Antimalarial Evaluation of the Chemical Constituents of Hairy Root Culture of Bixa orellana L.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structure Elucidation

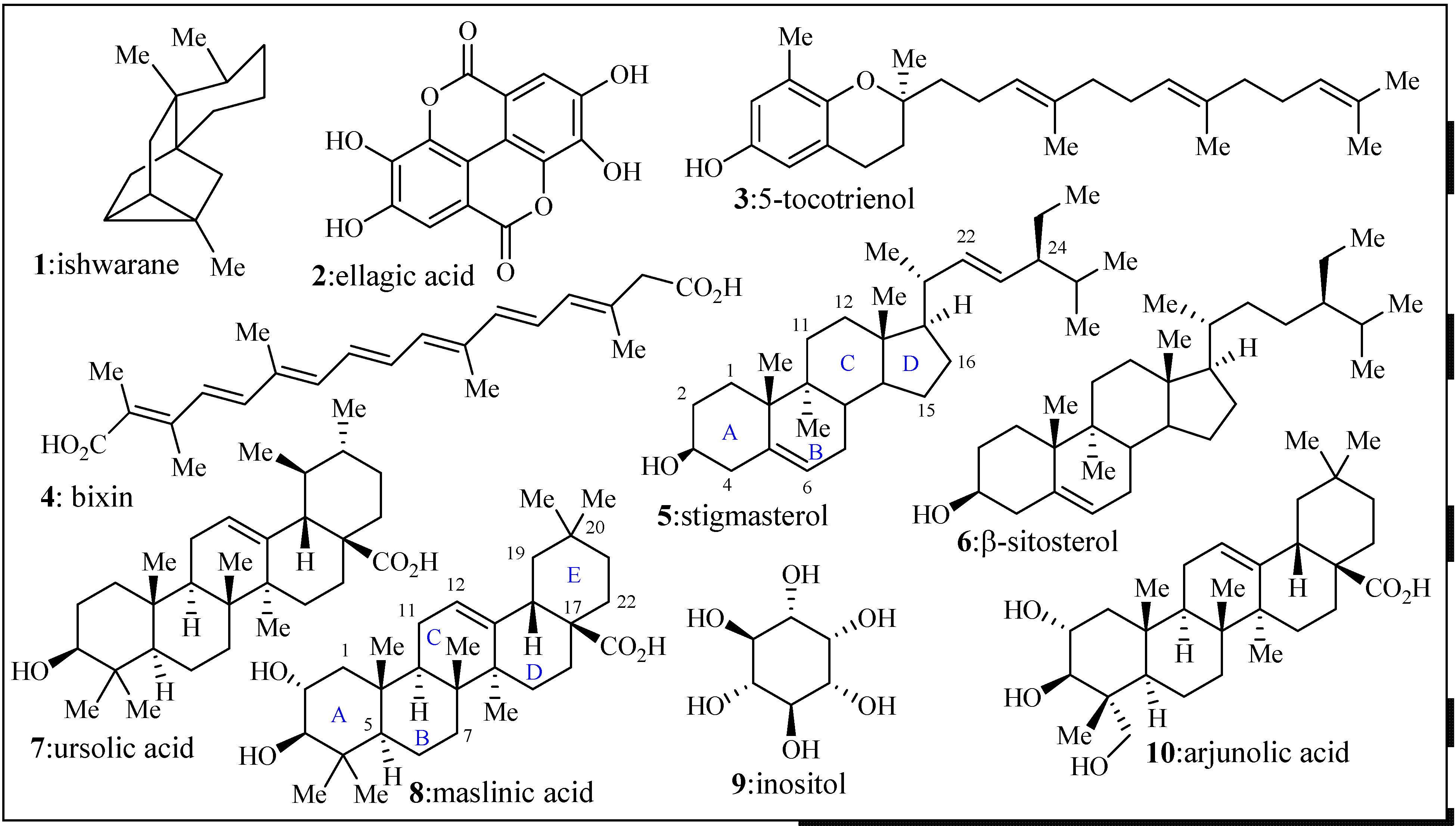

2.2. Anti-Malarial Activity

3. Experimental
3.1. Reagents and Instruments
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectral Data for Compound 1, 5, 13a and 13b
3.5. X-ray Crystallography of Compound 5
3.6. Antimalarial Test
Plasmodium falciparum
3.7. Cytotoxicity Assays
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- WHO (world health organization). Fact Sheet on Malaria. Available online: http://www.who.int/topics/malaria/en/ (accessed on 18 December 2013).
