Diastereoselective Three-Component Reactions of Chiral Nickel(II) Glycinate for Convenient Synthesis of Novel α-Amino-β-Substituted-γ,γ-Disubstituted Butyric Acids
Abstract
:1. Introduction

2. Results and Discussion
| Entry | Base | Solvent | Yield (%) b | de c |
|---|---|---|---|---|
| 1 | DBU d | CH3CN | 94 | 88% |
| 2 | TEA | CH3CN | 97 | 94% |
| 3 | DIEA | CH3CN | 94 | 96% |
| 4 | NMM | CH3CN | 63 | 92% |
| 5 | Piperidine | CH3CN | 89 | 96% |
| 6 | DIEA | DMF | 99 | 88% |
| 7 | DIEA | EA | 98 | 80% |
| 8 | DIEA | MeOH | 99 | 88% |
| 9 | DIEA | DCM | 97 | 58% |
| 10 | DIEA | dioxane | 91 | 97% |
| 11 | DIEA | CHCl3 | 59 | 90% |
| 12 | DIEA | DMSO | 91 | 76% |
| Entry | Product | R | EWG | Yield (%) b | de c |
|---|---|---|---|---|---|
| 1 | (S,2S,3R)-7a | Ph | CN | 91 | 97% |
| 2 | (S,2S,3R)-7b | 4-(t-Bu)-C6H4 | CN | 86 | >99% |
| 3 | (S,2S,3R)-7c | 3-Cl-C6H4 | CN | 52 | 90% |
| 4 | (S,2S,3R)-7d | 4-F-C6H4 | CN | 83 | 93% |
| 5 | (S,2S,3R)-7e | 4-Br-C6H4 | CN | 44 | 98% |
| 6 | (S,2S,3R)-7f | 3,4-di-Cl-C6H3 | CN | 84 | 97% |
| 7 | (S,2S,3R)-7g | 3-Br-C6H4 | CN | 38 | 95% |
| 8 | (S,2S,3R)-7h | 3-OMe-C6H4 | CN | 82 | >99% |
| 9 | (S,2S,3R)-7i | 2-naphthyl | CN | 80 | 98% |
| 10 | (S,2S,3R)-7j | 1-naphthyl | CN | 26 | 98% |
| 11 | (S,2S,3R)-7k | 3-OH-C6H4 | CN | 46 | 98% |
| 12 | (S,2S,3R)-7l | 2-F-4-Br-C6H3 | CN | 90 | >99% |
| 13 | (S,2S,3R,4S)-7m | Ph | COOEt | 78 | 98% |
| 14 | (S,2S,3R,4S)-7n | 2-Br-C6H4 | COOEt | 88 | >99% |
| 15 | (S,2S,3R,4S)-7o | 3-Cl-C6H4 | COOEt | 89 | 97% |
| 16 | (S,2S,3R,4S)-7p | 4-F-C6H4 | COOEt | 75 | 96% |
| 17 | (S,2S,3R,4S)-7q | 3,4-di-Cl-C6H3 | COOEt | 82 | 98% |
| 18 | (S,2S,3R,4S)-7r | 2,4-di-Cl-C6H3 | COOEt | 96 | 98% |
| 19 | (S,2S,3R,4S)-7s | 4-CH3-C6H4 | COOEt | 76 | >99% |
| 20 | (S,2S,3R,4S)-7t | 4-OCH3-C6H4 | COOEt | 77 | 97% |
| 21 | (S,2S,3R,4S)-7u | 4-NO2-C6H4 | COOEt | 69 | 97% |
| 22 | (S,2S,3R,4S)-7v | 4-(t-Bu)-C6H4 | COOEt | 51 | 97% |
| 23 | (S,2S,3R,4S)-7w | 1-naphthyl | COOEt | 67 | >99% |
| 24 | (S,2S,3R)-7x | 3-Br-thienyl | CN | NR d | NR d |
| 25 | (S,2S,3R)-7y | 4-Me-Furyl | CN | NR d | NR d |
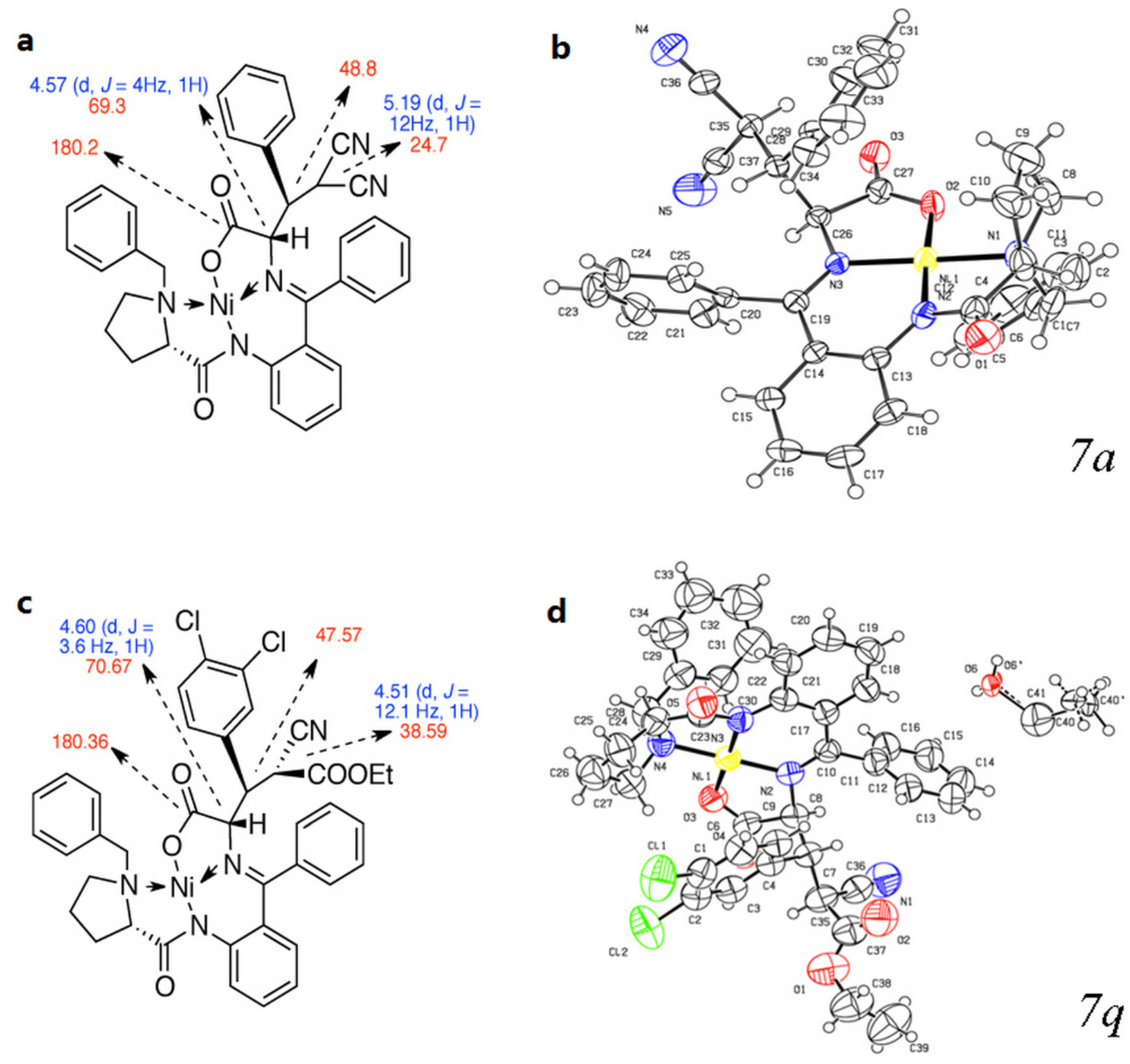

3. Experimental
3.1. General
3.2. Typical Procedure for the Synthesis of (S)-Nickel(II) Complex (5) [18]
3.3. General Procedure for the Synthesis of Ni(II) (7)
3.3.1. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-phenylbutyric Acid Schiff Base Complex (7a)
 = +1602 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8 Hz, 1H), 7.98 (d, J = 8 Hz, 2H), 7.68–7.63 (m, 6H), 7.39 (d, J = 8 Hz, 1H), 7.30–7.26 (m, 4H), 7.18–7.14 (m, 3H), 6.70 (d, J = 4 Hz, 1H), 5.19 (d, J = 12 Hz, 1H), 4.57 (d, J = 4 Hz, 1H), 4.15 (d, J = 12 Hz, 1H), 3.42 (d, J = 12 Hz, 1H), 3.29–3.21 (m, 2H), 2.97–2.91 (m, 1H), 2.32–2.20 (m, 1H), 2.06–2.02 (m, 1H), 1.95–1.91 (m, 1H), 1.85–1.83 (m, 1H), 1.56–1.50 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.2, 173.5, 143.4, 133.9, 133.5, 133.2, 132.6, 131.4, 130.9, 130.6, 130.1, 129.9, 129.5, 128.9, 128.9, 127.5, 127.0, 125.5, 123.3, 120.8, 111.5, 111.3, 70.5, 69.7, 63.9, 57.4, 48.8, 29.7, 24.7, 23.1. ESI-MS (m/z): calcd. 652.2, found 652.2 ([M+H]+); HRMS (m/z): calcd. C37H32N5NiO3 for 652.1859, found 652.1857 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.3 min, tminor = 13.4 min, de = 97%.
= +1602 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8 Hz, 1H), 7.98 (d, J = 8 Hz, 2H), 7.68–7.63 (m, 6H), 7.39 (d, J = 8 Hz, 1H), 7.30–7.26 (m, 4H), 7.18–7.14 (m, 3H), 6.70 (d, J = 4 Hz, 1H), 5.19 (d, J = 12 Hz, 1H), 4.57 (d, J = 4 Hz, 1H), 4.15 (d, J = 12 Hz, 1H), 3.42 (d, J = 12 Hz, 1H), 3.29–3.21 (m, 2H), 2.97–2.91 (m, 1H), 2.32–2.20 (m, 1H), 2.06–2.02 (m, 1H), 1.95–1.91 (m, 1H), 1.85–1.83 (m, 1H), 1.56–1.50 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.2, 173.5, 143.4, 133.9, 133.5, 133.2, 132.6, 131.4, 130.9, 130.6, 130.1, 129.9, 129.5, 128.9, 128.9, 127.5, 127.0, 125.5, 123.3, 120.8, 111.5, 111.3, 70.5, 69.7, 63.9, 57.4, 48.8, 29.7, 24.7, 23.1. ESI-MS (m/z): calcd. 652.2, found 652.2 ([M+H]+); HRMS (m/z): calcd. C37H32N5NiO3 for 652.1859, found 652.1857 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.3 min, tminor = 13.4 min, de = 97%.3.3.2. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(4-tert-butyl)-phenylbutyric Acid Schiff Base Complex (7b)
 = +1602 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.29 (d, J = 8.7 Hz, 1H), 7.95 (d, J = 7.5 Hz, 2H), 7.68 (dt, J = 13.0, 6.8 Hz, 3H), 7.60 (d, J = 8.1 Hz, 2H), 7.41 (d, J = 7.2 Hz, 1H), 7.29 (dd, J = 14.5, 7.1 Hz, 3H), 7.19 (d, J = 7.9 Hz, 3H), 7.16–7.09 (m, 2H), 6.70 (d, J = 2.9 Hz, 2H), 5.16 (d, J = 12.0 Hz, 1H), 4.58 (d, J = 3.4 Hz, 1H), 4.16 (d, J = 12.7 Hz, 1H), 3.79 (s, 1H), 3.49 (d, J = 12.7 Hz, 1H), 3.28–3.13 (m, 2H), 3.05–2.90 (m, 1H), 2.29–2.13 (m, 1H), 2.12–2.00 (m, 1H), 1.92 (dt, J = 27.0, 8.6 Hz, 1H), 1.78 (dt, J = 18.9, 9.3 Hz, 1H), 1.54 (s, 1H), 1.46–1.31 (m, 9H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 708.2, found 708.3 ([M+H]+); HRMS (m/z): calcd. C41H40N5NiO3 for 708.2485, found 708.2482 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 30.9 min, tminor = 17.3 min, de > 99%.
