The Phenolics from the Roots of Livistona chinensis Show Antioxidative and Obsteoblast Differentiation Promoting Activity
Abstract
:1. Introduction
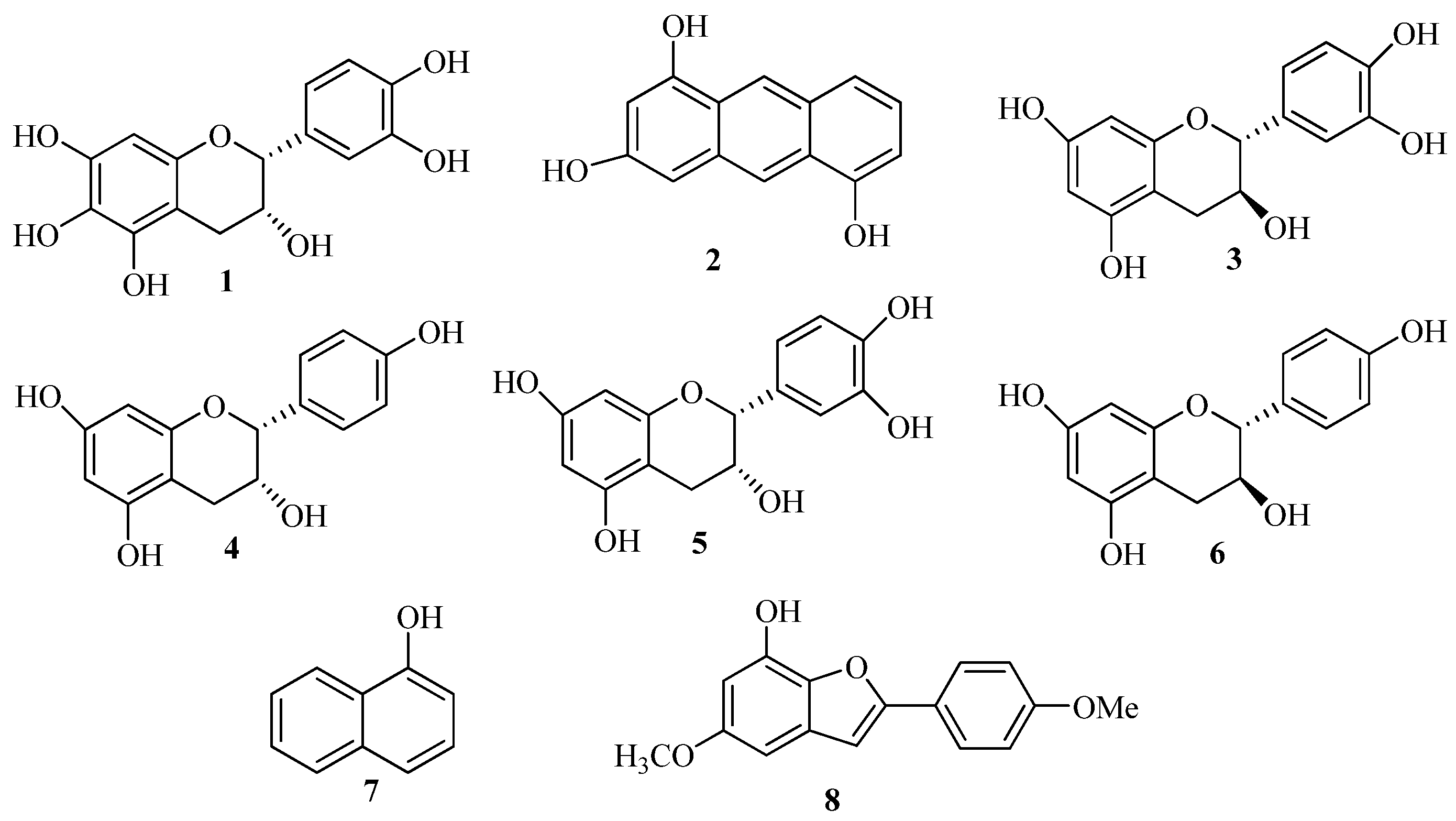
2. Results and Discussion
2.1. Structure Elucidation of the New Phenolics 1 and 2
 = −48.0 (c 0.30, MeOH)} indicated a 2R and 3R absolute configuration [14], the optical rotation value of 1 {
= −48.0 (c 0.30, MeOH)} indicated a 2R and 3R absolute configuration [14], the optical rotation value of 1 {  = −43.0 (c0.3, MeOH)} suggested the absolute configurations of C-2 and C-3 were (2R, 3R). Thus, the structure of 1 was elucidated to be 2R,3R-3,5,6,7,3',4'-hexahydroxyflavane.
