Structure and Antioxidant Activity of Polyphenols Derived from Propolis
Abstract
:1. Introduction
2. Propolis Composition
2.1. Phenolic Acids
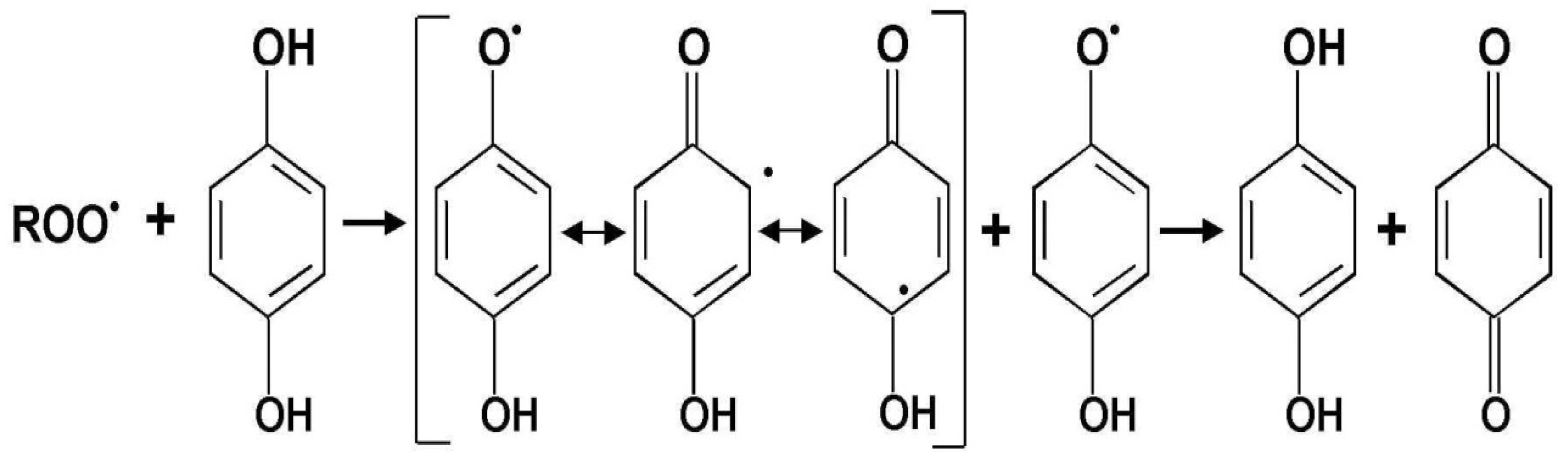
2.2. Flavonoids
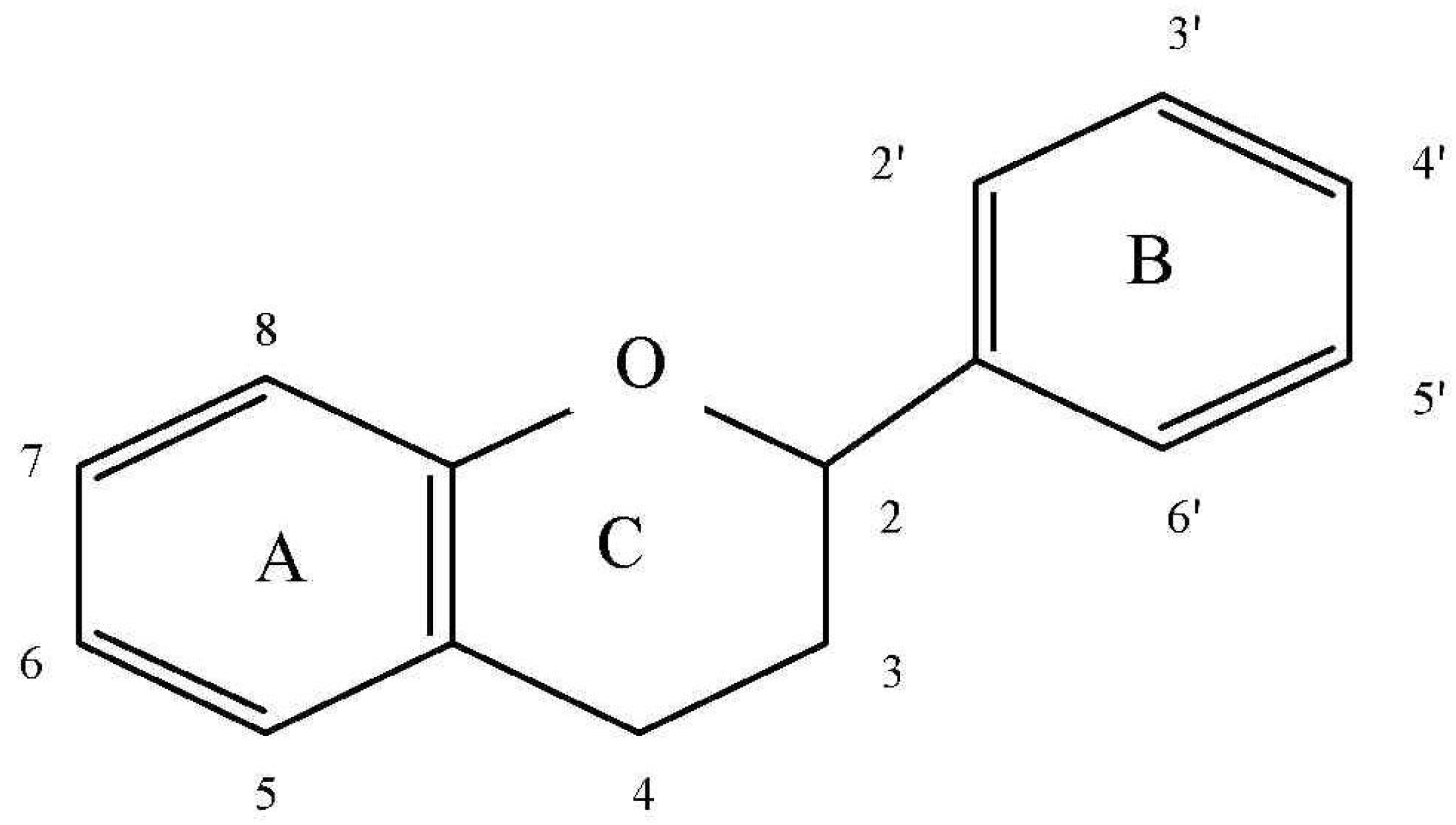
- the ortho-dihydroxy (catechol) group in B ring, which displays significant ability to “scavenge” oxygen (ROS) and nitrogen radicals (RNS) and ensures the high stability of the created phenoxyl radical. Hydroxyl groups in B ring are donors of electrons and nitrogen for radicals.