= +1602 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.29 (d, J = 8.7 Hz, 1H), 7.95 (d, J = 7.5 Hz, 2H), 7.68 (dt, J = 13.0, 6.8 Hz, 3H), 7.60 (d, J = 8.1 Hz, 2H), 7.41 (d, J = 7.2 Hz, 1H), 7.29 (dd, J = 14.5, 7.1 Hz, 3H), 7.19 (d, J = 7.9 Hz, 3H), 7.16–7.09 (m, 2H), 6.70 (d, J = 2.9 Hz, 2H), 5.16 (d, J = 12.0 Hz, 1H), 4.58 (d, J = 3.4 Hz, 1H), 4.16 (d, J = 12.7 Hz, 1H), 3.79 (s, 1H), 3.49 (d, J = 12.7 Hz, 1H), 3.28–3.13 (m, 2H), 3.05–2.90 (m, 1H), 2.29–2.13 (m, 1H), 2.12–2.00 (m, 1H), 1.92 (dt, J = 27.0, 8.6 Hz, 1H), 1.78 (dt, J = 18.9, 9.3 Hz, 1H), 1.54 (s, 1H), 1.46–1.31 (m, 9H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 708.2, found 708.3 ([M+H]+); HRMS (m/z): calcd. C41H40N5NiO3 for 708.2485, found 708.2482 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 30.9 min, tminor = 17.3 min, de > 99%.3.3.3. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(3-chlorophenyl) Butyric Acid Schiff Base Complex (7c)
 = +1622 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.5 Hz, 2H), 7.75–7.60 (m, 4H), 7.55 (d, J = 15.7 Hz, 1H), 7.42–7.34 (m, 2H), 7.32 (dd, J = 14.6, 6.9 Hz, 3H), 7.22–7.09 (m, 4H), 6.70 (d, J = 3.7 Hz, 2H), 5.14 (d, J = 11.9 Hz, 1H), 4.56 (d, J = 3.8 Hz, 1H), 4.15 (d, J = 12.7 Hz, 1H), 3.43 (d, J = 12.6 Hz, 1H), 3.25 (ddd, J = 16.2, 10.7, 5.6 Hz, 2H), 3.02 (dd, J = 9.8, 5.5 Hz, 1H), 2.25 (dd, J = 18.6, 10.0 Hz, 1H), 2.10–1.98 (m, 3H), 1.70–1.63 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 175.9, 173.7, 143.4, 136.3, 134.6, 133.9, 133.4, 133.4, 133.2, 131.3, 131.0, 131.0, 130.6, 130.4, 129.5, 129.0, 128.8, 127.4, 127.0, 125.4, 123.3, 120.8, 111.2, 110.9, 70.5, 69.4, 64.0, 57.6, 48.4, 30.7, 29.6, 24.6, 23.0. ESI-MS (m/z): calcd. 686.1, found 686.2 ([M+H]+). HRMS (m/z): calcd. C37H31ClN5NiO3 for 686.1469, found 686.1475 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.0 min, tminor = 13.0 min, de = 90%.
= +1622 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.5 Hz, 2H), 7.75–7.60 (m, 4H), 7.55 (d, J = 15.7 Hz, 1H), 7.42–7.34 (m, 2H), 7.32 (dd, J = 14.6, 6.9 Hz, 3H), 7.22–7.09 (m, 4H), 6.70 (d, J = 3.7 Hz, 2H), 5.14 (d, J = 11.9 Hz, 1H), 4.56 (d, J = 3.8 Hz, 1H), 4.15 (d, J = 12.7 Hz, 1H), 3.43 (d, J = 12.6 Hz, 1H), 3.25 (ddd, J = 16.2, 10.7, 5.6 Hz, 2H), 3.02 (dd, J = 9.8, 5.5 Hz, 1H), 2.25 (dd, J = 18.6, 10.0 Hz, 1H), 2.10–1.98 (m, 3H), 1.70–1.63 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 175.9, 173.7, 143.4, 136.3, 134.6, 133.9, 133.4, 133.4, 133.2, 131.3, 131.0, 131.0, 130.6, 130.4, 129.5, 129.0, 128.8, 127.4, 127.0, 125.4, 123.3, 120.8, 111.2, 110.9, 70.5, 69.4, 64.0, 57.6, 48.4, 30.7, 29.6, 24.6, 23.0. ESI-MS (m/z): calcd. 686.1, found 686.2 ([M+H]+). HRMS (m/z): calcd. C37H31ClN5NiO3 for 686.1469, found 686.1475 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.0 min, tminor = 13.0 min, de = 90%.3.3.4. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(4-fluorophenyl)butyric Acid Schiff Base Complex (7d)
 = +1734 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.6 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.73–7.62 (m, 3H), 7.34 (ddd, J = 14.8, 13.7, 7.2 Hz, 7H), 7.21–7.09 (m, 3H), 6.73–6.66 (m, 2H), 5.16 (d, J = 12.0 Hz, 1H), 4.55 (d, J = 3.7 Hz, 1H), 4.15 (d, J = 13.1 Hz, 1H), 3.48–3.37 (m, 1H), 3.32–3.22 (m, 2H), 3.02–2.90 (m, 1H), 2.33–2.23 (m, 1H), 2.13–2.05 (m, 1H), 1.97 (dd, J = 13.3, 7.4 Hz, 2H), 1.67–1.61 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.1, 173.6, 165.1, 162.6, 143.3, 133.9, 133.4, 133.3, 133.2, 131.3, 131.2, 130.9, 130.6, 129.5, 129.0, 128.8, 128.4, 128.4, 127.3, 127.0, 125.4, 123.3, 120.8, 117.1, 116.9, 111.3, 111.0, 70.5, 69.6, 64.0, 57.5, 48.1, 30.6, 24.7, 22.9. ESI-MS (m/z): calcd. 670.2, found 670.2 ([M+H]+). HRMS (m/z): calcd. C37H31FN5NiO3 for 670.1764, found 670.1801 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.0 min, tminor = 10.1 min, de = 93%.
= +1734 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.6 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.73–7.62 (m, 3H), 7.34 (ddd, J = 14.8, 13.7, 7.2 Hz, 7H), 7.21–7.09 (m, 3H), 6.73–6.66 (m, 2H), 5.16 (d, J = 12.0 Hz, 1H), 4.55 (d, J = 3.7 Hz, 1H), 4.15 (d, J = 13.1 Hz, 1H), 3.48–3.37 (m, 1H), 3.32–3.22 (m, 2H), 3.02–2.90 (m, 1H), 2.33–2.23 (m, 1H), 2.13–2.05 (m, 1H), 1.97 (dd, J = 13.3, 7.4 Hz, 2H), 1.67–1.61 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.1, 173.6, 165.1, 162.6, 143.3, 133.9, 133.4, 133.3, 133.2, 131.3, 131.2, 130.9, 130.6, 129.5, 129.0, 128.8, 128.4, 128.4, 127.3, 127.0, 125.4, 123.3, 120.8, 117.1, 116.9, 111.3, 111.0, 70.5, 69.6, 64.0, 57.5, 48.1, 30.6, 24.7, 22.9. ESI-MS (m/z): calcd. 670.2, found 670.2 ([M+H]+). HRMS (m/z): calcd. C37H31FN5NiO3 for 670.1764, found 670.1801 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.0 min, tminor = 10.1 min, de = 93%.3.3.5. Ni(II)-(S)-BPB/(2S,3R)-2-amino-4,4'-dicyano-3-(4-bromophenyl) Butyric Acid Schiff Base Complex (7e)
 = +1972 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.23 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.4 Hz, 2H), 7.78–7.58 (m, 3H), 7.38 (d, J = 6.8 Hz, 1H), 7.29 (dd, J = 14.9, 7.4 Hz, 3H), 7.25–7.08 (m, 7H), 6.70 (d, J = 13.7 Hz, 2H), 5.14 (d, J = 12.0 Hz, 1H), 4.53 (d, J = 3.2 Hz, 1H), 4.15 (d, J = 12.7 Hz, 1H), 3.40 (d, J = 12.6 Hz, 1H), 3.31–3.18 (m, 2H), 2.99 (dd, J = 10.5, 6.0 Hz, 1H), 2.23 (dt, J = 24.1, 12.1 Hz, 1H), 2.13–2.02 (m, 1H), 2.02–1.87 (m, 2H), 1.55 (dd, J = 12.1, 6.5 Hz, 1H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 730.1, found 730.2 ([M+H]+). HRMS (m/z): calcd. C37H31BrN5NiO3 for 730.0966, found 730.0978 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.0 min, tminor = 15.3 min, de = 98%.
= +1972 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.23 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.4 Hz, 2H), 7.78–7.58 (m, 3H), 7.38 (d, J = 6.8 Hz, 1H), 7.29 (dd, J = 14.9, 7.4 Hz, 3H), 7.25–7.08 (m, 7H), 6.70 (d, J = 13.7 Hz, 2H), 5.14 (d, J = 12.0 Hz, 1H), 4.53 (d, J = 3.2 Hz, 1H), 4.15 (d, J = 12.7 Hz, 1H), 3.40 (d, J = 12.6 Hz, 1H), 3.31–3.18 (m, 2H), 2.99 (dd, J = 10.5, 6.0 Hz, 1H), 2.23 (dt, J = 24.1, 12.1 Hz, 1H), 2.13–2.02 (m, 1H), 2.02–1.87 (m, 2H), 1.55 (dd, J = 12.1, 6.5 Hz, 1H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 730.1, found 730.2 ([M+H]+). HRMS (m/z): calcd. C37H31BrN5NiO3 for 730.0966, found 730.0978 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.0 min, tminor = 15.3 min, de = 98%.3.3.6. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(3,4-dichlorophenyl) Butyric Acid Schiff Base Complex (7f)
 = +1660 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.4 Hz, 2H), 7.69 (dd, J = 17.4, 7.8 Hz, 4H), 7.44 (s, 1H), 7.38 (d, J = 6.3 Hz, 1H), 7.31 (t, J = 7.4 Hz, 2H), 7.18 (dd, J = 15.9, 8.3 Hz, 2H), 7.09 (t, J = 7.8 Hz, 2H), 6.71 (d, J = 14.3 Hz, 2H), 5.15 (d, J = 11.9 Hz, 1H), 4.53 (s, 1H), 4.12 (dd, J = 17.0, 9.9 Hz, 1H), 3.42 (d, J = 12.6 Hz, 1H), 3.30 (t, J = 8.3 Hz, 1H), 3.22 (d, J = 11.9 Hz, 1H), 2.99 (d, J = 5.8 Hz, 1H), 2.33 (dd, J = 20.4, 8.8 Hz, 1H), 2.02 (qd, J = 13.8, 6.8 Hz, 3H), 1.77–1.66 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 175.8, 173.8, 143.4, 135.0, 134.6, 134.0, 133.5, 133.3, 132.7, 131.7, 131.3, 131.1, 130.7, 129.6, 129.0, 128.9, 127.2, 126.9, 125.2, 123.3, 120.8, 111.1, 110.6, 70.5, 69.3, 64.1, 57.8, 48.0, 30.7, 24.4, 22.8. ESI-MS (m/z): calcd. 720.1, found 720.2 ([M+H]+); HRMS (m/z): calcd. C37H30Cl2N5NiO3 for 720.1079, found 720.1090 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.0 min, tminor = 12.5 min, de = 97%.
= +1660 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.4 Hz, 2H), 7.69 (dd, J = 17.4, 7.8 Hz, 4H), 7.44 (s, 1H), 7.38 (d, J = 6.3 Hz, 1H), 7.31 (t, J = 7.4 Hz, 2H), 7.18 (dd, J = 15.9, 8.3 Hz, 2H), 7.09 (t, J = 7.8 Hz, 2H), 6.71 (d, J = 14.3 Hz, 2H), 5.15 (d, J = 11.9 Hz, 1H), 4.53 (s, 1H), 4.12 (dd, J = 17.0, 9.9 Hz, 1H), 3.42 (d, J = 12.6 Hz, 1H), 3.30 (t, J = 8.3 Hz, 1H), 3.22 (d, J = 11.9 Hz, 1H), 2.99 (d, J = 5.8 Hz, 1H), 2.33 (dd, J = 20.4, 8.8 Hz, 1H), 2.02 (qd, J = 13.8, 6.8 Hz, 3H), 1.77–1.66 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 175.8, 173.8, 143.4, 135.0, 134.6, 134.0, 133.5, 133.3, 132.7, 131.7, 131.3, 131.1, 130.7, 129.6, 129.0, 128.9, 127.2, 126.9, 125.2, 123.3, 120.8, 111.1, 110.6, 70.5, 69.3, 64.1, 57.8, 48.0, 30.7, 24.4, 22.8. ESI-MS (m/z): calcd. 720.1, found 720.2 ([M+H]+); HRMS (m/z): calcd. C37H30Cl2N5NiO3 for 720.1079, found 720.1090 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.0 min, tminor = 12.5 min, de = 97%.3.3.7. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(3-bromophenyl) Butyric Acid Schiff Base Complex (7g)
 = +1882 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.28 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.80 (d, J = 8.0 Hz, 1H), 7.75–7.60 (m, 3H), 7.49 (t, J = 7.8 Hz, 2H), 7.39 (d, J = 6.7 Hz, 1H), 7.31 (t, J = 7.6 Hz, 2H), 7.24–7.08 (m, 4H), 6.70 (d, J = 4.0 Hz, 2H), 5.14 (d, J = 11.9 Hz, 1H), 4.55 (d, J = 3.7 Hz, 1H), 4.20–4.09 (m, 1H), 3.42 (d, J = 12.6 Hz, 1H), 3.25 (ddd, J = 15.7, 10.7, 5.6 Hz, 2H), 3.03 (dd, J = 10.0, 5.6 Hz, 1H), 2.25 (dt, J = 16.4, 8.4 Hz, 1H), 2.15–1.91 (m, 4H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 730.1, found 730.2 ([M+H]+); HRMS (m/z): calcd. C37H31BrN5NiO3 for 730.0966, found 730.0966 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.0, tminor = 14.0 min, de = 95%.
