2.1. Effects of Quercetin and QS on Serum ALT and AST Activities in Mice
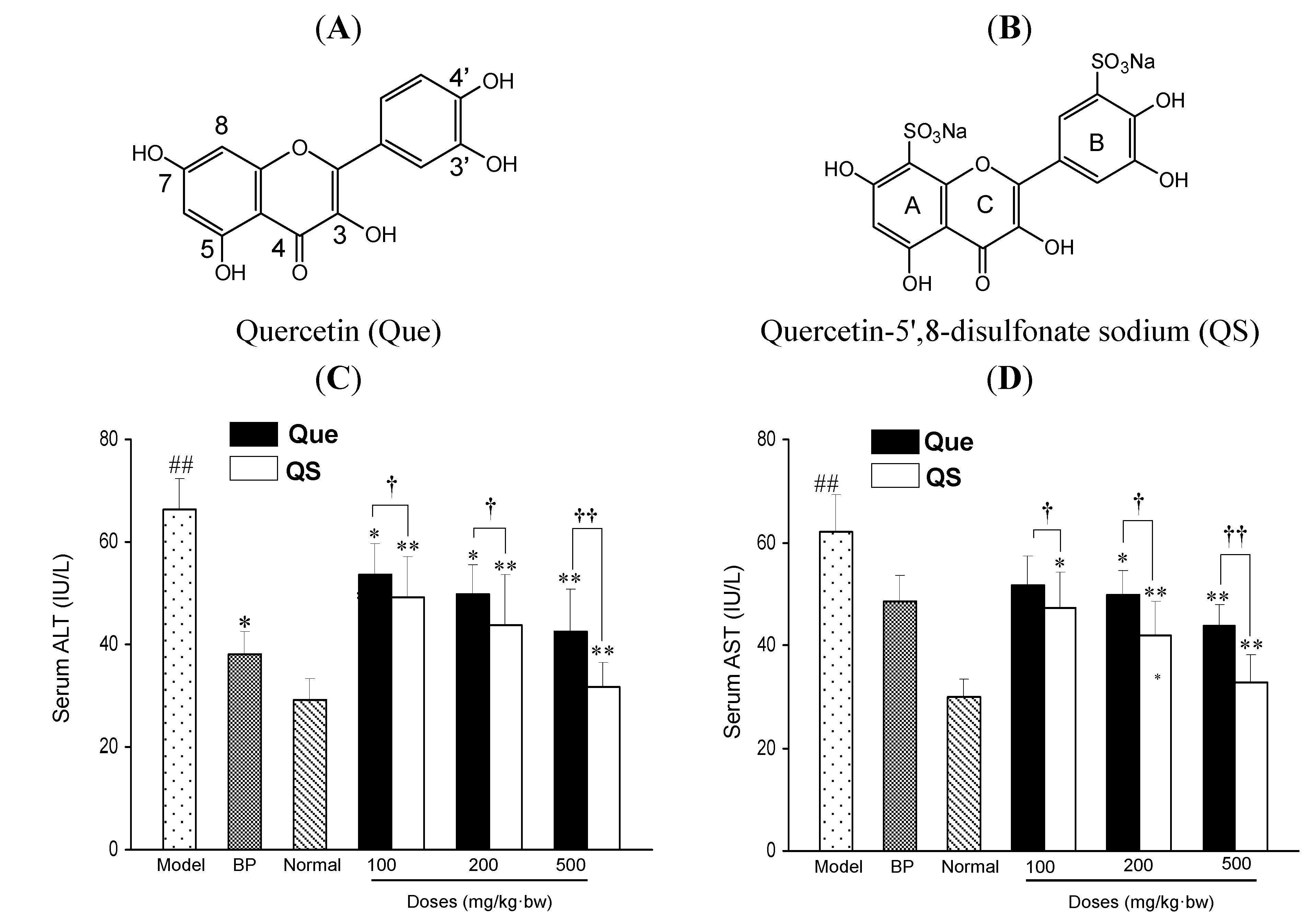
Figure 1C,D shows the effects of quercetin (Que) and QS on the enzymatic activities of serum ALT and AST in mice. In the normal group, the serum ALT and AST activities were 29.2 ± 4.2 and 30.0 ± 3.3 IU/L, respectively, whereas a single dose of CCl
4 injection in mice led to a rapid rise of serum ALT and AST activities up to 68.5 ± 6.0 and 62.1 ± 7.3 IU/L, respectively, with increases of 134.6% and 107.0%, compared to the normal mice (
p < 0.01), respectively. However, with the pretreatment of quercetin and QS before CCl
4 damage, the serum activities of ALT and AST were significantly decreased, compared to the CCl
4-intoxicated mice (
p < 0.05). Interestingly, the ALT and AST activities of QS-treated mice were remarkably lower than the same concentrations of Que-treated mice (
p < 0.05). At 100 mg/kg·bw, QS caused a 14.8% and 14.7% greater decrease in ALT and AST activities than quercetin (
p < 0.05).
