Optimized Ultrasound-Assisted Extraction of Phenolic Compounds from Polygonum cuspidatum
Abstract
:1. Introduction
2. Results and Discussion
2.1. Model Fitting
| Run | Independent variable a | Extraction yield (mg/g) b | ||||
|---|---|---|---|---|---|---|
| X1 (°C) | X2 (%) | X3 (W) | Piceid (Y1) | Resveratrol (Y2) | Emodin (Y3) | |
| 1 | +1 c (70) | −1 (40) | 0 (120) | 9.94 ± 0.57 | 3.67 ± 0.01 | 5.75 ± 0.07 |
| 2 | −1 (30) | −1 (40) | 0 (120) | 7.99 ± 0.47 | 3.11 ± 0.26 | 1.36 ± 0.17 |
| 3 | +1 (70) | 0 (60) | +1 (150) | 9.43 ± 0.12 | 3.73 ± 0.09 | 10.99 ± 0.13 |
| 4 | +1 (70) | +1 (80) | 0 (120) | 7.83 ± 0.12 | 3.66 ± 0.17 | 10.96 ± 0.32 |
| 5 | 0 (50) | 0 (60) | 0 (120) | 10.16 ± 0.04 | 3.72 ± 0.07 | 9.28 ± 0.02 |
| 6 | 0 (50) | +1 (80) | +1 (150) | 7.76 ± 0.02 | 3.39 ± 0.01 | 9.57 ± 0.08 |
| 7 | 0 (50) | −1 (40) | −1 (90) | 10.32 ± 0.17 | 3.45 ± 0.01 | 3.76 ± 0.10 |
| 8 | −1 (30) | +1 (80) | 0 (120) | 6.22 ± 0.35 | 2.95 ± 0.04 | 7.16 ± 0.30 |
| 9 | −1 (30) | 0 (60) | +1 (150) | 8.78 ± 0.07 | 3.50 ± 0.09 | 6.56 ± 0.07 |
| 10 | 0 (50) | 0 (60) | 0 (120) | 10.29 ± 0.20 | 3.70 ± 0.08 | 9.50 ± 0.04 |
| 11 | 0 (50) | 0 (60) | 0 (120) | 10.37 ± 0.02 | 3.73 ± 0.14 | 9.43 ± 0.02 |
| 12 | +1 (70) | 0 (60) | −1 (90) | 9.66 ± 0.13 | 3.70 ± 0.09 | 10.89 ± 0.22 |
| 13 | 0 (50) | −1 (40) | +1 (150) | 10.35 ± 0.12 | 3.41 ± 0.00 | 3.63 ± 0.18 |
| 14 | 0 (50) | +1 (80) | −1 (90) | 6.07 ± 0.01 | 2.99 ± 0.16 | 9.51 ± 0.47 |
| 15 | −1 (30) | 0 (60) | −1 (90) | 8.49 ± 0.14 | 3.10 ± 0.00 | 5.02 ± 0.10 |
| Factor a | Piceid (Y1) | Resveratrol (Y2) | Emodin (Y3) | |||
|---|---|---|---|---|---|---|