= +1882 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.28 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.80 (d, J = 8.0 Hz, 1H), 7.75–7.60 (m, 3H), 7.49 (t, J = 7.8 Hz, 2H), 7.39 (d, J = 6.7 Hz, 1H), 7.31 (t, J = 7.6 Hz, 2H), 7.24–7.08 (m, 4H), 6.70 (d, J = 4.0 Hz, 2H), 5.14 (d, J = 11.9 Hz, 1H), 4.55 (d, J = 3.7 Hz, 1H), 4.20–4.09 (m, 1H), 3.42 (d, J = 12.6 Hz, 1H), 3.25 (ddd, J = 15.7, 10.7, 5.6 Hz, 2H), 3.03 (dd, J = 10.0, 5.6 Hz, 1H), 2.25 (dt, J = 16.4, 8.4 Hz, 1H), 2.15–1.91 (m, 4H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 730.1, found 730.2 ([M+H]+); HRMS (m/z): calcd. C37H31BrN5NiO3 for 730.0966, found 730.0966 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 32.0, tminor = 14.0 min, de = 95%.3.3.8. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(3-methoxyphenyl) Butyric Acid Schiff Base Complex (7h)
 = +1678 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.18 (d, J = 8.7 Hz, 1H), 7.93 (d, J = 7.5 Hz, 2H), 7.59 (dd, J = 13.8, 7.2 Hz, 3H), 7.44 (t, J = 7.9 Hz, 1H), 7.32 (d, J = 6.9 Hz, 1H), 7.23 (t, J = 7.5 Hz, 2H), 7.07 (dd, J = 13.9, 6.8 Hz, 4H), 6.75 (d, J = 11.2 Hz, 2H), 6.62 (d, J = 4.0 Hz, 2H), 5.09 (d, J = 12.0 Hz, 1H), 4.47 (d, J = 3.4 Hz, 1H), 4.04 (dd, J = 15.5, 9.7 Hz, 1H), 3.71 (s, 3H), 3.34 (d, J = 12.6 Hz, 1H), 3.16 (dd, J = 16.1, 6.4 Hz, 2H), 2.89 (q, J = 10.2 Hz, 1H), 2.26–2.06 (m, 1H), 2.01 (dd, J = 13.8, 7.1 Hz, 1H), 1.87 (dd, J = 18.6, 6.4 Hz, 2H), 1.49 (dd, J = 14.2, 9.1 Hz, 1H). 13C-NMR (CDCl3) δ 180.1, 176.2, 173.3, 160.6, 143.3, 133.9, 133.8, 133.4, 133.3, 133.2, 131.4, 130.9, 130.9, 130.5, 129.4, 128.9, 128.8, 127.5, 127.0, 125.5, 123.2, 120.7, 115.7, 111.5, 111.2, 70.5, 69.6, 63.9, 57.4, 55.3, 48.6, 30.6, 24.6, 23.0. ESI-MS (m/z): calcd. 682.2, found 682.3 ([M+H]+); HRMS (m/z): calcd. C38H34N5NiO4 for 682.1964, found 682.1959 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 36.2, tminor = 16.5 min, de > 99%.
= +1678 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.18 (d, J = 8.7 Hz, 1H), 7.93 (d, J = 7.5 Hz, 2H), 7.59 (dd, J = 13.8, 7.2 Hz, 3H), 7.44 (t, J = 7.9 Hz, 1H), 7.32 (d, J = 6.9 Hz, 1H), 7.23 (t, J = 7.5 Hz, 2H), 7.07 (dd, J = 13.9, 6.8 Hz, 4H), 6.75 (d, J = 11.2 Hz, 2H), 6.62 (d, J = 4.0 Hz, 2H), 5.09 (d, J = 12.0 Hz, 1H), 4.47 (d, J = 3.4 Hz, 1H), 4.04 (dd, J = 15.5, 9.7 Hz, 1H), 3.71 (s, 3H), 3.34 (d, J = 12.6 Hz, 1H), 3.16 (dd, J = 16.1, 6.4 Hz, 2H), 2.89 (q, J = 10.2 Hz, 1H), 2.26–2.06 (m, 1H), 2.01 (dd, J = 13.8, 7.1 Hz, 1H), 1.87 (dd, J = 18.6, 6.4 Hz, 2H), 1.49 (dd, J = 14.2, 9.1 Hz, 1H). 13C-NMR (CDCl3) δ 180.1, 176.2, 173.3, 160.6, 143.3, 133.9, 133.8, 133.4, 133.3, 133.2, 131.4, 130.9, 130.9, 130.5, 129.4, 128.9, 128.8, 127.5, 127.0, 125.5, 123.2, 120.7, 115.7, 111.5, 111.2, 70.5, 69.6, 63.9, 57.4, 55.3, 48.6, 30.6, 24.6, 23.0. ESI-MS (m/z): calcd. 682.2, found 682.3 ([M+H]+); HRMS (m/z): calcd. C38H34N5NiO4 for 682.1964, found 682.1959 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 36.2, tminor = 16.5 min, de > 99%.3.3.9. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(2-naphthyl) Butyric Acid Schiff Base Complex (7i)
 = +1582 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.19 (d, J = 8.7 Hz, 1H), 8.08 (d, J = 8.4 Hz, 1H), 7.97 (d, J = 7.5 Hz, 3H), 7.93 (d, J = 7.7 Hz, 1H), 7.81 (s, 1H), 7.70 (d, J = 7.8 Hz, 3H), 7.60 (p, J = 6.7 Hz, 2H), 7.41 (d, J = 7.0 Hz, 1H), 7.32 (d, J = 7.0 Hz, 1H), 7.26 (t, J = 7.6 Hz, 3H), 7.20–7.09 (m, 2H), 6.70 (d, J = 14.1 Hz, 2H), 5.34 (d, J = 12.0 Hz, 1H), 4.62 (d, J = 3.1 Hz, 1H), 4.12 (d, J = 7.1 Hz, 1H), 4.02 (d, J = 12.5 Hz, 1H), 3.46 (dd, J = 12.0, 3.1 Hz, 1H), 3.23 (d, J = 12.5 Hz, 1H), 3.05 (t, J = 8.5 Hz, 1H), 2.64–2.48 (m, 1H), 2.04 (s, 1H), 1.74 (d, J = 10.0 Hz, 2H), 1.24 (dd, J = 16.0, 8.8 Hz, 1H). 13C-NMR (CDCl3) δ 180.1, 176.2, 173.4, 143.3, 134.1, 133.9, 133.5, 133.3, 133.2, 131.3, 130.9, 130.5, 129.8, 129.5, 128.9, 128.8, 128.5, 127.8, 127.5, 127.5, 127.3, 127.1, 125.5, 123.3, 120.7, 111.6, 111.3, 70.3, 70.0, 64.0, 57.7, 48.9, 30.0, 24.6, 22.3. ESI-MS (m/z): calcd. 702.2, found 702.3 ([M+H]+); HRMS (m/z): calcd. C41H34N5NiO3 for 702.2015, found 702.2022 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.5 min, tminor = 18.8 min, de = 98%.
= +1582 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.19 (d, J = 8.7 Hz, 1H), 8.08 (d, J = 8.4 Hz, 1H), 7.97 (d, J = 7.5 Hz, 3H), 7.93 (d, J = 7.7 Hz, 1H), 7.81 (s, 1H), 7.70 (d, J = 7.8 Hz, 3H), 7.60 (p, J = 6.7 Hz, 2H), 7.41 (d, J = 7.0 Hz, 1H), 7.32 (d, J = 7.0 Hz, 1H), 7.26 (t, J = 7.6 Hz, 3H), 7.20–7.09 (m, 2H), 6.70 (d, J = 14.1 Hz, 2H), 5.34 (d, J = 12.0 Hz, 1H), 4.62 (d, J = 3.1 Hz, 1H), 4.12 (d, J = 7.1 Hz, 1H), 4.02 (d, J = 12.5 Hz, 1H), 3.46 (dd, J = 12.0, 3.1 Hz, 1H), 3.23 (d, J = 12.5 Hz, 1H), 3.05 (t, J = 8.5 Hz, 1H), 2.64–2.48 (m, 1H), 2.04 (s, 1H), 1.74 (d, J = 10.0 Hz, 2H), 1.24 (dd, J = 16.0, 8.8 Hz, 1H). 13C-NMR (CDCl3) δ 180.1, 176.2, 173.4, 143.3, 134.1, 133.9, 133.5, 133.3, 133.2, 131.3, 130.9, 130.5, 129.8, 129.5, 128.9, 128.8, 128.5, 127.8, 127.5, 127.5, 127.3, 127.1, 125.5, 123.3, 120.7, 111.6, 111.3, 70.3, 70.0, 64.0, 57.7, 48.9, 30.0, 24.6, 22.3. ESI-MS (m/z): calcd. 702.2, found 702.3 ([M+H]+); HRMS (m/z): calcd. C41H34N5NiO3 for 702.2015, found 702.2022 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.5 min, tminor = 18.8 min, de = 98%.3.3.10. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(1-naphthyl) Butyric Acid Schiff Base Complex (7j)
 = +1614 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.8 Hz, 1H), 8.14 (d, J = 8.2 Hz, 1H), 8.05 (d, J = 8.2 Hz, 1H), 7.89 (d, J = 7.5 Hz, 2H), 7.80–7.65 (m, 4H), 7.57 (d, J = 7.3 Hz, 1H), 7.55–7.43 (m, 3H), 7.41 (d, J = 7.0 Hz, 1H), 7.29–7.21 (m, 3H), 7.21–7.07 (m, 3H), 6.80–6.67 (m, 2H), 5.36 (d, J = 11.8 Hz, 1H), 4.74 (d, J = 2.6 Hz, 1H), 4.20 (dd, J = 11.8, 2.2 Hz, 1H), 4.12 (q, J = 7.1 Hz, 1H), 3.97 (d, J = 12.6 Hz, 1H), 3.25 (d, J = 12.6 Hz, 1H), 2.95 (t, J = 8.7 Hz, 1H), 2.50 (dt, J = 11.4, 5.8 Hz, 1H), 1.86 (dd, J = 12.9, 9.2 Hz, 1H), 1.75 (dt, J = 22.7, 9.6 Hz, 2H), 0.97 (dt, J = 14.6, 7.4 Hz, 1H). 13C-NMR (CDCl3) δ 179.5, 176.1, 173.6, 143.4, 134.4, 134.0, 133.5, 133.4, 133.1, 133.0, 131.2, 130.6, 130.4, 130.1, 129.3, 129.2, 128.8, 128.7, 127.2, 127.1, 126.9, 126.7, 126.1, 126.0, 125.1, 123.0, 122.5, 120.5, 111.6, 111.1, 71.4, 70.3, 63.7, 57.2, 43.5, 30.2, 25.7, 22.9. ESI-MS (m/z): calcd. 702.2, found 702.3 ([M+H]+); HRMS (m/z): calcd. C41H34N5NiO3 for 702.2015, found 702.2019 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 55.7 min, tminor = 20.4 min, de = 98%.