- the double bond between C-2 and C-3 carbon and the presence of the 4-oxo group in the C ring is the reason for the dislocation of an electron in the B ring. The antioxidative properties result from the dislocation of an electron of the aromatic unit. During the reaction of the compounds with free radicals, phenoxyl radicals are created and stabilized by the effect of the aromatic ring resonance.
- hydroxyl groups near C-3 and C-5 carbon in the presence of 4-oxo groups in the A and C ring generate the maximum free radical scavenging effects.
3. The Biological Properties of Polyphenols, Including Their Antioxidative Activity
- inhibiting the activity of enzymes and thus inhibiting the appearance of reactive forms of oxygen (ROS)
- chelating ions of metals involved in the process of free radical creation
- scavenging reactive forms of oxygen (ROS), thus interrupting the cascade of reactions leading to the peroxidation of lipids
- synergistic action with other antioxidants
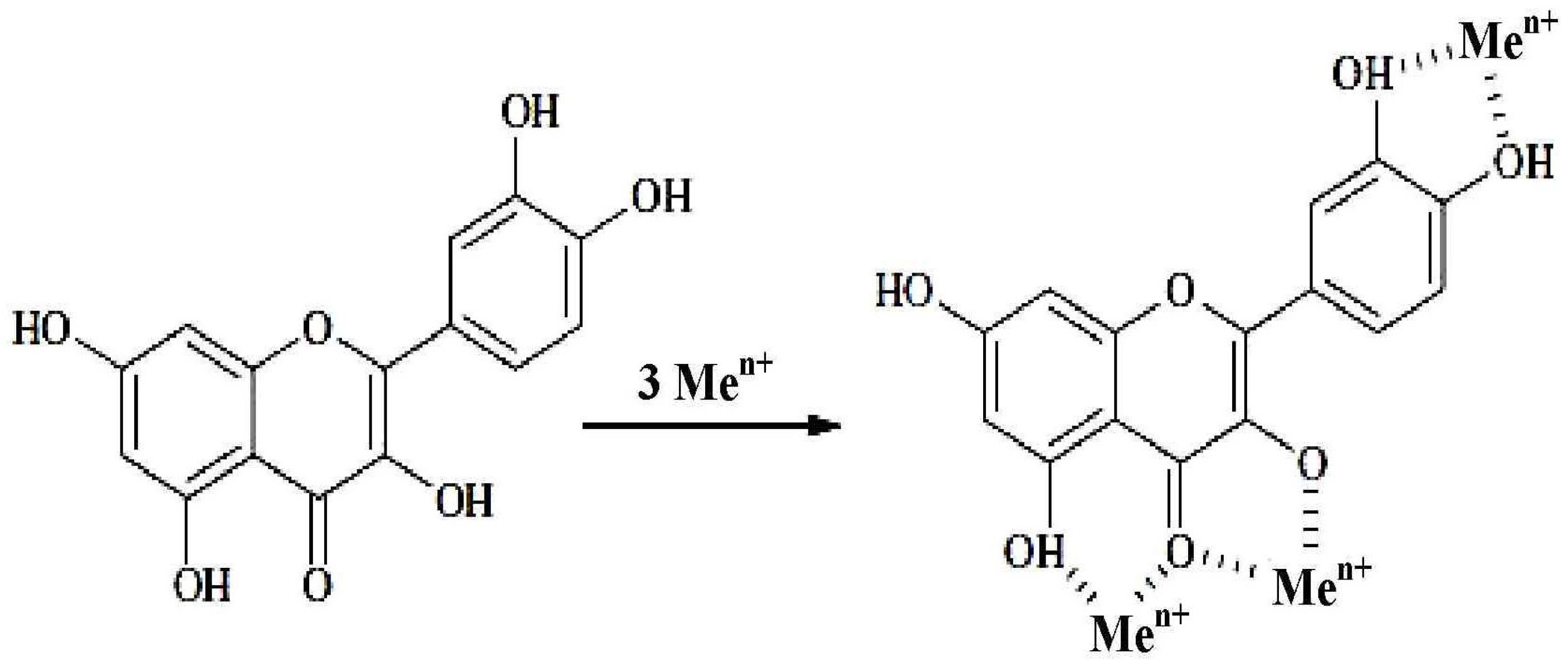
- initiation, as a result of the inactivation of the radical initiating the process (hydroxyl radical):
- propagation due to the inactivation of lipid superoxide radical (LOO●):
- termination as a result of the inactivation of lipid peroxide radical (LOO●), lipid radical (L●) and alkoxy radical (LO●), appearing as a result of lipid peroxidation by metal ions:
- FL-OH—flavonoids
- FL-O●—phenyloxyl radical [47]
3.1. Antibacterial Activity
3.2. Anti-Inflammatory Activity
3.3. Anticarcinogenic Effects
3.4. Antiatherogenic Effects
3.5. Effects on the Cardiovascular System
3.6. Estrogenic Effects
3.7. Antidiabetic Effects
3.8. Anti-HIV Activity
3.9. Reparative-Regenerative Effects
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Kędzia, B. Chemical composition of polish propolis. Part I. The initial period of investigations. Post. Fitoter. 2009, 1, 39–44. [Google Scholar]
- Kędzia, B. Chemical composition of polish propolis. Part II. New Studies. Post. Fitoter. 2009, 2, 122–128. [Google Scholar]
- Olczyk, P.; Komosińska-Vassev, K.; Olczyk, K. Propolis-chemical composition, properties and application. Farm. Pol. 2007, 63, 1102–1107. [Google Scholar]
- Kędzia, B.; Hołderna-Kędzia, E. Chemical composition of propolis in nowadays researches. Herba Pol. 1991, 38, 95–107. [Google Scholar]
- Khalil, M.L. Biological activity of bee propolis in health and disease. Asian Pac. J. Cancer Prev. 2006, 7, 22–31. [Google Scholar]
- De Castro, S.L. Propolis: Biological and pharmacological activities. Ann. Rev. Biomed. Sci. 2001, 3, 49–83. [Google Scholar]
- Hegazi, A.G.; Abd El Hady, F.K.; Abd Allah, F.A. Chemical composition and antimicrobial activity of European propolis. Z. Naturforsch. C 2000, 55, 70–75. [Google Scholar]
- Bankova, V. Recent trends and important developments in propolis research. Evid. Based. Complement. Alternat. Med. 2005, 2, 29–32. [Google Scholar] [CrossRef]
- Warakomska, Z.; Maciejewicz, W. Microscopic analysis of propolis from Polish regions. Apidologie 1992, 23, 277–283. [Google Scholar]
- Bankova, V.; de Castro, S.L.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef]
- Geckil, H.; Ates, B.; Durmaz, G.; Erdogan, S.; Yilmaz, I. Antioxidant, Free Radical Scavenging and Metal chelating characteristics of propolis. Am. J. Biochem. Biotech. 2005, 1, 27–31. [Google Scholar] [CrossRef]
- Popova, M.; Bankova, V.; Butovska, D.; Petkov, V.; Damyanova, B.; Sabatini, A.G.; Marcazzan, G.L.; Bogdanov, S. Poplar type propolis and analysis of its biologically active components. Honeybee Sci. 2003, 24, 61–66. [Google Scholar]
- Kędzia, B.; Hołderna-Kędzia, E. The bee products in the nutrition and supplementation. Post. Fitoter. 2006, 4, 213–221. [Google Scholar]
- Scheller, S.; Wilczok, T.; Imielski, S.; Król, W.; Gabryś, J.; Shani, J. Free radical scavenging by ethanol extract of propolis. Int. J. Radiat. Biol. 1990, 57, 461–465. [Google Scholar] [CrossRef]