| Sum of Squares | Prob > F | Sum of Squares | Prob > F | Sum of Squares | Prob > F | |
| Model | 28.56 | 0.015 * | 1.12 | 0.003 * | 130.65 | <0.001 * |
| Linear term | ||||||
| X1 | 3.61 | 0.026 * | 0.55 | <0.001 * | 42.70 | <0.001* |
| X2 | 14.38 | 0.002 * | 0.05 | 0.043 * | 64.36 | <0.001* |
| X3 | 0.40 | 0.345 | 0.08 | 0.022 * | 0.31 | 0.267 |
| Quadratic | ||||||
| X12 | 3.04 | 0.035 * | 0.03 | 0.111 | 1.68 | 0.034* |
| X22 | 6.95 | 0.007 * | 0.30 | 0.001 * | 21.70 | <0.001* |
| X32 | 0.29 | 0.419 | 0.06 | 0.038 * | 0.49 | 0.178 |
| Interactions | ||||||
| X1X2 | 0.03 | 0.789 | 0.01 | 0.419 | 0.08 | 0.544 |
| X1X3 | 0.07 | 0.686 | 0.03 | 0.084 | 0.52 | 0.167 |
| X2X3 | 0.69 | 0.231 | 0.05 | 0.047 * | 0.01 | 0.843 |
| R2 = 0.94 | R2 = 0.97 | R2 = 0.99 | ||||
2.2. Response Surface Analysis
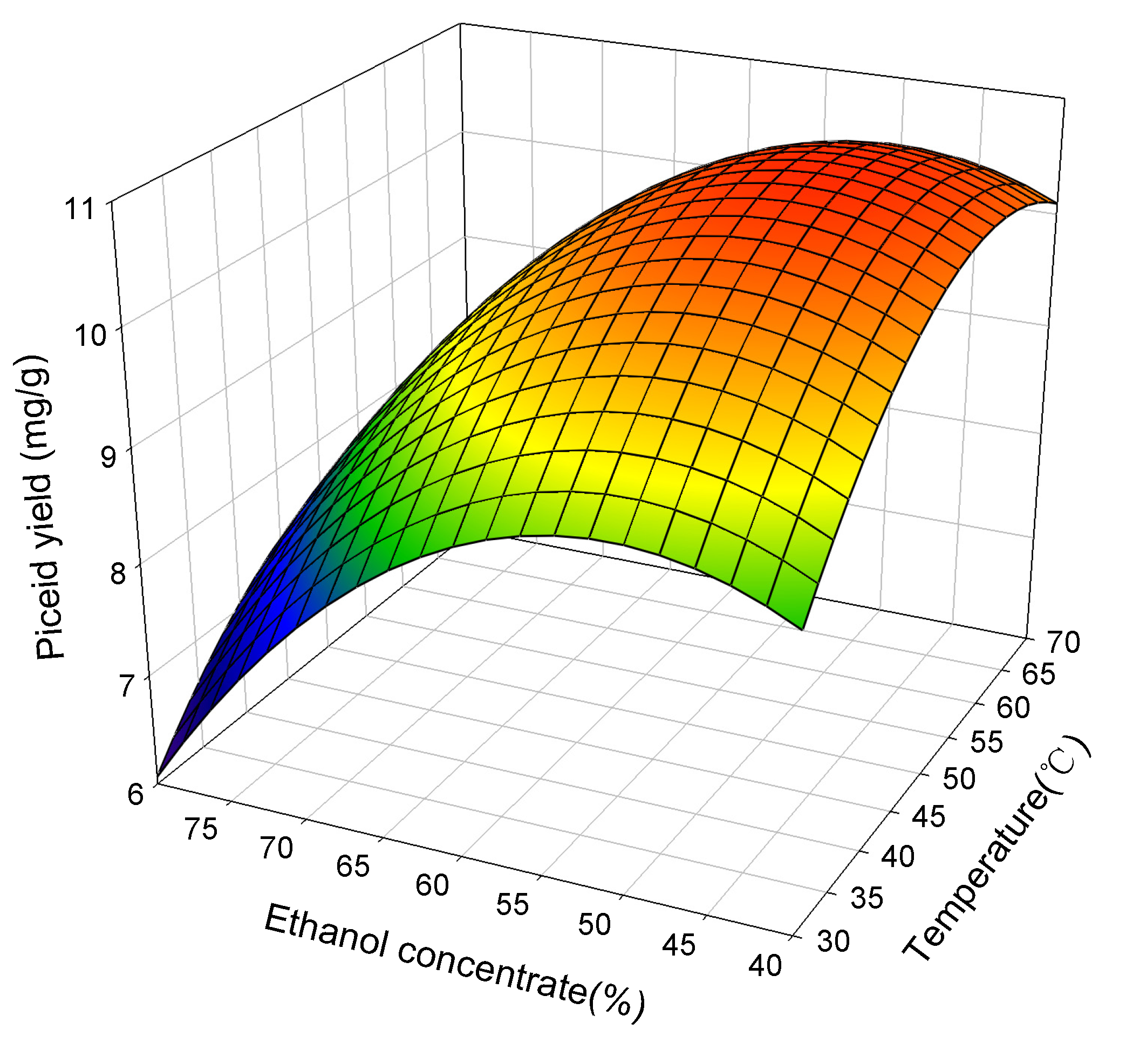
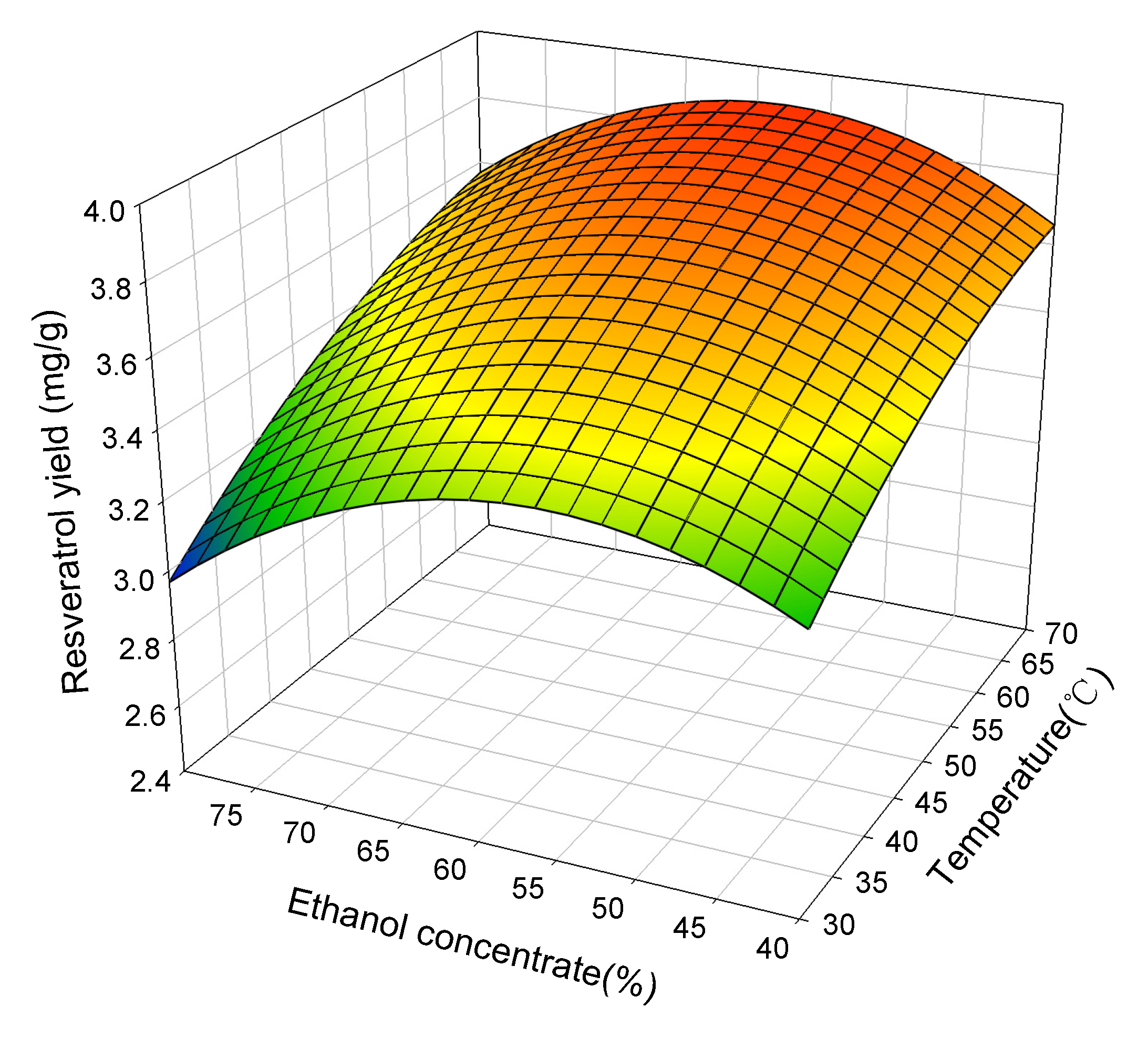
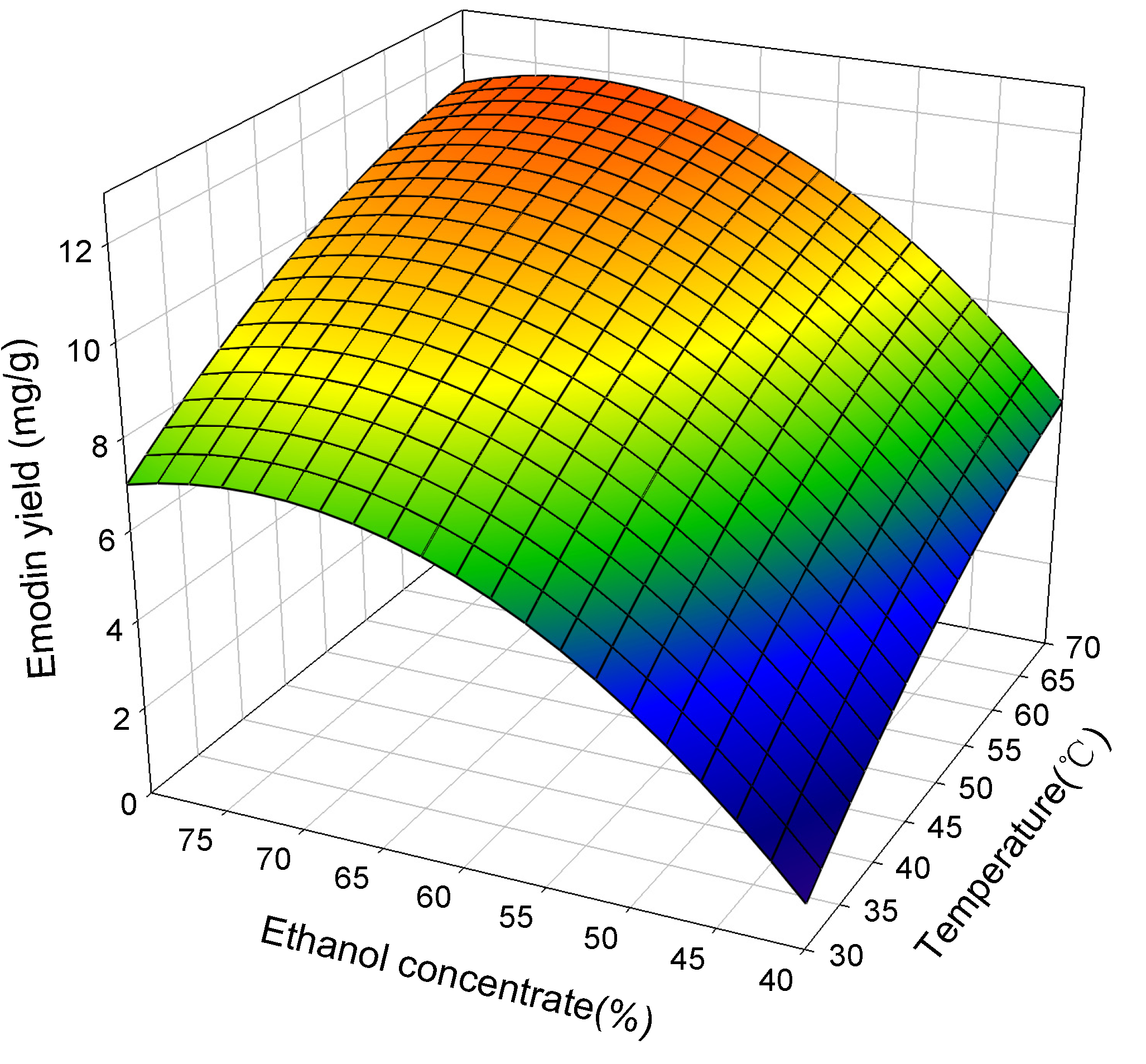
2.3. Attaining Optimum Extraction Condition
| Extraction target | Extraction conditions | Predicted Yield (mg/g) | Experimental Yield (mg/g) | ||
|---|---|---|---|---|---|
| Temp (°C) | EtOH (%) | Power (W) | |||
| Piceid | 57.8 | 49.75 | 120 | 10.74 | 10.77 ± 0.05 |
| Resveratrol | 70.0 | 58.51 | 120 | 3.90 | 3.82 ± 0.06 |
| Emodin | 70.0 | 71.06 | 120 | 11.79 | 11.72 ± 0.11 |
3. Experimental
3.1. Materials
3.2. Ultrasound-Assisted Extraction
3.3. Experimental Design
3.4. Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References and Notes
- Banerjee, S.; Bueso-Ramos, C.; Aggarwal, B.B. Suppression of 7, 12-dimethylbenz(a)anthracene-induced mammary carcinogenesis in rats by resveratrol role of nuclear factor-κB, cyclooxygenase 2, and matrix metalloprotease 9. Cancer Res. 2002, 62, 4945–4954. [Google Scholar]