= −43.0 (c0.3, MeOH)} suggested the absolute configurations of C-2 and C-3 were (2R, 3R). Thus, the structure of 1 was elucidated to be 2R,3R-3,5,6,7,3',4'-hexahydroxyflavane.2.2. In Vitro Effect of the Phenolics 1–3, 7, 8 in Osteoblast Viability
| Concentrations (μg/mL) | Cell Viability (%) | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 7 | Positive Control | |
| 0 | 101.73 ± 6.54 | 104.55 ± 8.78 | 103.29 ± 10.85 | 101.29 ± 9.85 | 101.34 ± 13.23 |
| 3.125 | 158.43 ± 15.44 * | 111.75 ± 12.19 * | 163.99 ± 10.50 * | 107.99 ± 10.50 | 107.21 ± 9.56 |
| 6.25 | 181.54 ± 8.19 * | 125.07 ± 10.23 * | 171.45 ± 15.44 * | 111.45 ± 11.44 * | 115.25 ± 14.14 * |
| 12.5 | 208.88 ± 12.95 * | 129.13 ± 8.19 * | 186.90 ± 16.78 * | 116.90 ± 9.78 * | 169.32 ± 16.00 * |
| 25 | 314.67 ± 21.48 * | 134.23 ± 11.97 * | 214.22 ± 20.60 * | 121.22 ± 12.60 * | 197.66 ± 16.57 * |
| 50 | 228.94 ± 11.58 * | 125.76 ± 11.60 * | 175.73 ± 16.59 * | 115.73 ± 8.59 * | 153.23 ± 16.65 * |