- Dharia, N.V.; Plouffe, D.; Bopp, S.E.R.; Gonzalez-Paez, G.E.; Lucas, C.; Salas, C.; Soberon, V.; Bursulaya, B.; Kochel, T.J.; Bacon, D.J.; et al. Genome scanning of Amazonian Plasmodium falciparum shows subtelomeric instability and clindamycin-resistant parasites. Genome Res. 2011, 20, 1534–1544. [Google Scholar]
- Dondorp, A.M.; Nosten, F.; Yi, P.; Das, D.; Phyo, A.P.; Tarning, J.; Lwin, K.M.; Ariey, F.; Hanpithakpong, W.; Lee, S.J.; et al. Artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med. 2009, 361, 455–467. [Google Scholar] [CrossRef]
- Mishra, B.B.; Tiwari, V.K. Natural products: An evolving role in future drug discovery. Eur. J. Med. Chem. 2011, 46, 4769–4807. [Google Scholar] [CrossRef]
- Bouvier, F.; Dogbo, O.; Camara, B. Biosynthesis of the food and cosmetic plant pigment bixin (annatto). Science 2003, 300, 2089–2091. [Google Scholar] [CrossRef]
- Frega, N.; Mozzon, M.; Bocci, F. Identification and estimation of tocotrienols in the annatto lipid fraction by gas chromatography-mass spectroscopy. J. Am. Oil Chem. Soc. 1998, 75, 1727–1723. [Google Scholar]
- Galindo-Cuspinera, V.; Lubran, M.B.; Rankin, S.A. Comparison of volatile compounds in water and oil soluble Annatto (Bixa Orellana L.) extracts. J. Agric. Food Chem. 2002, 50, 2010–2015. [Google Scholar] [CrossRef]
- Gómez-Barrio, A.; Martínez Grueiro, M.M.; Montero, D.; Nogal, J.J.; Escario, J.A.; Muelas, S.; Fernández, C.; Vega, C.; Rolón, M.; Martínez Fernández, A.R.; et al. In vitro antiparasitic activity of plant extracts from panama. Pharm. Biol. 2004, 42, 332–337. [Google Scholar]
- Fleischera, T.C.; Ameadea, E.P.K.; Mensaha, M.L.K.; Sawerb, I.K. Antimicrobial activity of the leaves and seeds of Bixa orellana. Fitoterapia 2003, 74, 136–138. [Google Scholar] [CrossRef]
- Ruiz, L.; Ruiz, L.; Maco, M.; Cobos, M.; Gutierrez-Choquevilca, A.-L.; Roumy, V. Plants used by native Amazonian groups from the Nanay River (Peru) for the treatment of malaria. J. Ethnopharmacol. 2011, 133, 917–921. [Google Scholar] [CrossRef]
- Medina-Bolivar, F.; Condori, J.; Rimandoc, A.M.; Hubstenberger, J.; Shelton, K.; O’Keefe, S.F.; Bennett, S.; Dolan, M.C. Production and secretion of resveratrol in hairy root cultures of peanut. Phytochemistry 2007, 68, 1992–2003. [Google Scholar] [CrossRef]
- Sivakumar, G.; Medina-Bolivar, F.; Lay, J.O.; Dolan, M.C.; Condori, J.; Grubbs, S.K.; Wright, S.M.; Baque, M.A.; Lee, E.J.; Paek, K.Y. Bioprocess and bioreactor: Next generation technology for production of potential plant-based antidiabetic and antioxidant molecules. Curr. Med. Chem. 2011, 18, 79–90. [Google Scholar] [CrossRef]
- Malatesta-Ferrand, M. Analyses of Constitutive and Inducible Specialized Metabolites in Tissues and Hairy Root Cultures of Annatto (Bixa orellana L.). Master Thesis, Arkansas State University, Jonesboro, AR, USA, 2008. [Google Scholar]