- Volpi, N. Separation of flavonoids and phenolic acids from propolis by capillary zone electrophoresis. Electrophoresis 2004, 25, 1872–1878. [Google Scholar]
- Maciejewicz, W.; Daniewski, M.; Dzido, T.H.; Bal, K. GC-MS and HPLC analysis of phenolic acids extracted from propolis and from Populus Nigra bud exudate. Chem. Anal. 2002, 47, 25–29. [Google Scholar]
- Dobrowolski, J.W.; Vohora, S.B.; Sharma, K.; Shah, S.A.; Naqvi, S.A.; Dandiya, P.C. Antibacterial, antifungal, antiamoebic, antiinflammatory, antipyretic studies on propolis bee products. J. Ethnopharmacol. 1991, 35, 77–82. [Google Scholar] [CrossRef]
- Sahinler, N.; Kaftanoglu, O. Natural product propolis: Chemical composition. Nat. Prod. Res. 2005, 19, 183–188. [Google Scholar]
- Maciejewicz, W. Isolation of flavonoid aglycones from propolis by a column chromatography method and their identification by GC-MS and TLC methods. J. Liq. Chrom. Rel. Tech. 2001, 24, 1171–1179. [Google Scholar] [CrossRef]
- Ellnain-Wojtaszek, M.; Hładoń, B.; Walkowiak, A.; Kowalewski, Z. Chemical composition of bees products and their application in therapy. Farm. Pol. 1992, 48, 1–9. [Google Scholar]
- Maciejewicz, W.; Daniewski, M.; Mielniczuk, Z. Gas chromatography-mass spectrometry investigation of propolis. Analysis of β-steroids. Acta Polon. Pharm. 1983, 40, 251–253. [Google Scholar]
- Erenia, N.; Tatiana, D. The content of micro- and macroelements in propolis. Bull. USAMV-CN. Anim. Sci. Biotechnol. 2007, 63/64, 176–178. [Google Scholar]
- Leja, M.; Mareczek, G.; Wyzgolik, G.; Klepacz-Baniak, J.; Czekońska, K. Antioxidative properties of bee pollen in selected plant species. Food Chem. 2007, 100, 237–240. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acid. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Budryn, G.; Nebesny, E. Structure and antioxidant properties of coffee bean polyphenols. Bromat. Chem. Toksykol. 2005, 3, 203–209. [Google Scholar]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidative activity and phenolic composition of pilot-plant and commercial extracts of sage and rosemary. J. Am. Oil. Chem. Soc. 1996, 73, 645–652. [Google Scholar] [CrossRef]
- Budryn, G.; Nebesny, E. Phenolic acids—their properties, Occurrence in plant materials, Absorption and metabolism. Bromat. Chem. Toksykol. 2006, 2, 103–110. [Google Scholar]
- Silva, F.A.; Borges, F.; Guimaraes, C.; Lima, J.L.; Matos, C.; Reis, S. Phenolic acids and derivatives studies on the relationship among structure, radical scavenging activity and physicochemical parameters. J. Agric. Food Chem. 2000, 48, 2122–2126. [Google Scholar] [CrossRef]
- Siquet, C.; Paiva-Martins, F.; Lima, J.L.; Reis, S.; Borges, F. Antioxidant profile of dihydroxy- and trihydroxyphenolic acids-a structure-activity relationship study. Free Radic. Res. 2006, 40, 433–442. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, E.; Berset, C.M. Use of free radical method to evaluate antioxidant activity. Lebensm-Wiss. U. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid). Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef]
- Makarska, E.; Michalak, M. Antioxidant activity of phenolic acids of spring barley. Annal. UMCS. Sec. E 2005, 60, 263–269. [Google Scholar]
- Gawlik-Dziki, U. The effect of cooking on composition and antioxidant activity of phenolic acids isolated from broccoli. Annal. UMCS. Sec. E 2004, 59, 1567–1575. [Google Scholar]
- Cos, P.; Rajan, P.; Vedernikova, I.; Calomme, M.; Pieters, L.; Vlietinck, A.J.; Augustyns, K.; Haemers, A.; Vanden Berghe, D. In vitro antioxidant profile of phenolic acid derivatives. Free Radic. Res. 2002, 36, 711–716. [Google Scholar] [CrossRef]
- Makowska-Wąs, J.; Janeczko, Z. Bioavailability of plant polyphenols. Post. Fitoter. 2004, 3, 126–137. [Google Scholar]
- Olszewska, M. Flavonoids and their use in therapy. Farm. Pol. 2003, 59, 391–402. [Google Scholar]