2.3. Effects of the Phenolics 1–3, 7 on ALP Activity of Rat Osteoblast
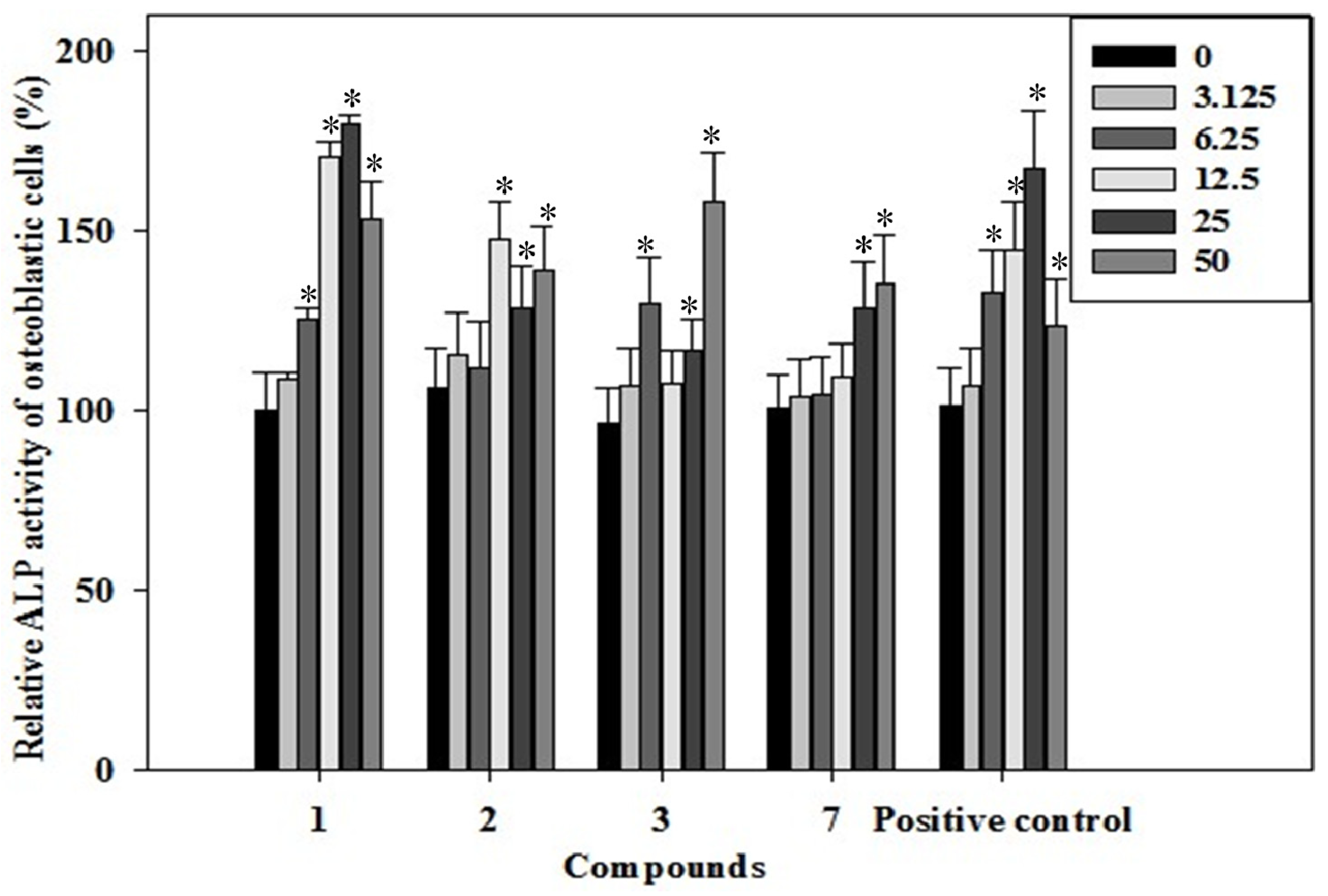
2.4. Effects of the Phenolics 1–3, 7 on Osteocalcin Content of the Rat Osteoblastic Cells
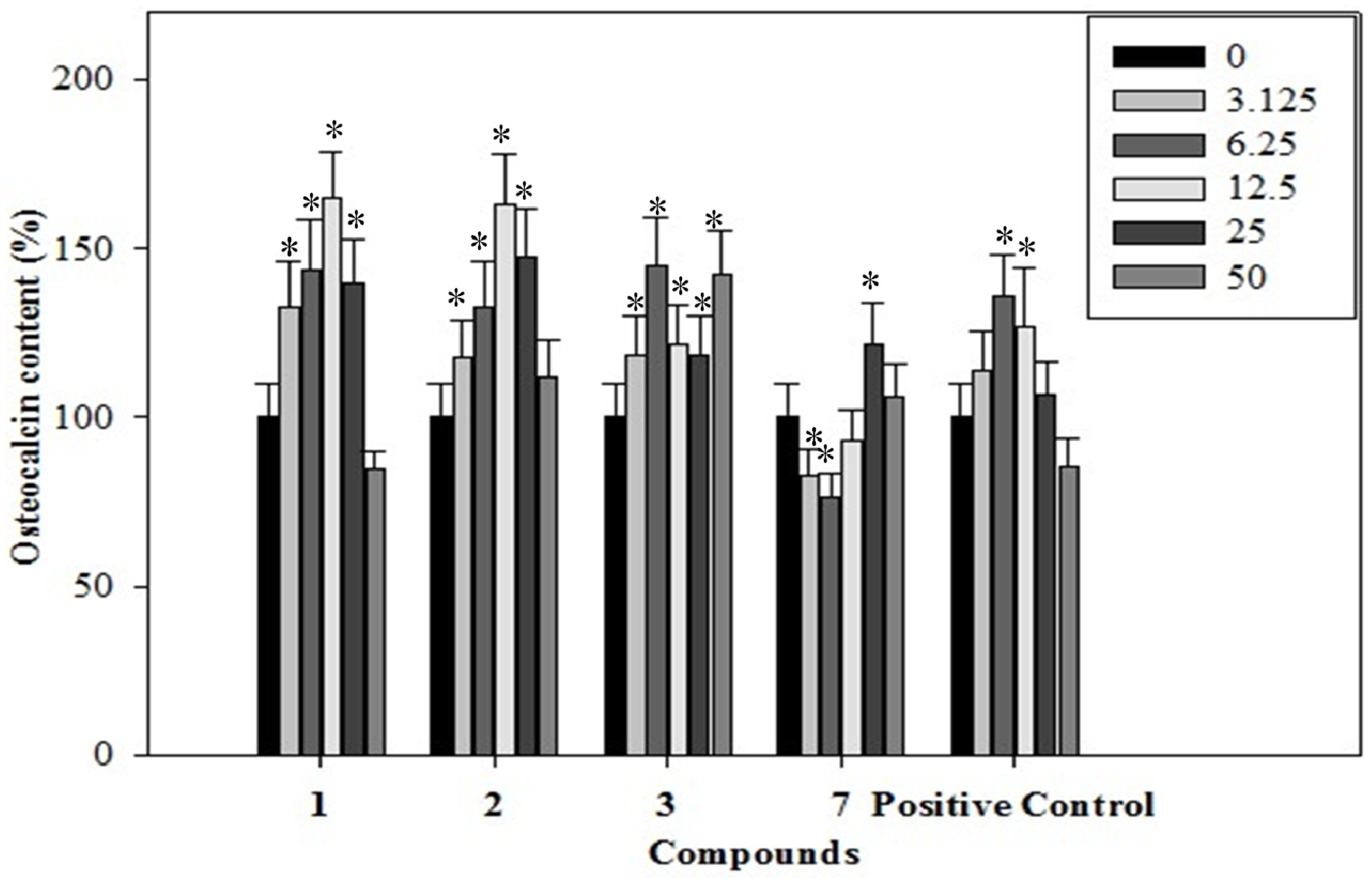
2.5. Effects of the Phenolics 1–3, 7 on Collagen Content of the Rat Osteoblastic Cells

2.6. Effects of the Phenolics 1–3, 7 on the Formation of Mineralized Bone Nodules
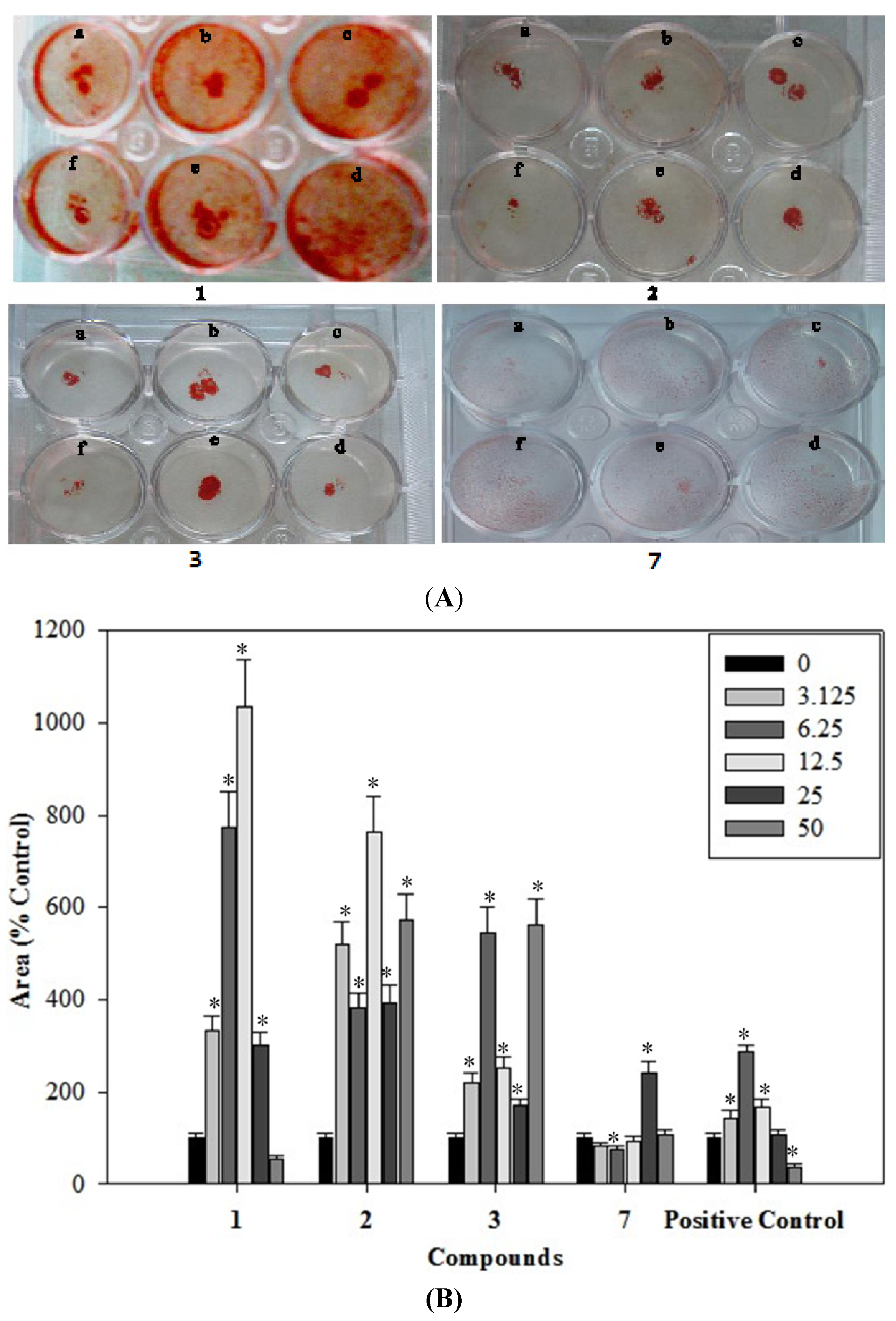
2.7. Antioxidantive Activity of the Phenolics 1–3, 7
2.7.1. Protective Effects against H2O2-Induced Cell Injury in C2C12 Mouse Myoblast Cells
| Concentrations (μg/mL) | (A) Cell Viability (%) | ||||
| 1 | 2 | 3 | 7 | Positive Control | |
| 0 | 211.23 ± 19.57 * | 205.92 ± 20.41 * | 211.45 ± 21.50 * | 197.99 ± 18.51 * | 210.12 ± 20.51 * |
| 100 μM H2O2 | 102.43 ± 8.34 | 103.66 ± 9.80 | 103.99 ± 8.08 | 102.50 ± 10.50 | 102.50 ± 9.65 |
| 100 μM H2O2 + 3.125 | 127.43 ± 6.34 | 113.66 ± 10.48 | 123.99 ± 10.09 | 106.81 ± 10.40 | 131.81 ± 12.40 |