- Creelman, R.A.; Mullet, J.E. Biosynthesis and action of jasmonates in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 355–381. [Google Scholar] [CrossRef]
- Turner, J.G.; Ellis, C.; Devoto, A. The Jasmonate signal pathway. Plant Cell 2002, 14, S153–S164. [Google Scholar]
- Dathe, W.; Schindler, C.; Schneider, G.; Schmidt, J.; Porzel, A.; Jensen, E.; Yamaguchi, I. Curcubic acid and its 6,7-stereoisomers. Phytochemistry 1991, 30, 1909–1914. [Google Scholar] [CrossRef]
- Miersch, O.; Regvar, M.; Wasternack, C. Metabolism of jasmonic acid in pisolithus tincto rius cultures. Phyton 1999, 39, 243–247. [Google Scholar]
- Mahendranath, G.; Venugopalan, A.; Parimalan, R.; Giridhar, P.; Ravishankar, G.A. Annatto pigment production in root cultures of Achiote (Bixa orellana L.). Plant Cell Tiss. Organ. Cult. 2011, 106, 517–522. [Google Scholar] [CrossRef]
- Ucho, V.T.; Paula, R.C.; Krettli, L.G.; Santana, A.E.G.; Krettli, A.U. Antimalarial activity of compounds and mixed fractions of Cecropia pachystachya. Drug Dev. Res. 2010, 71, 82–91. [Google Scholar]
- Guiguemde, W.A.; Shelat, A.A.; Bouck, D.; Duffy, S.; Crowther, G.J.; Davis, P.H.; Smithson, D.C.; Connelly, M.; Clark, J.; Zhu, F.; et al. Chemical genetics of Plasmodium falciparum. Nature 2010, 465, 311–315. [Google Scholar] [CrossRef]
- Condori, J.; Sivakumar, S.; Hubstenberger, J.; Dolan, M.C.; Sobolev, V.S.; Medina-Bolivar, F. Induced biosynthesis of resveratrol and the prenylated stilbenoids arachidin-1 and arachidin-3 in hairy root cultures of peanut: Effects of culture medium and growth stage. Plant Physiol. Biochem. 2010, 48, 310–318. [Google Scholar] [CrossRef]
- Raga, D.D.; Espiritu, R.A.; Shen, C.C.; Ragasa, C.Y. A bioactive sesquiterpene from Bixa orellana. J. Nat. Med. 2011, 65, 206–211. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhai, B.; Clark, J.; Ling, T.; Connelly, M.; Medina-Bolivar, F.; Rivas, F. Antimalarial Evaluation of the Chemical Constituents of Hairy Root Culture of Bixa orellana L.. Molecules 2014, 19, 756-766. https://doi.org/10.3390/molecules19010756
Zhai B, Clark J, Ling T, Connelly M, Medina-Bolivar F, Rivas F. Antimalarial Evaluation of the Chemical Constituents of Hairy Root Culture of Bixa orellana L.. Molecules. 2014; 19(1):756-766. https://doi.org/10.3390/molecules19010756
Chicago/Turabian StyleZhai, Bo, Julie Clark, Taotao Ling, Michele Connelly, Fabricio Medina-Bolivar, and Fatima Rivas. 2014. "Antimalarial Evaluation of the Chemical Constituents of Hairy Root Culture of Bixa orellana L." Molecules 19, no. 1: 756-766. https://doi.org/10.3390/molecules19010756
APA StyleZhai, B., Clark, J., Ling, T., Connelly, M., Medina-Bolivar, F., & Rivas, F. (2014). Antimalarial Evaluation of the Chemical Constituents of Hairy Root Culture of Bixa orellana L.. Molecules, 19(1), 756-766. https://doi.org/10.3390/molecules19010756