- Kohlmünzer, S. Farmakognozja, 5th ed.; PWN: Warszawa, Poland, 2003; pp. 150–156. [Google Scholar]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Lamer-Zarawska, E.; Kowal-Gierczyk, B.; Niedworok, J. Fitoterapia i leki roślinne, 1st ed.; PZWL: Warszawa, Poland, 2007; pp. 64–71. [Google Scholar]
- Czeczot, H. Flawonoidy—naturalne antyoksydanty w naszej diecie. [in Polish]. Żyw. Człow. Metab. 2000, 27, 372–382. [Google Scholar]
- Matławska, I. Farmakognozja, 1st ed.; Wydawnictwo Naukowe AM: Poznań, Poland, 2005; pp. 110–119. [Google Scholar]
- Majewska, M.; Czeczot, H. Flavonoids in prevention and therapy of diseases. Farm. Pol. 2009, 65, 369–377. [Google Scholar]
- Tapas, A.R.; Sakarkar, D.M.; Kakde, R.B. Flavonoids as nutraceuticals: A review. Trop. J. Pharm. Res. 2008, 7, 1089–1099. [Google Scholar]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar]
- Silva, M.; Santos, M.; Caroco, G.; Rocha, R.; Justino, G.; Mira, L. Stucture-antioxidant activity relationships of flavonoids: A re-examination. Free Radic. Res. 2002, 36, 1219–1227. [Google Scholar]
- Ostrowska, J.; Skrzydlewska, E. The biological activity of flavonoids. Post. Fitoter. 2005, 3–4, 71–79. [Google Scholar]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar]
- Dugas, A.J.; Castaneda-Acosta, J.; Bonin, G.C.; Price, K.L.; Fischer, N.H.; Winston, G.W. Evaluation of the total peroxyl radical-scavenging capacity of flavonoids: Structure-activity relationships. J. Nat. Prod. 2000, 63, 327–331. [Google Scholar]
- Rice-Evans, C. Flavonoids and isoflavones: Absorption, Metabolism, and bioactivity. Free Radic. Biol. Med. 2004, 36, 827–828. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomas-Barberan, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in Food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Mihai, C.M.; Marghitas, L.A. Antioxidant capacity of Transylvanian propolis. Anim. Sci. Biotech. 2010, 67, 266–270. [Google Scholar]
- Mihai, C.M.; Marghitas, L.A.; Dezmiren, D.S.; Barnutiu, L. Correlation between polyphenolic profile and antioxidant activity of propolis from Transylvania. Animal Sci. Biotech. 2011, 44, 100–103. [Google Scholar]
- Kurek-Górecka, A.; Sobczak, A.; Rzepecka-Stojko, A.; Górecki, M.T.; Wardas, M.; Pawłowska-Góral, K. Antioxidant activity of ethanolic fraction of Polish propolis. Z. Naturforsch. C 2012, 67, 545–550. [Google Scholar] [CrossRef]
- Olczyk, P.; Ramos, P.; Bernaś, M.; Komosińska-Vassev, K.; Stojko, J.; Pilawa, B. Application of electron paramagnetic resonance spectroscopy to comparative examination of different groups of free radicals in thermal injuries treated with propolis and silver sulphadiazine. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Wojtyczka, R.; Kubina, R.; Kabała-Dzik, A.; Bułdak, R. Antibacterial activity of ethanol extract of propolis. Ann. Acad. Med. Siles. 2012, 66, 39–48. [Google Scholar]
- Wojtyczka, R.; Kępa, M.; Idzik, D.; Kubina, R.; Kabała-Dzik, A.; Dziedzic, A.; Wąsik, T. In vitro antimicrobial activity of ethanolic extract of polish propolis against biofilm forming staphylococcus epidermidis strains. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Scheller, S.; Dworniczak, S.; Waldemar-Klimek, K.; Rajca, M.; Tomczyk, A.; Shani, J. Synergism between ethanolic extract of Propolis (EEP) and antituberculosis drugs on growth of mycobacteria. Z. Naturforsch. C 1999, 54, 549–553. [Google Scholar]
- Król, W.; Scheller, S.; Shani, J.; Pietsz, G.; Czuba, Z. Synergistic effect of ethanolic extract of propolis and antibiotics on the growth of Staphylococcus aureus. Arzneim-Forsch. 1993, 43, 607–609. [Google Scholar]
- Marcucci, M.C. Propolis: Chemical composition, Biological properties and therapeutic activity. Apidologie 1995, 26, 83–99. [Google Scholar] [CrossRef]
- Kabała-Dzik, A.; Szaflarska-Stojko, E.; Wojtyczka, R.; Stojko, A.; Stojko, R.; Pacha, J. Comparative studies on the antimibrobial activity of propolis balm and silver sulphadiazine applied to burn wounds in pigs. Bull. Vet. Inst. Pulawy. 2003, 47, 541–545. [Google Scholar]