| 100 μM H2O2 + 6.25 | 131.32 ± 11.57 * | 119.91 ± 10.50 | 126.02 ± 11.26 | 113.94 ± 11.33 | 163.94 ± 14.35 * |
| 100 μM H2O2 + 12.5 | 144.36 ± 18.83 * | 145.79 ± 12.61 * | 136.81 ± 13.33 * | 124.09 ± 12.64 * | 234.06 ± 21.63 * |
| 100 μM H2O2 + 25 | 198.44 ± 16.85 * | 154.01 ± 14.94 * | 188.02 ± 28.01 * | 134.24 ± 24.61 * | 499.24 ± 54.60 * |
| 100 μM H2O2 + 50 | 319.46 ± 23.20 * | 212.96 ± 19.07 * | 288.29 ± 22.14 * | 145.08 ± 16.06 * | 766.08 ± 79.15 * |
| Concentrations (μg/mL) | (B) Cell Apoptosis (%) | ||||
| 1 | 2 | 3 | 7 | Positive Control | |
| 0 | 2.50 ± 0.70 * | 2.55 ± 0.30 * | 2.40 ± 0.21 * | 2.04 ± 0.24 * | 2.35 ± 0.17 * |
| 100 μM H2O2 | 67.53 ± 4.56 | 66.87 ± 3.59 | 64.96 ± 6.73 | 68.67 ± 5.98 | 67.23 ± 4.39 |
| 100 μM H2O2 + 3.125 | 61.50 ± 5.73 | 62.50 ± 5.91 | 64.45 ± 3.34 | 66.50 ± 6.11 | 66.81 ± 6.41 |
| 100 μM H2O2 + 6.25 | 34.33 ± 3.44 * | 57.94 ± 5.34 * | 40.32 ± 3.57 * | 60.29 ± 6.07 | 61.94 ± 5.33 |