Figure 1.
The chemical structures of quercetin (Que) (A) and quercetin-5',8-disulfonate (QS) (B). Effects of Que and QS on serum enzymic activities of ALT (C) and AST (D) of mice after CCl4 treatment. Mice were treated intragastrically with Que or QS (100, 200 and 500 mg/kg·bw) once daily for fourteen consecutive days prior to the single administration of CCl4 (0.1%, ip). Data were expressed as mean ± SD. # p < 0.05, ## p < 0.01, compared to the normal group. * p < 0.05, ** p < 0.01, compared to the CCl4-intoxicated group. † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
Figure 1.
The chemical structures of quercetin (Que) (A) and quercetin-5',8-disulfonate (QS) (B). Effects of Que and QS on serum enzymic activities of ALT (C) and AST (D) of mice after CCl4 treatment. Mice were treated intragastrically with Que or QS (100, 200 and 500 mg/kg·bw) once daily for fourteen consecutive days prior to the single administration of CCl4 (0.1%, ip). Data were expressed as mean ± SD. # p < 0.05, ## p < 0.01, compared to the normal group. * p < 0.05, ** p < 0.01, compared to the CCl4-intoxicated group. † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
When the dosage increased to 200 mg/kg·bw, 21.4% and 26.7% decreases were observed, respectively (
p < 0.05), and a further decrease was achieved at 500 mg/kg·bw, where ALT and AST activities of QS-treated mice were 32.1% and 36.6% lower than those of Que-treated mice (
p < 0.01), respectively. It was also found that ALT and AST activities of QS-treated mice were close to that of the same concentration of the positive reference drug BP (
Figure 1C,D). These results suggest that QS at the tested concentrations of 100, 200 and 500 mg/kg·bw is more effective than the parent quercetin in lowering the CCl
4-induced hepatotoxicity in mice, and quercetin sulfation increases its hepatoprotective activity
in vivo.