- Xiao, K.; Xuan, L.; Xu, Y.; Bai, D.; Zhong, D. Constituents from Polygonum cuspidatum. Chem. Pharm. Bull. 2002, 50, 605–608. [Google Scholar] [CrossRef]
- Zhang, D.; Li, X.; Hao, D.; Li, G.; Xu, B.; Ma, G.; Su, Z. Systematic purification of polydatin, resveratrol and anthraglycoside B from Polygonum cuspidatum Sieb. et Zucc. Sep. Purif. Technol. 2009, 66, 329–339. [Google Scholar] [CrossRef]
- Pereira, A.P.; Ferreira, I.C.F.R.; Marcelino, F.; Valentão, P.; Andrade, P.B.; Seabra, R.; Estevinho, L.; Bento, A.; Pereira, J.A. Phenolic compounds and antimicrobial activity of olive (Olea europaea L. Cv. Cobrançosa) leaves. Molecules 2007, 12, 1153–1162. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Barros, L.; Soares, M.E.; Bastos, M.L.; Pereira, J.A. Antioxidant activity and phenolic contents of Olea europaea L. leaves sprayed with different copper formulations. Food Chem. 2007, 103, 188–195. [Google Scholar] [CrossRef]
- Bhat, K.P.L.; Kosmeder, J.W.; Pezzuto, J.M. Biological effects of resveratrol. Antioxid. Redox Signal. 2001, 3, 1041–1064. [Google Scholar] [CrossRef]
- Su, Y.C.; Li, S.C.; Wu, Y.C.; Wang, L.M.; Chao, K.S.C.; Liao, H.F. Resveratrol downregulates interleukin-6-stimulated sonic hedgehog signaling in human acute myeloid leukemia. Evid.-Based Complement Altern. Med. 2013, 2013, 547430. [Google Scholar]
- Rossi, D.; Guerrini, A.; Bruni, R.; Brognara, E.; Borgatti, M.; Gambari, R.; Maietti, S.; Sacchetti, G. Trans-resveratrol in nutraceuticals: Issues in retail quality and effectiveness. Molecules 2012, 17, 12393–12405. [Google Scholar] [CrossRef]