| 100 μM H2O2 + 12.5 | 9.56 ± 0.99 * | 46.09 ± 3.64 * | 13.36 ± 2.83 * | 55.13 ± 3.12 * | 13.09 ± 1.63 * |
| 100 μM H2O2 + 25 | 2.63 ± 0.19 * | 24.16 ± 2.15 * | 2.44 ± 0.19 * | 45.70 ± 4.13 * | 3.24 ± 0.20 * |
| 100 μM H2O2 + 50 | 1.92 ± 0.21 * | 10.68 ± 1.15 * | 1.46 ± 0.20 * | 23.89 ± 2.03 * | 0.68 ± 0.06 * |
2.7.2. DPPH Free Radical-Scavenging Activity
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. (2R,3R)-3,5,6,7,3',4'-Hexahydroxyflavane (1)
 = −43.0 (c 0.3, MeOH). ESI-MS (positive-ion mode) [M+Na]+ m/z 329. 1H-NMR (CD3OD) δH 4.77 (1H, brs, H-2), 4.14 (1H, m, H-3), 2.83 (1H, dd, J = 16.4, 4.8 Hz, H-4ax), 2.70 (1H, dd, J = 16.8, 2.8 Hz, H-4eq), 5.91 (1H, s, H-8), 6.94 (1H, d, J =1.6 Hz, H-2'), 6.72 (1H, d, J = 8.4 Hz, H-5'), 6.76 (1H, dd, J = 8.4, 2.0 Hz, H-6'); 13C-NMR (CD3OD): δC 79.9 (C-2), 67.6 (C-3), 29.5 (C-4), 157.7(C-5), 145.8 (C-6), 146.1 (C-7), 96.6 (C-8), 158.0 (C-9), 100.3 (C-10), 132.4 (C-1'), 115.5 (C-2'), 157.4 (C-3'), 157.2 (C-4'), 116.1 (C-5'), 119.6 (C-6').