2.2. Effects of Quercetin and QS on Serum LDH Activity and Hepatic MDA Level
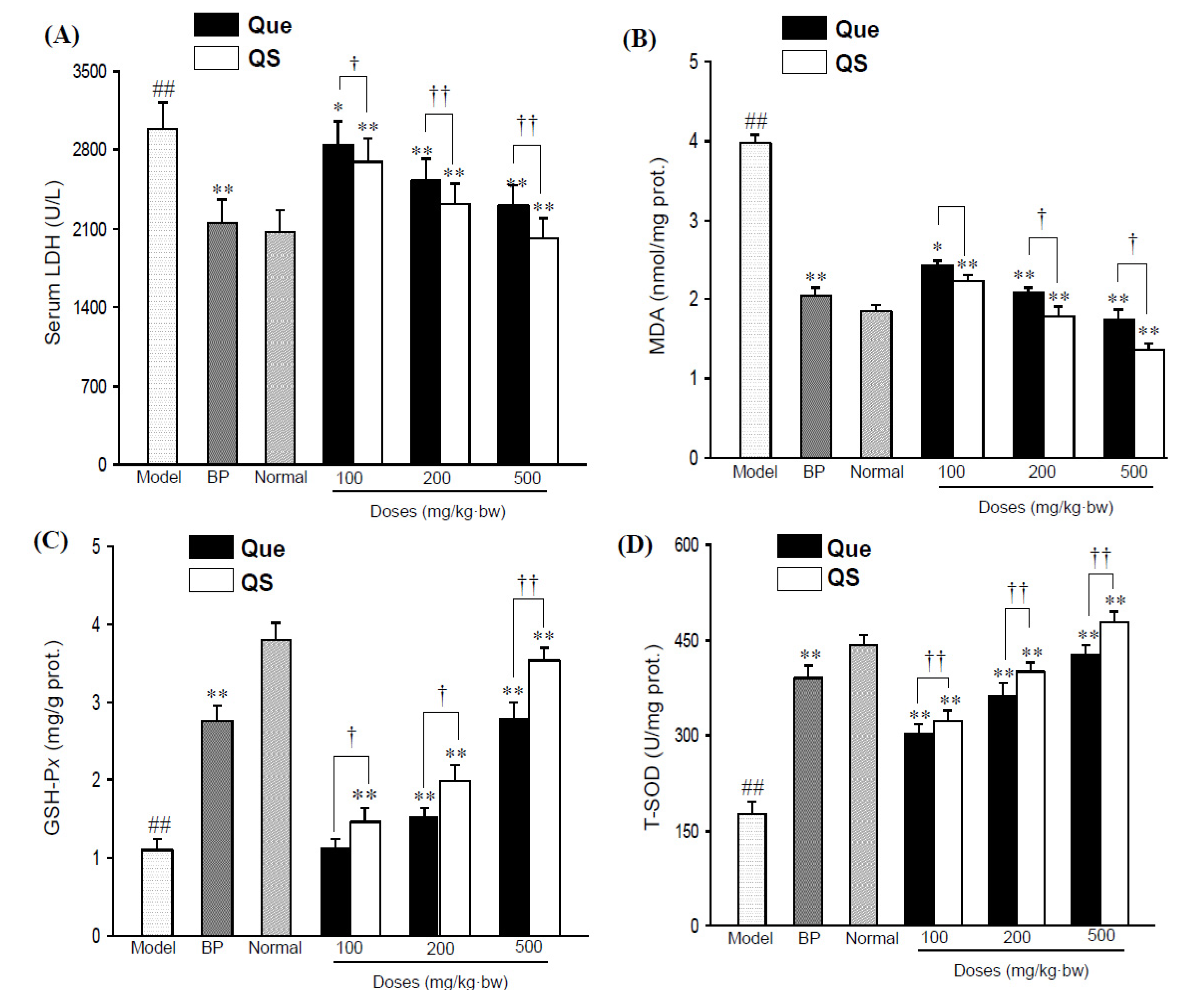
Figure 2A shows the change of serum LDH activity of all the tested mice. Administration of CCl
4 directly elevated serum LDH activity to 2,986.2 ± 241.6 U/L from 2,063.2 ± 200.7 U/L in the normal mice (
p < 0.01). However, pretreatment with both quercetin and QS effectively antagonized the CCl
4-induced elevation (
p < 0.05), and the LDH activities of Que- and QS-treated mice were 2844.1 ± 208.8 and 2698.6 ± 200.7 U/L at 100 mg/kg·bw (
p < 0.05,
p < 0.01), 2521.5 ± 203.1 and 2321.4 ± 185.9 U/L (
p < 0.01) at 200 mg/kg·bw (
p < 0.01), and 2307.1 ± 174.9 and 2014.7 ± 184.5 U/L at 500 mg/kg·bw (
p < 0.01), respectively.
Figure 2.