= +1614 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.8 Hz, 1H), 8.14 (d, J = 8.2 Hz, 1H), 8.05 (d, J = 8.2 Hz, 1H), 7.89 (d, J = 7.5 Hz, 2H), 7.80–7.65 (m, 4H), 7.57 (d, J = 7.3 Hz, 1H), 7.55–7.43 (m, 3H), 7.41 (d, J = 7.0 Hz, 1H), 7.29–7.21 (m, 3H), 7.21–7.07 (m, 3H), 6.80–6.67 (m, 2H), 5.36 (d, J = 11.8 Hz, 1H), 4.74 (d, J = 2.6 Hz, 1H), 4.20 (dd, J = 11.8, 2.2 Hz, 1H), 4.12 (q, J = 7.1 Hz, 1H), 3.97 (d, J = 12.6 Hz, 1H), 3.25 (d, J = 12.6 Hz, 1H), 2.95 (t, J = 8.7 Hz, 1H), 2.50 (dt, J = 11.4, 5.8 Hz, 1H), 1.86 (dd, J = 12.9, 9.2 Hz, 1H), 1.75 (dt, J = 22.7, 9.6 Hz, 2H), 0.97 (dt, J = 14.6, 7.4 Hz, 1H). 13C-NMR (CDCl3) δ 179.5, 176.1, 173.6, 143.4, 134.4, 134.0, 133.5, 133.4, 133.1, 133.0, 131.2, 130.6, 130.4, 130.1, 129.3, 129.2, 128.8, 128.7, 127.2, 127.1, 126.9, 126.7, 126.1, 126.0, 125.1, 123.0, 122.5, 120.5, 111.6, 111.1, 71.4, 70.3, 63.7, 57.2, 43.5, 30.2, 25.7, 22.9. ESI-MS (m/z): calcd. 702.2, found 702.3 ([M+H]+); HRMS (m/z): calcd. C41H34N5NiO3 for 702.2015, found 702.2019 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 55.7 min, tminor = 20.4 min, de = 98%.3.3.11. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(3-hydroxyphenyl) Butyric Acid Schiff Base Complex (7k)
 = +1775 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (DMSO) δ 9.89 (s, 1H), 8.42 (d, J = 7.5 Hz, 2H), 8.11 (d, J = 8.7 Hz, 1H), 7.78 (d, J = 5.3 Hz, 1H), 7.69 (s, 3H), 7.54 (d, J = 4.9 Hz, 1H), 7.46 (t, J = 7.8 Hz, 1H), 7.37 (t, J = 7.5 Hz, 2H), 7.13 (dd, J = 16.4, 6.8 Hz, 3H), 6.93–6.80 (m, 2H), 6.73 (t, J = 7.6 Hz, 1H), 6.63 (d, J = 8.3 Hz, 1H), 5.40 (d, J = 12.4 Hz, 1H), 4.40 (d, J = 3.6 Hz, 1H), 4.09 (q, J = 7.1 Hz, 1H), 3.91 (d, J = 12.2 Hz, 1H), 3.15 (dd, J = 12.3, 3.5 Hz, 1H), 2.96–2.84 (m, 1H), 2.56 (s, 2H), 2.23 (dd, J = 15.2, 8.6 Hz, 1H), 2.17–2.07 (m, 1H), 1.99 (dd, J = 25.2, 14.2 Hz, 2H), 1.70 (d, J = 6.5 Hz, 1H). 13C-NMR (DMSO) δ 180.0, 174.8, 171.9, 158.2, 143.2, 134.5, 134.1, 133.5, 133.1, 131.8, 131.5, 130.2, 130.1, 129.7, 128.9, 128.4, 128.1, 127.8, 127.4, 125.1, 122.8, 119.8, 116.4, 113.1, 112.1, 69.7, 69.6, 63.2, 57.7, 47.5, 30.4, 25.2, 22.6. ESI-MS (m/z): calcd. 668.2, found 668.2 ([M+H]+); HRMS (m/z): calcd. C37H32N5NiO4 for 668.1808, found 668.1819 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 14.7 min, tminor = 7.1 min, de = 98%.
= +1775 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (DMSO) δ 9.89 (s, 1H), 8.42 (d, J = 7.5 Hz, 2H), 8.11 (d, J = 8.7 Hz, 1H), 7.78 (d, J = 5.3 Hz, 1H), 7.69 (s, 3H), 7.54 (d, J = 4.9 Hz, 1H), 7.46 (t, J = 7.8 Hz, 1H), 7.37 (t, J = 7.5 Hz, 2H), 7.13 (dd, J = 16.4, 6.8 Hz, 3H), 6.93–6.80 (m, 2H), 6.73 (t, J = 7.6 Hz, 1H), 6.63 (d, J = 8.3 Hz, 1H), 5.40 (d, J = 12.4 Hz, 1H), 4.40 (d, J = 3.6 Hz, 1H), 4.09 (q, J = 7.1 Hz, 1H), 3.91 (d, J = 12.2 Hz, 1H), 3.15 (dd, J = 12.3, 3.5 Hz, 1H), 2.96–2.84 (m, 1H), 2.56 (s, 2H), 2.23 (dd, J = 15.2, 8.6 Hz, 1H), 2.17–2.07 (m, 1H), 1.99 (dd, J = 25.2, 14.2 Hz, 2H), 1.70 (d, J = 6.5 Hz, 1H). 13C-NMR (DMSO) δ 180.0, 174.8, 171.9, 158.2, 143.2, 134.5, 134.1, 133.5, 133.1, 131.8, 131.5, 130.2, 130.1, 129.7, 128.9, 128.4, 128.1, 127.8, 127.4, 125.1, 122.8, 119.8, 116.4, 113.1, 112.1, 69.7, 69.6, 63.2, 57.7, 47.5, 30.4, 25.2, 22.6. ESI-MS (m/z): calcd. 668.2, found 668.2 ([M+H]+); HRMS (m/z): calcd. C37H32N5NiO4 for 668.1808, found 668.1819 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 14.7 min, tminor = 7.1 min, de = 98%.3.3.12. Ni(II)-(S)-BPB/(2S,3R)-2-Amino-4,4'-dicyano-3-(2-fluoro-4-bromophenyl) Butyric Acid Schiff Base Complex (7l)
 = +1624 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.32 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.5 Hz, 2H), 7.66 (d, J = 6.2 Hz, 3H), 7.60 (d, J = 8.6 Hz, 2H), 7.35 (d, J = 5.2 Hz, 1H), 7.29 (dd, J = 13.5, 6.0 Hz, 3H), 7.24–7.11 (m, 3H), 6.68 (s, 2H), 5.23 (d, J = 12.0 Hz, 1H), 4.53 (d, J = 3.3 Hz, 1H), 4.20–4.05 (m, 1H), 3.80 (d, J = 9.4 Hz, 1H), 3.45 (d, J = 12.6 Hz, 1H), 3.30 (t, J = 8.5 Hz, 1H), 2.96 (q, J = 10.3 Hz, 1H), 2.44–2.25 (m, 1H), 2.25–2.08 (m, 1H), 2.02–1.89 (m, 2H), 1.79–1.64 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 748.1, found 748.1 ([M+H]+); HRMS (m/z): calcd. C37H30BrFN5NiO3 for 748.0869, found 748.0881 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 36.2 min, tminor = 15.3 min, de > 99%.
= +1624 (ca. 0.2 g/100 mL, CHCl3). 1H-NMR (CDCl3) δ 8.32 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.5 Hz, 2H), 7.66 (d, J = 6.2 Hz, 3H), 7.60 (d, J = 8.6 Hz, 2H), 7.35 (d, J = 5.2 Hz, 1H), 7.29 (dd, J = 13.5, 6.0 Hz, 3H), 7.24–7.11 (m, 3H), 6.68 (s, 2H), 5.23 (d, J = 12.0 Hz, 1H), 4.53 (d, J = 3.3 Hz, 1H), 4.20–4.05 (m, 1H), 3.80 (d, J = 9.4 Hz, 1H), 3.45 (d, J = 12.6 Hz, 1H), 3.30 (t, J = 8.5 Hz, 1H), 2.96 (q, J = 10.3 Hz, 1H), 2.44–2.25 (m, 1H), 2.25–2.08 (m, 1H), 2.02–1.89 (m, 2H), 1.79–1.64 (m, 1H). 13C-NMR (CDCl3) δ 180.2, 176.3, 173.3, 160.9, 143.2, 133.9, 133.4, 133.3, 133.2, 131.4, 130.8, 130.5, 129.4, 128.9, 128.8, 127.4, 127.0, 125.6, 124.1, 123.2, 120.8, 115.2, 111.6, 111.3, 70.6, 69.8, 64.0, 57.6, 55.4, 48.1, 30.5, 24.7, 22.9. ESI-MS (m/z): calcd. 748.1, found 748.1 ([M+H]+); HRMS (m/z): calcd. C37H30BrFN5NiO3 for 748.0869, found 748.0881 ([M+H]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 36.2 min, tminor = 15.3 min, de > 99%.3.3.13. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-5-oxo-3-phenylpentanoic Acid Schiff Base Complex (7m)
 = +2323 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.24 (d, J = 8.6 Hz, 1H), 7.99 (d, J = 7.1 Hz, 2H), 7.74–7.58 (m, 3H), 7.53 (s, 3H), 7.40 (d, J = 7.3 Hz, 1H), 7.35–7.26 (m, 4H), 7.22–7.08 (m, 3H), 6.70 (q, J = 7.7 Hz, 2H), 4.63 (s, 1H), 4.57 (d, J = 12.0 Hz, 1H), 4.18 (d, J = 12.6 Hz, 1H), 3.85 (q, J = 6.9 Hz, 2H), 3.39 (t, J = 12.9 Hz, 2H), 3.22 (t, J = 8.4 Hz, 1H), 2.93 (dt, J = 9.3, 4.6 Hz, 1H), 2.17 (dt, J = 16.0, 8.1 Hz, 1H), 2.02 (dd, J = 12.6, 6.5 Hz, 1H), 1.94 (dd, J = 18.3, 8.6 Hz, 1H), 1.82 (dt, J = 19.5, 6.8 Hz, 1H), 1.47 (ddd, J = 19.2, 12.4, 6.7 Hz, 1H), 0.90 (t, J = 6.9 Hz, 3H). 13C-NMR (CDCl3) δ 180.28, 176.58, 173.02, 164.34, 143.18, 134.17, 133.89, 133.71, 133.30, 132.85, 131.46, 130.59, 130.36, 129.32, 129.19, 129.12, 128.88, 128.80, 127.70, 127.12, 125.83, 123.20, 120.67, 114.69, 71.09, 70.50, 63.81, 62.42, 57.36, 48.45, 38.92, 30.62, 23.06, 13.48. HRMS (m/z): calcd. C39H36N4NaNiO5+ for 721.1931, found 721.1931 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.7 min, tminor = 8.0 min, de = 98%.
= +2323 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.24 (d, J = 8.6 Hz, 1H), 7.99 (d, J = 7.1 Hz, 2H), 7.74–7.58 (m, 3H), 7.53 (s, 3H), 7.40 (d, J = 7.3 Hz, 1H), 7.35–7.26 (m, 4H), 7.22–7.08 (m, 3H), 6.70 (q, J = 7.7 Hz, 2H), 4.63 (s, 1H), 4.57 (d, J = 12.0 Hz, 1H), 4.18 (d, J = 12.6 Hz, 1H), 3.85 (q, J = 6.9 Hz, 2H), 3.39 (t, J = 12.9 Hz, 2H), 3.22 (t, J = 8.4 Hz, 1H), 2.93 (dt, J = 9.3, 4.6 Hz, 1H), 2.17 (dt, J = 16.0, 8.1 Hz, 1H), 2.02 (dd, J = 12.6, 6.5 Hz, 1H), 1.94 (dd, J = 18.3, 8.6 Hz, 1H), 1.82 (dt, J = 19.5, 6.8 Hz, 1H), 1.47 (ddd, J = 19.2, 12.4, 6.7 Hz, 1H), 0.90 (t, J = 6.9 Hz, 3H). 13C-NMR (CDCl3) δ 180.28, 176.58, 173.02, 164.34, 143.18, 134.17, 133.89, 133.71, 133.30, 132.85, 131.46, 130.59, 130.36, 129.32, 129.19, 129.12, 128.88, 128.80, 127.70, 127.12, 125.83, 123.20, 120.67, 114.69, 71.09, 70.50, 63.81, 62.42, 57.36, 48.45, 38.92, 30.62, 23.06, 13.48. HRMS (m/z): calcd. C39H36N4NaNiO5+ for 721.1931, found 721.1931 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.7 min, tminor = 8.0 min, de = 98%.3.3.14. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-3-(2-bromophenyl)-4-cyano-5-ethoxy-5-oxopentanoic Acid Schiff Base Complex (7n)
 = +2120 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.44 (d, J = 8.7 Hz, 1H), 7.96 (d, J = 7.3 Hz, 2H), 7.88 (d, J = 7.8 Hz, 1H), 7.69–7.61 (m, 3H), 7.58–7.53 (m, 1H), 7.53–7.44 (m, 2H), 7.43–7.36 (m, 1H), 7.29 (dd, J = 10.5, 4.9 Hz, 3H), 7.16 (dt, J = 13.7, 5.3 Hz, 2H), 6.73–6.63 (m, 2H), 4.62 (d, J = 3.0 Hz, 1H), 4.49 (d, J = 12.2 Hz, 1H), 4.17 (d, J = 12.6 Hz, 1H), 4.06 (dd, J = 12.2, 3.0 Hz, 1H), 3.89 (qd, J = 7.1, 2.3 Hz, 2H), 3.43 (d, J = 12.6 Hz, 1H), 3.25 (t, J = 8.6 Hz, 1H), 2.85-2.75 (m, 1H), 2.19 (ddd, J = 19.3, 13.1, 7.3 Hz, 2H), 1.92 (dt, J = 11.2, 8.1 Hz, 1H), 1.70 (dt, J = 13.6, 7.5 Hz, 1H), 1.46 (dt, J = 18.8, 6.4 Hz, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.14, 176.38, 174.21, 163.71, 143.31, 134.68, 134.06, 133.87, 133.70, 133.24, 133.05, 131.42, 130.80, 130.31, 130.03, 129.76, 128.87, 128.79, 128.55, 128.06, 127.12, 127.05, 125.84, 122.92, 120.52, 113.84, 71.89, 70.74, 63.75, 62.67, 57.14, 46.61, 39.74, 30.89, 23.02, 13.50, 0.01. HRMS (m/z): calcd. C39H35BrN4NaNiO5+for 799.1037, found 799.1034 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor =42.8 min, tminor =10.6 min, de > 99%.