- Liu, Z.; Wei, F.; Chen, L.J.; Xiong, H.R.; Liu, Y.Y.; Luo, F.; Hou, W.; Xiao, H.; Yang, Z.Q. In vitro and in vivo studies of the inhibitory effects of emodin isolated from Polygonum cuspidatum on Coxsakievirus B4. Molecules 2013, 18, 11842–11858. [Google Scholar] [CrossRef]
- Xiang, M.X.; Xu, Z.; Su, H.W.; Hu, J.; Yan, Y.J. Emodin-8-O-β-D-glucoside from Polygonum Amplexicaule D. Don var. Sinense Forb. promotes proliferation and differentiation of osteoblastic MC3T3-E1 Cells. Molecules 2011, 16, 728–737. [Google Scholar] [CrossRef]
- Li, H.L.; Chen, H.L.; Li, H.; Zhang, K.L.; Chen, X.Y.; Wang, X.W.; Kong, Q.Y.; Liu, J. Regulatory effects of emodin on NF-kappaB activation and inflammatory cytokine expression in RAW 264.7 macrophages. Int. J. Mol. Med. 2005, 16, 41–47. [Google Scholar]
- Srinivas, G.; Anto, R.J.; Srinivas, P.; Vidhyalakshmi, S.; Senan, V.P.; Karunagaran, D. Emodin induces apoptosis of human cervical cancer cells through poly (ADP-ribose) polymerase cleavage and activation of caspase-9. Eur. J. Pharmacol. 2003, 473, 117–125. [Google Scholar] [CrossRef]
- Beňová, B.; Adam, M.; Pavlíková, P.; Fischer, J. Supercritical fluid extraction of piceid, resveratrol and emodin from Japanese knotweed. J. Supercrit. Fluids 2010, 51, 325–330. [Google Scholar] [CrossRef]