Serum LDH levels (A) and hepatic MDA levels (B) in CCl4-intoxicated mice under the effects of quercetin (Que) and QS. Hepatic GSH-Px (C) and T-SOD (D) activities of mice after oral administration of Que and QS for 2 weeks and subsequently CCl4 treatment. Data were expressed as mean ± SD. # p < 0.05, ## p < 0.01, compared to the normal group. * p < 0.05, ** p < 0.01, compared with CCl4-intoxicated group. † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
Figure 2.
Serum LDH levels (A) and hepatic MDA levels (B) in CCl4-intoxicated mice under the effects of quercetin (Que) and QS. Hepatic GSH-Px (C) and T-SOD (D) activities of mice after oral administration of Que and QS for 2 weeks and subsequently CCl4 treatment. Data were expressed as mean ± SD. # p < 0.05, ## p < 0.01, compared to the normal group. * p < 0.05, ** p < 0.01, compared with CCl4-intoxicated group. † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
Additionally, CCl
4 also had a significant impact on hepatic MDA (
Figure 2B), a lipid peroxidative product of cell membranes, as indicated by a significant increase in MDA content from 1.85 ± 0.2 nmol/mg prot. in the untreated normal group to 2.81 ± 0.3 nmol/mg prot. in the CCl
4-intoxicated mice group (
p < 0.01). However, the pretreatment of mice with QS at 100, 200 and 500 mg/kg·bw reduced the CCl
4-elevated hepatic MDA levels (
p < 0.01), with an decrese of MDA levels by 10.8%, 16.8% and 19.5% compared to that seen with the same dose of quercetin (
p < 0.05), respectively, indicating that QS has better antioxidant effects than the parent quercetin.
2.3. Effects of Quercetin and QS on Hepatic GSH-Px and T-SOD Activities
As shown in
Figure 2C,D, hepatic GSH-P
x and T-SOD activities in CCl
4-intoxicated mice sharply decreased by 70.5% and 60.2%, respectively, in comparison with the normal group (
p < 0.01). As expected, the pretreatment of quercetin and QS dose-dependently improved the GSH-P
x and T-SOD levels, relative to the CCl
4-injured mice. At 200 mg/kg·bw, hepatic GSH-P
x and T-SOD activities increased to 2.0 ± 0.2 mgGSH/g prot. and 401.4 ± 12.8 U/mg prot. (
p < 0.01), and the corresponding values were 3.5 ± 0.1 mgGSH/g prot. and 478.5 ± 16.9 U/mg prot. at 500 mg/kg·bw (
p < 0.01) from 1.1 ± 0.1 mgGSH/g prot. and 175.8 ± 20.9 U/mg prot. of CCl
4-injuried mice, respectively. It was worth noting that QS presented better hepatoprotective effects in enhancing antioxidant enzymes than quercetin (
p < 0.05), and this antioxidant protection was dose-dependent (
Figure 2C,D). When mice were administrated with low a dosis of QS (100 mg/kg·bw), a significant increase in GSH-Px and T-SOD levels started with a value of 8.8%, and 4.7% above that of same dose of quercetin (
p < 0.05), whereas the treatment of mice with 200 and 500 mg/kg·bw QS caused an increase of 12.1% and 22.9% for GSH-Px (
p < 0.05,
p < 0.01), and 9.0% and 11.8% for T-SOD compared to the effect seen for the same dose of quercetin (
p < 0.05), respectively, suggesting that QS has a better hepatoprotective effect than the parent quercetin.