= −43.0 (c 0.3, MeOH). ESI-MS (positive-ion mode) [M+Na]+ m/z 329. 1H-NMR (CD3OD) δH 4.77 (1H, brs, H-2), 4.14 (1H, m, H-3), 2.83 (1H, dd, J = 16.4, 4.8 Hz, H-4ax), 2.70 (1H, dd, J = 16.8, 2.8 Hz, H-4eq), 5.91 (1H, s, H-8), 6.94 (1H, d, J =1.6 Hz, H-2'), 6.72 (1H, d, J = 8.4 Hz, H-5'), 6.76 (1H, dd, J = 8.4, 2.0 Hz, H-6'); 13C-NMR (CD3OD): δC 79.9 (C-2), 67.6 (C-3), 29.5 (C-4), 157.7(C-5), 145.8 (C-6), 146.1 (C-7), 96.6 (C-8), 158.0 (C-9), 100.3 (C-10), 132.4 (C-1'), 115.5 (C-2'), 157.4 (C-3'), 157.2 (C-4'), 116.1 (C-5'), 119.6 (C-6').3.5. Anthracene-2,4,9-triol (2)
3.6. Rat Osteoblast Cell Culture
3.7. C2C12 Cell Culture
3.8. Cell Viability Assay
3.9. Alkaline Phosphatase (ALP) Activity
3.10. Osteocalcin Assay
3.11. Hydroxyproline Assay
3.12. Determination and Quantification of Mineralized Bone Nodules
3.13. Flow Cytometric Analysis for Apoptosis
3.14. DPPH Radical Scavenging Assay
3.15. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Ozgocmen, S.; Kaya, H.; Fadilliogl, E.; Aydogan, R.; Yilmaz, Z. Role of antioxidant systems, lipid peroxidation, and nitric oxide in postmenopausal osteoporosis. Mol. Cell. Biochem. 2007, 295, 45–52. [Google Scholar] [CrossRef]
- Overton, T.R.; Basu, T.K. Longitudinal changes in radial bone density in older men. Eur. J. Clin. Nutr. 1999, 53, 211–215. [Google Scholar]
- Linnane, A.W.; Eastwood, H. Cellular redox regulation and prooxidant signaling systems, a new perspective on the free radical theory of aging. Ann. N. Y. Acad. Sci. 2006, 1067, 47–55. [Google Scholar]
- Sendur, O.F.; Turan, Y.; Tastaban, E.; Serter, M. Antioxidant status in patients with osteoporosis: A controlled study. Joint Bone Spine 2009, 76, 514–518. [Google Scholar] [CrossRef]
- Mody, N.; Parhami, F.; Saraflan, T.A.; Demer, L.L. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radic. Biol. Med. 2001, 31, 509–519. [Google Scholar] [CrossRef]
- Fatokun, A.A.; Stone, T.W.; Smith, R.A. Responses of differentiated MC3T3-E1 osteoblast-like cells to reactive oxygen species. Eur. J. Pharm. 2008, 587, 35–41. [Google Scholar] [CrossRef]
- Yang, S.; Madyastha, P.; Bingel, S.; Ries, W.; Key, L. A new superoxide-generating oxidase in murine osteoclasts. J. Bio. Chem. 2001, 276, 5452–5458. [Google Scholar] [CrossRef]
- Sontakke, A.N.; Tare, R.S. A duality in the roles of reactive oxygen species with respect bone metabolism. Clin. Chim. Acta 2002, 318, 145–148. [Google Scholar] [CrossRef]
- Healthy Ministry of Guangzhou Force Logistics. Common Chinese Herbal Medicine Handbook; People’s Health Publishing House: Beijing, China, 1969. [Google Scholar]
- Zhao, G.P.; Dai, S.; Chen, E.S. Dictionary of Traditional Chinese Medicine; Shanghai Science and Technology Press: Shanghai, China, 2001. [Google Scholar]
- Chen, P.; Yang, J.S. Studies on chemical constituents of Livistona chinensis seeds. Chin. Tradit. Herb Drugs 2007, 8, 665–667. [Google Scholar]
- Chen, P.; Yang, J.S. Studies on chemical constituents of Livistona chinensis seeds. Chin. Pharm. J. 2008, 43, 1669–1670. [Google Scholar]
- Tao, Y.; Yang, S.P.; Zhang, H.Y.; Liao, S.G.; Wei, W.; Yan, W.; Wu, Y.; Tang, X.C.; Yue, J.M. Phenolic compounds with cell protective activity from the fruits of Livistona chinensis. J. Asian Nat. Prod. Res. 2009, 11, 243–249. [Google Scholar] [CrossRef]