- Wojtyczka, R.; Dziedzic, A.; Idzik, D.; Kępa, M.; Kubina, R.; Kabała-Dzik, A.; Smoleń-Dzirba, J.; Stojko, A.; Sajewicz, M.; Wąsik, T. Susceptibility of staphylococcus aureus clinical isolates to propolis extract alone or in combination with antimicrobial drugs. Molecules 2013, 18, 9623–9640. [Google Scholar] [CrossRef]
- Stojko, J.; Juszko-Piekut, M.; Rzepecka-Stojko, A.; Stojko, R.; Moździerz, A.; Olczyk, D.; Romaniuk, D.; Kasprzak, M.; Morawiec, T. Application of the preparation sepropol—in the bedsore prophylaxis and treatment. Pol. J. Environ. Stud. 2007, 16, 609–611. [Google Scholar]
- Bogdanov, S. Propolis: Composition, health, medicine: A review. Available online: http://www.bee-hexagon.net/en/propolis.htm (accessed on 5 September 2013).
- Rosa, G.M.; Mei, R.; DiCarlo, G.; Pacilio, M.; di Carlo, R. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 expression by flavonoids in macrophage J774A.1. Life Sci. 2001, 68, 921–931. [Google Scholar] [CrossRef]
- Mutoh, M.; Takashi, M.; Fukuda, K.; Komatsu, H.; Enya, T.; Masushima-Hibiya, Y.; Mutoh, H.; Sugimura, T.; Wakabayashi, K. Suppression by flavonoids of cyclooxygenase-2 promoter-dependent transcriptional activity in colon cancer cells: Structure activity relationship. Jpn. J. Cancer Res. 2000, 91, 686–691. [Google Scholar] [CrossRef]
- Iravani, S.; Zolfaghari, B. Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res. Pharm. Sci. 2011, 6, 1–11. [Google Scholar]
- Borrelli, F.; Maffia, P.; Pinto, L.; Ianaro, A.; Russo, A.; Capasso, F.; Ialenti, A. Phytochemical compounds involved in the anti-inflammatory effect of propolis extract. Fitoterapia 2002, 73, 53–63. [Google Scholar] [CrossRef]
- Rossi, A.; Ligresti, A.; Longo, R.; Russo, A.; Borrelli, F.; Sautebin, L. The inhibitory effect of propolis and caffeic acid phenethyl ester on cyclooxygenase activity in J774 macrophages. Phytomedicine 2002, 9, 530–535. [Google Scholar] [CrossRef]
- Mirzoeva, O.K.; Calder, P.C. The effect of propolis and its components on eicosanoid production during the inflammatory response. Prostaglandins Leucot. Essent. Fatty Acids 1996, 55, 441–449. [Google Scholar] [CrossRef]
- Błońska, M.; Bronikowska, J.; Pietsz, G.; Czuba, Z.P.; Scheller, S.; Krol, W. Effects of ethanol extract of propolis (EEP) and its flavones on inducible gene expression in J774A.1 macrophages. J. Ethnopharmacol. 2004, 91, 25–30. [Google Scholar]
- Song, Y.S.; Park, E.H.; Hur, G.M.; Ryu, Y.S.; Kim, Y.M.; Jin, C. Ethanol extract of propolis inhibits nitric oxide synthase gene expression and enzyme activity. J. Ethnopharmacol. 2002, 80, 155–161. [Google Scholar] [CrossRef]
- De Almeida, E.C.; Menezes, H. Anti-inflammatory activity of propolis extracts: A review. J. Venom. Anim. Toxins. 2002, 8, 191–212. [Google Scholar]
- Massaro, F.C.; Brooks, P.R.; Wallace, H.M.; Russel, F.D. Cerumen of Australian stingless bees (Tetragonula carbonaria): Gas chromatography-mass spectrometry fingerprints and potential anti-inflammatory properties. Naturwissenschaften 2011, 98, 329–337. [Google Scholar] [CrossRef]
- Reis, C.M.F.; Carvalho, J.C.T.; Caputo, L.R.G.; Patrício, K.C.M. Anti-inflammatory, anti-ulcer and subchronic toxicity ethanol extract of propolis. Rev. Bras. Farmacogn. 2000, 9, 43–52. [Google Scholar]
- Park, E.H.; Kahng, J.H. Suppressive effects of propolis in rat adjuvant arthritis. Arch. Pharm. Res. 1999, 22, 554–558. [Google Scholar] [CrossRef]
- Orsi, R.O.; Funari, S.R.C.; Soares, A.M.V.C.; Calvi, S.A.; Oliveira, S.L.; Sforcin, J.M.; Bankova, V. Immunomodulatory action of propolis on macrophage activation. J. Venom. Anim. Toxins 2000, 6, 205–219. [Google Scholar] [CrossRef]
- Moura, S.A.L.; Negri, G.; Salatino, A.; Lima, L.D.C.; Dourado, L.P.A.; Mendes, J.B.; Andrade, S.P.; Ferreira, M.A.N.D.; Cara, D.C. Aqueous extract of Brazilian green propolis: Primary components, evaluation of inflammation and wound healing by using subcutaneous implanted sponges. Evid. Based Complement. Alternat. Med. 2011, 2011. [Google Scholar] [CrossRef]