2.4. Histopathological Examination of Mice Liver
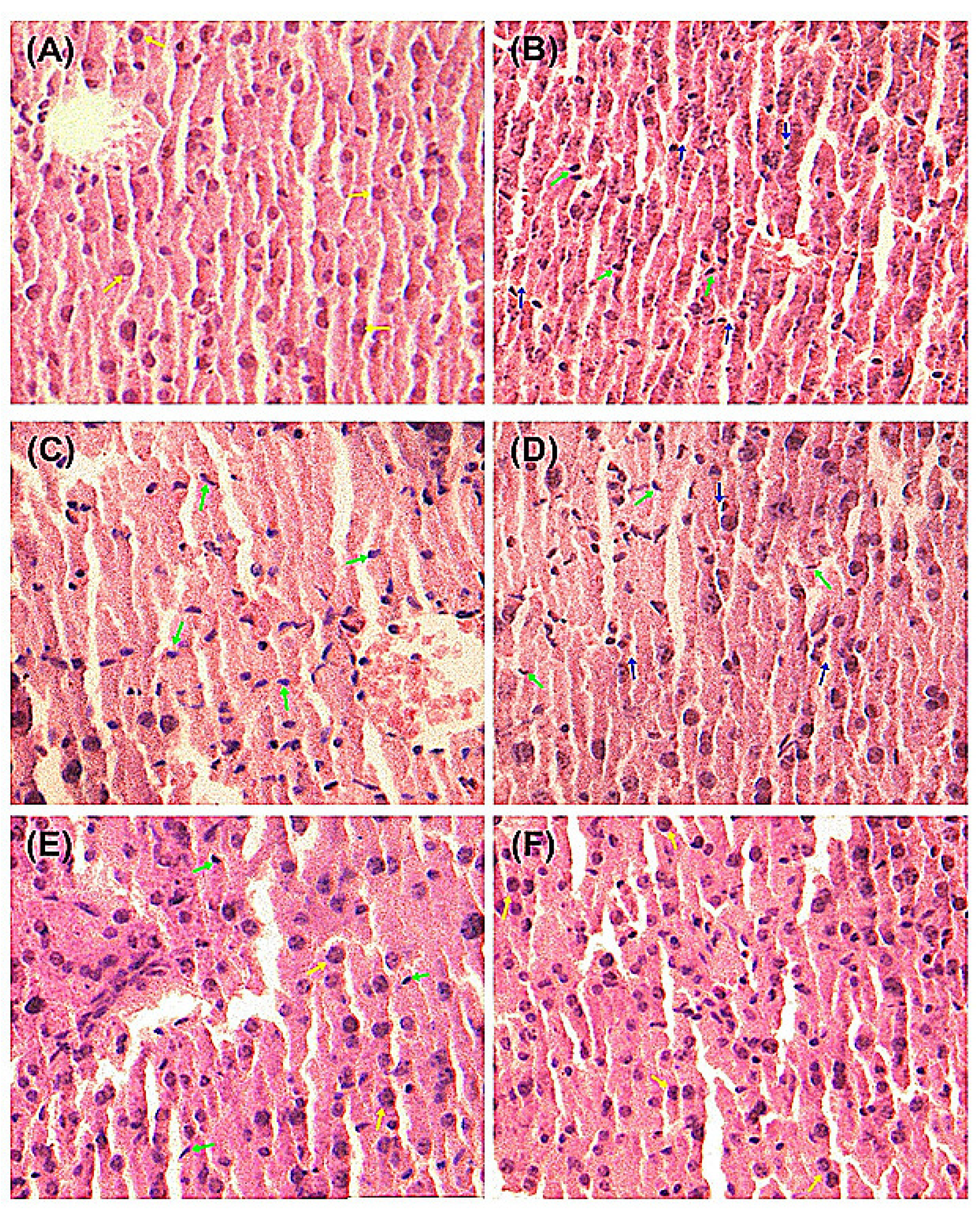
As demonstrated in
Figure 3, liver histopathological studies of hepatocyte morphological changes provided evidence to support the observed biochemical effects of quercetin and QS. In the Normal group, liver slices showed complete structure of cells with normal cell morphology, well-preserved cytoplasm and a clear plump nucleus (
Figure 3A). As shown in
Figure 3B, significant anomalies of liver cells were observed in CCl
4-injured mice, where the cytoplasm was significantly reduced and the nucleus become atrophic, suggesting that CCl
4 has induced severe liver cell injury. The pretreatment with quercetin and QS at 200 and 500 mg/kg·bw could effectively protect the liver from acute CCl
4-induced hepatocyte morphological damage (
Figure 3C–F). As depicted in
Figure 3E,F, oral administration of both quercetin and QS at high dose (500 mg/kg·bw) resulted in almost normal histopathology, with well-preserved cytoplasms and a clear nuclei. These results suggest that both quercetin and QS can protect the liver from CCl
4-induced acute liver damage.
