kim, N.-J.; Ahn, Y.-J. Larvicidal activity of compounds isolated from Asarum heterotropoides against Culex pipiens pallens, Aedes aegyti and Ochlerotatus togoi (Diptera: Culicidade). J. Med. Entomol. 2009, 46, 1420–1423. [Google Scholar]
- Cheng, S.-S.; Huang, C.-G.; Chen, Y.-J.; Yu, J.-J.; Chen, W.-J.; Chang, S.-T. Chemical compositions and larvicidal activities of leaf essential oils from two Eucaliptus species. Bioresour. Technol. 2009, 100, 452–456. [Google Scholar]
- Pontual, E.V.; Napoleão, T.H.; Dias de Assis, C.R.; de Souza Bezerra, R.; Xavier, H.S.; Navarro, D.M.A.F.; Coelho, L.C.B.B.; Paiva, P.M.G. Effect of Moringa oleifera flower extract on larval trypsin and acethylcholinesterase activities in Aedes aegypti. Arch. Insect Biochem. Physiol. 2012, 79, 135–152. [Google Scholar]
- Napoleão, T.H.; Pontual, E.V.; de Albuquerque Lima, T.; de Lima Santos, N.D.; Sá, R.A.; Coelho, L.C.B.B.; Navarro, D.M.A.F.; Paiva, P.M.G. Effect of Myracrodruon urundeuva leaf lectin on survival and digestive enzymes of Aedes aegypti larvae. Parasitol. Res. 2012, 110, 609–616. [Google Scholar]
- Blenau, W.; Rademacher, E.; Baumann, A. Plant essential oils and formamidines as insecticides/acaricides: What are the molecular targets? Apidologie 2012, 43, 334–347. [Google Scholar]
- Kostyukovsky, M.; Rafaeli, A.; Demchenko, N.; Shaaya, E. Activation of actopaminergic receptors by essential oil isolated from aromatic plants: Possible mode of action against insect pests. Pest Manag. Sci. 2002, 58, 1101–1106. [Google Scholar]
- Enam, E.E. Molecular and pharmacological analysis of an octopamine receptor from American cockroach and fruitfly in response to plant essential oils. Arch. Insect Biochem. Physiol. 2005, 59, 161–171. [Google Scholar]
- Khanikor, B.; Parida, P.; Yadav, R.N.S.; Bora, D. Comparative mode of action of some terpene compounds against octopamine receptor and acetyl cholinesterase of mosquito and human system by the help of homology modeling and Docking studies. J. Appl. Pharm. Sci. 2013, 3, 6–12. [Google Scholar]
- Anderson, J.A.; Coats, J.R. Acetylcholinesterase inhibition by nootkatone and carvacrol in arthropods. Pestic. Biochem. Physiol. 2012, 102, 124–128. [Google Scholar]
- Bentley, M.D.; Day, J.F. Chemical ecology and behavioral aspects of mosquito oviposition. Annu. Rev. Entomol. 1989, 34, 401–421. [Google Scholar]
- Autran, E.S.; Neves, I.A.; da Silva, C.S.B.; Santos, G.K.N.; da Câmara, C.A.G.; Navarro, D.M.A.F. Chemical composition, oviposition deterrent and larvicidal activities against Aedes aegypti of essential oils from Piper marginatum Jacq. (Piperaceae). Bioresour. Technol. 2009, 100, 2284–2288. [Google Scholar]
- Siriporn, P.; Mayura, S. The effects of herbal essential oils on the oviposition-deterrent and ovicidal activities of Aedes aegypti (Linn.), Anopheles dirus (Peyton and Harrison) and Culex quinquefasciatus (Say). Trop. Biomed. 2012, 29, 138–150. [Google Scholar]
- Van den Dool, H.; Dec. Kratz, P. A generalization of the retention index system including linear programmed gas–liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Compounds by Gas Chromatography and Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Navarro, D.M.A.F.; de Oliveira, P.E.S.; Potting, R.P.J.; Brito, A.C.; Fital, S.J.F.; Sant’Ana, A.E.G. The potential attractant or repellent effects of different water types on oviposition in Aedes aegypti L. (Dipt., Culicidae). J. Appl. Entomol. 2003, 127, 46–50. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Sample Availability: Samples of the compounds α-Phellandrene and 1,8-Cineole are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, G.K.N.; Dutra, K.A.; Lira, C.S.; Lima, B.N.; Napoleão, T.H.; Paiva, P.M.G.; Maranhão, C.A.; Brandão, S.S.F.; Navarro, D.M.A.F. Effects of Croton rhamnifolioides Essential Oil on Aedes aegypti Oviposition, Larval Toxicity and Trypsin Activity. Molecules 2014, 19, 16573-16587. https://doi.org/10.3390/molecules191016573
Santos GKN, Dutra KA, Lira CS, Lima BN, Napoleão TH, Paiva PMG, Maranhão CA, Brandão SSF, Navarro DMAF. Effects of Croton rhamnifolioides Essential Oil on Aedes aegypti Oviposition, Larval Toxicity and Trypsin Activity. Molecules. 2014; 19(10):16573-16587. https://doi.org/10.3390/molecules191016573
Chicago/Turabian StyleSantos, Geanne K. N., Kamilla A. Dutra, Camila S. Lira, Bheatriz N. Lima, Thiago H. Napoleão, Patrícia M. G. Paiva, Claudia A. Maranhão, Sofia S. F. Brandão, and Daniela M. A. F. Navarro. 2014. "Effects of Croton rhamnifolioides Essential Oil on Aedes aegypti Oviposition, Larval Toxicity and Trypsin Activity" Molecules 19, no. 10: 16573-16587. https://doi.org/10.3390/molecules191016573