- Ahmed, K.M.; Saleh, E.M.; Sayed, E.M.; Shalaby, K.A.F. Anti-Inflammatory effect of different propolis extracts in Thioacetamide-induced hepatotoxicity in male tat. Austral. J. Basic. Appl. Sci. 2012, 6, 29–40. [Google Scholar]
- Díaz-Carballo, D.; Malak, S.; Bardenheuer, W.; Freistuehler, M.; Reusch, H.P. The contribution of plukenetione A to the anti-tumoral activity of Cuban propolis. Bioorg. Med. Chem. 2008, 16, 9635–9643. [Google Scholar] [CrossRef]
- Benkovic, V.; Knezevic, A.H.; Dikic, D.; Lisicic, D.; Orsolic, N.; Basic, I.; Kosalec, I.; Kopjar, N. Radioprotective effects of propolis and quercetin in gamma-irradiated mice evaluated by the alkaline comet assay. Phytomedicine 2008, 15, 851–858. [Google Scholar] [CrossRef]
- Lugli, E.; Ferraresi, R.; Roat, E.; Troiano, L.; Pinti, M.; Nasi, M.; Nemes, E.; Bertoncelli, L.; Gibellini, L.; Salomoni, P.; et al. Quercetin inhibits lymphocyte activation and proliferation without inducing apoptosis in peripheral mononuclear cells. Leuk. Res. 2009, 33, 140–150. [Google Scholar] [CrossRef]
- Sánchez, Y.; Amrán, D.; Fernández, C.; de Blas, E.; Aller, P. Genistein selectively potentiates arsenic trioxide-induced apoptosis in human leukemia cells via reactive oxygen species generation and activation of reactive oxygen species-inducible protein kinases (p38-MAPK, AMPK). Int. J. Cancer 2008, 123, 1205–1214. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, X.H.; Wang, Z.J. Cytotoxicity of flavones and flavonols to a human esophageal squamous cell carcinoma cell line (KYSE-510) by induction of G2/M arrest and apoptosis. Toxicol. In Vitro 2009, 23, 797–807. [Google Scholar]
- Chan, W.S.; Wen, P.C.; Chiang, H.C. Structure-activity relationship of caffeic acid analogues on xanthine oxidase. Anticancer Res. 1995, 15, 703–707. [Google Scholar]
- Qin, W.; Zhu, W.; Shi, H.; Hewett, J.E.; Ruhlen, R.L.; MacDonald, R.S.; Rottinghaus, G.E.; Chen, Y.C.; Sauter, E.R. Soy isoflavones have an antiestrogenic effect and alter mammary promoter hypermethylation in healthy premenopausal women. Nutr. Cancer 2009, 61, 238–244. [Google Scholar] [CrossRef]
- Orsolić, N.; Benković, V.; Horvat-Knezević, A.; Kopjar, N.; Kosalec, I.; Bakmaz, M.; Mihaljević, Z.; Bendelja, K.; Basić, I. Assessment by survival analysis of the radioprotective properties of propolis and its polyphenolic compounds. Biol. Pharm. Bull. 2007, 30, 946–951. [Google Scholar] [CrossRef]
- Kimoto, T.; Arai, S.; Kohguchi, M.; Aga, M.; Nomura, Y.; Micallef, M.J.; Kurimoto, M.; Mito, K. Apoptosis and suppression of tumor growth by artepillin C extracted from Brazilian propolis. Cancer Detec. Prev. 1998, 22, 506–515. [Google Scholar]
- Slavin, J.; Marquart, L.; Jakobs, D., Jr. Consumption of whole–grain food and decreased risk of cancer: Proposed mechanisms. Cereal Foods World 2000, 45, 54–58. [Google Scholar]
- Kuźnicki, D. Antioxidants and cholesterol-reducing agents with antiatherogenic activity contained in plant raw materials. Post. Fitoter. 2006, 4, 206–212. [Google Scholar]
- Nijveldt, R.J.; Nood, E.; Hoorn, D.E.C.; Boelens, P.G.; Norren, K.; Leeuven, P.A.M. Flavonoids: A review of probable mechanisms of action and applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar]
- Li, Y.; Chen, M.; Xuan, H.; Hu, F. Effects of encapsulated propolis on blood glycemic control, lipid metabolism, and insulin resistance in type 2 diabetes mellitus rats. Evid. Based Complement. Alternat. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Fuliang, H.U.; Hepburn, H.R.; Xuan, H.; Chen, M.; Daya, S.; Radloff, S.E. Effects of propolis on blood glucose, blood lipid and free radicals in rats with diabetes mellitus. Pharmacol. Res. 2005, 51, 147–152. [Google Scholar] [CrossRef]
- Daleprane, J.B.; Freitas Vda, S.; Pacheco, A.; Rudnicki, M.; Faine, L.A.; Dörr, F.A.; Ikegaki, M.; Salazar, L.A.; Ong, T.P.; Abdalla, D.S. Anti-atherogenic and anti-angiogenic activities of polyphenols from propolis. J. Nutr. Biochem. 2012, 23, 557–566. [Google Scholar]