Figure 3.
Pathological changes of the liver tissues in mice with the H&E staining. (A) Normal control. (B) CCl4 alone treatment. (C) CCl4 treatment and administration of quercetin (Que) at 200 mg/kg·bw, ig. (D) CCl4 treatment and administration of QS at 200 mg/kg·bw, ig. (E) CCl4 treatment and administration of Que at 500 mg/kg·bw, ig. (F) CCl4 treatment and administration of QS at 500 mg/kg·bw, ig. The green arrow indicates hepatic cell necrosis and the nucleus contraction. The blue arrow indicates the enlarged sinusoids between the plates of hepatocytes and loss of cellular boundaries. The yellow arrow indicates normal cellular architecture with clear hepatic cells.
Figure 3.
Pathological changes of the liver tissues in mice with the H&E staining. (A) Normal control. (B) CCl4 alone treatment. (C) CCl4 treatment and administration of quercetin (Que) at 200 mg/kg·bw, ig. (D) CCl4 treatment and administration of QS at 200 mg/kg·bw, ig. (E) CCl4 treatment and administration of Que at 500 mg/kg·bw, ig. (F) CCl4 treatment and administration of QS at 500 mg/kg·bw, ig. The green arrow indicates hepatic cell necrosis and the nucleus contraction. The blue arrow indicates the enlarged sinusoids between the plates of hepatocytes and loss of cellular boundaries. The yellow arrow indicates normal cellular architecture with clear hepatic cells.
2.5. Sulfation of Quercetin Modulated its Hydrophilicity and Absorption in Mice
Quercetin with its low hydrophilicity is known to have minimal bioavailability [
13]. Hence, we further investigated the solubility of quercetin and QS, and their absorption in mice, respectively. As expected, quercetin is almost insoluble in water, and its solubility in alcohol is 3.5 mg/mL, whereas the solubility of QS in water is 186.2 mg/mL, and 130.4 mg/mL in alcohol. This result clearly suggests that sulfation of quercetin significantly improves the hydrophilicity of quercetin, indicating that this possibly meliorates the absorption
in vivo. In this study, a measure of total quercetin or QS metabolites, which are the markers of quercetin or QS consumption, was determined in fasting 24-hour urine and feces samples.
Figure 4 shows, as an example, the HPLC chromatograms of quercetin (
Figure 4A) and
QS (
Figure 4B) in urine, respectively. The urinary metabolites of quercetin or QS were detected by an enzyme-hydrolyzed method for cleavage of ester-bonds in the forms of glucuronides and/or sulfates [
13]. HPLC analysis of quercetin or QS showed a stable baseline and good resolution between the analytes and endogenous substances. The regression equation in the range of 5.0–50.0 μg/mL was as follows: Y = 0.1045X + 0.055,
R2 = 0.9994 for quercetin, and Y = 0.1865X − 0.143,
R2 = 0.9994 for QS. As shown in
Figure 4C, after oral administration of quercetin or QS at 100 mg/kg·bw, 24-hour urinary excretion amount of total quercetin and QS including glucuronides and/or sulfates and unconjugated aglycone was 0.22 mg and 0.84 mg, respectively. When mice were administered 200 and 500 mg/kg·bw, the average amount of quercetin excreted was 0.46 mg and 1.11 mg, whereas it was 1.53 mg and 2.10 mg for QS, respectively. Interestingly, QS metabolite concentrations were statistically higher after QS administration in comparison with quercetin intervention (
p < 0.01), with an increase by 2.82-, 2.33-, and 0.89-fold at 100, 200 and 500 mg/kg·bw, respectively. In contrast to the results obtained with urine, 24-hour faeces excretion amount of total quercetin and QS following 100 mg/kg·bw administration to the mice was 1.54 mg and 0.66 mg, respectively (
Figure 4D). When mice received 200 and 500 mg/kg·bw of QS, the amount of QS was 0.82 mg and 2.61 mg, which was significantly lower than the 1.1 mg and 3.8 mg, respectively, achieved with quercetin at the same dose. As a result, QS was shown to have an improved 24-hour urinary excretion and a decreased 24-hour fecal excretion in comparison with quercetin (
p < 0.05). In accordance with the finding that QS increased the hepatoprotective effect of parent quercetin, the selective 5',8-sulfation of quercetin also increased its absorption in mice.