= +2120 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.44 (d, J = 8.7 Hz, 1H), 7.96 (d, J = 7.3 Hz, 2H), 7.88 (d, J = 7.8 Hz, 1H), 7.69–7.61 (m, 3H), 7.58–7.53 (m, 1H), 7.53–7.44 (m, 2H), 7.43–7.36 (m, 1H), 7.29 (dd, J = 10.5, 4.9 Hz, 3H), 7.16 (dt, J = 13.7, 5.3 Hz, 2H), 6.73–6.63 (m, 2H), 4.62 (d, J = 3.0 Hz, 1H), 4.49 (d, J = 12.2 Hz, 1H), 4.17 (d, J = 12.6 Hz, 1H), 4.06 (dd, J = 12.2, 3.0 Hz, 1H), 3.89 (qd, J = 7.1, 2.3 Hz, 2H), 3.43 (d, J = 12.6 Hz, 1H), 3.25 (t, J = 8.6 Hz, 1H), 2.85-2.75 (m, 1H), 2.19 (ddd, J = 19.3, 13.1, 7.3 Hz, 2H), 1.92 (dt, J = 11.2, 8.1 Hz, 1H), 1.70 (dt, J = 13.6, 7.5 Hz, 1H), 1.46 (dt, J = 18.8, 6.4 Hz, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.14, 176.38, 174.21, 163.71, 143.31, 134.68, 134.06, 133.87, 133.70, 133.24, 133.05, 131.42, 130.80, 130.31, 130.03, 129.76, 128.87, 128.79, 128.55, 128.06, 127.12, 127.05, 125.84, 122.92, 120.52, 113.84, 71.89, 70.74, 63.75, 62.67, 57.14, 46.61, 39.74, 30.89, 23.02, 13.50, 0.01. HRMS (m/z): calcd. C39H35BrN4NaNiO5+for 799.1037, found 799.1034 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor =42.8 min, tminor =10.6 min, de > 99%.3.3.15. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-(3-chlorophenyl)-4-cyano-5-ethoxy-3-5-oxopentanoic Acid Schiff Base Complex (7o)
 = +2376 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.70 (dd, J = 11.3, 4.9 Hz, 1H), 7.67–7.59 (m, 2H), 7.53 (d, J = 8.2 Hz, 1H), 7.46 (t, J = 7.8 Hz, 1H), 7.42–7.34 (m, 2H), 7.31 (t, J = 7.6 Hz, 2H), 7.21–7.13 (m, 3H), 7.11 (d, J = 7.2 Hz, 1H), 6.76–6.64 (m, 2H), 4.62 (d, J = 3.7 Hz, 1H), 4.53 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.89 (q, J = 7.1 Hz, 2H), 3.42 (d, J = 12.6 Hz, 1H), 3.33 (dd, J = 12.2, 3.7 Hz, 1H), 3.26 (dd, J = 9.3, 7.7 Hz, 1H), 3.02 (dd, J = 10.6, 5.8 Hz, 1H), 2.23 (td, J = 17.0, 7.6 Hz, 1H), 2.09 (dd, J = 13.4, 7.4 Hz, 1H), 1.99 (dd, J = 10.8, 6.3 Hz, 1H), 1.93 (dd, J = 14.0, 7.2 Hz, 1H), 1.62 (d, J = 12.7 Hz, 1H), 0.96 (t, J = 7.1 Hz, 3H). 13C-NMR () δ 180.29, 176.27, 173.26, 164.17, 143.30, 136.44, 135.58, 133.91, 133.62, 133.34, 133.04, 131.45, 130.68, 130.43, 129.42, 129.26, 128.92, 128.83, 127.58, 127.06, 125.66, 123.25, 120.68, 114.35, 70.79, 70.54, 63.88, 62.61, 57.57, 48.07, 38.81, 30.76, 23.03, 13.51. HRMS (m/z): calcd. C39H35ClN4NaNiO5+for 755.1542, found 755.1541 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.6 min, tminor = 8.1 min, de = 97%.
= +2376 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.4 Hz, 2H), 7.70 (dd, J = 11.3, 4.9 Hz, 1H), 7.67–7.59 (m, 2H), 7.53 (d, J = 8.2 Hz, 1H), 7.46 (t, J = 7.8 Hz, 1H), 7.42–7.34 (m, 2H), 7.31 (t, J = 7.6 Hz, 2H), 7.21–7.13 (m, 3H), 7.11 (d, J = 7.2 Hz, 1H), 6.76–6.64 (m, 2H), 4.62 (d, J = 3.7 Hz, 1H), 4.53 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.89 (q, J = 7.1 Hz, 2H), 3.42 (d, J = 12.6 Hz, 1H), 3.33 (dd, J = 12.2, 3.7 Hz, 1H), 3.26 (dd, J = 9.3, 7.7 Hz, 1H), 3.02 (dd, J = 10.6, 5.8 Hz, 1H), 2.23 (td, J = 17.0, 7.6 Hz, 1H), 2.09 (dd, J = 13.4, 7.4 Hz, 1H), 1.99 (dd, J = 10.8, 6.3 Hz, 1H), 1.93 (dd, J = 14.0, 7.2 Hz, 1H), 1.62 (d, J = 12.7 Hz, 1H), 0.96 (t, J = 7.1 Hz, 3H). 13C-NMR () δ 180.29, 176.27, 173.26, 164.17, 143.30, 136.44, 135.58, 133.91, 133.62, 133.34, 133.04, 131.45, 130.68, 130.43, 129.42, 129.26, 128.92, 128.83, 127.58, 127.06, 125.66, 123.25, 120.68, 114.35, 70.79, 70.54, 63.88, 62.61, 57.57, 48.07, 38.81, 30.76, 23.03, 13.51. HRMS (m/z): calcd. C39H35ClN4NaNiO5+for 755.1542, found 755.1541 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 26.6 min, tminor = 8.1 min, de = 97%.3.3.16. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-3-(4-fluorophenyl)-5-oxopentanoic Acid Schiff Base Complex (7p)
 = +2250 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.5 Hz, 2H), 7.69 (t, J = 7.0 Hz, 1H), 7.67–7.59 (m, 2H), 7.39 (d, J = 7.3 Hz, 1H), 7.30 (dd, J = 14.3, 6.6 Hz, 4H), 7.22(d, J = 8.5, 2H), 7.16 (t, J = 7.3 Hz, 3H), 6.70 (q, J = 8.2 Hz, 2H), 4.62 (d, J = 3.4 Hz, 1H), 4.52 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.88 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.36 (dd, J = 12.2, 3.5 Hz, 1H), 3.29–3.22 (m, 1H), 2.99 (dd, J = 10.1, 5.7 Hz, 1H), 2.25 (td, J = 16.7, 7.6 Hz, 1H), 2.09 (dt, J = 16.1, 8.6 Hz, 1H), 1.96 (dt, J = 14.1, 9.6 Hz, 2H), 1.66–1.55 (m, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.32, 176.43, 173.22, 164.62, 164.23, 162.15, 143.22, 133.90, 133.68, 133.33, 132.97, 131.43, 130.65, 130.44, 130.04, 130.00, 129.23, 128.93, 128.83, 127.58, 127.09, 125.70, 123.22, 120.70, 116.40, 116.19, 114.44, 99.99, 70.99, 70.48, 63.91, 62.55, 57.48, 47.82, 38.99, 30.68, 22.94, 13.55. HRMS (m/z): calcd. C39H35FN4NaNiO5+for 739.1837, found 739.1837 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 53.2 min, tminor = 6.9 min, de = 96%.
= +2250 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.5 Hz, 2H), 7.69 (t, J = 7.0 Hz, 1H), 7.67–7.59 (m, 2H), 7.39 (d, J = 7.3 Hz, 1H), 7.30 (dd, J = 14.3, 6.6 Hz, 4H), 7.22(d, J = 8.5, 2H), 7.16 (t, J = 7.3 Hz, 3H), 6.70 (q, J = 8.2 Hz, 2H), 4.62 (d, J = 3.4 Hz, 1H), 4.52 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.88 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.36 (dd, J = 12.2, 3.5 Hz, 1H), 3.29–3.22 (m, 1H), 2.99 (dd, J = 10.1, 5.7 Hz, 1H), 2.25 (td, J = 16.7, 7.6 Hz, 1H), 2.09 (dt, J = 16.1, 8.6 Hz, 1H), 1.96 (dt, J = 14.1, 9.6 Hz, 2H), 1.66–1.55 (m, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.32, 176.43, 173.22, 164.62, 164.23, 162.15, 143.22, 133.90, 133.68, 133.33, 132.97, 131.43, 130.65, 130.44, 130.04, 130.00, 129.23, 128.93, 128.83, 127.58, 127.09, 125.70, 123.22, 120.70, 116.40, 116.19, 114.44, 99.99, 70.99, 70.48, 63.91, 62.55, 57.48, 47.82, 38.99, 30.68, 22.94, 13.55. HRMS (m/z): calcd. C39H35FN4NaNiO5+for 739.1837, found 739.1837 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 53.2 min, tminor = 6.9 min, de = 96%.3.3.17. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-3-(3,4-dichlorophenyl)-5-ethoxy-5-oxopentanoic Acid Schiff Base Complex (7q)
 = +2353 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.02 (d, J = 7.6 Hz, 2H), 7.70 (t, J = 7.1 Hz, 1H), 7.63 (dd, J = 13.5, 8.8 Hz, 3H), 7.44 (s, 1H), 7.38 (d, J = 7.4 Hz, 1H), 7.31 (t, J = 7.6 Hz, 2H), 7.20–7.11 (m, 3H), 7.08 (d, J = 7.6 Hz, 1H), 6.78–6.64 (m, 2H), 4.60 (d, J = 3.6 Hz, 1H), 4.51 (d, J = 12.1 Hz, 1H), 4.19 (d, J = 12.5 Hz, 1H), 3.94 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.35–3.25 (m, 2H), 3.00 (dt, J = 10.2, 5.2 Hz, 1H), 2.30 (dt, J = 16.8, 7.7 Hz, 1H), 2.09 (dd, J = 13.3, 6.0 Hz, 1H), 2.02 (dd, J = 11.8, 7.4 Hz, 1H), 1.95 (dd, J = 13.4, 6.3 Hz, 1H), 1.67 (dt, J = 13.0, 6.4 Hz, 1H), 1.02 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.36, 176.15, 173.45, 164.02, 143.31, 134.66, 133.94, 133.80, 133.56, 133.37, 133.17, 131.42, 131.08, 130.75, 130.50, 129.30, 128.96, 128.86, 127.48, 127.04, 125.53, 123.27, 120.75, 114.12, 70.67, 70.54, 64.03, 62.82, 57.78, 47.57, 38.59, 30.75, 22.83, 13.59. HRMS (m/z): calcd. C39H34Cl2N4NaNiO5+for 789.1152, found 789.1151 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 44.5 min, tminor = 7.6 min, de = 98%.