- Du, F.Y.; Xiao, X.H.; Li, G.K. Application of ionic liquids in the microwave-assisted extraction of trans-resveratrol from Rhizma Polygoni Cuspidati. J. Chromatogr. A 2007, 1140, 56–62. [Google Scholar] [CrossRef]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Achat, S.; Tomao, V.; Madani, K.; Chibane, M.; Elmaataoui, M.; Dangles, O.; Chemat, F. Direct enrichment of olive oil in oleuropein by ultrasound-assisted maceration at laboratory and pilot plant scale. Ultrason. Sonochem. 2012, 19, 777–786. [Google Scholar]
- Pingret, D.; Fabiano-Tixier, A.S.; Bourvellec, C.L.; Renard, C.M.; Chemat, F. Lab and pilot-scale ultrasound-assisted water extraction of polyphenols from apple pomace. J. Food Eng. 2012, 111, 73–81. [Google Scholar] [CrossRef]
- Lai, J.; Xin, C.; Zhao, Y.; Feng, B.; He, C.; Dong, Y.; Fang, Y.; Wei, S. Optimization of ultrasonic assisted extraction of antioxidants from black soybean (Glycine max var) sprouts using response surface methodology. Molecules 2013, 18, 1101–1110. [Google Scholar] [CrossRef]
- Lianfu, Z.; Zelong, L. Optimization and comparison of ultrasound/microwave assisted extraction (UMAE) and ultrasonic assisted extraction (UAE) of lycopene from tomatoes. Ultrason. Sonochem. 2008, 15, 731–737. [Google Scholar] [CrossRef]
- Li, Y.; Fabiano-Tixier, A.S.; Tomao, V.; Cravotto, G.; Chemat, F. Green ultrasound-assisted extraction of carotenoids based on the bio-refinery concept using sunflower oil as an alternative solvent. Ultrason. Sonochem. 2013, 20, 12–18. [Google Scholar] [CrossRef]
- Aybastıer, Ö.; Işık, E.; Şahin, S.; Demir, C. Optimization of ultrasonic-assisted extraction of antioxidant compounds from blackberry leaves using response surface methodology. Ind. Crop Prod. 2013, 44, 558–565. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Q.; Wu, Y.; Chen, G.; Yue, W.; Liang, Q. Response surface optimized ultrasonic-assisted extraction of flavonoids from Sparganii rhizoma and evaluation of their in vitro antioxidant activities. Molecules 2012, 17, 6769–6783. [Google Scholar] [CrossRef]