Figure 4.
HPLC-UV (320 nm) analysis for the detection of total quercetin (Que) and QS (conjugated and nonconjugated compounds derived from in vivo phase II) in 24-hour urine of mice. (A) HPLC profiles of Que in 24-hour urine of mice after oral administration of Que at 500 mg/kg·bw, ig. (B) HPLC profiles of QS in 24-hour urine of mice after oral administration of QS at 500 mg/kg·bw, ig. (C) The average contents of QS and Que in the 24-hour urine of mice. † p < 0.05, †† p < 0.01, compared to that of Que at the same dose. (D) The average contents of QS and Que in the 24-hour faeces of mice, † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
Figure 4.
HPLC-UV (320 nm) analysis for the detection of total quercetin (Que) and QS (conjugated and nonconjugated compounds derived from in vivo phase II) in 24-hour urine of mice. (A) HPLC profiles of Que in 24-hour urine of mice after oral administration of Que at 500 mg/kg·bw, ig. (B) HPLC profiles of QS in 24-hour urine of mice after oral administration of QS at 500 mg/kg·bw, ig. (C) The average contents of QS and Que in the 24-hour urine of mice. † p < 0.05, †† p < 0.01, compared to that of Que at the same dose. (D) The average contents of QS and Que in the 24-hour faeces of mice, † p < 0.05 and †† p < 0.01 versus the corresponding dose of Que group.
2.6. Discussion
Dietary quercetin and its derivatives have become a research focus because quercetin is a major flavonoid widely found in natural plants, and has been proved to have strong antioxidant, anti-cancer and the other beneficial effects [
7,
8,
9,
10,
11,
12,
13,
14]. The bioactivity of quercetin is attributed to its specific molecular structure, in which the oxygen active group, phenolic hydroxyls and 2,3-unsaturated double bond give quercetin strong antioxidant ability not only from accepting oxygen free radicals, but also by forming metal chelation compounds, and thus the excessive levels of metals such as iron and copper can be reduced [
15,
16]. However, quercetin's low hydrophilicity results in minimal absorption in the gastrointestinal tract, and its oral bioavailability is less than 17% in rats [
17] and perhaps less than 1% in man [
18]. As a result, the clinical application of quercetin is greatly restricted. In the human organism, the lipophilic quercetin molecule is largely metabolized and distributed as highly hydrophilic conjugated derivatives (sulfates, glucuronides, methylated species,
etc.). It has been reported that in animal models the synthetic compound 3'-O-methylquercetin was shown to retain an elevated level in plasma, as compared to its aglycon [
14]. Recently, we have established that QS (quercetin-5',8-disulfonate), a newly synthesized flavonoid derivative, had remarkably higher anti-tumor effects than quercetin [
9]. The sulfonation reaction is an electrophilic substitution reaction, and the 7-OH of A ring of quercetin (
Figure 1B) is the electron-donating group which increases the density of the
ortho electron cloud and 4'-OH of B ring is also an electron-donating group. This can increase the density of the electron cloud to allow access to the 5' and 8 sites of quercetin (
Figure 1A). As a result, the synthetic quercetin-5',8-disulfonate showed greater water solubility. Here, we employed a mice model of CCl
4-induced oxidative liver damage to show that both quercetin and QS had hepatoprotective effects, and QS exhibited a significantly higher effect than the parent quercetin.