= +2353 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.27 (d, J = 8.7 Hz, 1H), 8.02 (d, J = 7.6 Hz, 2H), 7.70 (t, J = 7.1 Hz, 1H), 7.63 (dd, J = 13.5, 8.8 Hz, 3H), 7.44 (s, 1H), 7.38 (d, J = 7.4 Hz, 1H), 7.31 (t, J = 7.6 Hz, 2H), 7.20–7.11 (m, 3H), 7.08 (d, J = 7.6 Hz, 1H), 6.78–6.64 (m, 2H), 4.60 (d, J = 3.6 Hz, 1H), 4.51 (d, J = 12.1 Hz, 1H), 4.19 (d, J = 12.5 Hz, 1H), 3.94 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.35–3.25 (m, 2H), 3.00 (dt, J = 10.2, 5.2 Hz, 1H), 2.30 (dt, J = 16.8, 7.7 Hz, 1H), 2.09 (dd, J = 13.3, 6.0 Hz, 1H), 2.02 (dd, J = 11.8, 7.4 Hz, 1H), 1.95 (dd, J = 13.4, 6.3 Hz, 1H), 1.67 (dt, J = 13.0, 6.4 Hz, 1H), 1.02 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.36, 176.15, 173.45, 164.02, 143.31, 134.66, 133.94, 133.80, 133.56, 133.37, 133.17, 131.42, 131.08, 130.75, 130.50, 129.30, 128.96, 128.86, 127.48, 127.04, 125.53, 123.27, 120.75, 114.12, 70.67, 70.54, 64.03, 62.82, 57.78, 47.57, 38.59, 30.75, 22.83, 13.59. HRMS (m/z): calcd. C39H34Cl2N4NaNiO5+for 789.1152, found 789.1151 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 44.5 min, tminor = 7.6 min, de = 98%.3.3.18. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-3-(2,4-dichlorophenyl)-5-ethoxy-5-oxopentanoic Acid Schiff Base Complex (7r)
 = +1960 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.42 (d, J = 8.8 Hz, 1H), 7.99 (d, J = 7.6 Hz, 2H), 7.71 (s, 1H), 7.69–7.59 (m, 3H), 7.44 (s, 2H), 7.41 (d, J = 3.5 Hz, 1H), 7.30 (dd, J = 13.9, 6.7 Hz, 3H), 7.19–7.11 (m, 2H), 6.72–6.63 (m, 2H), 4.60 (d, J = 3.1 Hz, 1H), 4.48 (d, J = 12.2 Hz, 1H), 4.18 (d, J = 12.6 Hz, 1H), 4.05 (dd, J = 12.2, 3.1 Hz, 1H), 3.99–3.88 (m, 2H), 3.43 (d, J = 12.6 Hz, 1H), 3.34–3.22 (m, 1H), 2.90 (dt, J = 11.0, 5.6 Hz, 1H), 2.33 (dt, J = 16.8, 7.7 Hz, 1H), 2.16 (td, J = 13.6, 7.7 Hz, 1H), 2.00 (dt, J = 10.9, 7.6 Hz, 1H), 1.88 (dt, J = 14.2, 7.4 Hz, 1H), 1.69–1.53 (m, 1H), 1.01 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.23, 176.30, 174.38, 163.63, 143.32, 137.69, 135.70, 134.05, 133.59, 133.31, 133.17, 131.56, 131.39, 130.83, 130.43, 130.19, 130.14, 129.65, 128.91, 128.83, 128.28, 127.08, 125.58, 122.92, 120.58, 113.73, 71.39, 70.71, 63.95, 62.85, 57.45, 43.63, 39.21, 30.98, 22.89, 13.56. HRMS (m/z): calcd. C39H34Cl2N4NaNiO5+for 789.1152, found 789.1151 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 77.3, tminor = 8.8 min, de = 98%.
= +1960 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.42 (d, J = 8.8 Hz, 1H), 7.99 (d, J = 7.6 Hz, 2H), 7.71 (s, 1H), 7.69–7.59 (m, 3H), 7.44 (s, 2H), 7.41 (d, J = 3.5 Hz, 1H), 7.30 (dd, J = 13.9, 6.7 Hz, 3H), 7.19–7.11 (m, 2H), 6.72–6.63 (m, 2H), 4.60 (d, J = 3.1 Hz, 1H), 4.48 (d, J = 12.2 Hz, 1H), 4.18 (d, J = 12.6 Hz, 1H), 4.05 (dd, J = 12.2, 3.1 Hz, 1H), 3.99–3.88 (m, 2H), 3.43 (d, J = 12.6 Hz, 1H), 3.34–3.22 (m, 1H), 2.90 (dt, J = 11.0, 5.6 Hz, 1H), 2.33 (dt, J = 16.8, 7.7 Hz, 1H), 2.16 (td, J = 13.6, 7.7 Hz, 1H), 2.00 (dt, J = 10.9, 7.6 Hz, 1H), 1.88 (dt, J = 14.2, 7.4 Hz, 1H), 1.69–1.53 (m, 1H), 1.01 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.23, 176.30, 174.38, 163.63, 143.32, 137.69, 135.70, 134.05, 133.59, 133.31, 133.17, 131.56, 131.39, 130.83, 130.43, 130.19, 130.14, 129.65, 128.91, 128.83, 128.28, 127.08, 125.58, 122.92, 120.58, 113.73, 71.39, 70.71, 63.95, 62.85, 57.45, 43.63, 39.21, 30.98, 22.89, 13.56. HRMS (m/z): calcd. C39H34Cl2N4NaNiO5+for 789.1152, found 789.1151 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 77.3, tminor = 8.8 min, de = 98%.3.3.19. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-5-oxo-3-(p-tolyl)pentanoic Acid Schiff Base Complex (7s)
 = +2570 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.24 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.3 Hz, 2H), 7.68 (dt, J = 9.7, 4.3 Hz, 1H), 7.61 (dd, J = 9.1, 5.4 Hz, 2H), 7.39 (d, J = 7.4 Hz, 1H), 7.31 (dd, J = 16.1, 8.0 Hz, 4H), 7.21–7.10 (m, 5H), 6.75–6.65 (m, 2H), 4.60 (d, J = 3.7 Hz, 1H), 4.54 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.86 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.33 (dd, J = 12.2, 3.7 Hz, 1H), 3.26–3.19 (m, 1H), 3.01–2.93 (m, 1H), 2.43 (s, 3H), 2.20 (dt, J = 16.7, 7.9 Hz, 1H), 2.04 (dt, J = 13.1, 6.8 Hz, 1H), 1.95 (dd, J = 11.1, 7.2 Hz, 1H), 1.83 (tt, J = 15.3, 7.6 Hz, 1H), 1.49 (tt, J = 12.8, 6.4 Hz, 1H), 0.94 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.19, 176.62, 172.87, 164.36, 143.12, 138.95, 133.86, 133.71, 133.31, 132.80, 131.45, 130.97, 130.55, 130.30, 129.97, 129.17, 128.87, 128.79, 127.70, 127.11, 125.83, 123.17, 120.65, 114.76, 71.12, 70.55, 63.87, 62.37, 57.40, 48.10, 38.85, 30.47, 22.78, 21.27, 13.51. HRMS (m/z): calcd. C40H38N4NaNiO5+for 735.2088, found 735.2089 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 46.3 min, tminor = 7.5 min, de > 99%.
= +2570 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.24 (d, J = 8.7 Hz, 1H), 8.00 (d, J = 7.3 Hz, 2H), 7.68 (dt, J = 9.7, 4.3 Hz, 1H), 7.61 (dd, J = 9.1, 5.4 Hz, 2H), 7.39 (d, J = 7.4 Hz, 1H), 7.31 (dd, J = 16.1, 8.0 Hz, 4H), 7.21–7.10 (m, 5H), 6.75–6.65 (m, 2H), 4.60 (d, J = 3.7 Hz, 1H), 4.54 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.86 (q, J = 7.1 Hz, 2H), 3.41 (d, J = 12.6 Hz, 1H), 3.33 (dd, J = 12.2, 3.7 Hz, 1H), 3.26–3.19 (m, 1H), 3.01–2.93 (m, 1H), 2.43 (s, 3H), 2.20 (dt, J = 16.7, 7.9 Hz, 1H), 2.04 (dt, J = 13.1, 6.8 Hz, 1H), 1.95 (dd, J = 11.1, 7.2 Hz, 1H), 1.83 (tt, J = 15.3, 7.6 Hz, 1H), 1.49 (tt, J = 12.8, 6.4 Hz, 1H), 0.94 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.19, 176.62, 172.87, 164.36, 143.12, 138.95, 133.86, 133.71, 133.31, 132.80, 131.45, 130.97, 130.55, 130.30, 129.97, 129.17, 128.87, 128.79, 127.70, 127.11, 125.83, 123.17, 120.65, 114.76, 71.12, 70.55, 63.87, 62.37, 57.40, 48.10, 38.85, 30.47, 22.78, 21.27, 13.51. HRMS (m/z): calcd. C40H38N4NaNiO5+for 735.2088, found 735.2089 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 46.3 min, tminor = 7.5 min, de > 99%.3.3.20. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-3-(4-methoxylphenyl)-5-oxopentanoic Acid Schiff Base Complex (7t)
 = +2376 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.23 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.5 Hz, 2H), 7.68 (t, J = 6.9 Hz, 1H), 7.65–7.58 (m, 2H), 7.38 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.21 (d, J = 8.1 Hz, 2H), 7.15 (dd, J = 13.2, 6.6 Hz, 3H), 7.04 (d, J = 8.5 Hz, 2H), 6.74–6.65 (m, 2H), 4.60 (d, J = 3.5 Hz, 1H), 4.52 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.91–3.86 (m, 2H), 3.85 (s, 3H), 3.40 (d, J = 12.6 Hz, 1H), 3.32 (dd, J = 12.2, 3.5 Hz, 1H), 3.24 (t, J = 8.5 Hz, 1H), 3.04–2.96 (m, 1H), 2.21 (dt, J = 16.3, 7.6 Hz, 1H), 2.08 (dd, J = 13.3, 6.2 Hz, 1H), 1.98 (dd, J = 11.0, 6.9 Hz, 1H), 1.90 (dd, J = 13.5, 6.8 Hz, 1H), 1.56–1.47 (m, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.28, 176.63, 172.88, 164.42, 160.34, 143.15, 133.85, 133.73, 133.37, 132.79, 131.46, 130.56, 130.32, 129.17, 128.88, 128.79, 127.67, 127.13, 125.85, 125.78, 123.18, 120.65, 114.73, 114.59, 71.20, 70.59, 63.92, 62.37, 57.55, 55.34, 47.89, 38.97, 30.62, 22.94, 13.56. HRMS (m/z): calcd. C40H38N4NaNiO6+for 751.2037, found 751.2037 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 71.2 min, tminor = 8.5 min, de = 97%.
= +2376 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.23 (d, J = 8.7 Hz, 1H), 8.01 (d, J = 7.5 Hz, 2H), 7.68 (t, J = 6.9 Hz, 1H), 7.65–7.58 (m, 2H), 7.38 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.21 (d, J = 8.1 Hz, 2H), 7.15 (dd, J = 13.2, 6.6 Hz, 3H), 7.04 (d, J = 8.5 Hz, 2H), 6.74–6.65 (m, 2H), 4.60 (d, J = 3.5 Hz, 1H), 4.52 (d, J = 12.2 Hz, 1H), 4.19 (d, J = 12.6 Hz, 1H), 3.91–3.86 (m, 2H), 3.85 (s, 3H), 3.40 (d, J = 12.6 Hz, 1H), 3.32 (dd, J = 12.2, 3.5 Hz, 1H), 3.24 (t, J = 8.5 Hz, 1H), 3.04–2.96 (m, 1H), 2.21 (dt, J = 16.3, 7.6 Hz, 1H), 2.08 (dd, J = 13.3, 6.2 Hz, 1H), 1.98 (dd, J = 11.0, 6.9 Hz, 1H), 1.90 (dd, J = 13.5, 6.8 Hz, 1H), 1.56–1.47 (m, 1H), 0.95 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.28, 176.63, 172.88, 164.42, 160.34, 143.15, 133.85, 133.73, 133.37, 132.79, 131.46, 130.56, 130.32, 129.17, 128.88, 128.79, 127.67, 127.13, 125.85, 125.78, 123.18, 120.65, 114.73, 114.59, 71.20, 70.59, 63.92, 62.37, 57.55, 55.34, 47.89, 38.97, 30.62, 22.94, 13.56. HRMS (m/z): calcd. C40H38N4NaNiO6+for 751.2037, found 751.2037 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 71.2 min, tminor = 8.5 min, de = 97%.3.3.21. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-3-(4-nitrophenyl)-5-oxopentanoic Acid Schiff Base Complex (7u)
 = +2163 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.40 (d, J = 8.5 Hz, 2H), 8.28 (d, J = 8.7 Hz, 1H), 7.98 (d, J = 7.5 Hz, 2H), 7.72 (t, J = 7.1 Hz, 1H), 7.69–7.60 (m, 2H), 7.48 (d, J = 8.4 Hz, 2H), 7.41 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.21–7.11 (m, 3H), 6.75–6.67 (m, 2H), 4.67 (d, J = 3.5 Hz, 1H), 4.62 (d, J = 12.1 Hz, 1H), 4.17 (d, J = 12.6 Hz, 1H), 3.96–3.86 (m, 2H), 3.48 (dd, J = 12.1, 3.5 Hz, 1H), 3.40 (d, J = 12.6 Hz, 1H), 3.23 (dd, J = 9.7, 7.1 Hz, 1H), 2.95–2.87 (m, 1H), 2.17 (dt, J = 17.7, 8.9 Hz, 1H), 1.97–1.86 (m, 2H), 1.68 (dd, J = 17.8, 10.7 Hz, 1H), 1.55–1.46 (m, 1H), 1.00 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.25, 176.03, 173.78, 163.86, 148.69, 143.31, 142.00, 134.01, 133.59, 133.32, 133.25, 131.36, 130.83, 130.64, 129.33, 129.00, 128.89, 127.47, 127.03, 125.46, 124.23, 123.26, 120.85, 113.96, 70.71, 70.32, 63.95, 62.93, 57.36, 47.97, 38.63, 30.64, 22.83, 13.61. HRMS (m/z): calcd. C39H35N5NaNiO7+for 766.1782, found 766.1782 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 69.0 min, tminor = 8.6 min, de = 97%.