- De Lima, R.O.A.; Bazo, A.P.; Said, R.A. Modifying effect of propolis on dimethylhydrazine-induced DNA damage but not colonic aberrant crypt foci in rats. Environ. Mol. Mutagen. 2005, 45, 8–16. [Google Scholar]
- Lio, A.; Ohguchi, K.; Maruyama, H.; Tazawa, S.; Araki, Y.; Ichihara, K.; Nozawa, Y.; Ito, M. Ethanolic extracts of Brazilian red propolis increase ABCA1 expression and promote cholesterol efflux from THP-1 macrophages. Phytomedicine 2012, 19, 383–388. [Google Scholar] [CrossRef]
- Chen, T.G.; Lee, J.J.; Lin, K.H.; Shen, C.H.; Chou, D.S.; Sheu, J.R. Antiplatelet activity of caffeic acid phenethyl ester is mediated through a cyclic GMP-dependent pathway in human platelets. Chin. J. Physiol. 2007, 50, 121–126. [Google Scholar]
- Roos, T.U.; Heiss, E.H.; Schwaiberger, A.V.; Schachner, D.; Sroka, I.M.; Oberan, T.; Vollmar, A.M.; Dirsch, V.M. Caffeic acid phenethyl ester inhibits PDGF-induced proliferation of vascular smooth muscle cells via activation of p38 MAPK, HIF-1α, and heme oxygenase-1. J. Nat. Prod. 2011, 74, 352–356. [Google Scholar] [CrossRef]
- Packer, L.; Rimbach, G.; Virgii, F. Antioxidant activity and biologic properties of a procyanidin — rich extract from pine (Pinus maritime) bark, pycnogenol. Free Radic. Biol. Med. 1999, 27, 704–724. [Google Scholar] [CrossRef]
- Rohdewald, P. A review of the French maritime pine bark extract (Pycnogenol), a herbal medication with a diverse clinical pharmacology. Int. J. Clin. Pharmacol. Ther. 2002, 40, 158–168. [Google Scholar] [CrossRef]
- Konishi, Y. Transepithelial transport of artepillin C in intestinal Caco-2 cell monolayers. Biochim. Biophys. Acta 2005, 1713, 138–144. [Google Scholar] [CrossRef]
- Yoko, K.; Keizo, U.; Kyokok, K. Anti-hypertensive effects of propolis in spontaneosly hypertensive rats. Clin. Pharmacol. Physiol. 2004, 32, 29–30. [Google Scholar]
- Chopra, S.; Pillai, K.K.; Husain, S.Z.; Ciri, D.K. Propolis protects against doxorubicin – induced cardiopathy in rats. Exp. Mol. Pathol. 1995, 62, 190–198. [Google Scholar]
- Song, Y.S.; Jin, C.; Jung, K.J.; Park, E.H. Estrogenic effects of ethanol and ether extract of propolis. J. Ethnopharmacol. 2002, 82, 89–95. [Google Scholar]
- Jung, B.; Kim, M.; Kim, H.A.; Kim, D.; Yang, J.; Her, S.; Son, Y.S. Caffeic acid phenethyl ester, a component of beehive propolis is a novel selective estrogen receptor modulator. Phytother. Res. 2010, 24, 295–300. [Google Scholar]
- Kao, Y.C.; Zhou, C.; Sherman, M.; Laughton, C.A.; Chen, S. Molecular basis of the inhibition of human aromatase (estrogen synthetase) by flavone and isoflavone phytoestrogens: A site-directed mutagenesis study. Environ. Health Perspect. 1998, 106, 85–92. [Google Scholar]
- Adlercreutz, H.; Bannwart, C.; Wähälä, K.; Mäkelä, T.; Brunow, G.; Hase, T.; Arosemena, P.J.; Kellis, J.T., Jr.; Vickery, L.E. Inhibition of human aromatase by mammalian lignans and isoflavonoid phytoestrogens. J. Steroid Biochem. Mol. Biol. 1993, 44, 147–153. [Google Scholar]
- Jachak, S.M. Herbal drugs an antidiabetic: An overview. CRIPS 2002, 3, 9–13. [Google Scholar]
- Sanderson, J.; McLauchlan, R.W.; Williamson, G. Quercetin inhibits hydrogen peroxide-induced oxidation of the rat lens. Free Radic. Biol. Med. 1999, 26, 639–645. [Google Scholar]
- Matsui, T.; Ebuchi, S.; Fujise, T.; Abesundara, K.J.M.; Doi, S.; Yamada, H.; Matsumoto, K. Strong antihyperglycemic effects of water-soluble fraction of Brazilian propolis and its bioactive constituent, 3,4,5-tri-o-caffeoylquinic acid. Biol. Pharm. Bull. 2004, 27, 1797–1803. [Google Scholar]
- Fuliang, H.V.; Hepburn, H.R.; Xuan, H. Effects of propolis on blood glucose, blood lipids and free radical in rats with Diabetes mellitus. Pharmacol. Res. 2005, 51, 1255–1261. [Google Scholar]
- Wang, H.K.; Xia, Y.; Yang, Z.Y.; Natschke, S.L.; Lee, K.H. Recent advances in the discovery and development of flavonoids and their analogues as antitumor and anti-HIV agents. Adv. Exp. Med. Biol. 1998, 439, 191–225. [Google Scholar]