In this study, a well-known hepatotoxin, CCl
4, was used to induce the acute liver damage in mice [
19]. It is reported that CCl
4 generates two active microsomal radicals or peroxides (
•CCl
3 or CCl
3OO
•) by cytochrome P450 during the toxic metabolic process of CCl
4 in the liver [
20,
21]. These substances may cause lipid peroxidation with liver cell membranes and subcellular structures, and undermine the integrity of the cell membrane structure [
22], which eventually may lead to the liver cell death [
23]. It is also well known that the increased ALT enzyme activity is an indicator of the degree of liver cell membrane damage, and elevated AST level is another indicator of liver mitochondrial damage, which are therefore the most important and effective index for evaluating liver cell damage [
24,
25]. In our study, the injection of CCl
4 in mice directly led to the rise of serum ALT and AST activities (
p < 0.01). After the pretreatment of quercetin and QS, the liver was protected, as indicated by the decreased serum enzyme activities of ALT and AST, as well as serum LDH activity, suggesting that quercetin and QS may effectively protect hepatocytes against the toxic effects of CCl
4. On this basis, it is suggested that both quercetin and QS, not only stabilize the hepatic cellular membrane, but also have a protective effects on the mitochondria. It should be noted that water soluble QS is more effective than quercetin in lowering the CCl
4-caused ALT and AST releases from liver cells (
Figure 1C,D), suggesting that quercetin sulfation as QS successfully modulated hepatoprotective effects against acute CCl
4-caused liver injury.
CCl
4-induced liver damage is considered a consequence of oxidative stress, which can lead to damage of liver cell biomolecules in rats and humans [
26,
27]. Antioxidants can reduce the cellular oxidative stress by inhibiting the formation of ROS/reactive nitrogen species through upregulation of cellular defense mechanisms, such as SOD, catalase, or GSH-P
x [
27]. In our experiment, we found that the hepatotoxicity of CCl
4 was associated with the formation of MDA and the decline of SOD and GSH-P
x activities. The present investigation demonstrated that CCl
4-induced oxidative stress in the liver was substantially attenuated by quercetin- or QS-pretreatment as it is evident that quercetin or QS, not only inhibited MDA formation, but also enhanced GSH-Px and SOD activities in CCl
4-induced mice, suggesting that both quercetin and QS had beneficial effects in inhibiting oxidative stress and might act as modulators of CCl
4-associated liver oxidative damage.
The ability of quercetin to exert its action
in vivo is dependent on the extent of its absorption post- ingestion and its ability to be distributed in various body tissues [
13]. To further investigate whether the difference in hepatoprotective effects between quercetin and QS in mice was due to disparate absorption and solubility, we also measured urinary and fecal accumulation of quercetin and QS in mice. Interestingly, quercetin or QS excretion in urine and faeces followed a dose-dependent response, and the relative absorption of QS was also higher in QS-treated mice than quercetin-treated mice. Urine and faeces samplings are particularly useful measures of polyphenols with relatively short half-lives, for which plasma measurements may fail to represent accurate intake [
13]. The proposed HPLC analysis has indicated that the fecal and urinary levels of quercetin and QS were parallel with the observed hepatoprotective effects in mice. In this study, QS has superior hepatoprotective effects over quercetin in mice. Although the structure and activity relationships of the new quercetin derivative, QS, are far from clear, these findings suggest that 5',8-sulfated groups in QS played an important part in enhancing hepatoprotective activities. Furthermore, the enhanced bioavailability owing to quercetin sulfation may be attributed to improved water solubility of QS, and subsequently may contribute to the higher hepatoprotective effects in mice. These findings suggest the potential of QS for the development of a novel hepatoprotective supplement.