= +2163 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.40 (d, J = 8.5 Hz, 2H), 8.28 (d, J = 8.7 Hz, 1H), 7.98 (d, J = 7.5 Hz, 2H), 7.72 (t, J = 7.1 Hz, 1H), 7.69–7.60 (m, 2H), 7.48 (d, J = 8.4 Hz, 2H), 7.41 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.21–7.11 (m, 3H), 6.75–6.67 (m, 2H), 4.67 (d, J = 3.5 Hz, 1H), 4.62 (d, J = 12.1 Hz, 1H), 4.17 (d, J = 12.6 Hz, 1H), 3.96–3.86 (m, 2H), 3.48 (dd, J = 12.1, 3.5 Hz, 1H), 3.40 (d, J = 12.6 Hz, 1H), 3.23 (dd, J = 9.7, 7.1 Hz, 1H), 2.95–2.87 (m, 1H), 2.17 (dt, J = 17.7, 8.9 Hz, 1H), 1.97–1.86 (m, 2H), 1.68 (dd, J = 17.8, 10.7 Hz, 1H), 1.55–1.46 (m, 1H), 1.00 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.25, 176.03, 173.78, 163.86, 148.69, 143.31, 142.00, 134.01, 133.59, 133.32, 133.25, 131.36, 130.83, 130.64, 129.33, 129.00, 128.89, 127.47, 127.03, 125.46, 124.23, 123.26, 120.85, 113.96, 70.71, 70.32, 63.95, 62.93, 57.36, 47.97, 38.63, 30.64, 22.83, 13.61. HRMS (m/z): calcd. C39H35N5NaNiO7+for 766.1782, found 766.1782 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 69.0 min, tminor = 8.6 min, de = 97%.3.3.22. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-3-(4-(tert-butyl)phenyl)-4-cyano-5-ethoxy-5-oxopentanoic Acid Schiff Base Complex (7v)
 = +2260 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.29 (d, J = 8.7 Hz, 1H), 7.95 (d, J = 7.4 Hz, 2H), 7.72–7.66 (m, 1H), 7.66–7.59 (m, 2H), 7.50 (d, J = 8.3 Hz, 2H), 7.40 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.17 (dt, J = 15.7, 7.6 Hz, 5H), 6.75–6.66 (m, 2H), 4.63 (d, J = 3.7 Hz, 1H), 4.53 (d, J = 12.2 Hz, 1H), 4.20 (d, J = 12.7 Hz, 1H), 3.86–3.76 (m, 2H), 3.49 (d, J = 12.7 Hz, 1H), 3.32 (dd, J = 12.2, 3.7 Hz, 1H), 3.17 (dd, J = 9.8, 7.4 Hz, 1H), 3.00 (dd, J = 10.3, 6.8 Hz, 1H), 2.18 (dt, J = 18.1, 8.3 Hz, 1H), 2.06 (dd, J = 11.2, 6.6 Hz, 1H), 1.89 (dd, J = 19.4, 8.6 Hz, 1H), 1.80 (dd, J = 10.3, 6.2 Hz, 1H), 1.48 (d, J = 8.3 Hz, 1H), 1.34 (s, 9H), 0.79 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.08, 176.69, 173.06, 164.52, 151.93, 143.13, 133.93, 133.77, 133.17, 132.84, 131.41, 130.88, 130.56, 130.34, 129.16, 128.85, 128.80, 127.82, 127.07, 126.29, 126.25, 125.92, 123.10, 120.69, 114.80, 70.98, 70.48, 63.49, 62.22, 56.56, 48.08, 39.24, 34.76, 31.35, 30.72, 22.77, 13.35. HRMS (m/z): calcd. C43H44N4NaNiO5+for 777.2557, found 777.2557 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 58.6 min, tminor = 8.7 min, de = 97%.
= +2260 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.29 (d, J = 8.7 Hz, 1H), 7.95 (d, J = 7.4 Hz, 2H), 7.72–7.66 (m, 1H), 7.66–7.59 (m, 2H), 7.50 (d, J = 8.3 Hz, 2H), 7.40 (d, J = 7.4 Hz, 1H), 7.30 (t, J = 7.6 Hz, 2H), 7.17 (dt, J = 15.7, 7.6 Hz, 5H), 6.75–6.66 (m, 2H), 4.63 (d, J = 3.7 Hz, 1H), 4.53 (d, J = 12.2 Hz, 1H), 4.20 (d, J = 12.7 Hz, 1H), 3.86–3.76 (m, 2H), 3.49 (d, J = 12.7 Hz, 1H), 3.32 (dd, J = 12.2, 3.7 Hz, 1H), 3.17 (dd, J = 9.8, 7.4 Hz, 1H), 3.00 (dd, J = 10.3, 6.8 Hz, 1H), 2.18 (dt, J = 18.1, 8.3 Hz, 1H), 2.06 (dd, J = 11.2, 6.6 Hz, 1H), 1.89 (dd, J = 19.4, 8.6 Hz, 1H), 1.80 (dd, J = 10.3, 6.2 Hz, 1H), 1.48 (d, J = 8.3 Hz, 1H), 1.34 (s, 9H), 0.79 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 180.08, 176.69, 173.06, 164.52, 151.93, 143.13, 133.93, 133.77, 133.17, 132.84, 131.41, 130.88, 130.56, 130.34, 129.16, 128.85, 128.80, 127.82, 127.07, 126.29, 126.25, 125.92, 123.10, 120.69, 114.80, 70.98, 70.48, 63.49, 62.22, 56.56, 48.08, 39.24, 34.76, 31.35, 30.72, 22.77, 13.35. HRMS (m/z): calcd. C43H44N4NaNiO5+for 777.2557, found 777.2557 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 58.6 min, tminor = 8.7 min, de = 97%.3.3.23. Ni(II)-(S)-BPB/(2S,3R,4S)-2-Amino-4-cyano-5-ethoxy-3-(naphthalen-1-y)-5-oxopentanoic Acid Schiff Base Complex (7w)
 = +1793 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.7 Hz, 1H), 8.00 (dd, J = 16.3, 8.0 Hz, 2H), 7.90 (d, J = 7.5 Hz, 2H), 7.71 (dd, J = 16.1, 8.1 Hz, 3H), 7.66–7.56 (m, 3H), 7.54–7.44 (m, 2H), 7.41 (d, J = 7.7 Hz, 1H), 7.23 (d, J = 7.4 Hz, 2H), 7.18–7.07 (m, 3H), 6.77 (d, J = 8.2 Hz, 1H), 6.71 (t, J = 7.5 Hz, 1H), 4.80 (s, 1H), 4.76 (d, J = 12.1 Hz, 1H), 4.30 (d, J = 10.7 Hz, 1H), 4.02 (d, J = 12.6 Hz, 1H), 3.80–3.65 (m, 2H), 3.25 (d, J = 12.5 Hz, 1H), 2.94 (t, J = 8.7 Hz, 1H), 2.51 (dt, J = 11.5, 5.9 Hz, 1H), 1.89–1.72 (m, 2H), 1.24 (dt, J = 13.6, 6.8 Hz, 2H), 0.94 (dt, J = 21.1, 7.7 Hz, 1H), 0.67 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 179.70, 176.53, 173.06, 164.07, 143.34, 134.19, 133.94, 133.72, 133.47, 133.23, 133.08, 131.36, 131.22, 130.96, 130.45, 129.86, 129.42, 128.98, 128.76, 128.69, 127.37, 126.94, 126.86, 126.59, 126.02, 125.80, 125.42, 122.98, 122.94, 120.40, 114.48, 72.69, 70.32, 63.60, 62.39, 57.23, 43.09, 39.99, 30.34, 22.94, 13.28. HRMS (m/z): calcd. C43H38N4NaNiO5+for 771.2088, found 771.2088 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.7 min, tminor = 12.3 min, de > 99%.
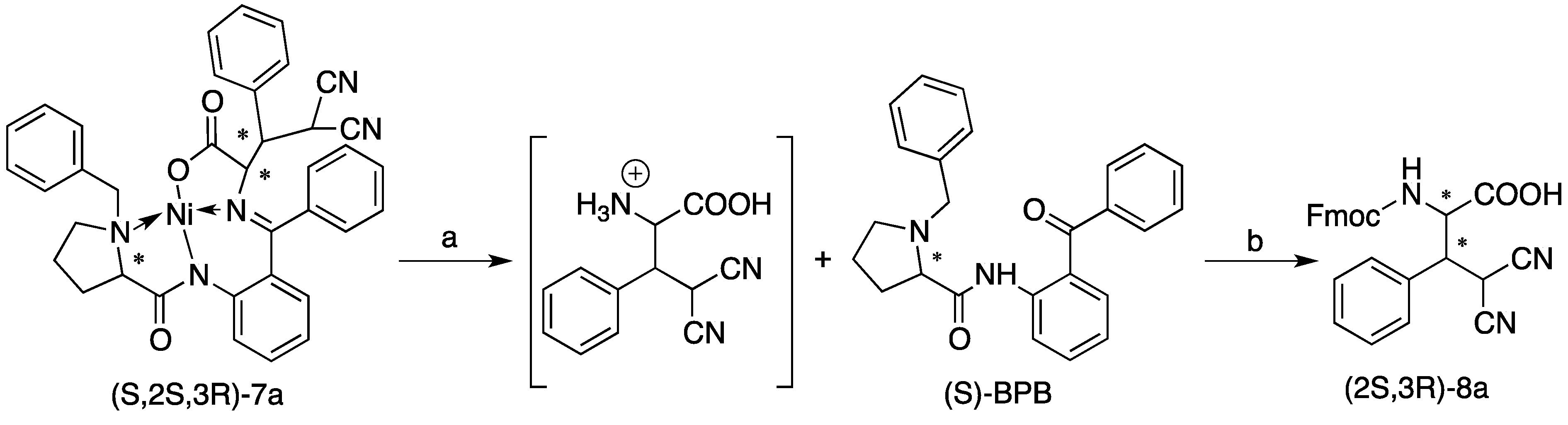
= +1793 (ca. 0.03 g/100 mL, CH2Cl2). 1H-NMR (CDCl3) δ 8.25 (d, J = 8.7 Hz, 1H), 8.00 (dd, J = 16.3, 8.0 Hz, 2H), 7.90 (d, J = 7.5 Hz, 2H), 7.71 (dd, J = 16.1, 8.1 Hz, 3H), 7.66–7.56 (m, 3H), 7.54–7.44 (m, 2H), 7.41 (d, J = 7.7 Hz, 1H), 7.23 (d, J = 7.4 Hz, 2H), 7.18–7.07 (m, 3H), 6.77 (d, J = 8.2 Hz, 1H), 6.71 (t, J = 7.5 Hz, 1H), 4.80 (s, 1H), 4.76 (d, J = 12.1 Hz, 1H), 4.30 (d, J = 10.7 Hz, 1H), 4.02 (d, J = 12.6 Hz, 1H), 3.80–3.65 (m, 2H), 3.25 (d, J = 12.5 Hz, 1H), 2.94 (t, J = 8.7 Hz, 1H), 2.51 (dt, J = 11.5, 5.9 Hz, 1H), 1.89–1.72 (m, 2H), 1.24 (dt, J = 13.6, 6.8 Hz, 2H), 0.94 (dt, J = 21.1, 7.7 Hz, 1H), 0.67 (t, J = 7.1 Hz, 3H). 13C-NMR (CDCl3) δ 179.70, 176.53, 173.06, 164.07, 143.34, 134.19, 133.94, 133.72, 133.47, 133.23, 133.08, 131.36, 131.22, 130.96, 130.45, 129.86, 129.42, 128.98, 128.76, 128.69, 127.37, 126.94, 126.86, 126.59, 126.02, 125.80, 125.42, 122.98, 122.94, 120.40, 114.48, 72.69, 70.32, 63.60, 62.39, 57.23, 43.09, 39.99, 30.34, 22.94, 13.28. HRMS (m/z): calcd. C43H38N4NaNiO5+for 771.2088, found 771.2088 ([M+Na]+). HPLC (Chiralpak IA, n-hexane/i-propanol = 50/50, flow rate 1.0 mL/min, λ = 220 nm), tmajor = 31.7 min, tminor = 12.3 min, de > 99%.3.4. Procedure for the Synthesis of (2S,3R)-8a
3.5. 2-(((9H-Fluoren-9-yl)methoxy)carbonyl)-4,4-dicyano-3-phenylbutanoic Acid (8a)
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Or, Y.S.; Ying, L.; Wang, C.; Long, J.; Qui, Y.L. Preparation of Substituted Pyrrolidine Derivatives for Use as Anti-Infective Agents. WO 2009003009, 31 December 2008. [Google Scholar]
- Edwards, D.L.; Berens, M.E.; Beaudry, C. Identification of Chemical Useful for Treating e.g., Cancer, Comprising Depositing Cancer Cells on Substrate Using Guided Cell Sedimentation, Treating Cancer Cells with Library of Chemicals, and Measuring Cell Migration Rate. WO 2003102153, 11 December 2003. [Google Scholar]
- Jones, D.T.; Harris, A.L. Identification of novel small-molecule inhibitors of hypoxia-inducible factor-1 transactivation and DNA binding. Mol. Cancer Ther. 2006, 5, 2193–2202. [Google Scholar] [CrossRef]
- Dandia, A.; Jain, A.K.; Laxkar, A.K.; Bhati, D.S. Synthesis and stereochemical investigation of highly functionalized novel dispirobisoxindole derivatives via [3+2] cycloaddition reaction in ionic liquid. Tetrahedron 2013, 69, 2062–2069. [Google Scholar] [CrossRef]
- Erdbrink, H.; Peuser, I.; Gerling, U.I.M.; Lentz, D.; Koksch, B.; Czekelius, C. Conjugate hydrotrifluoromethylation of α,β-unsaturated acyl-oxazolidinones: Synthesis of chiral fluorinated amino acids. Org. Biomol. Chem. 2012, 10, 8583–8586. [Google Scholar] [CrossRef]
- Aouadi, K.; Jeanneau, E.; Msaddek, M.; Praly, J.P. 1,3-Dipolar cycloaddition of a chiral nitrone to (E)-1,4-dichloro-2-butene: A new efficient synthesis of (2S,3S,4R)-4-hydroxyisoleucine. Tetrahedron Lett. 2012, 53, 1817–1821. [Google Scholar]
- Tong, B.M.K.; Chiba, S. Diamine-catalyzed conjugate addition to acrylate derivatives. Org. Lett. 2011, 13, 2948–2951. [Google Scholar]