- Zeng, X.B.; Qiu, Q.; Jiang, C.G.; Jing, Y.T.; Qiu, G.F.; He, X.J. Antioxidant flavanes from Livistona chinensis. Fitoterpia 2011, 82, 609–614. [Google Scholar] [CrossRef]
- Zeng, X.B.; Wang, Y.H.; Qiu, Q.; Jiang, C.G.; Jing, Y.T.; Qiu, G.F.; He, X.J. Bioactive phenolics from the fruits of Livistona chinensis. Fitoterapia 2012, 83, 104–109. [Google Scholar] [CrossRef]
- Zeng, X.B.; Xiang, L.M.; Li, C.Y.; Wang, Y.H.; Qiu, G.F.; Zhang, Z.X.; He, X.J. Cytotoxic ceramides and glycerides from the roots of Livistona chinensis. Fitoterapia 2012, 83, 609–616. [Google Scholar] [CrossRef]
- Zeng, X.B.; Li, C.Y.; Wang, H.; Qiu, Q.; Qiu, G.F.; He, X.J. Unusual lipids and acylglucosylsterols from the roots of Livistona chinensis. Phytochem. Lett. 2013, 6, 36–40. [Google Scholar] [CrossRef]
- Waterman, P.G.; Faulkner, D.F. (−)-Epiafzelechin from the Root Bark of Cassia sieberiana. Planta Med. 1979, 37, 178–179. [Google Scholar] [CrossRef]
- Jin, G.Z.; Jin, H.S.; Jin, L.L. Synthesis and antiproliferative activity of 1,4-bis(dimethylamino)-9,10-anthraquinone derivatives against P388 mouse leukemic tumor cells. Arch. Pharm. Res. 2011, 34, 1071–1076. [Google Scholar] [CrossRef]
- Aubin, J.E. Advances in osteoblast lineage. Biochem. Cell. Biol. 1998, 76, 899–910. [Google Scholar] [CrossRef]
- Lian, B.J.; Stein, G.S.; Canalis, E.; Robey, P.G.; Boskey, A.L. Bone Formation: Osteoblast Lineage Cells, Growth Factors, Matrix Proteins and the Mineralization Process. In Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism; Favus, M.J., Ed.; Lippincott Williams & Williams: Philadelphia, PA, USA, 1999; p. 461. [Google Scholar]
- Tong, A.L.; Chen, L.L.; Ding, G.Z. Progress of mechanism of osteoblastic bone formation. Chin. J. Osteoporos. 1999, 5, 60–64. [Google Scholar]
- Lee, N.K.; Sowa, H.; Hinoi, E.; Ferron, M.; Ahn, J.D.; Confavreux, C.; Dacquin, R.; Mee, P.J.; McKee, M.D.; Jung, D.Y.; et al. Endocrine regulation of energy metabolism by the skeleton. Cell 2007, 130, 456–469. [Google Scholar] [CrossRef]
- Sakano, S.; Murata, Y.; Miura, T. Collagen and alkaline phosphatase gene expression during bone morphogenetic protein (BMP)-induced cartilage and bone differentiation. Clin. Orthop. Relat. Res. 1993, 292, 337–344. [Google Scholar]
- Riggs, B.L.; Melton, L.J. The prevention and treatment of osteoporosis. N. Engl. J. Med. 1992, 327, 620–627. [Google Scholar] [CrossRef]
- Nohl, H. Involvement of free radicals in ageing: A consequence or cause of senescence. Br. Med. Bull. 1993, 49, 653–657. [Google Scholar]
- Basu, K.; Michaëlsson, H.; Olofsson, H.; Johansson, S.; Melhus, H. Association between oxidative stress and bone mineral density. Biochem. Biophys. Res. Commun. 2001, 288, 275–279. [Google Scholar] [CrossRef]
- Muthusami, S.; Ramachandran, I.; Muthusamy, B.; Vasudevan, G.; Prabhu, V.; Subramaniam, V.; Jagadeesan, A.; Narasimhan, S. Ovariectomy induces oxidative stress and impairs bone antioxidant system in adult rats. Clin. Chim. Acta 2005, 360, 81–86. [Google Scholar] [CrossRef]