- Ng, T.B.; Huang, B.; Fong, W.P.; Yeung, H.W. Anti-human immunodeficiency virus (anti-HIV) natural products with special emphasis on HIV reverse transcriptase inhibitors. Life Sci. 1997, 61, 933–934. [Google Scholar]
- Middleton, E.J. Effect of plant flavonoids on immune and inflammatory cell function. Adv. Exp. Med. Biol. 1998, 439, 175–182. [Google Scholar]
- Veljkovic, V.; Mouscadet, J.F.; Veljkovic, N.; Glisic, S.; Debyser, Z. Simple criterion for selection of flavonoid compounds with anti-HIV activity. Bioorg. Med. Chem. Lett. 2007, 17, 1226–1232. [Google Scholar]
- Ito, J.; Chang, F.R.; Wang, H.K.; Park, Y.K.; Ikegaki, M.; Kilgore, N.; Lee, K.H. Anti-AIDS agents. 48. (1) Anti-HIV activity of moronic acid derivatives and the new melliferone-related triterpenoid isolated from Brazilian Propolis. J. Nat. Prod. 2001, 64, 1278–1281. [Google Scholar]
- Barbosa, M.H.; Zuffi, F.B.; Maruxo, H.B.; Jorge, L.L.R. Therapeutic properties of propolis for treatment of skin lesion. Acta Paul. Enfer. 2009, 22, 318–322. [Google Scholar]
- Burdock, G.A. Review of the biological properits and toxocity of bee propolis. Food Chem. Toxicol. 1998, 36, 347–363. [Google Scholar]
- Olczyk, P.; Wisowski, G.; Komosińska-Vassev, K.; Stojko, J.; Klimek, K.; Olczyk, M.; Koźma, E.M. Propolis modifies collagen types I and III accumulation in the matrix of burnt tissue. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Olczyk, P.; Komosińska-Vassev, K.; Winsz-Szczotka, K.; Stojko, J.; Klimek, K.; Koźma, E. Propolis induces chondroitin/dermatan sulphate and hyaluronic acid accumulation in the skin of burned wound. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Stojko, J.; Juszko-Piekut, M.; Rzepecka-Stojko, A.; Stojko, R.; Moździerz, A.; Olczyk, D.; Romaniuk, D.; Kasprzak, M.; Morawiec, T.; Paruzel, K.; et al. Clinical Study on the Use of the Propol in the Therapy of Burn Wounds. In Środowiskowe źródła Zagrożeń Zdrowotnych, 1st ed.; Kaczor, A., Borzęcki, A., Iskra, M., Eds.; Polihymnia: Lublin, Poland, 2007; pp. 1076–1080. [Google Scholar]
- Scheller, S.; Ilewicz, L.; Luciak, M.; Skobidurska, D.; Stojko, A.; Matuga, W. Biological properties and clinical application of propolis. IX. Experimental observation on the influence of EEP on dental pulp regeneration. Arzneim-Forsch. 1978, 28, 289–291. [Google Scholar]
- Scheller, S.; Stojko, A.; Szwarnowiecka, I.; Tustanowski, J.; Obuszko, Z. Biological properties and clinical application of propolis. VI. Investigation of the influence of ethanol extracts of propolis (EEP) on cartilaginous tissue regeneration. Arzneim-Forsch. 1977, 27, 2138–2140. [Google Scholar]
- Buczek, E.; Stojko, A.; Zając, M.; Breguła, S.; Olczyk, D. The case of gas gangrene in the course of post-operative fracture proximal end of the tibia. Kwart. Ortop. 1998, 4, 441–443. [Google Scholar]
- Sample Availability: Not available.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kurek-Górecka, A.; Rzepecka-Stojko, A.; Górecki, M.; Stojko, J.; Sosada, M.; Świerczek-Zięba, G. Structure and Antioxidant Activity of Polyphenols Derived from Propolis. Molecules 2014, 19, 78-101. https://doi.org/10.3390/molecules19010078
Kurek-Górecka A, Rzepecka-Stojko A, Górecki M, Stojko J, Sosada M, Świerczek-Zięba G. Structure and Antioxidant Activity of Polyphenols Derived from Propolis. Molecules. 2014; 19(1):78-101. https://doi.org/10.3390/molecules19010078
Chicago/Turabian StyleKurek-Górecka, Anna, Anna Rzepecka-Stojko, Michał Górecki, Jerzy Stojko, Marian Sosada, and Grażyna Świerczek-Zięba. 2014. "Structure and Antioxidant Activity of Polyphenols Derived from Propolis" Molecules 19, no. 1: 78-101. https://doi.org/10.3390/molecules19010078
APA StyleKurek-Górecka, A., Rzepecka-Stojko, A., Górecki, M., Stojko, J., Sosada, M., & Świerczek-Zięba, G. (2014). Structure and Antioxidant Activity of Polyphenols Derived from Propolis. Molecules, 19(1), 78-101. https://doi.org/10.3390/molecules19010078