- Zhang, G.; He, L.; Hu, M. Optimized ultrasonic-assisted extraction of flavonoids from Prunella vulgaris L. and evaluation of antioxidant activities in vitro. Innov. Food Sci. Emerg. Technol. 2011, 12, 18–25. [Google Scholar] [CrossRef]
- Pan, G.; Yu, G.; Zhu, C.; Qiao, J. Optimization of ultrasound-assisted extraction (UAE) of flavonoids compounds (FC) from hawthorn seed (HS). Ultrason. Sonochem. 2012, 19, 486–490. [Google Scholar] [CrossRef]
- Chemat, F.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef]
- Pingret, D.; Durand, G.; Fabiano-Tixier, A.S.; Rockenbauer, A.; Ginies, C.; Chemat, F. Degradation of edible oil during food processing by ultrasound: electron paramagnetic resonance, physicochemical, and sensory appreciation. J. Agric. Food Chem. 2012, 60, 7761–7768. [Google Scholar] [CrossRef]
- Pingret, D.; Fabiano-Tixier, A.S.; Chemat, F. Degradation during application of ultrasound in food processing: a review. Food Control 2013, 31, 593–606. [Google Scholar] [CrossRef]
- Gu, F.; Xu, F.; Tan, L.; Wu, H.; Chu, Z.; Wang, Q. Optimization of enzymatic process for vanillin extraction using response surface methodology. Molecules 2012, 17, 8753–8761. [Google Scholar] [CrossRef]
- Kuo, C.H.; Hsiao, F.W.; Chen, J.H.; Hsieh, C.W.; Liu, Y.C.; Shieh, C.J. Kinetic aspects of ultrasound-accelerated lipase catalyzed acetylation and optimal synthesis of 4'-acetoxyresveratrol. Ultrason. Sonochem. 2013, 20, 546–552. [Google Scholar] [CrossRef]