- Dugave, C.; Cluzeau, J.; Menez, A.; Gaudry, M.; Marquet, A. Chemo-enzymic synthesis of protected cyano derivatives of glutamate. Tetrahedron Lett. 1998, 39, 5775–5778. [Google Scholar] [CrossRef]
- Heimgartner, H. 3-Amino-2H-azirines. Synthons for α,α-disubstituted α-amino acids in heterocycle and peptide synthesis. Angew. Chem. Int. Ed. 1991, 30, 238–264. [Google Scholar] [CrossRef]
- Duthaler, R.O. Recent developments in the stereoselective synthesis of α-amino acids. Tetrahedron 1994, 50, 1539–1650. [Google Scholar] [CrossRef]
- Wirth, T. New strategies to alpha-alkylated alpha-amino acids. Angew. Chem. Int. Ed. 1997, 36, 225–227. [Google Scholar] [CrossRef]
- Gibson, S.E.; Guillo, N.; Tozer, M.J. Towards control of chi-space: Conformationally constrained analogues of Phe, Tyr, Trp and His. Tetrahedron 1999, 55, 585–615. [Google Scholar] [CrossRef]
- Cativiela, C.; Diaz-de-Villegas, M.D. Stereoselective synthesis of quaternary alpha-amino acids. Part 1: Acyclic compounds. Tetrahedron Asymmetry 1998, 9, 3517–3599. [Google Scholar] [CrossRef]
- Cativiela, C.; Diaz-de-Villegas, M.D. Stereoselective synthesis of quaternary alpha-amino acids. Part 2. Cyclic compounds. Tetrahedron Asymmetry 2000, 11, 645–732. [Google Scholar] [CrossRef]
- Flores-Conde, M.I.; Reyes, L.; Herrera, R.; Rios, H.; Vazquez, M.A.; Miranda, R.; Tamariz, J.; Delgado, F. Highly regio- and stereoselective diels-alder cycloadditions via two-step and multicomponent reactions promoted by infrared irradiation under solvent-free conditions. Int. J. Mol. Sci. 2012, 13, 2590–2617. [Google Scholar] [CrossRef]
- Belokon, Y.N. Chiral complexes of Ni(II), Cu(II), and Cu(I) as reagents, catalysts and receptors for asymmetric-synthesis and chiral recognition of amino-acids. Pure Appl. Chem. 1992, 64, 1917–1924. [Google Scholar] [CrossRef]
- Belokon, Y.N.; Bespalova, N.B.; Churkina, T.D.; Cisarova, I.; Ezernitskaya, M.G.; Harutyunyan, S.R.; Hrdina, R.; Kagan, H.B.; Kocovsky, P.; Kochetkov, K.A.; et al. Synthesis of alpha-amino acids via asymmetric phase transfer-catalyzed alkylation of achiral nickel(II) complexes of glycine-derived Schiff bases. J. Am. Chem. Soc. 2003, 125, 12860–12871. [Google Scholar]
- Belokon, Y.N.; Tararov, V.I.; Maleev, V.I.; Saveleva, T.F.; Ryzhov, M.G. Improved procedures for the synthesis of (S)-2-N-(N'-benzylprolyl)amino benzophenone (BPB) and Ni(II) complexes of Schiff’s bases derived from BPB and amino acids. Tetrahedron Asymmetry 1998, 9, 4249–4252. [Google Scholar] [CrossRef]
- Hayashi, T.; Kishi, E.; Soloshonok, V.A.; Uozumi, Y. Erythro-selective aldol-type reaction of N-sulfonylaldimines with methyl isocyanoacetate catalyzed by gold(I). Tetrahedron Lett. 1996, 37, 4969–4972. [Google Scholar] [CrossRef]
- Soloshonok, V.A.; Fokina, N.A.; Rybakova, A.V.; Shishkina, I.P.; Galushko, S.V.; Sorochinsky, A.E.; Kukhar, V.P.; Savchenko, M.V.; Svedas, V.K. Biocatalytic approach to enantiomerically pure beta-amino acids. Tetrahedron Asymmetry 1995, 6, 1601–1610. [Google Scholar] [CrossRef]
- Soloshonok, V.A.; Soloshonok, I.V.; Kukhar, V.P.; Svedas, V.K. Biomimetic transamination of alpha-alkyl beta-keto carboxylic esters. Chemoenzymatic approach to the stereochemically defined alpha-alkyl beta-fluoroalkyl beta-amino acids. J. Org. Chem. 1998, 63, 1878–1884. [Google Scholar] [CrossRef]
- Deng, G.H.; Wang, J.; Zhou, Y.; Jiang, H.L.; Liu, H. One-pot, large-scale synthesis of Nickel(II) complexes derived from 2-N-(alpha-picolyl)amino benzophenone (PABP) and α- or β-amino acids. J. Org. Chem. 2007, 72, 8932–8934. [Google Scholar] [CrossRef]
- Lin, D.Z.; Deng, G.H.; Wang, J.; Ding, X.; Jiang, H.L.; Liu, H. Efficient Synthesis of Symmetrical alpha,alpha-Disubstituted beta-Amino Acids and alpha,alpha-Disubstituted Aldehydes via Dialkylation of Nucleophilic beta-Alanine Equivalent. J. Org. Chem. 2010, 75, 1717–1722. [Google Scholar] [CrossRef]
- Wang, J.; Lin, D.Z.; Shi, J.M.; Ding, X.; Zhang, L.; Jiang, H.L.; Liu, H. Highly enantio- and diastereoselective mannich reaction of a chiral Nickel(II) glycinate with an alpha-imino ester for asymmetric synthesis of a 3-aminoaspartate. Synthesis 2010, 7, 1205–1208. [Google Scholar]
- Wang, J.; Zhou, S.B.; Lin, D.Z.; Ding, X.; Jiang, H.L.; Liu, H. Highly diastereo- and enantioselective synthesis of syn-beta-substituted tryptophans via asymmetric Michael addition of a chiral equivalent of nucleophilic glycine and sulfonylindoles. Chem. Commun. 2011, 47, 8355–8257. [Google Scholar]
- Wang, J.; Ji, X.; Shi, J.M.; Sun, H.F.; Jiang, H.L.; Liu, H. Diastereoselective Michael reaction of chiral nickel(II) glycinate with nitroalkenes for asymmetric synthesis of beta-substituted alpha,gamma-diaminobutyric acid derivatives in water. Amino Acids 2012, 42, 1685–1694. [Google Scholar] [CrossRef]
- Wang, J.; Liu, H.; Acena, J.L.; Houck, D.; Takeda, R.; Moriwaki, H.; Sato, T.; Soloshonok, V.A. Synthesis of bis-α,α'-amino acids through diastereoselective bis-alkylations of chiral Ni(II)-complexes of glycine. Org. Biomol. Chem. 2013, 11, 4508–4515. [Google Scholar] [CrossRef]
- Smith, D.J.; Yap, G.P.A.; Kelley, J.A.; Schneider, J.P. Enhanced stereoselectivity of a Cu(II) complex chiral auxiliary in the synthesis of Fmoc-L-γ-carboxyglutamic acid. J. Org. Chem. 2011, 76, 1513–1520. [Google Scholar] [CrossRef]
- Xie, X.; Peng, C.; He, G.; Leng, H.-J.; Wang, B.; Huang, W.; Han, B. Asymmetric synthesis of a structurally and stereochemically complex spirooxindole pyran scaffold through an organocatalytic multicomponent cascade reaction. Chem. Commun. 2012, 48, 10487–10489. [Google Scholar]
- He, J.; Ouyang, G.; Yuan, Z.; Tong, R.; Shi, J.; Ouyang, L. A facile synthesis of functionalized dispirooxindole derivatives via a three-component 1,3-dipolar cycloaddition reaction. Molecules 2013, 18, 5142–5154. [Google Scholar] [CrossRef]
- Hou, X.; Luo, H.; Zhong, H.; Wu, F.; Zhou, M.; Zhang, W.; Han, X.; Yan, G.; Zhang, M.; Lu, L.; et al. Analysis of furo 3,2-c tetrahydroquinoline and pyrano 3,2-c tetrahydroquinoline derivatives as antitumor agents and their metabolites by liquid chromatography/electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectr. 2013, 27, 1222–1230. [Google Scholar] [CrossRef]
- Li, X.; Yang, L.; Peng, C.; Xie, X.; Leng, H.-J.; Wang, B.; Tang, Z.-W.; He, G.; Ouyang, L.; Huang, W.; et al. Organocatalytic tandem Morita-Baylis-Hillman-Michael reaction for asymmetric synthesis of a drug-like oxa-spirocyclic indanone scaffold. Chem. Commun. 2013, 49, 8692–8694. [Google Scholar] [CrossRef]
- Wu, G.; Ouyang, L.; Liu, J.; Zeng, S.; Huang, W.; Han, B.; Wu, F.; He, G.; Xiang, M. Synthesis of novel spirooxindolo-pyrrolidines, pyrrolizidines, and pyrrolothiazoles via a regioselective three-component 3+2 cycloaddition and their preliminary antimicrobial evaluation. Mol. Diver. 2013, 17, 271–283. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 7a–y and 8a are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhou, R.; Guo, L.; Peng, C.; He, G.; Ouyang, L.; Huang, W. Diastereoselective Three-Component Reactions of Chiral Nickel(II) Glycinate for Convenient Synthesis of Novel α-Amino-β-Substituted-γ,γ-Disubstituted Butyric Acids. Molecules 2014, 19, 826-845. https://doi.org/10.3390/molecules19010826
Zhou R, Guo L, Peng C, He G, Ouyang L, Huang W. Diastereoselective Three-Component Reactions of Chiral Nickel(II) Glycinate for Convenient Synthesis of Novel α-Amino-β-Substituted-γ,γ-Disubstituted Butyric Acids. Molecules. 2014; 19(1):826-845. https://doi.org/10.3390/molecules19010826
Chicago/Turabian StyleZhou, Rui, Li Guo, Cheng Peng, Gu He, Liang Ouyang, and Wei Huang. 2014. "Diastereoselective Three-Component Reactions of Chiral Nickel(II) Glycinate for Convenient Synthesis of Novel α-Amino-β-Substituted-γ,γ-Disubstituted Butyric Acids" Molecules 19, no. 1: 826-845. https://doi.org/10.3390/molecules19010826