- Isomura, H.; Fujie, K.; Shibata, K.; Inoue, N.; Iizuka, T.; Takebe, G.; Takahashi, K.; Nishihira, J.; Izumi, H.; Sakamoto, W. Bone metabolism and oxidative stress in postmenopausal rats with iron overload. Toxicology 2004, 197, 93–100. [Google Scholar]
- Yalin, S.; Bagis, S.; Aksit, S.C.; Arslan, H.; Erdogan, C. Effect of free radicals and antioxidants on postmenopausal osteoporosis. Asian J. Chem. 2006, 18, 1091–1096. [Google Scholar]
- Zhang, D.W.; Cheng, Y.; Wang, N.L.; Zhang, J.C.; Yang, M.S.; Yao, X.S. Effects of total flavonoids and flavonol glycosides from Epimedium koreanum Nakai on the proliferation and differentiation of primary osteoblasts. Phytomedicine 2008, 15, 55–61. [Google Scholar] [CrossRef]
- Zeng, X.B.; Su, Y.J.; Zheng, Y.Y.; Cui, L. Osteogenic effects of the flavanes from green tea polyphenols. Acta Pharmacol. Sin. 2013, S2, 34. [Google Scholar]
- Declercq, H.; Vreken, N.V.; Maeyer, E.D.; Verbeeck, R.; Schacht, E.; Ridder, L.D.; Cornelissen, M. Isolation, proliferation and differentiation of osteoblastic cells to study cell/biomaterial interactions: Comparison of different isolation techniques and source. Biomaterials 2004, 25, 757–768. [Google Scholar] [CrossRef]
- He, X.J.; Liu, R.H. Triterpenoids isolated from apple peels maybe responsible for their anticancer activity. J. Agric. Food Chem. 2007, 55, 4366–4370. [Google Scholar] [CrossRef]
- Rao, L.G.; Liu, L.J.; Murray, T.M.; McDermott, E.; Zhang, X. Estrogen added intermittently but not continuously, stimulates differentiation and bone formation in SaOS-2 cells. Biol. Pharm. Bull. 2003, 26, 936–945. [Google Scholar] [CrossRef]
- Hale, L.V.; Ma, Y.F.; Santerre, R.F. Semi-quantitative fluorescence analysis of calcein binding as a measurement of in vitro mineralization. Calcif. Tissue Int. 2000, 67, 80–84. [Google Scholar] [CrossRef]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food. Chem. 1995, 43, 27–32. [Google Scholar] [CrossRef]
- Manolagas, S.C. De-fense! De-fense! De-fense: Scavenging H2O2 while making cholesterol. Endocrinology 2008, 149, 3264–3266. [Google Scholar] [CrossRef]
- Garrett, J.R.; Boyce, B.F.; Oreffo, R.O.; Bonewald, L.; Poser, J.; Mundy, G.R. Oxygen-derived free radicals stimulate osteoclastic bone resorption in rodent bone in vitro and in vivo. J. Clin. Invest. 1990, 85, 632–639. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds (1–8) are available from the authors.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zeng, X.; Tian, J.; Cui, L.; Wang, Y.; Su, Y.; Zhou, X.; He, X. The Phenolics from the Roots of Livistona chinensis Show Antioxidative and Obsteoblast Differentiation Promoting Activity. Molecules 2014, 19, 263-278. https://doi.org/10.3390/molecules19010263
Zeng X, Tian J, Cui L, Wang Y, Su Y, Zhou X, He X. The Phenolics from the Roots of Livistona chinensis Show Antioxidative and Obsteoblast Differentiation Promoting Activity. Molecules. 2014; 19(1):263-278. https://doi.org/10.3390/molecules19010263
Chicago/Turabian StyleZeng, Xiaobin, Jun Tian, Liao Cui, Yang Wang, Yanjie Su, Xin Zhou, and Xiangjiu He. 2014. "The Phenolics from the Roots of Livistona chinensis Show Antioxidative and Obsteoblast Differentiation Promoting Activity" Molecules 19, no. 1: 263-278. https://doi.org/10.3390/molecules19010263