- Cho, Y.J.; Hong, J.Y.; Chun, H.S.; Lee, S.K.; Min, H.Y. Ultrasonication-assisted extraction of resveratrol from grapes. J. Food Eng. 2006, 77, 725–730. [Google Scholar] [CrossRef]
- Romero-Pérez, A.I.; Lamuela-Raventós, R.M.; Andrés-Lacueva, C.; Torre-Boronat, M.C. Method for the quantitative extraction of resveratrol and piceid isomers in grape berry skins. Effect of powdery mildew on the stilbene content. J. Agric. Food Chem. 2001, 49, 210–215. [Google Scholar] [CrossRef]
- Chen, R.S.; Wu, P.L.; Chiou, R.Y.Y. Peanut roots as a source of resveratrol. J. Agric. Food Chem. 2002, 50, 1665–1667. [Google Scholar] [CrossRef]
- Sample Availability: Samples of P. cuspidatum roots are available from the authors.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kuo, C.-H.; Chen, B.-Y.; Liu, Y.-C.; Chang, C.-M.J.; Deng, T.-S.; Chen, J.-H.; Shieh, C.-J. Optimized Ultrasound-Assisted Extraction of Phenolic Compounds from Polygonum cuspidatum. Molecules 2014, 19, 67-77. https://doi.org/10.3390/molecules19010067
Kuo C-H, Chen B-Y, Liu Y-C, Chang C-MJ, Deng T-S, Chen J-H, Shieh C-J. Optimized Ultrasound-Assisted Extraction of Phenolic Compounds from Polygonum cuspidatum. Molecules. 2014; 19(1):67-77. https://doi.org/10.3390/molecules19010067
Chicago/Turabian StyleKuo, Chia-Hung, Bao-Yuan Chen, Yung-Chuan Liu, Chieh-Ming J. Chang, Tzu-Shing Deng, Jiann-Hwa Chen, and Chwen-Jen Shieh. 2014. "Optimized Ultrasound-Assisted Extraction of Phenolic Compounds from Polygonum cuspidatum" Molecules 19, no. 1: 67-77. https://doi.org/10.3390/molecules19010067






